Abstract
The MAPK-interacting kinases 1 and 2 (MNK1/2) have generated increasing interest as therapeutic targets for many cancers with little known in osteosarcoma. This study evaluated the efficacy of eFT508, a highly selective inhibitor of MNK1/2, as single drug alone and in combination with paclitaxel in preclinical models of osteosarcoma. EFT508 is active against multiple osteosarcoma cell lines via inhibiting growth, survival and migration. It also demonstrates anti-osteosarcoma selectivity with much less toxicity on normal osteoblastic than osteosarcoma cells. Consistent with in vitro findings, eFT508 at non-toxic dose significantly arrested tumor growth in mice throughout the whole duration of treatment. Mechanistically, eEFT508 is highly effective in blocking eIF4E phosphorylation and eIF4E-mediated protein translation. Combination index shows that eFT508 and paclitaxel is synergistic in osteosarcoma cells. Our findings highlight the therapeutic value of MNK1/2 inhibition and suggest eFT508 as a promising candidate for the treatment of osteosarcoma.
Introduction
Osteosarcoma is the most common type of cancer that initiates at the bone and is characterized by widespread and recurrent chromosomal and genetic abnormalities.1,2
Although surgery combined with systemic chemotherapy using doxorubicin and paclitaxel has significantly improved outcome, the clinical management of recurrent and refractory disease is still challenging. In addition, the treatment for osteosarcoma has not changed over the past 30 years. 3 Targeted therapy has being hotly investigated for the treatment of osteosarcoma and approximately 30% of related clinical trials involve targeted drugs using inhibitors of cyclin-dependent kinases (CDK), receptor tyrosine kinase (RTK), mammalian target of rapamycin (mTOR), the mitogen-activated protein kinase (MAPK) and histone deacetylase (HDAC). 4
MAPK signaling pathway is aberrantly activated in osteosarcoma and plays a role in the aggressive progression.5,6 MAPK-interacting kinases 1 and 2 (MNK1/2) are downstream effectors of this pathway, which phosphorylates eukaryotic translation initiation factor 4E (eIF4E) at serine 209 and activates eIF4E-mediated protein translation. eIF4E is upregulated and is closely linked to malignant cell transformation, growth, survival, migration and chemoresistance in many cancers, including osteosarcoma.7–9 MNK knock-out mice display no adverse effects on normal cells or phenotypes, 10 suggesting that inhibiting eIF4E via targeting MNK is a potential strategy to inhibit cancer without causing toxicity in normal cells. Several MNK inhibitors have been identified and evaluated in preclinical and clinical trials for cancer treatment for the last decade. 11 We hypothesized that MNK inhibition is effective against osteosarcoma and enhances efficacy of chemotherapy. EFT508 is a potent, highly selective and orally bioavailable inhibitor of both MNK1 and MNK2, with half-maximal inhibitory concentration (IC50) of 2.4 nM and 1 nM, respectively. 11
In this work, we aimed to investigate the therapeutic potential of MNK inhibition in osteosarcoma and whether eFT508 is effective against osteosarcoma. We systematically evaluated the efficacy of eFT508 alone and its combination with paclitaxel using multiple pre-clinical osteosarcoma models, and confirmed MNK inhibition as the underlying mechanism of eFT508 in osteosarcoma.
Materials and methods
Cell culture, generation of cell lines and compounds
Human osteosarcoma lines (MG-63, U2OS and OST) and the human normal osteoblastic cell line OB-6 (the Cell Bank of Shanghai Institute of Biological Science) were cultured in RPMI-1640 medium supplemented with 2 mM L-glutamine, 100 U/ml of penicillin, 100 µg/ml of streptomycin and 10% FBS in humidified 5% CO2 at 37 0C. Cells were authenticated using the human 9-Marker STR DNA profile analysis (Xpbiomed Biotechnology). EFT508 and paclitaxel (Selleckchem) were reconstituted in dimethyl sulfoxide (DMSO) and were stored in aliquots at −20 0C.
