Abstract
Thymoquinone (TQ) is one of the components extracted from Nigella sativa seeds and has antioxidant, anti-inflammatory, and anticancer effects. We evaluated the effect of TQ on 5-fluorouracil (5-FU) pharmacokinetics (PK) in vivo and in vitro on human colorectal cancer cell line. Ten Adult male Wistar rats were assigned to two groups. TQ treated group received intraperitoneal TQ once daily for 14 consecutive days (5 mg/kg). Both groups received intraperitoneal 5-FU (50 mg/kg) on day 15 and blood samples were collected from retro-orbital plexus. The pharmacokinetics parameters were analyzed using high-performance liquid chromatography (HPLC). Moreover, various concentrations of 5-FU, TQ, and combination of 5-FU and TQ were added to the HT-29 cell line and cell viability was measured using 3- (4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide colorimetric assay. The maximum serum concentration (Cmax), area under the curve (AUC), and time of maximum concentration (Tmax) of 5-FU in TQ treated group were significantly increased approximately by 61, 60, and 24% compared to the control group, respectively. The combination of 5-FU with TQ (0.284 mM) showed a greater inhibitory effect on HT-29 cell growth compared to the alone 5-FU (0.027 and 0.055 mM) administration. TQ increases the AUC, Cmax, and Tmax of 5-FU and has a synergistic effect on the PK of 5-FU. Moreover, low concentration of TQ enhances the inhibitory effects of 5-FU on cell growth in colorectal cancer cell line. This synergistic effect might enhance the anticancer effects of low concentration of 5-FU, leading to drug dose reduction and reduced systemic toxicity of this chemotherapeutic agent.
Introduction
Cancer remains one of the leading causes of death worldwide. Colorectal cancer is the third most common cancer worldwide and the second most common cause of cancer-related death in both genders. 1 Overall 5 years survival from diagnosis is about 60% considering all stages of disease. 2 In recent years, the overall incidence and mortality of colorectal cancer have dropped in the US and Europe, due to advances in screening tests. Since the mid-2000s, the incidence of colorectal cancer has decreased by 2–3% per year in both genders in the US. 2 However, early-onset colorectal cancer, defined as diagnosis in patients younger than 50, is increasing in the US 3 and other high-income countries 4 over the past few decades. The incidence of early-onset colorectal cancer is projected to increase and more than double by 2030. 5 There are several options for cancer treatment based on the type of the condition, including surgery, radiation therapy, and especially chemotherapy. Chemotherapy includes several classes of agents. Anthracyclines, alkylating agents, and antimetabolites are some examples. 5-FU is an antimetabolite chemotherapeutic agent introduced more than 50 years ago, but it is still widely used, especially in colorectal (most significant efficacy), 6 breast, 7 and head and neck malignancies. 8 Antimetabolite drugs inhibit biosynthetic pathways or damage macromolecules such as DNA and RNA. The drug has both of the mentioned functions, inhibits thymidylate synthase and causes severe damages to DNA. 9
Herbal medicines are commonly used as a treatment for various diseases. Scientists have targeted many traditional herbal medicines in parallel with modern medicines for developing drugs with more efficacy and likely fewer side effects. Over time, many herbs showed interactions with chemical drugs, and several and severe interactions were reported. 10 Herbal medicines can affect the metabolism of drugs and their elimination in many ways, such as their inhibitory effect on P450 (CYP) enzyme.11,12 Nigella Sativa seeds or black cumin seeds are useful herbs in traditional medicine for their role in alleviating many diseases, including allergy, asthma, headache, infections, obesity, musculoskeletal pains, and high blood pressure. 13 TQ is one of the components extracted from Nigella sativa seeds and it has been reported to have antioxidant, 14 anti-inflammatory, 15 and anticancer effects. 16 Moreover, studies revealed that TQ could inhibit cancer cells growth and induce apoptosis in many malignancies such as breast, 17 lung, 18 colon, 19 pancreas, 20 prostate, 21 ovary cancers, 22 and leukemia. 23 However, along with its benefits, it showed inhibitory effects on CYP2C19, CYP2C9, CYP2D6, and CYP3A4, and it can cause herb-drug interactions.11,12
Moreover, it has been shown that TQ could increase 5-FU effects by molecular and cellular pathways in colorectal cancer and experimental osteosarcoma models. TQ downregulates precancerous pathways (β-catenin, Wnt, Cox-2, and VEGF) and upregulates anti-tumorigenesis pathways (DKK1, CDNK-1A, and TGF-β1).24,25 Furthermore, TQ enhances the 5-FU effect on cancer cells apoptosis in vitro. 26 Nevertheless, it is not clear whether TQ could affect 5-FU pharmacokinetics or not. Understanding the plausible synergistic effect may reduce the prescribed doses of 5-FU and its cytotoxic effects on normal cells. We aimed to conduct an experimental study on the TQ effect on the PK of 5-FU in vivo in rat and in vitro effect of 5-FU on human colorectal cancer cell line to find the possible synergistic effect between TQ and 5-FU.
