Abstract
This present study was designed to investigate ameliorating potential of thymol (THY) on hexachlorobenzene (HBC)-induced epididymal and testicular toxicities in adult male rats. Forty adult male rats were orally treated by gavage daily for 28 consecutive days and divided into four groups; control group administered with corn oil, HBC-treated group (16 mg/kg b. wt), thymol-treated group (30 mg/kg b. wt), and HBC + THY-treated group. The results revealed that HBC exposure caused a significant decrease in the body weight change, organ weights, sperm functional parameters, serum testosterone level with widespread histological abnormalities. Furthermore, HBC-treated rats showed increased in the serum levels of luteinizing hormone (LH) and follicle stimulating hormone (FSH), epididymal and testicular myeloperoxidase activity, tumor necrosis-α, interleukin-1β level and caspase-3 activity, induced oxidative damage as evidenced by elevated malondialdehyde (MDA), reactive oxygen species (RONS) levels and significant reduction in antioxidant enzyme activities and reduced glutathione (GSH). However, co-treatment of THY with HBC alleviated the HBC-induced epididymal and testicular toxicities. Our findings revealed that HBC acts as a reproductive toxicant in rats and thymol could be a potential remedial agent for HBC-induced reproductive toxicity.
Introduction
The harmful effect of environmental toxicants on male reproductive system has become a global concern over the last few decades. These toxicants cause alterations of epididymal and testicular functions that may result into male infertility.1,2 Persistent organic pollutants are the major class of active pesticides that are used for crop protection against pests.
Hexachlorobenzene (HBC) is one of the widespread lipophilic persistent environmental pollutant that was originally used as fungicide and pesticide for treatment of seeds of wheat, sorghum and grains. 3 Although prohibited in many countries owing to its adverse effects on the environment and human health, 4 it is still release as by-product of various chlorinated pesticides and in many industrial processes 5 and human chronic exposure to low HCB doses is still a concern due to its persistent nature, recorded environmental and occupational exposure. 6 HBC-contamination is majorly from the environment through food, inhalation from air and dermal. 4 Several studies have reported that prolonged exposure to HCB can induce neurotoxic, immunotoxic, hepatorenotoxic, dermal, thyroid and reproductive toxic effects in animals and humans.5,7 Also, significant decrease in male sex hormones, sperm count, motility and viability accompanied by epididymal and testicular histological damage have been reported with HCB.4,8 These male reproductive disorders could be associated to excessive generation of free reactive oxygen species (ROS), increased lipid peroxidation, inflammation and apoptosis in the testis induced by HCB. 9
Many natural products are currently being investigated for their potential efficacy in alleviating hazardous effects of environmental pollutant exposure.10,11 Amongst these natural products is thymol (THY) a dietary phenolic monoterpene with pharmacological benefits. Thymol is found in essential oil of thyme and many plants such as
Although, study by Jafari et al. 20 reported the protective effect of THY on titanium dioxide nanoparticles-induced testicular damage, its influence on HCB-induced testicular damage has not been reported. Therefore, this present study sought to investigate the potential effect of THY on HCB-induced testicular damage in rats by determining the sperm quality, levels of male reproductive hormone, oxidative stress parameters, inflammatory and apoptotic markers as well as histopathological examinations of the testis.
Materials and methods
Chemical and reagents
Hexachlorobenzene (HCB; >99% purity), thymol powder (THY; C10H14O, 99.99%), thiobarbituric acid (TBA), 50,50-dithiobis-2-nitrobenzoic acid (DTNB), 1-chloro-2,4-dinitrobenzene (CDNB), hydrogen peroxide (H2O2), glutathione (GSH) and epinephrine were purchased from Sigma Chemical Co (St Louis, MO). Enzyme-linked Immunosorbent Assay (ELISA) kits for tumor necrosis factor alpha (TNF-α), interleukins (IL-1β and IL-10), caspase-3 (CASP-3) activity, follicle stimulating hormone (FSH), luteinizing hormone (LH) and testosterone were purchased from Elabscience Biotechnology Company, Beijing, China. All other chemicals were of analytical grade and were obtained from the British Drug Houses (Poole, Dorset, UK).
