Abstract
Introduction
Osteoarthritis (OA) is one of the most common joint diseases in the elderly population. Proinflammatory cytokines, such as Interleukin-1β (IL-1β), play an important role in the development and progression of OA. Dapansutrile is a specific inhibitor of the NOD-like receptor protein 3 (NLRP3) inflammasome and exhibits anti-inflammatory properties.
Methods
In this study, we investigated the protective effect and the underlying mechanism of dapansutrile on cartilage degeneration in vitro and in vivo. In the present study, chondrocytes were isolated from rats and then were treated with dapansutrile. After that, the expression of (Cox-2, inducible nitric oxide synthase (iNOS), Mmp-3, Mmp-9, Mmp-13 and IL-10) were evaluated at RNA level, then the expression of (COX-2, MMP-3, MMP-9, MMP-13, SOX-9 and COL2) were evaluated at protein level. Subsequently, the activation of the mitogen-activated protein kinase (MAPK) pathway was tested using western blotting (WB). Additionally, the rat OA model was developed to evaluate the protective effects of dapansutrile in vivo.
Results
The results showed that dapansutrile had no obvious cytotoxicity on rat chondrocytes at 24 h (0, 1, 2, 5 and 10 μM). Dapansutrile significantly decreased IL-1β-induced upregulation of COX2, iNOS, matrix metalloproteinase 3 (MMP3), 9 (MMP9) and 13 (MMP13), and reversed IL-1β-induced the downregulation of IL-10, SOX9 and COL2. Dapansutrile also inhibited IL-1β-induced upregulation of the MAPK signaling pathway by downregulating the expression levels of phospho-ERK, and phospho-P38 in a concentration dependent manner. In addition, dapansutrile exhibited protective effects in rat OA model with lower Mankin’s score and Osteoarthritis Research Society International (OARSI) score.
Conclusion
Our study suggested that dapansutrile effectively inhibited chondrocyte inflammation by suppressing MAPK signaling pathway in vitro, and ameliorated cartilage degeneration in vivo, indicating an anti-inflammatory effect in OA treatment.
Keywords
Introduction
Osteoarthritis (OA) is a degenerative joint disease, mainly characterized by the progressive destruction of articular cartilage and joint inflammation, resulting in physical disability and reduced quality of life. 1 This disease affects 80% of the population aged 60 years and older and has high annual hospitalization rates in many countries.2,3
Chondrocytes are the main cells of articular cartilage, and they govern the pathological progression of OA through apoptosis and cytokine production.2,4 Chondrocytes play a crucial role in maintaining normal synthesis and regeneration of cartilage matrix, but this function may be delayed in the inflammatory microenvironment. 5 Therefore, controlling inflammation and chondrocyte bioactivity can potentially be an effective therapeutic approach in OA patients.
Many recent studies focus on the importance of inflammation in OA progression.5–7 In fact, the levels of some pro-inflammatory cytokines in synovial fluid may serve as diagnostic indicators of joint damage. These cytokines, which are secreted by activated synovial membranes and cartilage, stabilize chronic joint inflammation, whereas catabolic/anabolic imbalances promote cartilage degradation.8,9 Interleukin-1β (IL-1β), an important therapeutic target in OA, has been an attractive conception for many years.10,11 It is a very potent inducer of cartilage degradation, controlling the bioactivity of OA-associated proteases, such as matrix metalloproteinase 3 (MMP3) and 13 (MMP13). 5 In addition, IL-1β plays a crucial role in regulating the expression of chondrocyte-specific proteins, such as SOX9 and type II collagen, during OA.5,7
After stimulation by pro-inflammatory cytokines in OA, inducible nitric oxide synthase (iNOS) produces large amounts of nitric oxide (NO), resulting in increasing of cyclooxygenase 2 (COX2) and prostaglandin E2 (PGE2). Prostaglandin E2 induces collagen degradation by increasing MMP13 production. The inhibition of the inflammatory response induced by IL-1β may control the development of OA.5,12
The mitogen-activated protein kinase (MAPK) signaling pathway is a major signaling pathway that participates in the development of OA, and therefore has become one of the main therapeutic targets in the treatment of OA.7,12
