Abstract
In this study, we aimed to evaluate possible toxic effects of thimerosal, aluminum and combination of thimerosal and aluminum in SH-SY5Y cells. Inhibitory concentrations were determined by MTT assay; reactive oxygen species (ROS) were determined by a fluorometric kit and antioxidant/oxidant parameters were measured by spectrophotometric kits. Nuclear factor erythroid 2-associated factor 2 (Nrf2), norepinephrine (NE), dopamine transporter (DAT) and dopamine beta β-hydroxylase (DBH) levels were measured by sandwich ELISA kits while 8-hydroxy deoxyguanosine (8-OHdG) and dopamine levels were determined by competitive ELISA kits. Thimerosal (1.15 μM) and aluminum (362 μM) were applied to cells at inhibitory concentrations 20 (IC20s) for 24 h. ROS increased significantly in cells aluminum- and aluminum+thimerosal-treated cells. Glutathione levels decreased in aluminum group while total antioxidant capacity and protein oxidation levels increased significantly in aluminum and aluminum+thimerosal groups. Lipid peroxidation increased significantly in groups treated with aluminum and aluminum+thimerosal. Nrf2 levels and DNA damage were significantly higher in all groups while dopamine levels significantly increased in cells treated with thimerosal and aluminum+thimerosal, DAT levels were found to be higher in all experimental groups compared to the control. These findings showed that both thimerosal and aluminum can change oxidant/antioxidant status, cause DNA damage, alter dopamine and DAT levels. Changes seen in cells treated with combined exposure to aluminum and thimerosal are more pronounced. Special care should be taken while vaccinating sensitive populations and safer alternatives for aluminum and thimerosal should used.
Introduction
Vaccines are pharmaceutical preparations that are developed to prevent disability, decrease in quality of life and death due to infectious agents.1–3 Many infectious diseases have been eradicated with the application of vaccines around the world. 4
Vaccines consist of five basic components: antigens, adjuvants, suspension fluids, stabilizers and preservatives. 5 Vaccines are divided into two groups according to their antigenic contents: live attenuated and inactivated vaccines. As inactive vaccines cannot produce the same level of immune response in a single dose, there is need for booster dosing or co-administration of the antigen with an adjuvant in inactivated vaccines. 6
Adjuvants are not usually immunogenic compounds. However, they can provide a stronger and longer-lasting immune response in a shorter time and reduce the amount of antigen or the number of vaccinations required to achieve a primary immune response. Thus, they provide reducing in the cost of vaccination. 6 Aluminum salts are one of the Food and Drug Administration (FDA)-approved adjuvants. 7 Today, the most commonly used aluminum salt adjuvants are aluminum hydroxide Al(OH)3 and aluminum phosphate AlPO4. 8
Aluminum is found in many pharmaceutical products, the human food chain and the content of various consumer products. 9 Aluminum salt-based adjuvants are generally referred to as “alum”. 10 Alum is included in sevral licensed human vaccines. 11 However, based on different in vitro and in vivo studies, aluminum intoxication has been associated with Alzheimer’s disease and amyotrophic lateral sclerosis (ALS).12–15
Preservatives are used in vaccines to prevent bacterial and fungal contamination. Thimerosal (sodium ethyl mercury salicylate) is a broad spectrum compound that is usually added to multiple dose vaccines at a concentration of 0.01%.16–18 Thimerosal contains 49.6% mercury (Hg) by weight. It is rapidly metabolized to ethyl Hg and thiosalicylate. 19 Hg is a neurotoxic heavy metal that causes global environmental pollution. 17 It is known that thimerosal-containing vaccines can pass through the blood-brain barrier and cause neurotoxicity when given in repetitive low doses, especially in the prenatal and postnatal period. 20
The increase in the production of intracellular reactive oxygen species (ROS) and the consecutively oxidative stress can play a role in the pathogenesis of many neurodegenerative diseases.21–23 To overcome oxidative stress, the endogenous antioxidant response pathway increases the expression of cytoprotective enzymes, which scavenge free radicals and reduce the risk of cellular damage caused by ROS. This endogenous antioxidant pathway is regulated by the binding of the nuclear factor erythroid 2-associated factor 2 (Nrf-2) to antioxidant response elements in the promoters of antioxidant genes.24,25 In addition, there is evidence that Nrf-2 exerts anti-inflammatory effects and it has also been shown to be associated with mitochondrial function. Given that mitochondrial dysfunction and neuroinflammation are features of many neurodegenerative diseases, these properties of Nrf-2 may make this protein a therapeutic target. 26
It has been shown in many publications that most of neurological and neurodegenerative disorders are associated with disturbances in the homeostasis of neurotransmitters. Dopamine is a neurotransmitter that plays a key role in the cardiovascular and central nervous systems. Dopamine release at physiological concentrations does not appear to be toxic. However, dopamine-related disorders in can lead to neuropathologies. These mechanisms underlying these disorders are still unclear although few evidence has shown that oxidative stress, neuroinflammation and apoptosis can be observed after disturbances in dopamine release/metabolism.27,28
Considering the available data, this study was designed to evaluate the toxic effects of thimerosal and aluminum hydroxide in human neuroblastoma cell line, by determining cytotoxicity and levels of ROS, antioxidant enzymes, cellular antioxidants, nuclear factor erythroid 2–related factor 2 (Nrf2) levels, oxidative DNA damage and neurotransmitters/neurotransmitter-related proteins.