Methods
Reagents and cell lines
TQ, 5-FU, 5-chlorouracil (5-ClU), and water (HPLC grade) were purchased from Sigma-Aldrich (St Louis, MO, USA). Acetonitrile and ethyl acetate (HPLC grade) were purchased from Carlo ERBA Reagents (Sabadell, Spain). Methyl alcohol (HPLC grade) was purchased from Daejung chemicals (Siheung-si, South Korea). The HT-29 cell line (human colorectal adenocarcinoma) was purchased from Pasteur Institute Cell Bank (Iran, Tehran) and was maintained according to the American Type Culture Collection instructions.
Animals
Adult male Wistar rats weighing 150–200 g were provided by the University animal house, Babol, Iran. Animals were randomly assigned to two groups (five per group): control and pretreated with TQ. The rats were maintained at a temperature of 22 ± 2°C, 12 h/12 h light/dark cycle, and relative humidity of 60 ± 10%. In addition, rats had free access to food and water. The handling of animals and all experimental procedures were conducted in compliance with the guideline for the care and use of laboratory animals in Iran, and the protocol was approved by the Institutional Review Board of Babol University of Medical Sciences.
Study protocol
TQ was administered intraperitoneally once daily for 14 consecutive days at a dose of 5 mg/kg. On the morning of day 15, 5-FU was administered intraperitoneally at a dose of 50 mg/kg to the control rats (n = 5), and pretreated rats with TQ (n = 5). After 5-FU injection, each rat was anesthetized with ketamine (75 mg) and xylazine (25 mg), and blood samples (0.5 mL) were collected from the retro-orbital plexus of rats into the heparinized tubes at 0, 5, 15, 30, 60, 120, and 240 min. The samples were centrifuged immediately, and plasma samples were stored at −20°C.
Pharmacokinetic study in vivo
Each plasma sample (100 μL) was transferred to a microtube. Moreover, 100 μL of internal standard (5-ClU) (50 μ/mL) and 150 μL of acetonitrile were added and mixed by vortex mixer. The samples were shaken for 1 min and then centrifuged at 8000 rpm for 15 min at 4°C. The organic layer was separated and moved to a microtube and dried under a stream of nitrogen at 40°C. The remains were reconstituted in 200 μL of mobile phase and a 10 μL sample was analyzed using HPLC. Moreover, we used standard samples (1, 2, 4, 8, and 10 μg/mL) to construct the calibration curve. The HPLC system consisted of a guard column (ODS 4 mm 3.0 mm I.D, security guard, Phenomenex, Torrance, CA, USA) and a C18 column (Prodigy ODS 250 4.6 mm, 5 μm, Phenomenex, Torrance, CA, USA). The mobile phase included ammonium acetate (0.01 M, pH = 3.5) and acetonitrile. The flow rate of the solvent delivery system was 0.75 mL/min and the UV wavelength was 264 nm. 27 The pharmacokinetic parameters were estimated using PKSolver software (an add-on software of Microsoft Excel) based on a two-compartment model.
Cell culture
The human colon cancer cell line HT-29 was cultured in RPMI 1640 medium supplemented with 10% (v/v) FBS (fetal bovine serum) and 1 (v/v) penicillin-streptomycin at 37°C in a humidified atmosphere containing 5% CO2. Cells were seeded on 96-well microplates. After 24 h, cells were exposed to the drugs. Various concentrations of 5-FU (0.027, 0.055, 0.11, 0.22, and 0.44 mM), TQ (0.11, 0.142, 0.284, and 0.568 mM), 5-FU combined with 0.284 mM TQ, and TQ combined with 0.11 mM 5-FU were added and cells were incubated at 37°C for 48 h. Each group was set up in quadruplicate.
Cell viability measurement or MTT assay
Following the treatment with 5-FU or TQ alone and 5-FU in combination with TQ, cell viability was measured by an 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) colorimetric assay. 3- (4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide is a yellow dye, which is reduced to the blue product formazan by cellular enzymes.