Cell proliferation and combination index analysis
Cells at 5000/well were seeded in 96-well plate. After 3 days drug treatment, proliferation was measured using BrdU Cell Proliferation Assay (Abcam) as per manufacturer’s instructions. Combination studies were designed based on the median-effect equation method. 12 Briefly, cells were treated with increasing doses of paclitaxel or eFT508 alone or an equipotent constant-ratio concentration of paclitaxel and eFT508. The CI at 0% to 100% growth inhibition was calculated using the CalcuSyn software.
Flow cytometry of Annexin V and 7-AAD
Cells at 106/well were seeded in 6-well plate. After 3 days drug treatment, cells were harvested for Annexin V and 7-AAD double staining (Abcam), followed by flow cytometry analysis (Miltenyibiotec Inc.). Stained cells were analyzed on Beckman Coulter FC500 cytometer. Annexin V+/7-AAD+ and Annexin V+/7-AAD- were considered as apoptosis.
Cell migration assay
Cell migration assay was performed using Cell Biolabs CytoSelect cell migration assay kit with slight modification. Cells at 104/well together with drug were seeded into insert and placed in the upper chamber. Medium containing 2% FBS was placed onto upper chamber and medium containing 10% FBS were placed onto lower chamber. After 24-hour incubation in a cell culture incubator, non-migrated cells were wiped out by cotton swab and migrated cells on the lower surface of inserts were stained with Giemsa (Sigma). The migrated cells were photographed and counted under light microscopy.
Western blot (WB) analyses
Cells at 106/well were seeded in 6-well plate. After 24 hours drug treatment, cell lysates were generated by lysing cells in cell lysis buffer (Cell Signaling) on ice, followed by centrifugation. Supernatants were separated for WB analyses using standard protocol. 13 Antibodies against p-eIF4E (#9741), eIF4E (#9742), Mcl-1 (#94296), VEGF (#65373) and cyclins D1 (#2978) were obtained from Cell Signaling Technologies. Anti-β-actin (#A1978) antibody was from Sigma. Immunoreactive bands were detected using chemiluminescence (GE Healthcare).
Real-time RT-PCR
Expression levels of Cyclin D1, Mcl-1 and VEGF were analyzed using quantitative PCR. After 24 hours drug treatment, total RNA was extracted using TRIzol (Invitrogen). First-strand cDNA was synthesized using iScript cDNA Synthesis Kit (Bio-rad, CA) and mixed with a SsoFast EvaGreen Supermix (Bio-rad, CA) for real-time PCR analysis (CFX96). The primers are: Cyclin D1 5’-AGA AGC TGT GCA TCT ACA CCG ACA-3’ and 5’-TGA TCT GTT TGT TCT CCT CCG CCT-3’; VEGF, 5’-GTA CCT CCA CCA TGC CAA GT-3’ and 5’-AAT AGC TGC GCT GGT AGA CG-3’; Mcl-1 5’-GGA CAC AAA GCC AAT GGG CAG GT-3’ and 5’-GCA AAA GCC AGC AGC ACA TTC CTG’3; GAPDH 5´-GCA CCG TCA AGG CTG AGA AC-3´ and 5´-TGG TGA AGA CGC CAG TGG A-3´.
Osteosarcoma xenograft mouse model
Male severe combined immunodeficiency (SCID) mice were purchased from Hunan Slaccas Jingda Laboratory Animal Co. and housed in a pathogen-free environment. MG-63 cells at 107 in 100 µl PBS were subcutaneously implanted into mice flank. When tumour reached ∼100 mm3 after a week of tumour inoculation, mice were divided into three groups (n = 8 per group) for treatment: 20%/80% DMSO/saline, eFT508 at 1 mg/kg and eFT508 at 15 mg/kg. Drugs were given once per day via oral gavage for 31 days. Tumour volume was calculated using the formula 0.52 X length X (width2). Tumour volume and mice body weight were monitored throughout the treatment duration.
