Abstract
Objectives
The study aimed to examine long-term survival of patients with acute paraquat poisoning using computed tomography (CT) images and spirometry.
Methods
A total of 36 patients with long-term survival after paraquat poisoning were followed-up and divided into mild (11 patients), moderate (17 patients), and severe (8 patients) paraquat poisoning groups. Differences among the groups were compared using clinical indicators, such as peripheral capillary oxygen saturation, arterial partial pressure of oxygen and 6-min walk test (6-MWT), chest CT, spirometry, and serum immunoglobulin E (IgE).
Results
The 6-MWT distance was significantly shorter in the severe paraquat poisoning group than that in the mild and moderate paraquat poisoning groups. In the mild paraquat poisoning group, CT revealed no obvious lung injury, and spirometry showed normal lung function in most patients. In moderate or severe paraquat poisoning group, CT images showed fibrotic lesions as cord-like high-density shadows, reticulations, and honeycombs. In addition, other pulmonary changes, including bronchiectasis, increased lung transparency, and pulmonary bullae, were discovered. In moderate or severe paraquat poisoning group, obvious obstructive ventilation dysfunction with slight restrictive and diffuse impairment were observed in some patients, with positive bronchial relaxation test and high serum IgE level.
Conclusion
In the long-term follow-up, patients with severe paraquat poisoning showed the lowest exercise endurance. In moderate or severe paraquat poisoning group, CT images revealed diversified changes, not only dynamic evolution of pulmonary fibrosis process, but also signs of bronchiectasis, and chronic obstructive pulmonary disease. Some patients with moderate or severe paraquat poisoning developed obstructive ventilatory dysfunction with airway hyperresponsiveness.
Introduction
While patients with acute paraquat poisoning often die within 2 weeks from acute respiratory distress syndrome (ARDS) or multiple organ failure, those who survive have varying degrees of pulmonary fibrosis that clinically manifest as hypoxemia, dyspnea, and decreased exercise tolerance. 1 Although toxicants like paraquat are cleared from the body within a relatively short period after acute exposures, paraquat-induced acute toxicity can result in longer-term onset of potential pulmonary injury, up to and including lung fibrosis. However, combined assessments of imaging and lung function on progression or recovery of acute lung injury is not known. In this study, patients with at least 5 years survival after acute paraquat poisoning were followed-up. In addition, lung images and spirometric parameters were summarized to understand the sequence and impact of initial poisoning intensity on long-term paraquat poisoning adverse outcomes.
Methods
Study design
Patients with acute paraquat poisoning treated in the emergency department of Northern Jiangsu People’s Hospital from July 2012 to July 2016 were included in the study. The patients were followed-up via telephonic communication and asked to return to the hospital for further evaluation. The study met the medical ethical standards and was approved by the ethical committee of our hospital (approval number: 2021ky134). Written informed consent was obtained for follow-up and blood sample utilization from each patient, their relatives, or caretakers.
In the present study, acute paraquat poisoning was diagnosed based on the following: (1) Medical history of oral paraquat ingestion for suicide provided by patients or their family members. (2) Urine samples were collected from patients with suspected paraquat poisoning at admission. These samples underwent semi-quantitative detection using the sodium disulfite chromatographic method, which was compared with the standard colorimetric card. The blue color of the urine–reagent mixture indicated positive result, diagnosing the patient with paraquat poisoning.
The inclusion criteria were as follows: (1) Oral ingestion of paraquat for suicide and positive urine semi-quantitative detection; (2) age >14 years; and (3) patients with complete computed tomography (CT) imaging data during hospitalization.
The exclusion criteria were as follows: (1) Patients diagnosed with tuberculosis and chronic lung diseases, such as asthma, chronic obstructive pulmonary disease (COPD), bronchiectasis, and malignant lung tumors, during the period of acute paraquat poisoning; (2) patients with severe cardiac, hepatic, renal, or cerebrovascular disease; (3) oral ingestion of more than one kind of pesticide or herbicide besides paraquat according to the patients or their families, and unexplained clinical presentation by paraquat intoxication alone; (4) pregnant women from the time of acute paraquat poisoning (2012–2016) until follow-up (2021); and (5) patients who lost contact, refused follow-up, or were intolerant to chest CT and spirometry.
Grade of acute paraquat poisoning according to the Chinese national diagnostic classification method in the “diagnosis of occupational acute paraquat poisoning” (GBZ246–2013).

GI: gastrointestinal; PO2: arterial partial pressure of oxygen; ARDS: Acute Respiratory Distress Syndrome.