Experimental animals
Forty healthy adult male Wistar rats (10 weeks) weighing 180–200 g were purchased from the Faculty of Veterinary Medicine, University of Ibadan. The rats were kept in plastic cages located in a well-ventilated rat house, under standard laboratory conditions of a natural photoperiod of 12 h light-dark cycle at a controlled temperature of 22–27°C and a relative humidity (48–60%) and provided with commercial rat feed and water
Experimental design
The animals were assigned into four groups of 10 rats in each experimental group and were treated orally by gavage once daily for 28 consecutive days. The first group (control group) received corn oil as a vehicle (1 mL/100 g body weight). The second group (HCB-treated group) orally received 16 mg/kg body weight HCB dissolved in corn oil. The third group (HCB + THY-treated group) was simultaneously treated with HCB (16 mg/kg body weight) and THY (30 mg/kg body weight). The fourth group (THY-treated group) was orally administered with thymol (30 mg/kg body weight) dissolved in corn oil. The doses and duration of HCB and THY administration were chosen from the preliminary range-finding experiments to determine the sub-lethal and environmentally relevant doses and previously established data.8,18
Sample preparation
Twenty four hours after the last treatment, the over-night fasted rats were weighed and blood sample were obtained by retro-orbital sinus under ketamine-xylazine anesthesia (50 mg/kg− mg/kg, i.p) into plain sample bottles and left to coagulate at room temperature, then centrifuged at 3000 × g for 10 min to obtain serum that was kept at −20°C. All rats were sacrificed by cervical dislocation. The testis and epididymis of rats were immediately excised, cleared of fats and connective tissues, washed with physiological saline, weighed using a sensitive Mettler weighing balance (Mettler Toledo, Switzerland) and kept separately. Each right testis was homogenized in ice-cold 10% w/v phosphate buffer saline (PBS, pH 7.4). The homogenate was centrifuged at 10,000 × g for 15 min at 4°C separately and the supernatant obtained preserved at −20°C for biochemical analyses. Using sharp scissors, each right caudal epididymal was minced to release the semen in 1 mL of ice-cold PBS (pH = 7.4). The diluted sperm suspensions from each rat were then homogenized at 4°C with a glass homogenizer for 10 s and centrifuged at 2000 × g for 10 min. The seminal supernatant obtained was stored at 20°C before the biochemical assays.
Sperm motility assay
Individual sperm motility was evaluated from the control, HCB, and HCB-THY-treated and THY by the method of Juárez-Rojas et al. 21 Briefly, 10 μL of the sperm suspension were taken, placed on a pre-warmed slide and covered with a pre-heated glass cover slide at 37°C. Total sperm motility (progressive and non-progressive motility) was assessed from 200 sperm cells per slide and expressed as the percentage of motile sperm of the total sperm counted. Sperm were observed under an optic microscope (Olympus Light Microscope CX 41) at ×100 magnification.
Sperm count
The epididymal sperm count (ESC) from the control, HCB, HCB-THY treated and THY rats was estimated by the method of Arisha et al. 22 Briefly, the sperm solution was filtered through a nylon mesh followed by addition of 4 drops of formalin (40%). Subsequently, 10 μL of the resultant sperm solution was transferred to the hemocytometer counting chamber and allowed to stand for 5 min in a humid chamber to prevent drying before they were counted using the advanced Neubauer hemocytometer (Deep 1/10 m; LAB ART, Munich, Germany) counting chamber. The sperm cell count is equal to the total number of spermatozoa in four secondary squares × 2500 × dilution factor.
Sperm viability and morphological abnormalities
A portion of the sperm suspension from the control, HCB, HCB-THY and THY rats were placed on a glass slide, smeared out with another slide and stained with Wells and Awa’s reagent containing 0.2 g eosin and 0.6 g fast green dissolved in ethanol in a ratio of two to one (2:1) for morphological examination. A total of 400 sperm cells from each rat were used for morphological examination. 23
For sperm viability, sperm from each rat was staining with 1% eosin and 5% nigrosin in 3% sodium citrate dehydrate solution, according to Ju´arez-Rojas et al. 21 method as modified by Ommati et al. 24 Briefly, 10 μL of the sperm suspension and 10 μL of the stain solution were mixed, analyzed on pre-warmed slide and observed under microscope (Olympus Light Microscope CX 41) at ×400 magnification. Different fields were randomly analyzed until 200 sperm cells were counted per slide. Stained sperm cells were considered dead and unstained sperm cells were considered viable. Viable sperm was reported as viable spermatozoa percentage of total sperm count.