Dapansutrile is a specific inhibitor of the NOD-like receptor protein 3 (NLRP3) inflammasome. 13 NOD-like receptor protein 3 inflammasome is considered to play an important role in many human diseases such as type 2 diabetes, gout, and multiple sclerosis.14–16 Recently, some research data suggest that dapansutrile is effective in the treatment of endotoxin-induced inflammation and joint arthritis. 17
In vitro, nanomolar concentrations of dapansutrile is able to reduce IL-1β and IL-18 release following canonical and noncanonical NLRP3 inflammasome activation. 18 In LPS-stimulated human blood-derived macrophages, dapansutrile decreases IL-1β levels by 60% and IL-18 by 70% at concentrations 100-fold lower in vitro than plasma concentrations safely reached in humans. Dapansutrile also reduces IL-1β release and caspase-1 activity in freshly obtained human blood neutrophils. Furthermore, in monocytes isolated from patients with cryopyrin-associated periodic syndrome (CAPS), dapansutrile inhibits LPS-induced IL-1β release by 84% and 36%. 19
In vivo, treatment of mice with dapansutrile limits MSU crystal articular inflammation (p > 0.0001), which is associated with decreased synovial IL-1β, IL-6, myeloperoxidase, and CXCL1 levels (p < 0.01) compared with vehicle-treated mice. Oral dapansutrile is highly effective in ameliorating reactive as well as gouty arthritis. 17
However, there have been no reports on how dapansutrile inhibits OA or the underlying mechanism. Therefore, our study discussed the action of dapansutrile in inhibiting IL-1β-induced chondrocyte inflammation and the potential mechanism.
Materials and methods
Chemicals and materials
Dapansutrile was obtained from MedChemExpress (Shanghai, China). Dulbecco’s modified eagle medium (DMEM), fetal bovine serum (FBS), penicillin, streptomycin and 0.25% pancreatic enzyme were all obtained from Gibco, NY, USA. Recombinant rat IL-1β was purchased from R&D Systems, Abingdon, UK. DMSO, BSA and collagenase II were obtained from Sigma-Aldrich, Merck KGaA, MO, USA. Radioimmunoprecipitation assay buffer, bicinchoninic acid (BCA) assay kit, protease and phosphatase inhibitors were purchased from Beyotime Institute of Biotechnology, Shanghai, China.
Isolation, culture, and treatment of chondrocytes
The isolation, culture and treatment of chondrocytes were conducted as previously described. 5 Briefly, cartilage was extracted from the hip joints of 15 rats (4 week-old male Sprague-Dawley, weighing 140 ± 20 g). The rats were put at room temperature (25°C) with 12 h light/12 h dark cycles under 50–60% relative humidity, and food and water were routinely provided. 100% CO2 was used at a chamber volume of 20% per minute to euthanized the rats. Loss of breathing and fading of eye color were observed within 2-3 min of the procedure. Articular cartilage was harvested under sterile conditions and digested using 0.25% pancreatic enzyme for 30 min. Chondrocytes were isolated from the harvested tissues by digesting the tissues for an additional 4 h using DMEM containing 0.2% collagenase at 37°C, followed by centrifugation of the cell suspension. The isolated cells were subsequently incubated in DMEM (10% FBS and penicillin-streptomycin) at 37°C with 5% CO2 until confluent, and the medium was replaced every 24 h. Then, the cells were treated with or without IL-1β (10 ng/mL) for an additional 24 h or 30 min post treatment with different concentrations of dapansutrile for 1 h. After, the cells were harvested using trypsin.
The study protocol was approved by the Ethics Committee of The Second Affiliated Hospital Zhejiang University School of Medicine, Hangzhou, China.
Cell counting kit (CCK-8) analysis
To analyze the cytotoxicity of dapansutrile on chondrocyte, CCK-8 assay (Nanjing KeyGen Biotech Co., Ltd.) was performed according to the manufacturer’s protocol. Chondrocytes (5 × 103 cells/well) were seeded into 96-well plates, and then treated with different concentrations of dapansutrile (0, 1, 2, 5, and 10 μM) for 24 h. The medium was replaced with fresh medium containing 10% CCK-8 solution, and then the cells were incubated at 37°C for 3 h. Optical density (OD) was determined at a wavelength of 450 nm. Triplicate experiments were performed independently and the data were presented as the mean ± SD.