Materials and methods
Chemicals and kits
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and thimerosal were purchased from Sigma-Aldrich (Mannheim, Germany). Dimethyl sulfoxide (DMSO) was purchased from Duchefa Biochemicals (RV, Haarlem, The Netherlands). Superoxide dismutase (SOD), catalase (CAT), glutathione (GSH), protein carbonyl, oxidative DNA/RNA damage (8-hydroxy deoxyguanosine, 8-OHdG), thiobarbituric acid reactive substances (TBARS), total antioxidant capacity (TAOC) and total protein assay kits were obtained from Cayman Chemical (Ann Arbor, MI). Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM-F12), Dulbecco’s Phosphate Buffered Saline (DPBS), fetal bovine serum (FBS) and penicillin/streptomycin were purchased from Biowest (Riverside, MO). ROS assay kit was obtained from Sigma-Aldrich (Mannheim, Germany). DNA isolation kit was from Zymo Researh (Irvine, CA). Human dopamine beta β-hydroxylase (DBH), human norepinephrine (NE) and human Nrf2 assay kits were purchased from BT LAB (Shanghai, China). Human dopamine assay kit was obtained from Cusabio (Wuhan, China) and human dopamine transporter (DAT) assay kit was purchased from Elabscience (Houston, TX).
Cell line
Studies were performed on SH-SY5Y cell line (ATCC CRL-2266™, Manassas, VA). The cells are neuroblastoma cells in epithelial morphology and they were obtained from bone marrow tissue from a 4-year-old girl. Originally derived from a metastatic bone tumor biopsy, this cell line is a subset of SK-N-SH (ATCC® HTB-11™) line and consist of both adherent and suspended cells. Cells were grown in flasks in DMEM/F-12 medium by adding 10% FBS and 1% penicillin-streptomycin. The incubation process was carried out in an incubator at 37oC and 5% CO2. The medium was changed 2–3 times a week. Cells were split when they reached to 70–90% confluency.
Cytotoxicity
MTT assay was performed to evaluate the cell viability. Cells (1x10 4 ) were plated in media that contained thimerosal (0; 1; 1,1; 1,25; 1,3; 1,5; 1,75; 2; 2,5; 3; 4 µM) or aluminum hydroxide (0; 10; 25; 50; 100; 250; 500; 750; 1000; 1500; 2000; 2500 µM) at different concentrations for 24 h. The test substance-containing solutions were removed at the end of the incubation time and 100 µl MTT solution (0.5 mg/ml) was added to each well and incubated at 37oC for 3 h. Later, MTT solution was then withdrawn from each well and 150 µl of DMSO was added. To dissolve the formazan crystals, the plate was put on a horizontal shaker for 5–10 min. The absorbance values were measured at 570 nm. The viability of the control cells was considered to be 100%. The viability of the aluminum and/or thimerosal cells was calculated as a percentage of the viability of the control cells.
Experimental groups
1. Control Group (C): Cells were grown in cell medium. 2. Thimerosal Group (T): The inhibitory concentration 20 (IC20 = 1.15 μM) was applied for 24 h. 3. Aluminum Hydroxide Group (A): IC20 (362 μM) of aluminum hydroxide was applied for 24 h. 4. Aluminum Hydroxide + Thimerosal Group (AT): Thimerosal (1.15 μM) and aluminum hydroxide (362 μM) were applied to the cells in this group at IC20 concentrations for 24 h.
Intracellular reactive oxygen species
A one-step precision fluorometric intracellular ROS kit was used to detect intracellular ROS levels. A cell-permeable sensor reacts with cytoplasmic ROS to produce a fluorometric product proportional to the amount of whole-cell ROS. Fluorescence was measured at λexcitation = 540 nm and λemission = 570 nm. The ROS levels of control cells were assumed to be 100% and the ROS levels of study groups were expressed as a percentage of the control.