Statistical analyses
Statistical analyses of the differences between two groups were performed using the one-way analysis of variance (ANOVA) and subsequently by unpaired Student’s t test. p values < 0.05 is considered statistically significant.
Results
EFT508 is effective and selective in inhibiting osteosarcoma cells
We investigated the effects of eFT508 on osteosarcoma cell growth, survival and migration using three human osteosarcoma cell lines: MG-63, U2SO and OST. These cell lines are representative of in vitro osteosarcoma models and have been shown to be useful for functional drug screening.
14
We found that eFT508 significantly inhibited proliferation of all tested osteosarcoma cell lines, with IC50 < 10 µM (Figure 1(a)). In addition, eFT508 at the same concentrations either did not or inhibited less proliferation in normal osteoblastic cell line OB-6 than osteosarcoma cells. Similarly, eFT508 induced apoptosis in osteosarcoma cells, and to a more extent than in OB-6 cells (Figure 1(b)). These results clearly demonstrate that eFT508 has anti-osteosarcoma selectivity. We further found that Z-VAD-fmk, a pan-caspase inhibitor, completely reversed the pro-apoptotic effect of eFT508 in osteosarcoma cells (Figure 1(c)), indicating that eFT508 induces apoptosis via caspase-dependent pathways. We showed that eFT508 significantly inhibited osteosarcoma cell migration (Figures 1(d) and (e)), as assessed by Boyden chamber assay. Collectively, our results demonstrate that eFT508 is effective in inhibiting osteosarcoma cell growth, survival and migration while spraining normal osteoblastic cells. EFT508 is effective in inhibiting osteosarcoma cells while sparing normal osteoblastic cells. The anti-proliferative (a) and pro-apoptotic (b) effects of eFT508 on human osteosarcoma cell lines MH-63, U2OS and OST, and human normal osteoblastic cell line OB-6. (c) A pan-caspase inhibitor Z-VAD-fmk completely reversed the pro-apoptotic effect of eFT508 (50 µM) on osteosarcoma cells. Z-VAD-fmk at 10 µM was used. Z + T is Z-VAD-fmk and eFT508. (d) Representative images of cell migration after control (DMSO) and eFT508 (50 µM) treatment. (e) EFT508 significantly decreases osteosarcoma cell migration. Results were obtained from at least three time-independent experiments and presented as mean ± SD. EFT508 at 1, 5, 10 and 50 µM were used. *p < 0.05, compared to control.
EFT508 acts synergistically with paclitaxel in osteosarcoma cells
As chemoresistance is the main cause of treatment failure in patients with osteosarcoma, it is important to address the issue whether eFT508 overcome chemoresistance by enhancing efficacy of chemotherapeutic agent. We used two approaches to address this question. The first approach was to perform combination studies using drug concentration that induces <50% inhibition as single drug alone. We found that eFT508 or paclitaxel alone inhibited proliferation by 40% to 50% and induced apoptosis by 30% to 40% in three osteosarcoma cell lines (Figures 2(a) and (b)). However, the combination of eFT508 and paclitaxel resulted in 80% to 90% growth inhibition and apoptosis induction, suggesting that the combination is much more effective than single drug alone in osteosarcoma cells. The second approach was to perform studies based on the Chou-Talalay method using equipotent concentration of each drugs, followed by calculation of combination index (CI) which offers quantitative definition for additive effect (CI = 1), synergism (CI < 1), and antagonism (CI > 1) in drug combination.