RNA extraction and qRT-PCR
Primer sequences for target genes.

Protein extraction and western blotting
Chondrocytes were treated with dapansutrile, and then whole protein was extracted using radioimmunoprecipitation assay (RIPA) lysis buffer containing protease and phosphatase inhibitors (1% phenylmethylsulfonyl fluoride and 0.1% phosphorylated proteinase inhibitor, Invitrogen, Thermo Fisher Scientific, Inc.). The sample proteins were quantified using a BCA assay kit, and then denatured at 95–100°C for 10 min. Equal volumes of protein samples were separated by Sodium Dodecyl Sulfate PolyAcrylamide Gel Electrophoresis SDS-PAGE with 8% or 10% gels, and transferred onto membranes. After blocking with 5% BSA (Sigma-Aldrich; Merck KGaA) for 1 h, the membranes were incubated with corresponding primary antibodies against target proteins overnight at 4°C. The primary antibodies for target proteins were as follows: MMP3 (rabbit, cat. no. #ab52915; Abcam), MMP13 (rabbit, cat. no. #ab39012; Abcam), COX2 (rabbit, cat. no. #12282; Cell Signaling Technology, Inc.), SOX9 (rabbit, cat. no. #ab185966Ab; Abcam), COL2 (rabbit, cat. no. #ab34712; Abcam), (P38 ERK). β-ACTIN (mouse, cat. no. #ab8226; Abcam) or GAPDH (rabbit, cat. no. #ab70699; Abcam) were used as the endogenous controls. All antibodies were used at 1/500-1000 dilution. After washing three times with TBS-T for 30 min, the membranes were incubated with secondary antibody (cat. no. A0208; Beyotime Institute of Biotechnology) for 2 h at 4°C. After washing three times with TBS-T for 30 min, the membranes were visualized using an ECL kit (Immobilon Western Chemiluminescent HRP Substrate; cat. no. WBKLS0050; Merck KGaA), and quantified using a Bio-Rad ChemiDoc system (Bio-Rad Laboratories, Inc.).
Safranin O staining of chondrocytes
Chondrocytes were seeded into 12-well plates (5 × 105 cells/well), and then treated with dapansutrile for 1 h followed with IL-1β stimulation for 24 h. Next, the cells were fixed using 4% paraformaldehyde solution for 15 min, and then stained with safranin O for 7 min at room temperature. After washing three times with PBS, the images were captured with a gross camera and a microscope. The experiment was independently repeated three times.
Animal experiments
A total of 21 rats (6 weeks-old, male, Sprague Dawley, 180–240 g) were used to investigate the role of dapansutrile on OA in vivo. The rats were randomly divided into 3 groups (seven rats per group). Housing condition, food and water and light/dark cycles were the same as mentioned above. The rats received medial meniscus resection to develop OA. 5 Briefly, the rats were anesthetized with pentobarbital (40 mg/kg), and the knee joints were opened with medial parapatellar approach. Then, the medial meniscus was carefully resected without any articular cartilage or ligament damage. The sham group rats received an arthrotomy without medial meniscus transection.
After a week, dapansutrile was injected into the knee joints (intra-articular injection) every 7 days. After four weeks, the rats were sacrificed with 100% CO2, and the knee joint samples were fixed with 4% paraformaldehyde solution. The knee joint samples were decalcified for two months with 10% EDTA, and then dehydrated using an ascending series of ethanol, and finally embedded in paraffin blocks. The study protocol was approved by the Ethics Committee of The Second Affiliated Hospital Zhejiang University School of Medicine, Hangzhou, China.
Histological analysis
The knee joint samples were cut into 5 mm slices, and deparaffinized with xylene. The sections were rehydrated using a graded ethanol series, and then stained with safranin O/Fast Green and hematoxylin-eosin staining solutions. Three independent graders assessed the histological samples in a blinded manner using the Osteoarthritis Research Society International (OARSI) scoring system.