Preparation of cell lysates
Aluminum and/or thimerosal at IC20 doses were applied to the study groups and the cells were incubated in flasks at 37oC and 5% CO2 for 24 h. At the end of the incubation period, the medium was removed and cell lysates were prepared by adding 800 μl of lysis buffer, which contained 1 ml of protease inhibitor and a protease inhibitor cocktail. After adding lysis buffer and centrifuging at 13000 r/min for 15 min, the supernatant was discarded and the lysates were stored at −80oC until analysis.
Oxidative stress parameters
Antioxidant enzymes
Superoxide dismutases (SOD) are metalloenzymes that facilitate the reduction of superoxide anion, a reactive oxygen compound that is exceedingly harmful to living organisms, to molecular oxygen and H2O2. 29 The kit was worked by detecting superoxide radicals produced by hypoxanthine and xanthine oxidase using tetrazolium salt. At 460 nm, the absorption of the formazan dye was measured. The quantity of Cu,Zn-SOD required to convert half of the superoxide anion to products was represented as U/mg protein.
In most aerobic species, CAT is an antioxidant enzyme found in practically every tissue and cell. It is essential for the detoxification of H2O2, one of the most toxic ROS products produced by both normal aerobic metabolism and pathological alterations in cells. 30 The assay technique was based on the production of formaldehyde from methanol in the presence of H2O2 using CAT. Purpald (4-amino-3-hidrazino-5-merkapto-1,2,4-triazol), which was utilized as a chromogen, interacted with the formaldehyde to produce a purple color. At 540 nm, the absorbance was measured. The amount of enzyme that allows the formation of 1 nmol formaldehyde per minute was expressed as nmol/min/mg protein.
Cellular oxidants/antioxidants
Lipid peroxidation is an indicator of oxidative stress and malondialdehyde (MDA) and by-products are formed as a result. 31 As a result of the reaction between thiobarbituric acid (TBA) and MDA in an acidic environment at high temperature, a complex was formed. The amount of MDA was determined by spectrophotometric measurement of this complex at 530 nm. 32 MDA levels were expressed as nmol/mg protein.
Protein carbonyls formed as a result of protein oxidation are one of the indicators of oxidative stress. Protein carbonyl levels were measured based on the principle that the 2,4-dinitrophenhydrazine (DNPH) compound and carbonyls react to form a hydrazone compound. Absorbance of the product was measured spectrophotometrically at 370 nm to calculate the amount of hydrazone compound. 33 Protein carbonyl levels were expressed as nmol/mg protein.
Glutathione is the most important thiol in higher organisms. Glutathione transferase, which is involved in the detoxification of xenobiotics in the cell, uses GSH as a co-substrate. The sulfhydryl groups of GSH react with 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) and the disulfide bond is broken to produce 5-thio-2-nitrobenzoic acid (TNB). As a result of this reaction, GSH is converted into oxidized glutathione (GSSG). In order to maintain the detoxification of free radicals in the cell, GSSG must be converted back to its reduced form by glutathione reductase. This recycling reaction is directly proportional to the TNB production rate, which is directly proportional to the GSH concentration in the sample. 34 By measuring the absorbance of TNB at 414 nm, the level of GSH in the sample was determined and expressed as nmol/mg protein.
The sum of endogenous and food-derived antioxidants constitutes the total antioxidant activity (TAOC). The assay was based on the ability of metmyoglobin to cause 2,2′-azino-di(3-etilbenztiyazolin sulfonate (ABTS) to get oxidized. Cellular antioxidants prevent this reaction. 35 By using Trolox, TAOC levels were determined at 750 nm and expressed as nmol/mg protein.
Nrf2 levels
Nrf2 is one of the most important transcription factors for antioxidant responses. 36 Nrf2 levels in the samples were measured based on sandwich ELISA principle. The kit plate was coated with human NRF2 antibody. Nrf2 in the samples was bond to the antibodies in the wells. Biotinylated human NRF2 antibody and Streptavidin-horse radish peroxidase (HRP) were then added, respectively. The color intensity, which changed proportionally to the Nrf2 levels in the samples, was measured at 450 nm and TAOC levels were expressed as ng/mg protein.