28
After adding the 5-FU or/and TQ, the contents of wells were removed carefully, and MTT (thiazolyl blue tetrazolium bromide) dye was added. After four hours of incubation under standard condition, the MTT dye was separated, and the formazan crystals were dissolved using DMSO (dimethyl sulfoxide). The samples’ absorbance was determined using an ELISA (enzyme-linked immunosorbent assay) reader at 570 nm. The mentioned steps were repeated at least three times for each concentration. Half-maximal inhibitory concentration (IC-50) and cell viability (%) were calculated (Figure 1).
Statistical analysis
Mean ± standard deviation (SD) was used to express all values. Values were compared using one-way ANOVA. The difference between groups was considered statistically significant at p value < 0.05. R version 4.0.2 was used for statistical analysis. The graphs were visualized using GraphPad Prism 9.
Results
Pharmacokinetics of 5-FU
The calibration curve of 5-FU plasma concentration was linear over the concentration range of 0.1–50 μg/mL (Supplementary material). The representative regression equation was y = 45,462x + 1704.1, where y was the peak area ratio of drug to internal standard and x was the plasma concentration (μg/ml) of 5-FU. Intra-day and inter-day assay variations were determined by analyzing a set of low, medium, and high concentration standards 6 times in 1 day and on 6 different days. The inter-day and intra-day coefficients of variation (CV%) were all less than 5%. The accuracies ranged from 90% to 110% on the same day and between days. The retention time of 5-FU was 5.13 min and that of the internal standard was 8.18 min (Supplementary material). The limit of detection was 0.06 μg/mL based on a signal‐to‐noise ratio of 3:1, whereas the limit of quantitation was 0.07 μg/mL based on a signal‐to‐noise ratio of 6:1. The mean percentage recovery of 5-FU in plasma was 97.2 ± 4.84%. In the pharmacokinetics study, rats were randomly assigned to two groups. Thymoquinone treated group received intraperitoneal thymoquinone (5 mg/kg) for 14 days. Both groups received intraperitoneal 5-fluorouracil (50 mg/kg) at day 15 and blood samples were collected from retro-orbital plexus. The pharmacokinetics parameters were analyzed using HPLC. In the cellular study, HT-29 cell line was exposed to various concentrations of 5-fluorouracil and thymoquinone and the cell viability was measured using MTT colorimetric assay.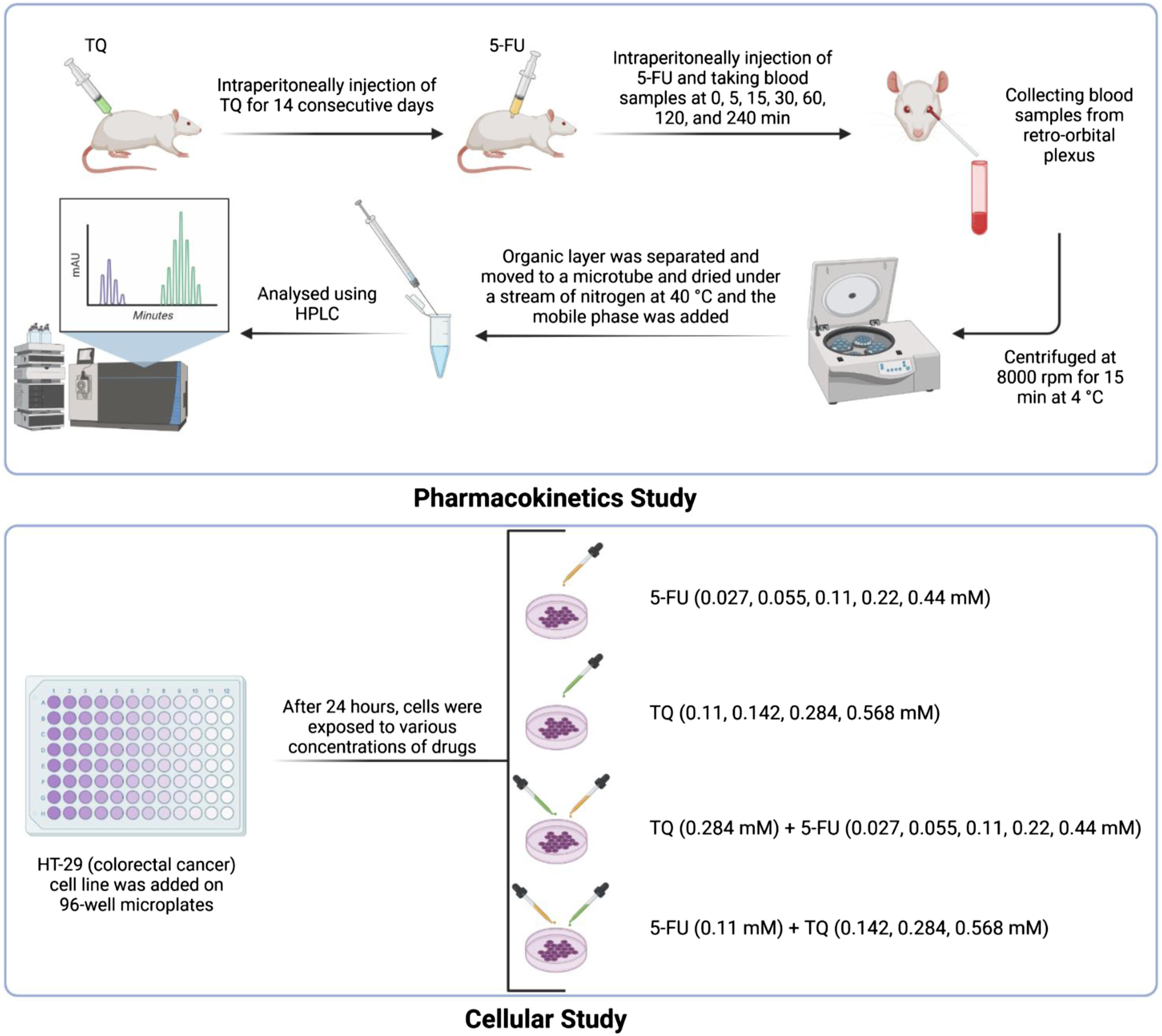