Monitoring clinical indexes
During follow-up, the peripheral capillary oxygen saturation (SpO2) was measured using the 512-F oxygen probe of the PM9000 monitor (Mindray, Shenzhen, China). Arterial blood gas analysis was performed using Cobas b221 automatic blood gas analyzer (Roche, Shanghai, China). Results of the arterial partial pressure of oxygen (PO2) were recorded. The exercise tolerance of patients who survived paraquat poisoning were reflected using the 6 min walk test (6-MWT) distance. The patient was asked to walk as fast as possible in a straight corridor. The walking distance (m) in 6 min was measured.
Chest CT
United imaging uCT780 machine was used with 1-mm conventional scanning layer thickness and 512 × 512 matrix. The scan ranged from the bilateral pulmonary apex to the rib diaphragm angle. Two attending physicians reviewed the images and determined increased lung texture, ground-glass opacity, pulmonary nodules, cord-like high-density shadow, reticulations, honeycombs, bronchiectasis, increased lung field transparency or bullae, pleura or interlobar fissure thickening, and pleural effusion.
Spirometry
According to the European Respiratory Association/American Thoracic Society standards, 2 pulmonary function in patients was measured using a Jaeger spirometer (MasterScreen, Jaeger, Germany). Pulmonary function measurements were affected by factors including sex, age, body weight, and height, therefore, the test indicators were calculated by percentages of measured and predicted values. Lung function test indices included restrictive index as vital capacity (VC; %), obstructive indices as forced expiratory volume in the first second (FEV1; %), forced VC (FVC; %), FEV1/FVC; %, maximal mid expiratory flow (MMEF75/25; %), and diffuse index as carbon monoxide in a single breath (%). A bronchial dilation test was performed to evaluate airway hyperresponsiveness. Patients were divided into mild, moderate, and severe ventilatory dysfunction groups according to the pulmonary function test indices. 3
Serum immunoglobulin E (IgE) level determination
Blood samples taken from the patients during follow-up were centrifuged and frozen at −80°C. The serum concentration of IgE was detected using the enzyme-linked immunosorbent assay (Cat #: MB00131, HOB Biotech, Suzhou, China). The normal range of serum IgE level was defined as <60 KU/L. All samples were measured in duplicates.
Statistical analysis
The IBM SPSS Statistics for Windows version 22.0 (IBM Corp., Armonk, NY, USA) software was used for statistical analysis. The normal distribution data were expressed as mean ± standard deviation, non-normal distribution data were expressed as median (quartile interval), and classification data were expressed as the number of patients and percentages (%). Among the three groups, the normal distribution data were compared using analysis of variance and the non-normal distribution data were compared using the Kruskal–Wallis test. The chi-square test was used to compare categorical variables. p < .05 was considered statistically significant.
Results
Survival survey of patients with paraquat poisoning
A total of 162 patients with acute paraquat poisoning, including 18 with mild, 35 with moderate, and 109 with severe paraquat poisoning, were retrospectively analyzed from July 2012 to July 2016. All patients with mild and moderate paraquat poisoning survived at discharge. Of the patients with severe paraquat poisoning, 98 died, while 11 survived at discharge. In July 2021, all discharged patients were telephonically followed-up. All 11 patients with mild, 17 with moderate, and 8 with severe paraquat poisoning were successfully followed-up using CT images and spirometry (Figure 1). The follow-up time ranged from 5 to 8 years, with mean length of 6.6 years. Flowchart of follow up for patients who survived paraquat poisoning.
Oxygenation index and exercise tolerance of patients during follow-up
Demographic parameters, exercise tolerance and oxygenation index of patients.