DNA isolation and oxidative DNA damage
One of the most important causes of DNA damage is oxidative stress. The one of the highly measured biomarkers of DNA damage is 8-OHdG, which was determined by using a competitive ELISA kit after isolation of DNA from samples by using a DNA isolation kit. DNA amount of the samples were given as μg/ml. The assay for 8-OH-dG was based on competitive binding of oxidatively damaged guanine species and the 8-OH-dG-acetylcholinesterase conjugate was employed as a tracer to a DNA/RNA oxidative damage monoclonal antibody. When all of the steps in the kit method were completed, the color development was measured at 412 nm. Standard curves were used in the calculations. The results were given in pg/μg DNA.
Neurotransmitters
Norepinephrine and dopamine, which are neurotransmitters of the sympathetic nervous system, show adaptation to acute and chronic stress in the body. 37 NE levels in cell culture media were measured based on the “Double Antibody Sandwich ELISA” principle. The plate was coated with human norepinephrine antibody. NE in the samples were attached to the antibodies in the wells. Afterwards, biotinylated human NE antibody and Streptavidin-HRP were added respectively. Color change was observed in proportion to the NE in the samples. NE levels were determined by measuring this color intensity at 450 nm and expressed as pg/ml.
Dopamine levels in cell culture media were measured using a Competitive ELISA kit. First, the 96-well plate was coated with antibody. Samples were added to a 96-well plate with an antibody specific for dopamine and HRP conjugated dopamine. A competitive inhibition reaction between HRP-labeled dopamine and antibody-unlabeled dopamine resulted in a color change inversely proportional to the amount of dopamine in the samples. Dopamine levels were calculated by measuring the intensity of this color change at 450 nm in the spectrophotometer and expressed ng/ml.
Dopamine transporter is a membrane-spanning protein, which mainly pumps dopamine out of the synaptic cleft back into cytosol. Dopamine reuptake via DAT provides the primary mechanism through which dopamine is cleared from synapses. 38 DAT levels were measured using a Sandwich ELISA kit. The plate in the kit was coated with the specific Human DAT antibody and the DAT in the samples is conjugated with these DAT antibodies. Afterwards, the wells containing DAT turned blue with the added Avidin-HRP conjugate and biotinylated detection antibody. The reaction ended with the addition of the stop solution. The DAT concentrations in the samples were measured at 450 nm and expressed as pg/mg protein.
Dopamine beta hydroxylase is an enzyme that catalyzes the conversion of dopamine to norepinephrine. 39 DBH levels were measured using the Sandwich ELISA kit. The 96-well plate was precoated with human DBH antibody. DBH in the samples bind to the antibody in the wells. Biotinylated human DBH antibody was then added and attached to DBH in the sample. Streptavidin-HRP later binds to the biotinylated DBH antibody. DBH levels in the samples were determined at 450 nm and expressed as ng/mg protein.
Total protein levels
Protein quantification was based on the Bradford method. 40 In the Bradford method, the blue dye turns brown when bound to the protein in an acidic medium. By measuring this colored product at 595 nm, the total protein amount in the medium and lysates was calculated and expressed as mg/ml.
Statistical analysis
Statistical Package for Social Sciences Program (SPSS) version 17.0 (SPSS Inc., Chicago, IL) was used for statistical analysis. The comparison of the results obtained from all study groups was performed by Kruskal–Wallis one-way analysis of variance (ANOVA). The difference between the groups was analyzed by using Student’s t-test. The results were given as mean±standard deviation (SD). p < 0.05 was considered statistically significant.
Results
Cell viability
Considering the MTT assay results (Figure 1(a)) for aluminum hydroxide, IC50, IC30 and IC20 values were determined as 2.59, 1.75 and 1.15 μM, respectively. For thimerosal (Figures 1(b)), IC50, IC30 and IC20 values were determined as 2812.89, 889.81 and 362 μM, respectively (Table 1). IC20 values were applied to the study groups. Cell viability observed at different aluminum hydroxide (a) and thimerosal (b) concentrations. IC50, IC30 and IC20 values of aluminum hydroxide and thimerosal. IC50: inhibitory concentration 50; IC30: inhibitory concentration 30; IC20: inhibitory concentration 20.

Intracellular reactive oxygen species
Intracellular ROS levels of the study groups are given in Figure 2. Intracellular ROS levels in the thimerosal group (T) increased by 5.67% compared to the control group (p = 0.06). The ROS levels in aluminum hydroxide group (A) increased by 17.33% and this difference was significant compared to the control group (p = 0.00001). Co-administration of aluminum and thimerosal (AT) increased intracellular ROS production by 11.26% (p = 0.001). The lesser increase in the AT group compared to group A may be attributed to the increased cytotoxicity with the co-administration of aluminum and thimerosal. Intracellular ROS levels of study groups. C: control group; T: thimerosal group; A: aluminum group; AT: aluminum+thimerosal The amount of intracellular ROS produced by the control cells was assumed as 100% and the amount of ROS produced by the other cells was calculated as % compared with the control. ROS: reactive oxygen species. a, bBars that do not share same letters (superscripts) are significantly different from each other (p < 0.05).