TUNEL staining
In order to detect the degree of cartilage DNA damage in vivo, we performed TUNEL staining. The samples were deparaffinized and rehydrated. Next, the samples were incubated with 0.1% Triton X-100 for 30 min, and then stained with In Situ Cell Death Detection Kit for 30 min at 37°C according to the manufacturer’s instructions. The nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI), and visualization of images were conducted using fluorescence microscope.
Immunohistochemical analysis
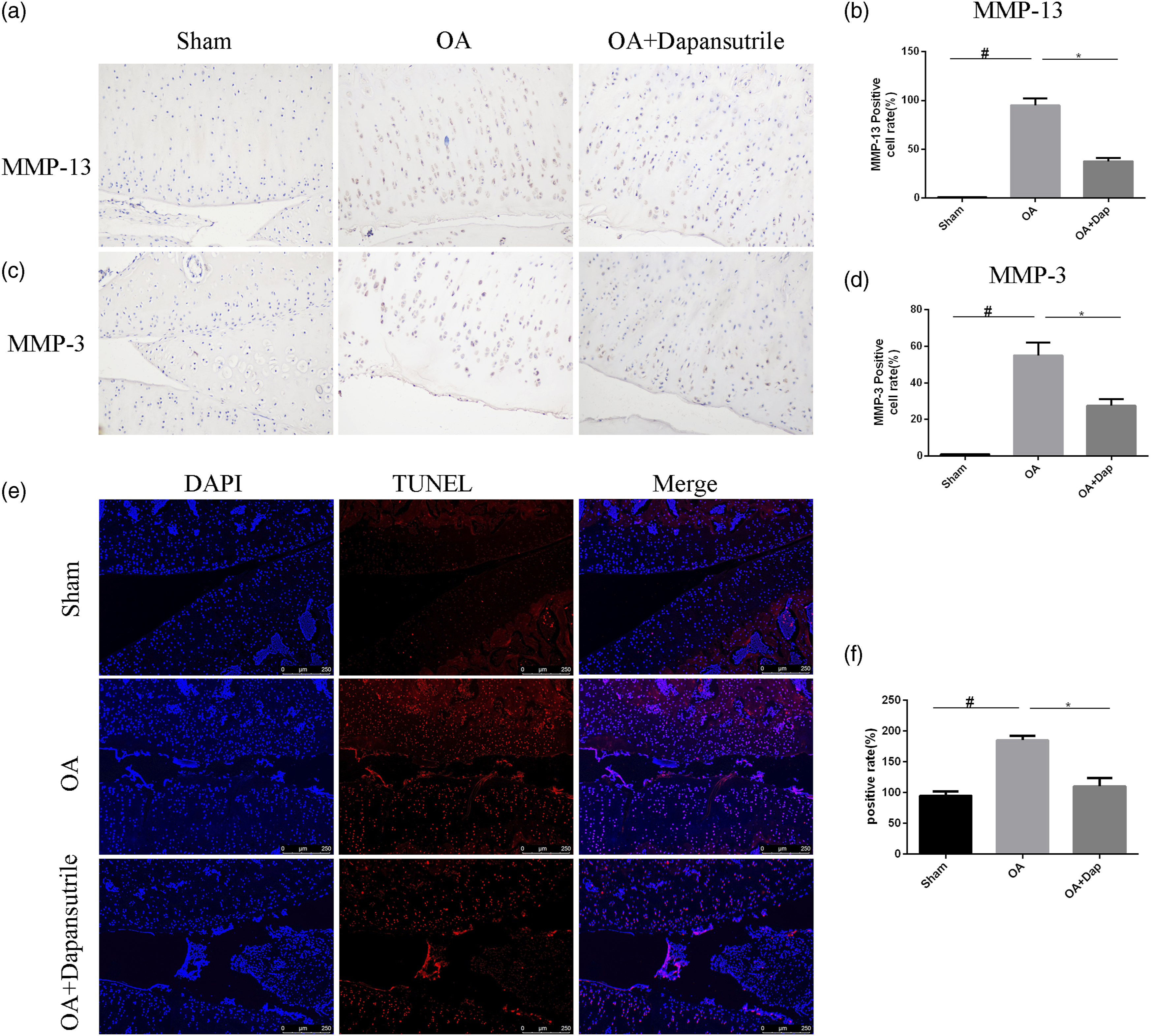
We performed immunohistochemical staining to investigate the role of dapansutrile on OA treatment in the sagittal sections of the knee joints. The sections were prepared as described above, and then subjected to antibodies against MMP3 (rabbit, cat. no. #ab52915; Abcam) and MMP13 (rabbit, cat. no. #ab39012; Abcam).