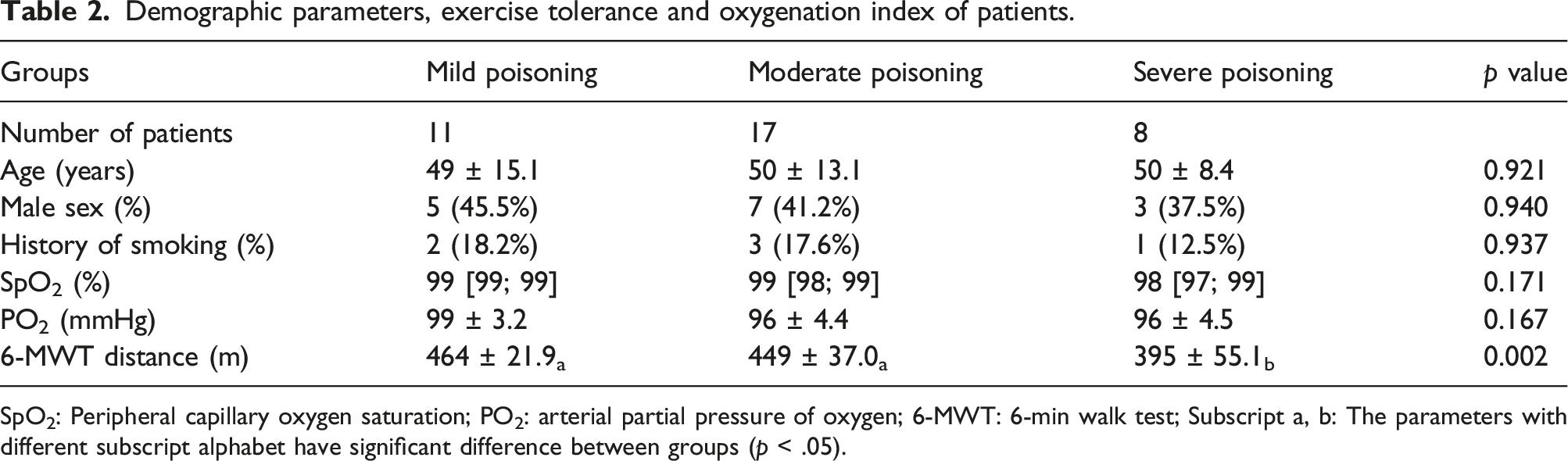
SpO2: Peripheral capillary oxygen saturation; PO2: arterial partial pressure of oxygen; 6-MWT: 6-min walk test; Subscript a, b: The parameters with different subscript alphabet have significant difference between groups (p < .05).
CT images and pulmonary function parameters of patients with long-term survival after paraquat poisoning
CT image, lung function and IgE level of patients in three groups during follow-up period.


Chest computed tomography (CT) findings of four patients with moderate or severe paraquat poisoning during acute and follow-up periods. a1- a2: CT scan showed multiple patchy dense and strip shadows in the right lung during acute period (red arrow) (a1). CT scan showed cord-like high-density shadow and reticulations during follow-up period (red arrow) (a2). b1-b2: CT showed interstitial inflammation, with localized fibrosis (red arrow) and mediastinal pneumatosis during acute period (green arrow) (b1). CT showed that the range of interstitial lesions decreased with time, and the density of ground-glass lesions decreased during follow-up period (red arrow) (b2). c1-c2: CT showed multiple ground-glass opacities and fibrous streak shadows in both the lungs during acute period (red arrow) (c1). CT showed multiple patches and streaks in both the lungs, partially with grid-like changes during follow-up period (red arrow). The translucent brightness in the bilateral lung fields was increased (green arrow) (c2). d1 - d2: CT showed multiple ground-glass opacities in both the lungs during acute period (red arrow) (d1). CT showed reticular lesions in both the lungs with bronchiectasis during follow-up period (red arrow) (d2).
Serum IgE concentration
As an airway hyperresponsiveness index, serum IgE was measured during follow-up. Most patients showed normal serum IgE concentrations in the three groups. Two patients with moderate paraquat poisoning and three with severe paraquat poisoning had higher serum IgE levels than normal range (Table 3). These five patients suffered from severe obstructive ventilation dysfunction and had positive bronchial dilation tests.
Discussion
In this study, patients with a 5-years survival after acute paraquat poisoning were followed-up using CT images and spirometry. CT showed that patients with mild paraquat poisoning had no significant abnormality; however, patients with moderate or severe paraquat poisoning had fibrotic lesions and other lung structural changes, including bronchiectasis, increased lung transparency, and pulmonary bullae. Although most patients reported good pulmonary function recovery and normal exercise tolerance, a few patients with moderate or severe paraquat poisoning showed apparent ventilatory dysfunction with elevated IgE levels.
Paraquat poisoning primarily targets the lungs. In the early stage of paraquat poisoning, inflammatory cell infiltration and pulmonary edema form, showing lung texture thickening, pulmonary exudation, and consolidation in chest CT. In the middle stage of paraquat poisoning with fibroblast proliferation, CT images show more changes of pulmonary consolidation and ground-glass opacities than pulmonary exudation. In the delayed stage of paraquat poisoning, collagen deposition and pulmonary fibrosis occurs, showing pulmonary interstitial fibrosis in CT.4,5 Studies have demonstrated that patients with immensely severe paraquat poisoning often die from ARDS in the early stage. Patients with mild paraquat poisoning could live well, entirely reversing the lung injury. 6 Some patients with moderate-to-severe paraquat poisoning can survive, with pulmonary interstitial fibrosis as the primary manifestation. 7 Long-term changes in pulmonary imaging features and pulmonary function in patients with over 1 year of survival after paraquat poisoning are rarely reported. Lee et al. showed that CT images of patients with 3–12 months of survival after paraquat poisoning had irregular pulmonary consolidation, ground-glass opacity, or honeycomb lesions, while the consolidation gradually dissipated. 8 In the present study, 36 patients with paraquat poisoning were followed up over 5 years. Pulmonary interstitial fibrotic lesions were sustainable and could not be removed entirely in some cases, manifesting as cord-like high-density shadows, reticulations, and honeycombs. However, the area of fibrotic lesions gradually shrunk to a steady-state, and the density of ground-glass opacity slowly decreased.