The plasma concentration-time curves of 5-FU after a single intraperitoneal injection of 50 mg/kg 5-FU to control group and TQ pretreated group are presented in Figure 2. The Cmax was significantly increased from 7.2 to 11.6 μg/mL in the TQ pretreated group compared to the control, which was approximately a 61% increase. The AUC was 638 and 1025.6 in the control and the TQ pretreated groups, respectively, which was approximately a 60% increase in TQ receiving group compared to the control. Plasma concentration-time curves of 5-FU after a single intraperitoneal injection of 50 mg/kg 5-FU to control group (n = 5) and TQ pretreated group (n = 5). There were significant differences in the plasma concentration of 5-FU between the control group and TQ pretreated group at 15, 30, 60, and 120 min (**p < 0.01 compared to the control group; Values were compared using one-way ANOVA).
Values are mean ± standard deviation of five rats.

ap < 0.05 compared with the control group.
Cytotoxicity assay
HT-29 (colorectal cancer) cell line was chosen to detect cytotoxicity. The result of cells viability test showed that the 5-FU and TQ have significant inhibitory effects (p < 0.05, p < 0.01, and p < 0.001) on cells growth, which increased with increasing the concentration of 5-FU and TQ from 0.027 to 0.44 mM and 0.11 to 0.568 mM, respectively (Figure 3(a, b)). Compared with the singular inhibitory effect of 5-FU, the combination of 5-FU and TQ showed a significant inhibitory effect at the 5-FU concentrations of 0.027 and 0.055 mM, which show that TQ might increase the inhibitory effects of 5-FU on cell growth. Although higher concentrations of 5-FU with TQ (0.284 mM) decreased the cell viability, it was not statistically significant compared to the 5-FU singular treatment (Figure 3(c)). It is presented in Figure 3(d) that the TQ concentration of 0.142 mM with 5-FU was statistically significant compared to the singular inhibitory effects of TQ (p < 0.05). However, this significant effect was not observed in higher concentrations of TQ Figure 3(d). The cell viability of HT-29 (colorectal cancer) cell line treated with 5-FU, TQ, and combination of 5-FU and TQ. Each value represents the mean ± standard deviation. * p < 0.05, ** p < 0.01, and ** p < 0.001: (a, b) compared with no treatment group; (c, d) combination group compared with alone group.
Discussion
We found that TQ increases the AUC, Cmax, and Tmax of 5-FU. TQ does not affect half-life parameters of 5-FU. In addition, TQ showed inhibitory effects on cell growth. Moreover, low concentration of TQ enhances the inhibitory effect of 5-FU on cell growth in colorectal cancer cell line.