12
Isobologram analysis showed that CI value of eFT508 and paclitaxel combination at 50% and 80% was all less than 1 (Figures 2(c) to (e)), demonstrating that the combination is synergistic in osteosarcoma cells. EFT508 acts synergistically with paclitaxel on osteosarcoma cells. The combination of eFT508 and paclitaxel is significantly more effective in inhibiting proliferation (a) and inducing apoptosis (b) on osteosarcoma cells than paclitaxel alone. Paclitaxel at 0.1 µM and 0.5 uM was used for proliferation and apoptosis analysis, respectively. Combination index (CI) vs fraction affected (Fa) for MG-63 (c), U2OS (d) and OST (e) at combination of eFT508 and paclitaxel. CI50 < 1 indicates synergism and CI50 > 1 indicates antagonism. Results were obtained from at least three time-independent experiments and presented as mean ± SD. *p < 0.05, compared to paclitaxel.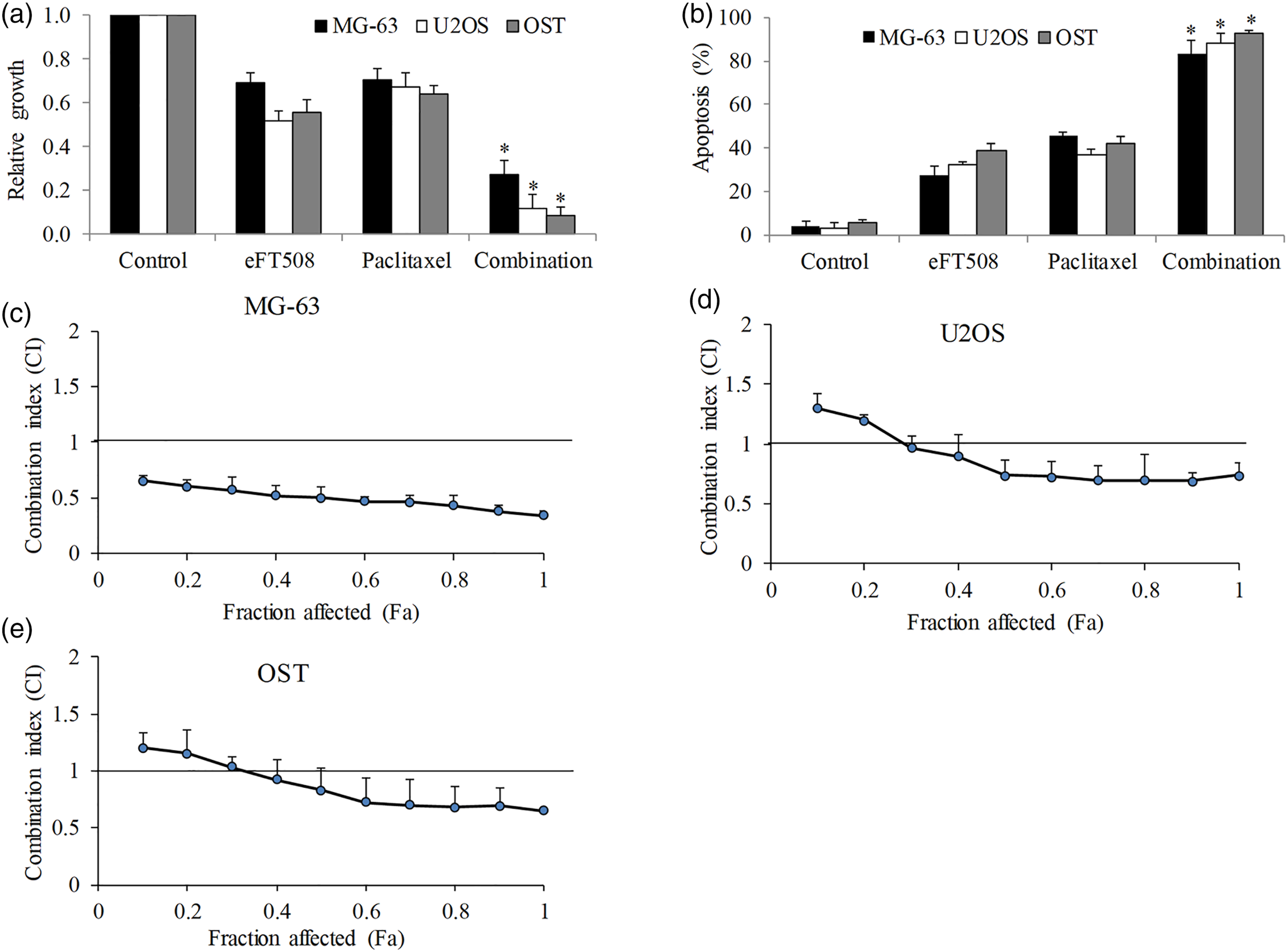
EFT508 inhibits osteosarcoma cells via targeting eIF4E
We next analyzed the phosphorylation of Mnk effector eIF4E and its mediated protein translation in osteosarcoma cells exposed to eFT508. Consistent with the previous findings,15,16 we observed the remarkable reduction of p-eIF4E at ser209 site in osteosarcoma cells after eFT508 treatment even at the lowest concentration (Figures 3(a) and (b)). We further observed a significant reduction of protein level but not mRNA level of eIF4E-targeted genes: VEGF, Cyclin D1 and Mcl-1 (Figures 3(b) and (c)), indicating that eFT508 inhibits eIF4E-mediated protein translation. EFT508 inhibits osteosarcoma cells in an eIF4E-dependent manner. Image (a) and quantification (b) of western blot analysis of p-eIF4E, VEGF, Cyclin D1 and Mcl-1 in MG-63 cells after eFT508 treatment. Quantification of western blot was performed using imaging J and normalized with β-actin. (c) mRNA level of VEGF, Cyclin D1 and Mcl-1 in MG-63 cells after eFT508 treatment. Results were obtained from at least three time-independent experiments and were presented as mean ± SD. *p < 0.05, compared to control.