Evaluation of testicular enzyme
Testicular activities of acid and alkaline phosphatase (ACP and ALP) were determined in the testis’s supernatant according to the established method based on the hydrolysis of p-nitrophenyl-phosphate in acid and alkaline medium as described by Malymy and Horecker; Vanha-Perttula respectively.25,26 The testicular activity of glucose-6-phosphate dehydrogenase (G6PD) activity was determined using NADP+ and glucose-6-phosphate as substrates following the modified method as described by Wolf et al. 27 and lactate dehydrogenase (LDH) activity was estimated based on the inter-conversion of pyruvate and lactate as described by Vassault. 28
Evaluation of serum hormones
Follicle stimulating hormone (FSH; E-EL-R0371), luteinizing hormone (LH; E-EL-R0046) and testosterone (E-EL-R0031) levels were estimated in the serum by rat enzyme-linked immunosorbent assay (ELISA) commercial kits purchased from Elabscience Biotechnology, according to manufacturer’s protocol.
Assessment of testicular and epididymal antioxidant/oxidative stress biomarkers
The testicular and epididymal supernatants obtained were used separately for the estimation of antioxidant/oxidative stress assays. Superoxide dismutase (SOD) activity was determined by the method of Sun and Zigma, 29 catalase (CAT) activity was determined using exogenous H2O2 as substrate according to the method of Clairborne, 30 glutathione peroxidase (GPx) activity was measured as described by Moin, 31 reduced glutathione (GSH) was estimated according to the method of Jollow et al., 32 glutathione-S-transferase (GST) activity was quantified by the method of Habig et al. 33 Xanthine oxidase (XO) activity was measured spectrophotometrically by the formation of uric acid from xanthine through the increase in absorbency at 293 nm, according to the method of Prajda and Weber. 34 Malondialdehyde (MDA) content, an index of lipid peroxidation was measured as described by Driessen et al. 35 The level of protein oxidation (PCO) was determined by measuring the level of protein-bound carbonyl as a biomarker for oxidative stress based on the method of Hosseinzadeh et al. 36 Protein concentration was determined according to the method described by Popovic et al. 37 using bovine serum albumin as standard. Total antioxidant capacity (TAC) was determined using a commercially available kit (Randox Laboratories, Crumlin, UK) according to the manufacturer’s protocol. The assay is based on the incubation of 2,2- azino-di-(3-ethylbenzthiazoline sulfonate) (ABTS) with methmyoglobin (a peroxidase) and H2O2 to produce the radical cation ABTS, with a relatively stable blue-green colour measured spectrophotometrically at 600 nm as described by Quintanilha et al. 38
Quantification of reactive oxygen and nitrogen species level
Epididymal and testicular RONS levels from the control, HCB, HCB-THY-treated and THY rats were quantified by the RONS-dependent oxidation of 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) to dichlorofluorescein (DCF) as described by Owumi and Dim. 39 The reaction mixture containing 35 μL distilled water, 150 μL 0.1 M potassium phosphate buffer; 10 μL sample and 5 μL freshly prepared DCFH-DA was constituted with minimal exposure to air. Fluorescence emission of DCF generated from DCFH-DA oxidation was analyzed spectrophotometrically for 10 min at 30 s intervals using a Spectra Max 384 multimodal plate reader (Molecular Devices). DCF produced was expressed as a percentage over control.