Statistical analysis
All data are presented as mean ± SD. To determine the statistical significance between test groups, we used one-way analysis of variance followed by Tukey’s post hoc test (Prism 7.00; GraphPad Software, Inc.,). p < 0.05 were considered statistically significant.
Results
The effect of dapansutrile on chondrocyte viability
To investigate the cytotoxic effect of dapansutrile on chondrocyte, we assessed cell viability using the CCK8 kit. As shown in Figure 1(b)–(d) low dose (0, 1, 2, 5, and10 μM) of dapansutrile had no obvious cytotoxicity on rat chondrocyte at 24 h, 48 h and 72 h. And then, we analyzed cytotoxicity of high dose (100, 200, 300, 400 and 500 μM) of dapansutrile. The IC50 of dapansutrile at 24 h, 48 h and 72 h were 611.7 μM, 442.8 μM and 328.3 μM, respectively. Therefore, we selected concentrations of 5 and 10 μM for further research in vitro. The effect of dapansutrile on the viability of chondrocyte. (a) Molecular structure of dapansutrile. (b, c and d) Chondrocytes were treated with various concentrations of dapansutrile, and the viability of chondrocyte was detected with CCK-8 at 24, 48, and 72 h. (e) The IC50 value of chondrocytes treated with different concentrations of dapansutrile. (f) Gross camera view of safranin O staining of IL-1β-induced chondrocytes treated with various concentrations of dapansutrile for 24 h. N = 3; data are expressed as the mean ± standard deviation; #p <0.05 versus control group; *p <0.05 versus model group.
Dapansutrile inhibited the expression levels of COX-2, iNOS, MMP-3, MMP-9, MMP-13 and upregulated the expression levels of IL-10, SOX-9 and COL2
The role of dapansutrile on the expression of major genes associated with IL-1β-induced chondrocyte inflammation and matrix degradation was investigated with qRT-PCR and western blotting (WB). qRT-PCR results revealed that dapansutrile significantly decreased IL-1β-induced upregulation of Cox2, iNOS, Mmp3, Mmp9 and Mmp13 in a concentration dependent manner (Figure 2(a)). Furthermore, dapansutrile is able to revere the effect of IL-1β-induced downregulation of IL-10. Western blotting analysis also showed that pretreatment with dapansutrile decreased the protein expression levels of COX2, MMP3 MMP9 and MMP13 (Figure 2(b) and (c)). In addition, dapansutrile significantly increased the expression levels of SOX9 and COL2 at concentrations greater than 5 μM (Figure 2(b) and (c)). In addition, we detected glycosaminoglycan, a cartilage matrix component, using safranin O staining. Dapansutrile treatment restored the fading of safranin O staining induced by IL-1β in rat chondrocytes (Figure 1(c)). Collectively, these results indicated that dapansutrile is able to protect rat chondrocytes by downregulating inflammatory genes and upregulating chondrocyte-specific protein expression in vitro. The effect of dapansutrile on IL-1β-induced chondrocyte inflammation and cartilage-specific proteins. Dapansutrile pretreated chondrocytes were subjected to IL-1β (10 ng/mL) for 24 h. (a) The expression of Cox2, iNOS, IL-10, Mmp-3, Mmp-9 and Mmp-13 of rat chondrocytes were evaluated at mRNA levels using qRT-PCR. (b and c) Western Blotting was used to evaluate the expression of COX2, MMP3, MMP9, MMP13, SOX9 and COL2. N = 3; data are expressed as the mean ± standard deviation; #p <0.05 versus control group; *p <0.05 versus model group. Abbreviations: GAPDH, glyceraldehyde 3-phosphate dehydrogenase; COX2, Cyclooxygenase-2; MMP, matrix metalloproteinase; SOX9, Sex-determining region Y-Box 9; COL2, type II collagen; qRT-PCR, quantitative real-time polymerase chain reaction.