eFT508 suppresses osteosarcoma growth in mice without causing toxicity
To investigate the translational potential of eFT508, we challenged eFT508 using xenograft osteosarcoma mouse model.
17
We inoculated mice with MG-63 cells and initialized eFT508 treatment after the development of palpable tumours. We found that oral eFT508 at 1 mg/kg displayed a slight inhibition of osteosarcoma growth in mice. EFT508 at 15 mg/kg significantly inhibited osteosarcoma growth throughout the duration of treatment (Figure 4(a)). In addition, eFT508 at 15 mg/kg delayed tumour growth beginning at 5 days of the initial treatment and inhibited growth by 50% after 30 days treatment. We monitored overall mouse health, such as ungroomed fur, change in fur colour and weight loss in mice given eFT508 and did not observe any toxicity signs (Figure 4(b) and data not shown). Collectively, our results show that eFT508 dose-dependently suppresses osteosarcoma growth in mice without causing toxicity. EFT508 significantly inhibits osteosarcoma growth. (a) EFT508 at 15 mg/kg significantly inhibits osteosarcoma growth in mice. MG-63 cells were inoculated into the flanks of SCID mice and treated with either eFT508 or vehicle (20%/80% DMSO/saline) via oral gavage. (b) Mice body weight in control and eFT508 groups on day 0 and day 37. Tumor volume was measured at the indicated timepoints. Results were presented as mean ± SD. *p < 0.05, compared to control.
Discussion
In the present study, we are the first to find that MNK/eIF4E inhibition by eFT508 has activity in osteosarcoma while spraining normal counterpart. Whereas the conventional chemotherapy has limited effectiveness and high toxicity, the combination of eFT508 with paclitaxel results in lower doses of each drug to minimize toxicity meanwhile maximizing efficacy in our tested preclinical osteosarcoma models. The ability of eFT508 to extinguish normal and malignant osteoblastic cells and synergize with chemotherapeutic agent may contribute to improved control of osteosarcoma in patients.