Determination of inflammatory and apoptotic biomarkers
Testicular and epididymal myeloperoxidase (MPO) activity was evaluated by the method described by Granell et al. 40 and nitric oxide (NO) level was determined using the Griess assay as described by Hosseinzadeh et al. 36 Additionally, testicular and epididymal IL-10 (cat. no. CSB-E08047r), IL-1β (cat. no. CSB-E08045r), TNF-α (cat. no. CSB-E11967r) levels, caspases −3, and −10 activities (cat. no. CSB-E08854r; cat. no. CSB-E08856r) were analyzed using commercially available ELISA kits (Cusabio Biotech CO., Wuhan, China) according to the manufacturer’s instruction with the aid of a SpectraMaxTM 384 multimodal plate reader (Molecular Devices, CA, USA).
Histopathology
Microscopic evaluation of the testis and epididymis-sections was performed by the method of Bancroft and Gamble. 41 The testes and epididymis were carefully removed, cleaned of fats and fixed with Bouin’s solution. The samples were embedded in paraffin after dehydration processes. The tissues were after that cut with a microtome to 4–5 μm sections, fixed on charged-slides and then stained with hematoxylin and eosin (H&E) stain. The tissue histology slides were blinded and examined under a light microscope (Leica DM 500, Germany) by a pathologist who was unaware of the various treatments. The histopathological aberrations were scored and reported accordingly. Representative images were captured with the aid of a digital camera (Leica ICC50E, Germany) attached to the microscope.
Statistical analysis
Data were analyzed by one-way analysis of variance (ANOVA) and followed by post hoc Tukey test using GraphPad Prism version 7 Software, (www.graphpad.com; La Jolla, California, USA). Statistically significant differences were set at values of
Results
Effect of THY on the body weight gain and relative organ weights of rats treated with HCB
Body weight gain and relative testis and epididymis weights of rats in HCB-treated rats.
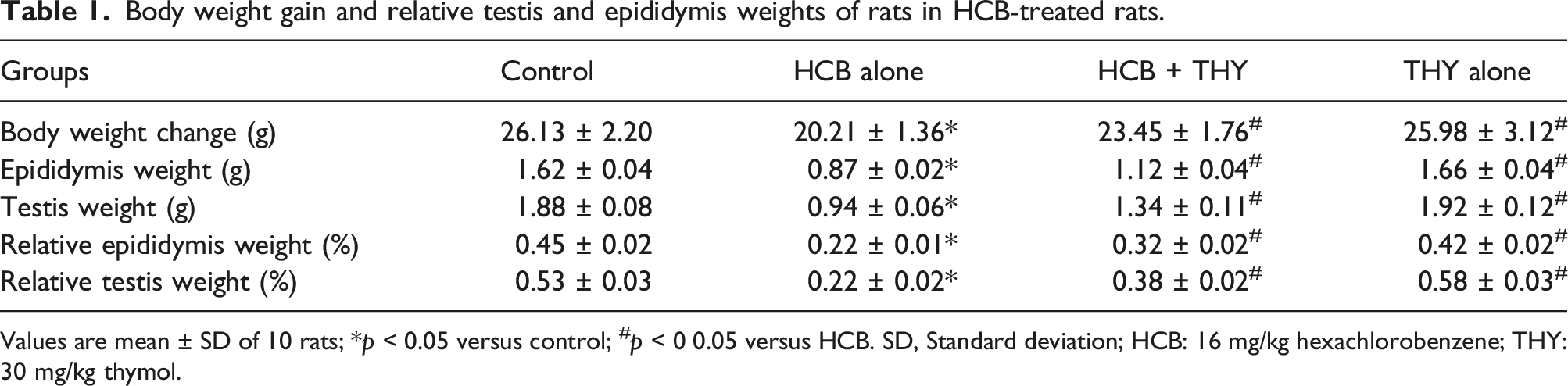
Values are mean ± SD of 10 rats; *
Effect of thymol on sperm functional parameters in HCB-treated rats
Rats exposed to HCB alone showed a significant ( Effect of co-administration of hexachlorobenzene and thymol on sperm motility, sperm count, sperm viability and total sperm abnormality in rats. Values are expressed as mean ± SD; 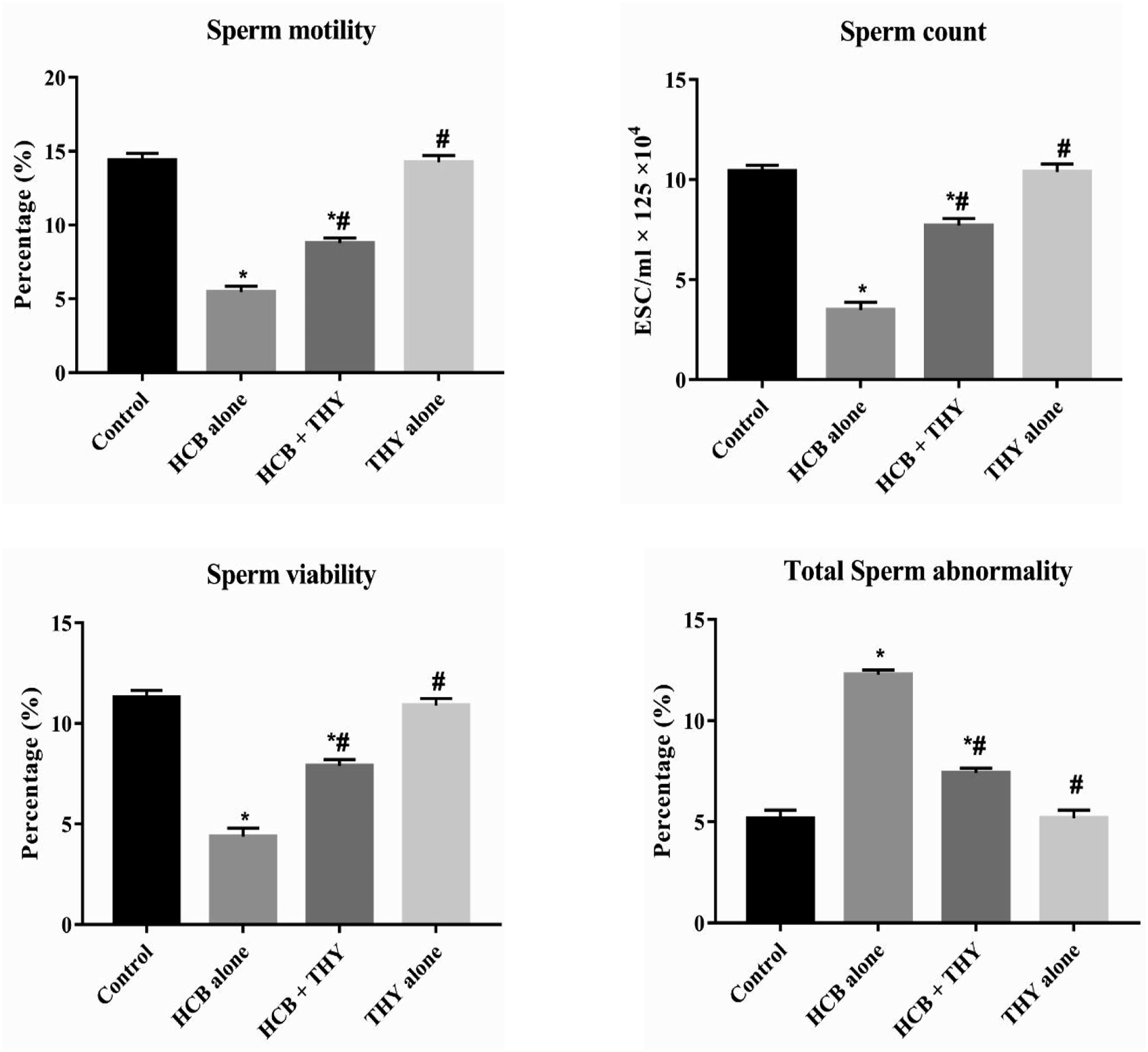
Effect of thymol on testicular enzyme activity in HCB-treated rats
Rats exposed to HCB alone revealed significant reduction ( Effect of co-administration of hexachlorobenzene and thymol on testicular ACP, ALP, G-6PD and total LDH activities in rats. Values are expressed as mean ± SD; 