Dapansutrile suppressed MAPK signaling pathway stimulated by IL-1β in chondrocytes
ERK, and P38 are major proteins of the MAPK signaling pathway. We analyzed the activation of the MAPK pathway by detecting the phosphorylation of ERK, and P38 using WB. Dapansutrile effectively downregulated the expression levels of phospho-ERK, and phospho-P38 in a concentration dependent manner (Figure 3(a)–(c)). Western blotting analysis results indicated that dapansutrile could inhibit MAPK signaling pathway in rat chondrocytes. Dapansutrile suppressed MAPK signaling pathway stimulated by IL-1β in chondrocytes. Chondrocytes were treated with different concentrations of dapansutrile for 1 h, and then stimulated with IL-1β (10 ng/mL) for 30 min. (a) Western blot analysis (b) and relevant quantitative analysis of p-P38 and p-ERK. N = 3; data are expressed as the mean ± standard deviation; #p <0.05 versus control group; *p <0.05 versus model group. Abbreviations: GAPDH, glyceraldehyde 3-phosphate dehydrogenase; MAPK, mitogen-activated protein kinase; p-P38, phosphor-P38; p-ERK, phosphor-ERK.
Dapansutrile exerted protective effects in rat OA model
To evaluate the protective effects of dapansutrile in vivo, we developed the rat OA model by surgical resection of the medial meniscus of 14 rats, which were randomly divided into two groups (OA group and dapansutrile treatment group). Sham surgery was performed on 7 rats for the normal group. Dapansutrile injection (knee joint injection) was initiated one-week post-surgery, and once a week for 4 times. After that, the rats were sacrificed and the knee joints were collected for histological analysis. The OA group exhibited typical OA characteristics compared with the sham group. Compared to the OA-group, cartilage degeneration was obviously ameliorated in the dapansutrile treatment group (Figure 4(a) and (b)). Furthermore, the results of Mankin’s score and OARSI score analysis showed that dapansutrile had significant protective effects on OA (Figure 4(c) and (d)). In addition, the results of TUNEL staining showed that dapansutrile alleviated destructive changes in cartilage matrix (Figure 5(e) and (f)). Immunohistochemistry staining results also revealed that the expression levels of MMP3 and MMP13 were significantly decreased in the dapansutrile treatment group compared with the OA group (Figure 5(a)–(d)). These results indicated that dapansutrile has protective effects on OA in vivo. The protective effect of dapansutrile in rat OA model. Articular cartilage slices stained with (a) safranin O/Fast Green and (b) hematoxylin-eosin staining; scale bar = 200 μM. (c) Mankin’s score and (d) OARSI score in sham group, OA group and OA + dapansutrile group. N = 3; data are expressed as the mean ± standard deviation; #p <0.05 versus sham group; ∗p <0.05 versus OA group. Abbreviations: OARSI, Osteoarthritis Research Society International; OA, osteoarthritis. The protective effects of dapansutrile in rat OA model. Immunohistochemistry for antibody against (a) MMP13 and (c) MMP3. Quantitative analysis (b, d) of MMP-13 and MMP-3 and (e, f) TUNEL staining; scale bar = 200 μM. N = 3; data are expressed as the mean ± standard deviation; #p <0.05 versus sham group; ∗p < 0.05 versus OA group. Abbreviations: OA, osteoarthritis; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling.
Discussion
Osteoarthritis is the most common form of arthritis worldwide, and the number of OA patients are increasing every year. 10 Nowadays, the main types of treatment for OA include surgical treatment and non-surgical treatment. 20 However, although pharmacological treatments (including steroids and NSAIDs) can relieve some symptoms of OA, they fail to prevent articular cartilage damage, and side effects such as damage to the gastrointestinal tract often occur. Therefore, it is of great importance to find and use compounds that can effectively inhibit cartilage degeneration.