The anti-cancer activities of eFT508 have been shown in many solid cancers and leukemia.15,16,18–20 Various clinical trials have been actively undergoing to investigate the efficacy of eFT508 as single drug or in combination with conventional toxicity drugs or anti-PD-1 therapy in prostate cancer, breast cancer, lymphoma and non-small cell lung cancer. In lines with these efforts, we revealed the selective anti-proliferative and pro-apoptotic effects of eFT508 in osteosarcoma cells regardless of cellular origin and genetic background. Because osteosarcoma metastasis is the main clinical issue in osteosarcoma patients, the anti-migratory effect of eFT508 suggests that eFT508 might inhibit osteosarcoma metastasis, which is worthy of future investigation on osteosarcoma metastasis using orthotopic mouse model. Of note, the selectivity and efficacy of eFT508 observed in vitro are all reproducible in vivo. Using osteosarcoma subcutaneous mouse model, we observed the osteosarcoma growth arrest in eFT508-treated mice without causing significant toxicity throughout the whole treatment duration.
Our combination index analysis shows synergism between eFT508 and paclitaxel in osteosarcoma cells. This is consistent with the previous finding that combination of eFT508 and venetoclax resulted in synergistic anti-leukemic responses in AML cells. 15 In addition, eFT508 preferentially sensitizes gastric cancer to chemotherapy. 19 Synergistic effect of eFT-508 and Adriamycin were reported in breast tumor xenografts. 20 It is worthy of evaluating the combinatory effects of eFT508 with chemotherapy in osteosarcoma patient-derived xenograft mouse models. The preclinical evidence obtained from our work and others suggest that eFT508 is highly likely to act synergistically with anti-cancer therapies, and support the relevant clinical trials for the treatment of osteosarcoma.
To understand the molecular mechanisms of eFT508’s action in osteosarcoma, we investigated eIF4E phosphorylation and eIF4E-mediated translational activity after eFT508 treatment. We confirm that the action of eFT508 in osteosarcoma is via MNK/eIF4E. This is consistent with the previous work on the fact that eFT508 is a highly selective MNK1/2 inhibitor.16,19 As eIF4E modulates the translation of tumor associated-mRNAs rather than global protein synthesis such as VEGF, cyclin D1 and Mcl-1 as shown in our study, eIF4E inhibition is an attractive therapeutic strategy in many cancers.21–24 Inhibiting eIF4E via ribavirin has demonstrated to augment efficacy of doxorubicin in osteosarcoma. 7 MNK is the only kinase known to phosphorylate eIF4E at serine 209 and MNK1/2 is dispensable for normal cell growth and development. 10 This is supported by our findings that eFT508 displays anti-osteosarcoma selectivity with much less toxicity on normal osteoblastic than osteosarcoma cells. Inhibition of the MNK1/2-eIF4E signaling axis has advantage than eIF4E inhibition to provide a safer and more specific option for cancer treatment. Diverse MNK inhibitors with varying MNK selectivity have been developed but all show robust anti-tumor response.25–27 Yang et al.’s work demonstrates that β-catenin is the downstream target of MNK/eIF4E inhibition by eFT508. 19 Besides eFT508, a panel of small molecule MNK kinase inhibitors have been shown to prevent eIF4E phosphorylation and β-catenin activation. 28 β-catenin inhibition sensitizes osteosarcoma to standard chemotherapy, 29 and this is supported by our findings that eFT508 and paclitaxel is synergistic in osteosarcoma cells.
In summary, we show that eFT508 selectively targets osteosarcoma cells through inhibition of MNK/eIF4E, and acts synergistically with paclitaxel. eFT508 single arm is effective against osteosarcoma growth in mice without causing toxicity. Hence, targeting MNK/eIF4E may represent a new therapeutic strategy against osteosarcoma.
Footnotes
Author contributions
BH and PCJ performed the experiments, analyzed the data and wrote the manuscript. KJY performed the experiments and analysed the data. JHD supervised the study and edited the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hubei University of Medicine (CG2019066J).
Ethical approval
Animal work was approved by the Institutional Animal Care and Use Committee of Hubei University of Medicine (Approved Number IACUC-081). This study does not involve human participants. The procedures were carried out in accordance with the guidelines approved by the Ethics Committees of Hubei University of Medicine.
Data availability
Data will be made available upon reasonable request from the corresponding author.