Effect of thymol on male reproductive hormones in HCB-treated rats
The serum FSH and LH levels were significantly increased ( Effect of co-administration of hexachlorobenzene and thymol on serum FSH, LH and testosterone levels in rats. Values are expressed as mean ± SD; 
Effect of THY on epididymal and testicular antioxidant/oxidative stress biomarkers
Rats exposed to HCB alone showed significant diminution ( Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular SOD, CAT and GPx activities in rats. Values are expressed as mean ± SD; Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular GST activity and levels of GSH and TAC in rats. Values are expressed as mean ± SD; Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular LPO, PCO and RONS and XO activity in rats. Values are expressed as mean ± SD; 


Effect of THY on epididymal and testicular inflammatory markers
Rats administered with HCB alone revealed significant elevated ( Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular MPO activity and NO level in rats. Values are expressed as mean ± SD; Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular TNF-α, IL-1β and IL-10 levels in rats. Values are expressed as mean ± SD; 

Effect of THY on epididymal and testicular caspase-3 activities
Rats exposed to HCB alone exhibited marked increase ( Effect of co-administration of hexachlorobenzene and thymol on epididymal and testicular caspase-3 in rats. Values are expressed as mean ± SD; 
Effect of thymol on testicular histological lesions in IMI-treated rats
The representative epididymal and testicular photomicrographs of all the experimental rats are depicted in Figure 10. The epididymis and testis of control and THY-alone groups show normal histological features that include normal seminiferous tubules lined with healthy interstitial Leydig cells, Sertoli cells resting on basal layer and typical stages of maturation with and presence of spermatozoa inside their lumen. The epididymis of HCB alone treated rats’ revealed degeneration of germ cells, vacuolation, inflammatory cells infiltration and reduced spermatozoa. The epididymis of rats co-treated with HCB and THY showed improvement in the damage caused by HCB administration to rats. The testis of HCB alone rats revealed degenerative alterations of seminiferous tubules with reduced germ cells, vacuolation, infiltration of inflammatory cells, interstitial edema, Leydig cells hyperplasia. Testis of rats co-administered with HCB and THY revealed restoration of the testicular