Antioxidant enzymes
The superoxide dismutase activities of the study groups are also in Figure 3(a). The Cu,Zn-SOD activity of the thimerosal group was not different from control. The Cu,Zn-SOD activity of the aluminum hydroxide applied group increased significantly (81%, p = 0.01) compared to the control group. Although the Cu,Zn-SOD activity of the AT group decreased compared to the control group, this difference was not significant (10%, p = 0.08). Superoxide dismutase and catalase activities of study groups. a: SOD activity in the study groups; b. CAT activity in the study groups. a,b, cBars that do not share same letters (superscripts) are significantly different from each other (p < 0.05). CAT: catalase; SOD: superoxide dismutase.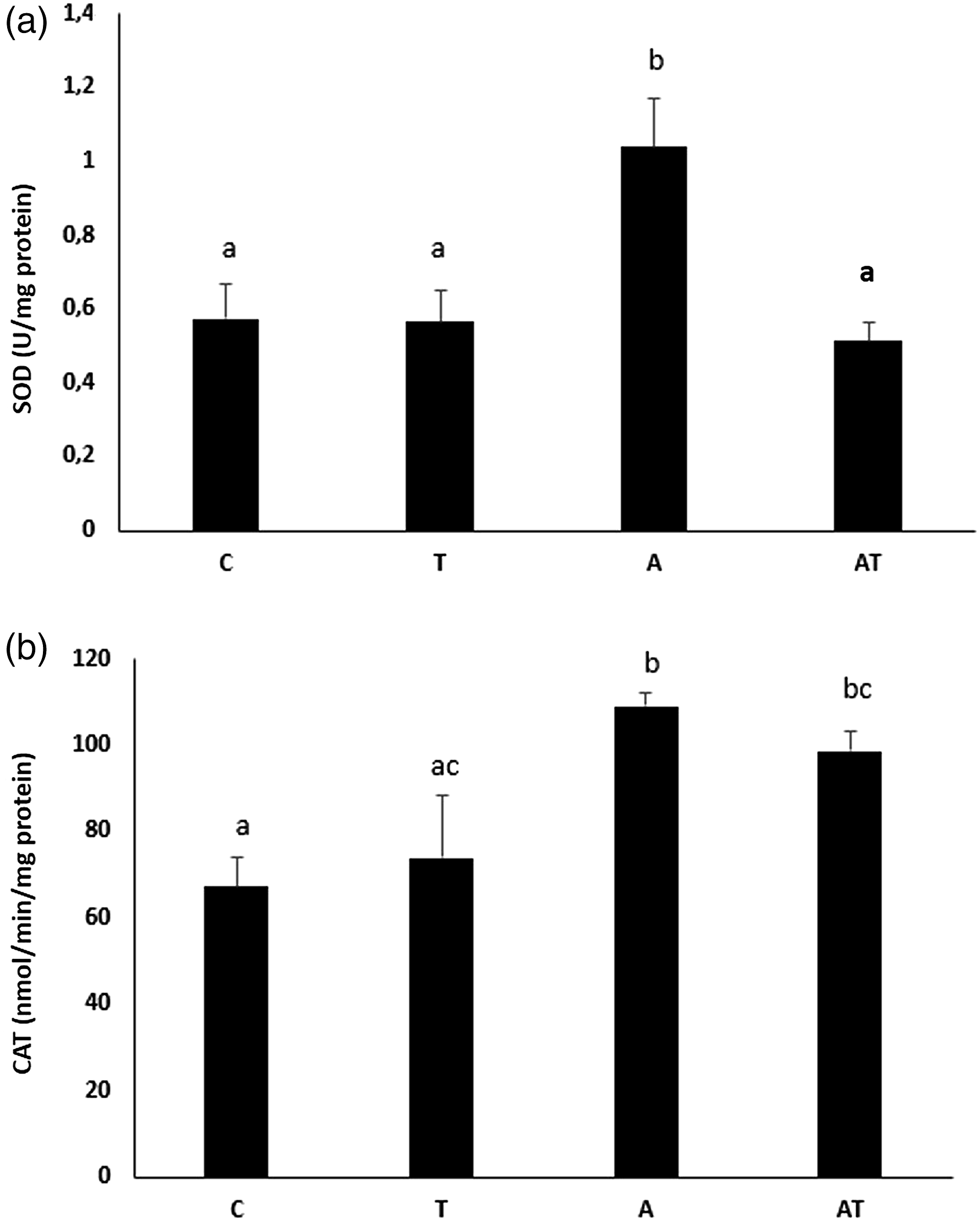
Catalase activity of the study groups is given in Figure 3(b). Although the CAT activity of the T group was slightly higher than the control group, this difference was not significant (9.7%; p = 0.07). The CAT activity of groups A and AT was significantly higher than the control group [62% (p = 0.01) and 46%, (p = 0.02), respectively]. CAT activity of group A was significantly higher than T group (48%; p < 0.02).