Statins, also known as HMG-CoA reductase inhibitors, are lipid-lowering medications generally used to reduce the risk of cardiovascular disease. They also reveal pleiotropic effects, including immunomodulation, improvement of endothelial function, increased antioxidant properties, and inhibition of inflammatory responses. The positive role of simvastatin and/or ezetimibe-loaded nanofibers on the healing of bone formation has been demonstrated in earlier investigations. 21 In this process, statins mainly exert their effects through inhibition of the mevalonate pathway, thereby reducing osteoclastic activity, as well as by increasing the expression of bone morphogenetic protein 2 (BMP-2). Statins also stimulate the production of osteoprotegerin (OPG). Osteoprotegerin is a natural decoy binding protein produced by the activated osteoblasts which antagonizes the effects of receptor activator of nuclear factor-κB ligand (RANKL), a molecule that enhances the osteoclast formation. 22
Dapansutrile is a specific inhibitor of the NLRP3 inflammasome, which has anti-inflammatory properties, and has been reported to be effective in the treatment of various inflammatory diseases. 14 In the present study, we found that low dose (0, 1, 2, 5, and 10 μM) of dapansutrile has no cytotoxicity on chondrocytes at 24, 48 and 72 h, and we confirmed IC50 of dapansutrile in rat chondrocytes at 24, 48 and 72 h (611.7 μM, 442.8 μM and 328.3 μM, respectively).
Interleukin-1β, one of the pro-inflammatory cytokines in OA, plays an important role in the development and progression of OA.10,23,24 According to clinical trials, IL-1β inhibition therapy has shown significant benefits in knee and hand OA patients.25–27 Interleukin-1β can induce matrix MMP3 and MMP13 at RNA and protein levels in chondrocytes. 28 In addition, IL-1β inhibits the expression of type II collagen in chondrocytes by inducing phosphorylation of c-Jun, activation of AP-1, and subsequent suppression of SOX9. 29 Our results indicated that dapansutrile can inhibit IL-1β-induced upregulation of inflammatory genes and degrading genes including Cox-2, iNOS, Mmp-3, Mmp-9 and Mmp-13, and it is able to reverse the expression of IL-10 (Figure 2). Moreover, dapansutrile could revive the expression of type II collagen and Sox-9 (Figure 2). Dapansutrile is also capable of restoring IL-1β-induced reduction of safranin O staining in chondrocytes (Figure 1(c)). These results indicated that dapansutrile may exhibit antidegenerative action in vitro.
The mechanism of chondrocyte inflammation and OA is complicated, and there are many signal pathways involved in its occurrence and progression.
Mitogen-activated protein kinase signaling pathway is also recognized as one of classical pathways participated in this progress, and this signal pathway was focused in present study. 7
The MAPK signaling pathway mainly involves three major proteins including ERK (phospho-ERK), JNK (phospho-JNK) and P38 (phospho-P38).30,31 According to WB analysis, dapansutrile is able to suppress the expression of phospho-ERK, and phospho-P38, leading to the inhibition of the MAPK signaling pathway (Figure 3(a)–(c)). This result suggested that dapansutrile could exert antidegenerative action through inhibition of MAPK signaling pathway.
Additionally, the protective action of dapansutrile on articular cartilage was evaluated with rat OA model. In the dapansutrile treatment group, cartilage inflammation and degeneration were alleviated, and synovium hyperplasia was also inhibited (Figure 4). TUNEL staining results showed that dapansutrile alleviated destructive changes in cartilage matrix (Figure 5(e) and (f)). In addition, the immunohistochemistry analysis results also showed that dapansutrile was able to inhibit the expression of MMP3 and MMP13 in vivo (Figure 5(a)–(d)). Furthermore, dapansutrile decreased Mankin’s score and OARSI score in vivo.
Further preclinical studies are needed to determine the potential of dapansutrile as an anti-inflammatory drug for the treatment of OA.
Conclusion
The present study demonstrated that dapansutrile inhibited chondrocyte associated inflammation by suppressing MAPK signaling pathway in vitro, and ameliorated cartilage degeneration in vivo. These findings indicate dapansutrile as a potentially effective drug candidate for the treatment of OA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Zhejiang Province (LD19H060001).