histoarchitecture similar to that of control rats. Representative photomicrograph of the epididymis and testis from control, HCB alone, HCB + THY co-treated and THY alone. Control (a) and THY (d) alone rats for epididymis and testis show normal histoarchitectures: epididymal tubules (red arrow) filled with spermatozoa. Epididymis of HCB alone (b) rats shows vacuolation (black arrow), infiltration of inflammatory cells (red arrow head) with reduced spermatozoa. HCB + THY (c) treated rat shows increased spermatozoa with vacuolation almost disappearing. HCB alone testis (b) shows testicular degeneration with defective spermatogenesis progression in the lumen (grey arrow), vacuolation, degeneration of germinal epithelium (blue arrow), mild hyperplasia of Leydig cells (orange arrow), inflammatory cell’s infiltration (red arrow head), interstitial edema and formation of multinucleated giant cell in seminiferous tubule (arrow head). HCB + THY testis shows improved morphological structure without distortion (H and E × 400).
Discussion
Uncontrollable and unlawful use of HCB in some developing countries including Nigeria as pesticides has caused untoward detrimental effects on numerous non-target organisms and their organs. HCB has been reported to cause male reproductive toxicity. 42 However, the mechanism of HCB-induced male reproductive toxicity has not been fully investigated. Thus, this study was carried out to unravel the possible effects of HCB administration on reproductive functions and the capability of THY in mitigating against HCB-induced testicular toxicity in male Wistar rats.
The present results revealed a considerable reduction in body weight of the rats treated with HCB alone compared to the control group. This result is similar to the previous reports,4,42 and may suggest anorexia, fed malabsorption resulting to breakdown of body cell mass. Furthermore, HCB-induced significant reductions in relative epididymal and testicular weights possibly due to alteration in metabolic routes that could possibly lead to tissue breakdown 43 and also, could be linked to reduced serum testosterone level that is required for growth and improved function of male sexual reproductive organs. Relative testicular weight is an index of spermatogenesis and is usually measured in male reproductive toxicity. 44 The results are comparable to the earlier report. 8 Besides, administration of THY to HCB-treated rats reversed decrease in the body weight, and the epididymal and testicular weights possibly by improving the metabolic processes via its antioxidant properties.