Oxidative stress parameters and oxidative DNA damage
Total GSH levels in the experimental groups are shown in Figure 4(a). While the total GSH levels decreased by 47% in the aluminum hydroxide applied group (p = 0.010), no statistically significant change was observed in the total GSH levels in the other experimental groups. Total glutathione, lipid peroxidation, protein carbonyl, total antioxidant capacity and oxidative DNA damage levels of study groups. a: GSH levels; b: MDA levels; c: Protein carbonyl levels; d: TAOC levels; E: 8-OHdG levels. GSH: total glutathione; MDA: malondialdehyde. TAOC: total antioxidant capacity; 8-OHdG: 8-hydroxy two deoxyguanosine a,b, cBars that do not share same letters (superscripts) are significantly different from each other (p < 0.05).
Lipid peroxidation levels are shown in Figure 3(b). MDA levels increased by 36% in A group (p = 0.048). MDA levels ∼2.7 times in group AT versus control (p = 0.002).
Protein carbonyl levels of the experimental groups are shown in Figure 4(c). Protein oxidation increased by 40% in T group versus control group (p = 0.22). Protein carbonyl levels were 78% higher in group A group compared to control (p = 0.03). In the AT group, protein carbonyl levels increased ∼2.6 times (p = 0.01) vs. control group. However, there were no significant differences between the AT group versus A and T groups.
TAOC levels in the experimental groups are shown in Figure 4(d). TAOC levels increased significantly in aluminum hydroxide-exposed cells versus control (43%; p = 0.03). TAOC levels increased ∼2.7 times in the AT group compared to control group (p = 0.001).
8-OHdG levels showing the amount of oxidative DNA damage in the experimental groups are shown in Figure 4(e). While 8-OHdG levels increased by 13% in the T group (p = 0.50), the increase was approximately ∼2.2 times in the A group (p = 0.01). 8-OHdG levels increased approximately 3.3 times in the AT group (p = 0.0003).
Nrf2 levels
Nrf2 protein levels in the experimental groups are shown in Figure 5. While Nrf2 levels increased ∼44% in the T group (p = 0.00001), the increase was 55% in the A group (p = 0.02). Nrf2 levels increased almost 2.2 times in the AT group (p = 0.002, vs. control). The difference in Nrf2 protein levels was also significant between AT group and T group (52%, p = 0.008). Nuclear factor erythroid 2-related factor 2 (Nrf2) levels of study groups. a,b, cBars that do not share same letters (superscripts) are significantly different from each other (p < 0.05). Nrf2: Nuclear factor erythroid 2-related factor 2.
Neurotransmitters
Norepinephrine levels in the experimental groups are shown in Figure 6(a). There were no statistically significant changes in the study groups versus control (p > 0.05, all). Norepinephrine, dopamine, dopamine transporter protein and dopamine β-hydroxylase levels of study groups. a,b,c,d Bars that do not share same letters (superscripts) are significantly different from each other (p < 0.05). A: Norepinephrine levels; B: Dopamine levels; C: DAT levels; D: DBH levels DAT: Dopamine transporter; DBH: Dopamine β-hydroxlase.
Dopamine levels in the experimental groups are shown in Figure 6(b). While dopamine levels increased in the T group (47%; p = 0.10 vs. control), we observed a non-significant decrease in A group (4.5%; p = 0.8). In the AT group, dopamine levels increased 3.3 times (p = 0.03 vs. control).
Dopamine transporter protein (DAT) levels in the experimental groups are shown in Figure 6(c). DAT levels increased significantly in T, A and AT groups versus control [28% (p = 0.048), 38% (p = 0.041) and 29% (p = 0.043), respectively; p < 0.05 in all]. However, the differences between T and A, T and AT and A groups were not significant.