The major amount (80–85%) of administered 5-FU is catabolized to inactive metabolites and about 1–3% mediates the cytotoxic and anticancer effects by inhibiting DNA synthesis and RNA processing and function. 29 5-FU is converted to three main active metabolites: (1) fluorouridine triphosphate (FUTP) that replaces instead of uridine triphosphate into RNA; (2) fluorodeoxyuridine triphosphate (FdUTP) that replaces deoxythymidine triphosphate into DNA; and (3) Fluorodeoxyuridine monophosphate (FdUMP), which forms a ternary complex with 5,10-methylene tetrahydrofolate and thymidylate synthase, which inhibits the DNA synthesis. 30 FUTP disrupts RNA processing and function and FdUTP and FdUMP damage the DNA, leading to cytotoxic effects and cell death. 30 In the series of reactions within 24 h, 60–90% of intravenous 5-FU is excreted in urine as α-fluoro-β-alanine (FBAL). 31 Dihydropyrimidine dehydrogenase (DPD) deficiency, the initial and rate-limiting enzyme in the catabolism of 5-FU, increases the risk of severe toxicities, including mucositis, cardiotoxicity, diarrhea, neurotoxicity, and even death. 29 Although 5-FU remains one of the most effective anticancer drugs, the resistance of cancer cells to the drug is observed frequently. Several mechanisms were suggested, including genetic and epigenetic modification within the cells, decreased drug delivery to cancerous cells either by decreased uptake, increased efflux, or changes in enzymes involved in its mechanism of action and signaling pathways and cell cycle disturbance. 32
Thymoquinone is a natural compound and one of the active components of Nigella Sativa seeds. TQ showed anticancer effects, anti-inflammatory, and antioxidant efficacy. 16 The TQ is rapidly eliminated and slowly absorbed, resulting in low bioavailability. 33 Biodistribution study showed that the main uptake of TQ occurred in the liver and kidney following intravenous administration and the intestine following oral administration. 34 Moreover, compromised liver function is associated with decreased systemic clearance of TQ following intravenous administration. 35 It seems that TQ applies anticancer effects through several mechanisms, including inducing apoptosis, surviving healthy cells, regulating cell cycle, preventing metastasis, 36 inhibiting inflammatory mediators, detoxification, and antioxidant effects. 37
Although cytochrome P450 (CYP) enzymes play a central role in detoxifying xenobiotics and biosynthesis of antioxidants, activation of these enzymes, particularly CYP1 family members, might biotransform the polycyclic aromatic hydrocarbons into highly reactive carcinogenic intermediates. 38 TQ inhibits carcinogen-metabolizing enzymes such as CYP family members and it is a biochemical mechanism of anticancer effect. 39 TQ exerts anticancer effects via several signaling pathways. For example, Raut et al. in 2021 showed that the TQ induces apoptosis of SK-MEL-28 cell line (melanoma) by inhibition of the Jak/STAT3 signaling pathway through reactive oxygen species (ROS) production. 40 Moreover, TQ arrests cell proliferation by inhibiting cell cycle via different signaling pathways, such as STAT3 signaling pathway. 36
Furthermore, TQ combined with other agents, including 5-FU, doxorubicin, cisplatin, bortezomib, mesalazine, and paclitaxel showed promising efficacy. 33 Many studies noted that the TQ enhances 5-FU efficacy and the cancer cells chemosensitization to 5-FU-induced apoptosis in the early stages of cancer. Ballout et al. in 2020 showed that low concentrations of TQ could enhance the 5-FU action on resistant cancer HCT116 cell lines through inducing apoptosis, reducing invasion and migration, increasing DNA damage, and reducing DNA repair via inhibition of NF-κB, MEK, and CXCR4 signaling pathways, downregulation of CD44, and upregulation of p53 and p21, suggesting an assuring effect of TQ on 5-FU resistant cancer cells. 41 Combination of TQ and 5-FU downregulates the WNT/β-catenin and PI3K/AKT signaling pathways, which targets a broader spectrum of oncogenic pathways, compared to 5-FU alone treatment, leading to effective eradication of colorectal cancer stem cells. 25 Kensara et al. in 2016 showed that the combination of TQ and 5-FU restrains the tumor growth and increases the cells susceptibility to 5-FU in an early colorectal tumor model in rats via downregulation of precancerous pathways, including NF-κB, VEGF, WNT/β-catenin, iNOS, COX-2, and TBRAS as well as upregulation of anticancer pathways, including TGF-β1, TGF-βRII, CDNK-1A, DKK-1, and GPx. 24 Lei et al. in 2012 showed that the combination of TQ and 5-FU upregulates the bax (a pro-apoptotic protein), downregulates the bcl-2 (an anti-apoptotic protein), and activates the caspase-3 and caspase-9, thus resulting in the augmentation of gastric cancer cells apoptosis. 42 In addition, Sarman et al. in 2016 showed that the TQ and 5-FU combination induces apoptosis and decreases the osteosarcoma cell viability (in MG63 osteosarcoma cell line) compared to 5-FU alone administration (percent of early apoptosis increased from 35% to 60.35%). 26 Our results are in line with mentioned studies that the TQ increases the inhibitory effect of 5-FU on cell growth in colorectal cancer cells. However, this is the first study, which showed the synergistic effect of TQ on 5-FU pharmacokinetics.