Reproductive performance in human and experimental animals is commonly assessed by evaluation of sperm. 45 The significant decrease in the sperm count, viability and motility accompanied by significant increase in percentage of morphological abnormal sperms in the HCB alone rat’s compared to control rats in this study may indicates spermatogenesis impairment as evidenced by the progressive deterioration and necrotic changes in the seminiferous tubules of the testes and may justify the increase in sperm morphological abnormalities. Also, the reduction in sperm motility may be associated with the decrease in steroid-regulated antioxidant defense in the epididymis. Furthermore, the decrease in the quantity and quality of sperm of HCB alone rats may be linked to the diminution of testosterone that is crucial to sperm viability and spermatogenesis.46,47 The detrimental effects of HCB on the quality and quantity of sperm in rats in the study correspond to the earlier reports of Batiri et al. 8 However, administration of THY to HCB rats ameliorated the toxic impact of HCB suggesting the positive influence of THY on the epididymal and testicular wellbeing. Our results similar to the previous report of Guvec et al. 19
Activities of testicular ACP, ALP, G6PD and LDH are use as indices of spermatogenesis48,49 and partake in testicular energy metabolism and stabilization. Testicular ACP and ALP are involved in the secretion of gonadotropins and in the intracellular and intercellular transportation of metabolites during steroidogenesis. 50 ALP catalyzes the release of free glucose use in proliferation and differentiation of spermatogenic cells from 6-phosphoglucose. ACP participates in protein synthesis whereas spermatogenic epithelium growth is closely linked to LDH. 51 Additionally, generation of reduced nicotinamide adenine dinucleotide (NADPH) via G6PD activity in hexose monophosphate pathway in rat testes is crucial to hydroxylation of steroids required for spermatogenesis. The reductions in the testicular ACP and ALP activities after the administration of HCB alone to rats in this present study connotes impairment in the formation and transportation of required metabolites within and outside the cells of the testes during steroidogenesis with consequential unfavorable effect on spermatogenesis. 52 Also, the reduction in G6PD and LDH activities in HCB alone connotes diminished generation of nicotinamide adenine dinucleotide phosphate (NADPH) that is essential for the conversion of cholesterol to testosterone and impairment in the ability of the rats to convert spermatocyte to spermatozoa. However, co-administration of THY with HCB to rats, restored the activities of these enzymes thus suggesting the mitigating effect of THY on HCB-induced testicular injury.
Spermatogenesis regulation in the testes occurs at the hypothalamic-pituitary-testicular axis. Gonadotropin-releasing hormone (GnRH) regulates the secretion of LH and FSH by the pituitary gland. GnRH release is controlled by testosterone in a negative feedback loop. Leydig cells are stimulated by LH to synthesize testosterone and Sertoli cells are stimulated by FSH to produce androgen-binding protein (ABP). Androgen-binding protein binds to testosterone for spermatogenesis. 53 In the current research, elevated serum levels of FSH and LH in rats exposed to HCB alone revealed an undesirable response from hypothalamus-pituitary-gonadal axis leading to impaired spermatogenesis and the reduction in the serum testosterone level suggests injury to the Leydig cells possibly due to ROS generation 54 resulting to suppression of spermatogenesis and subsequently, testicular dysfunction. Our result agreed with the reports of Elissalde and Clark 42 who reported reduction in serum testosterone levels in male mice fed diets containing approximately 30 mg HCB/kg-day) for 21 days, and Bitri et al. 8 The reduction in the serum levels of FSH and LH with simultaneous increase in the testosterone level following concomitant administration of THY to rats suggests protective effect of THY on HCB-induced hormonal disruption and improved spermatogenesis.