Dopamine β-hydroxylase levels in the experimental groups are shown in Figure 6(d). While DBH levels increased by 48% in the T group (p = 0.131), the increase was 63% in the A group (p = 0.06) compared to control group. In the AT group, DBH levels increased by 93% versus control (p = 0.005).
Discussion
Vaccines are biological drugs that are obligatory to be used in order to protect the health and well-being of all individuals of all ages in the society today. In particular, adverse reaction concerns and social judgements are the most important factors leading to “vaccine rejection” or “vaccine hesitancy”. Apart from the adverse effects of vaccines, studies show that thimerosal, which is used as a preservative in multiple-dose vials, and aluminum, which is the most frequently used adjuvant compound, are causes serious rejection and uncertainty in infant and child vaccinations due to worries about their neurotoxic effects.41,42
Many studies, which were later determined to be biased and most of which were withdrawn from publications, claimed that thimerosal may be associated with autism spectrum disorders (ASDs) in children.42,43 On the other hand, the neurotoxic effects of aluminum have been known for mny years. It has been stated that many neurodegenerative diseases, especially AD, are caused by the accumulation of heavy metals, including aluminum and Hg in the brain.41-43 Both aluminum and Hg are suggested to cause oxidative stress and their neurotoxic effects, in part, may arise from the imbalance between oxidants and antioxidants.44,45 However, both ASDs and AD are multi-factorial diseases and it should not be forgotten that not only environmental exposures but also genetic predispositions may be effective in their etiology. 46
In the present study, the oxidative stress inducing effects of thimerosal, aluminum hydroxide, and their combined exposure on neuroblastoma cells were determined by evaluating the levels of intracellular ROS, total GSH, TAOC, lipid peroxidation, protein oxidation and DNA damage. Intracellular ROS production increased in thimerosal applied cells. While thimerosal did not significantly change total GSH, TAOC and MDA levels, it markedly increased protein carbonyl levels. In one study, researchers administered 200 μg/kg thimerosal to spontaneously hypertensive rats (SHR) and Sprague-Dawley (SD) rats during pregnancy between gestation day 10–15 (G10-G15) and lactation (P5-P10). It was determined that prenatal exposure to thimerosal can trigger cerebellar oxidative stress on rats. 47 On the other hand, in the present study, ROS production increased significantly, total GSH levels did not change, TAOC, MDA and protein oxidation levels increased in neuroblastoma cells treated with aluminum. These results indicate that aluminum application significantly increases oxidative stress in SH-SY5Y cell line. In one study, 100–600 µM aluminum maltolate was administered to SH-SY5Y cells for 1, 3, 6 and 24 h. Aluminum caused oxidative stress by increasing ROS production and intracellular calcium levels, along with a decrease in intracellular GSH levels in neuroblastoma cells. The effects of aluminum were dose and time dependent. Although it was shown in the aforementioned study that GSH levels decreased, the increase in GSH levels within the current study may be due to the differences in the application time or a reaction to acute exposure to aluminum in order to protect cellular integrity. Aluminum application may suppress total GSH levels at first; however, when cell defense mechanisms are activated, total GSH levels may increase. The same study concluded that aluminum induces endoplasmic reticulum stress and ROS generation, which compromises the antioxidant defense of neuronal cells, thereby promoting neuronal apoptosis in the p53-independent pathway. 48 Increasing 8-OHdG levels with aluminum application showed that aluminum could cause DNA damage. In a study conducted by Ahmad Rather et al. 49 (2018), researchers stated that the application of 400 µM aluminum maltolate for 24 h may cause DNA damage in neuroblastoma cells.
In our study, the most dramatic changes observed in SH-SY5Y cells were in the combined exposure group. Co-administration of aluminum and thimerosal significantly increased intracellular ROS production. The lesser increase in the AT group compared to the A group may be attributed to the increased cytotoxicity with the co-administration of aluminum and thimerosal as the cell count clearly decreases after exposure to both of the metals. The fact that the total GSH levels did not decrease in the AT group can be interpreted as an increase in the GSH synthesis of the cells due to increased cellular thiol production after co-exposure to both aluminum and Hg. However, it may be possible to reach a definite interpretation by measuring the activity and levels of the GSH synthesis pathway proteins and GSH-related enzymes. In a study by Waly et al. 50 (2016), exposure of cells to 100 nM of aluminum, Hg or thimerosal (ethyl Hg) for 1 h caused a decrease in GSH levels and inhibition of methionine synthase activity by reducing cysteine uptake, which is a rate-limiting step for GSH synthesis.