As previously mentioned, DPD is the initial and rate-limiting enzyme in the catabolism of 5-FU. Guimbaud et al. in 2000 showed that the activity of DPD in colon increases in the situation of inflammation. 43 Moreover, Horiguchi et al. in 2004 revealed that the activity of DPD in breast cancer tissue is higher compared to normal breast tissue. 44 In addition, indomethacin, an anti-inflammatory drug, reduces the activity of DPD and increases the concentration of 5-FU; thereby, the increasing concentration of 5-FU could enhance the tumor cells toxicity. 45 Likewise, Zhang et al. in 2017 showed that the high activity of DPD in gastric cancer cells is associated with a negative impact on sensitivity to 5-FU. 46 Accordingly, due to the anti-inflammatory effect of TQ, the decreasing activity of DPD could be expected. However, it is not a definite finding. Similar to the aforementioned findings, the decrease in DPD activity might justify the accumulation of 5-FU during administration of TQ and more anticancer effects in the 5-FU and TQ group (besides the role of signaling pathways). Nevertheless, we highly recommend more experimental studies to define the exact role of TQ in the modulation of DPD enzyme activity to determine the underlying mechanism of this synergistic effect between TQ and 5-FU.
Although many studies explored the anticancer effect of TQ alone or concomitant with other anticancer agents, none investigated the pharmacological synergism between TQ and 5-FU. The current study evaluated the impact of TQ on the pharmacokinetics of 5-FU for the first time. However, we had some limitations. First, we only measured the concentration of 5-FU, not its metabolites which are responsible for its anticancer effects. Therefore, we could not definitely state that the increase in 5-FU concentration is responsible for enhancing the anticancer effects. In the same way, despite the increase in 5-FU concentration, we did not examine the 5-FU-related toxicity. Second, we cannot determine the underlying mechanism of TQ and 5-FU synergism for sure and more studies are necessary to define the impact of TQ on catabolizing enzymes of 5-FU. We administered the 5-FU intraperitoneally. Intraperitoneal injection is safer and more effective compared to intravenous injection in peritoneal metastasis and abdominal malignancies. Actually, this method can achieve high concentration in target tissue with limited systemic penetration.47–49 The utilization of intraperitoneal injection of chemotherapy agents is expected to increase in the future.
In summary, we determined that the TQ increases the AUC, Cmax, and Tmax of 5-FU and has synergistic effects on 5-FU pharmacokinetics. Moreover, low concentration of TQ enhances the inhibitory effects of 5-FU on cell growth in colorectal cancer cell line. However, we cannot exactly determine the underlying mechanism. This synergistic effect might enhance the anticancer effects of low concentration of 5-FU, leading to drug dose reduction and reduced systemic toxicity of this chemotherapeutic agent. More studies are necessary to determine the precise mechanism of this synergistic effect.
Supplemental Material
Supplemental Material - Effect of thymoquinone on pharmacokinetics of 5-fluorouracil in rats and its effect on human cell line in vitro
Supplemental Material for Effect of thymoquinone on pharmacokinetics of 5-fluorouracil in rats and its effect on human cell line in vitro by Seyed Parsa Eftekhar, Sohrab Kazemi and Ali Akbar Moghadamnia in Human & Experimental Toxicology
Footnotes
Author contributions
AAM and SK conceptualized the study. SPE prepared the initial draft of the paper. SPE and SK revised and edited the manuscript. SPE conducted the statistical tests. AAM critically revised the project. All authors participated in the preparation of the data. All authors read the final draft of the manuscript and approved it.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was approved (IR.MUBABOL.REC.1399.465) by the Ethics Committee of Babol University of Medical Sciences (Babol, Iran).
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author and with permission from Babol University of Medical Sciences, Babol, Iran.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