Oxidative and nitrosative stresses have been linked to pathologic outcomes in male reproductive organs. 55 Excessive oxidative stress caused serious changes in epididymis and developing spermatozoa leading to impaired sperm quality. 56 HCB metabolism usually produced free radical and reactive metabolites that form covalent bonds with cellular constituents resulting to irreversible cellular damage. 57 Enzymatic and non-enzymatic antioxidants such as SOD, CAT, GPx, GST and GSH level in the reproductive organs are usually employ to neutralize ROS generated during the initiation of spermatogenesis and epididymal maturation.58,59 The reduction in the SOD, CAT, GPx, GST and GSH and TAC levels in the epididymis and testes of rats exposed to HCB alone in our current study revealed an increased RONS generation that overwhelmed the antioxidant capacity accompanied by simultaneous increase in the levels of LPO, PCO, RONS and XO activity possibly due to enhanced oxidations of lipids in spermatozoa membrane phospholipids and proteins with purine metabolism suggesting oxidative damage. The decrease in the antioxidant system may also be ascribed to the decrease in steroid-regulated antioxidant defense in the epididymis and testes. 60 Nonetheless, co-treatment of THY with HCB increase the antioxidant status and decrease the LPO, PCO, RONS levels and XO activity in the epididymis and testes of rats signifying antioxidant ability of THY 20 due to the presence of phenolic hydroxyl group in its chemical structure that scavenge and neutralize the RONS by donating electrons and disrupting the radical chain reaction of lipid peroxidation, stimulates endogenous antioxidant enzymes and augments their antioxidant capacity.18,61
Inflammation has been linked to reproductive toxicity with ROS generation as the main culprit. 62 Cytokine-related MPO activity stimulates neutrophils to produce RONS including hypochlorous acid, causing tissue injuries 63 and subsequently inflammation. Superoxide radical (O2-) produced by HCB metabolism interact with NO to form peroxynitrite (ONOO−), a potent radical involved in modification of cellular macromolecules including protein nitration and impairment of signal transduction. 64 ROS-induced oxidative stress may also stimulate secretion of pro-inflammatory cytokine such as TNF-α and IL-1β, which are known activators of NF-kB. 65 In this present study, HCB-induced reprotoxicity was evidenced by the increase in MPO activity, and levels of NO, TNF-α and IL-1β with concomitant diminution in IL-10 level in the epididymis and testes of rats. The anti-inflammatory cytokine; IL-10 is a potent negative feedback regulator that regulates inflammation through autocrine and paracrine mechanisms. 66 Our results suggest oxidative-mediated inflammatory process that may leads to activation NF-kB signaling pathways. However, co-treatment of rats with HCB and THY downgraded the pro-inflammatory markers and upgraded the anti-inflammatory cytokine, thus, validating the anti-inflammatory capability of THY.
Apoptosis in the germ cells of rats testes is associated directly or indirectly to dysfunctional spermatogenesis and disruption of sperm count and viability due to excessive ROS generation and inflammation. 67 Caspase-3 play a major role in apoptosis activation as the main effector caspase that is activated by oxidative stress or inflammation. 68 The elevated activity of caspase-3 in the epididymis and testes of rats exposed to HCB alone in this study may be due to induction of apoptosis via activation of caspase-3. This observation was affirmed by the histological examination that revealed epididymal tissue degeneration, decreased spermatozoa, vacuolation, interstitial edema, varying degree of testicular fragmentation, mild hyperplasia of Leydig cells, spermatogenesis arrest, vacuolation and inflammatory cells infiltration of interstitial connective tissues. However, co-treatment of HCB with THY down-regulated the caspase-3 activity and restored the histoarchitecture of the examined organs, thus, suggesting the antiapoptotic effect of THY through the inhibition of the executioner caspase-3 activity in the epididymis and testes of rats. Our histological results also corroborate our biochemical results on the mechanism of actions of THY to prevent reproductive toxicity induced by HCB in the epididymis and testes of Wistar rats.
In conclusion, the results of this study indicate that HCB exposure can cause epididymal and testicular damage, and the possible mechanisms are induction of oxidative stress, inflammation and apoptosis and subsequent spermatogenesis impairment. But, THY co-administration alleviated HCB-induced reprotoxicity by enhancing the sperm quality and functionality, restoring antioxidant status and histological alterations, suppressing inflammation and apoptosis. The pharmacological benefits displayed by THY could be due to its ability to scavenge free radicals and interacts with several intracellular signaling pathways by modulating ROS-mediated NFkB/TNF-α signaling pathways. Further studies are needed to explore additional molecular mechanisms involved in these processes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