Furthermore, this study found that some patients who survived moderate-to-severe paraquat poisoning had lung structural changes, such as bronchiectasis. Others showed increased lung field transparency and pulmonary bullae, which are characteristics of COPD. The potential mechanism may be due to fibroblast proliferation induced by paraquat poisoning, leading to the deposition of extracellular matrix fibers. 9 The fibrous tissues surrounding the bronchus and alveoli promote irreversible airway remodeling. Moreover, paraquat poisoning may form multiple fibrotic lung lesions. Fibrous lesions cause continuous bronchus mechanical traction and abnormal intratracheal pressure, resulting in bronchiectasis. 10 Sustained stress on the alveoli also accelerates emphysema formation. 11
In this follow-up study, a certain degree of pulmonary function improvement was observed in most patients who survived paraquat poisoning. Some patients completely recovered, with normal exercise tolerance. This result was in accordance with Lee’s study. 8 However, some patients in the moderate or severe paraquat poisoning group suffered from mixed pulmonary dysfunction. These patients showed apparent obstructive ventilatory injury with mild restrictive and diffuse impairment. The results were different from previous reports, which considered that restrictive type of pulmonary dysfunction was mainly left in survivors of paraquat poisoning.12,13 Five patients with severe ventilatory dysfunction were further analyzed. The MMEF 75%/25% was significantly decreased, indicating increased small airway resistance. The bronchial diastolic test results were positive, and the serum IgE levels were elevated, with or without pulmonary structural changes in CT images. Obstructive ventilation dysfunction with airway hyperresponsiveness was first discussed in patients with paraquat poisoning; however, its pathological mechanism is unclear. Presumably, paraquat poisoning can cause inflammatory response and oxidative stress, inducing allergic inflammatory factors and releasing smooth muscle contractions, 14 leading to airway hyperresponsiveness.15–17 In addition, the elevated serum IgE level required significant attention. Previous study reported that farmers who occupationally exposed to a variety of pesticides and herbicides including paraquat had a high prevalence of allergic rhinitis and elevated concentrations of IgE. 18 Our follow-up also found high level of serum IgE accompanied with airway hyperresponsiveness in some patients surviving paraquat poisoning. Hence, we assume that IgE might be a crucial mediator between paraquat poisoning and airway hyperresponsiveness. More clinical evidence and underlying cell signals deserve further investigation.
Limitations
The study has some limitations. First, this was a single-center study with a small number of cases. The follow-up showed characteristic changes not previously reported, including long-term pulmonary structural lesions of paraquat poisoning, manifesting as bronchiectasis or COPD in addition to fibrosis. This finding enriched our understanding of the long-term prognosis of patients with paraquat poisoning. However, we aim to conduct a multiple-center study with a substantial number of cases for a more conclusive result. Second, the follow-up period ranged from 5 to 8 years, resulting in possibly different imaging and pulmonary functional changes. Therefore, the present study discovered and summarized new phenomena. However, more cases and time horizons need to be studied to obtain a clearer view of the long-term pulmonary changes associated with paraquat poisoning.
Conclusion
In the long-term follow-up, patients with severe paraquat poisoning showed the lowest exercise endurance. In moderate or severe paraquat poisoning group, CT images showed diversified changes, not only the dynamic evolution of the pulmonary fibrosis process, but also signs of bronchiectasis and chronic obstructive pulmonary disease. A few patients with moderate or severe paraquat poisoning complained of obstructive ventilation dysfunction with airway hyperresponsiveness. Hence, dynamic follow-up of CT images and spirometry is necessary for the survival of patients with paraquat poisoning. In addition, prompt treatment and rehabilitation guidance should be provided to patients with pulmonary function damage.
Footnotes
Author contributions
Conception and design: Mingfeng Lu.
Performing the literature search, data collection and analysis: Lu Fan, Xuejie Wang, Tianyi Lv, Fei Xue, Benhe Wu, Aiwen Ma.
Drafting the manuscript: all authors.
Approving the final version: all authors.
Mingfeng Lu is responsible for the integrity of the work as a whole.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Municipal Society Development Project of Yangzhou City (YZ2020091).
Ethics approval and consent to participate
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study met the medical ethical standards and was approved by the ethical committee of Northern Jiangsu People’s hospital (approval number: 2021ky134). Written informed consent of follow-up and blood sample utilization was obtained from all individual participants.