High intracellular ROS levels may lead to damage to nucleic acids, lipids and proteins and effect different cellular organelles. Oxidative stress can cause apoptosis and may be an underlying factor for aging, diabetes, cancer and neurodegenerative diseases.21–23 Several strategies have been tried against the toxic effects of organic Hg compounds. Raeeszadeh et al. (2021) determined the antioxidant effect of Medicago sativa L. (250, 500 and 750 mg/kg) ethanolic extract against mercury chloride (HgCl2, 2 mg/kg) in rats. Researchers determined increases in lipid peroxidation and decreases in the activities of glutathione peroxidase (GPx), CAT and SOD and levels of TAOC HgCl2. In addition, HgCl2 caused glomerular and tubular damage and hepatic necrosis. Medicago sativa L. (750 mg/kg) reduced the oxidative damage caused by HgCl2 in a dose-dependent manner. 51 Medicago sativa L. extracts were shown to exert protective effects against several other toxic compounds including nicotine in different organs of rats. Therefore, antioxidant and anti-inflammatory herbal extracts can provide significant improvement after inorganic Hg exposure. However, their safety and protective against should also be asses against organic Hg compounds as well.52,53
Nrf2 is an important regulator of cellular resistance to oxidants. Nrf2 controls the basal and stimulated expression of a number of antioxidant response element-dependent genes to regulate the physiological and pathophysiological consequences of oxidant exposure. Nrf2 functions as a xenobiotic-activated receptor (XAR) to regulate the adaptive response towards oxidants and electrophiles. 54 In this study, we observed that thimerosal increased Nrf2 levels by 44% while Nrf2 levels increased by 55% in the aluminum administered group. The reason for these increases may be that the Nrf2 pathway might be triggered in the presence of high oxidative stress. Mustafa Rizvi et al. 48 (2014) found that Nrf2 levels decreased after 24 h with aluminum application to neuroblastoma cells. Researchers have stated that the decrease in Nrf2 levels, which regulates the protective mechanism in cells against oxidative stress by transcription of antioxidant enzymes, may be associated with different neurodegenerative diseases. In our study, Nrf2 levels increased markedly in the AT group. The high levels of oxidation due to combined exposure might have triggered the intracellular protective mechanisms. On the other hand, the increase in lipid peroxidation, protein oxidation and oxidative DNA damage clearly suggests high levels of intracellular oxidative stress after co-exposure (p < 0.05).
Disturbances in the dopaminergic system play an important role in the development of various diseases, including schizophrenia, addiction and Parkinson’s disease.55,56 One of the key proteins involved in regulating dopaminergic tone is DAT. DAT is located on the cell membrane of dopaminergic neurons and functions to rapidly uptake dopamine from the extracellular gap to the presynaptic neuron. DAT does not only control the amplitude and duration of the extracellular dopamine signal but also acts to maintain intracellular dopamine levels. 57 When the changes in neurotransmitters between the groups were evaluated, we observed that both dopamine, DAT and DBH levels increased in the thimerosal group while norepinephrine levels did not change. Since there is no previous study on this subject, it was not possible to compare the data obtained with the literature. After aluminum application, while dopamine and NE levels did not change significantly, DAT levels were increased markedly. On the other hand, DBH levels increased markedly. The increase in DAT levels can lead to loss of dopamine-producing neurons and oxidative stress over time. 55 Co-administration of aluminum and thimerosal increased dopamine levels ∼3.3 times while DAT levels snd DBH also showed significant increment. These changes reflect the negative effects of AT administration, particularly on the dopaminergic system. 58
Conclusion
We have determined that thimerosal, aluminum and their co-exposure may lead to oxidative stress in neurons. Moreover, the alterations in neurotransmitter levels and related proteins clearly suggests thimerosal and/or aluminum leads to neurotoxic effects. The increase in the incidence of different neurological disorders might be due to high exposure to heavy metals, such as Hg and aluminum as well as co-exposure to other environmental neurotoxic substances. Our results clearly indicate both Hg and aluminum exposure may lead to serious consequences, the importance of which should be clarified with mechanistic in vivo toxicological data.
Footnotes
Author contributions
Pınar Erkekoğlu: Writing and reviewing the manuscript, Conceptualization, Supervision
Mehmet Evren Öztürk, Anıl Yirün, Aylin Balcı Özyurt: Experimental analysis, Data curation
Selinay Başak Erdemli Köse and Deniz Arca Çakır: Experimental analysis, Writing- original draft
Didem Oral: Writing- original draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hacettepe University Research Fund. Funding ID: TYL-2020–18729.
