Abstract
Background
Dysfunction of airway epithelial cells in patients with asthma is closely with the occurrence and development of allergic asthma. Finding the differences of airway epithelium between asthmatic patients and normal patients is helpful to find out new treatment strategies.
Methods
First, three original microarray datasets (GSE89809, GSE41861, GSE104468) from the Gene Expression Omnibus (GEO) dataset were used to assess differentially expressed genes in the epithelial tissues between patients with allergic asthma and healthy controls. Then, 10 ng/mL TGF-β1 treated BEAS-2B cells and rats with ovalbumin induced allergic asthma were performed to confirm our assumption from the gene expression analysis with microarrays.
Results
Top ten hub significant difference genes were obtained by Cytohubba plug-in from GSE41861, and found that androgen receptor (AR) was closely associated with the mitogen-activated protein kinase (MAPK) pathway, especially MAPK1 and MAPK14. After treated with the TGF-β1 treated BEAS-2B cells and rats with allergic asthma, we found that 5α-dihydrotestosterone (5α-DHT), AR agonist, significantly decreased the Th2 inflammation (IL-25 and IL-33), MAPK1 and MAPK14 proteins expression in vitro and in vivo. The roles of 5α-DHT were similar with the results of chicanine (a p38 MAPK and ERK1/2 inhibitor), but the roles of 5α-DHT were masked by the C16-PAF (a MAPK and MEK/ERK activator) treatment.
Conclusion
Androgen receptor limits the secretion of Th2 inflammatory factors by downregulating MAPK1 and MAPK14 in the TGF-β1 treated BEAS-2B cells and rats with ovalbumin induced allergic asthma, which plays a critical role for the therapeutics of patients with asthma.
Introduction
Allergic asthma is characterized by helper type 2 (Th2) airway inflammation, eosinophil infiltration, mucus production and airway hyperreactivity (AHR).1,2 Interleukin (IL)-33 signaling through its receptor, ST2, is the initiator of Th2 inflammatory cascade, resulting in increased production of IL-4, IL-5 and IL-13 by CD4+ Th2 cells, group 2 innate lymphocytes (ILC2s), mast cells, eosinophils and other cells.1–5 A large number of studies have shown that dysfunction of airway epithelial cells in patients with asthma is closely with the occurrence and development of asthma.6–8 Airway epithelial cells, the first barrier of the airway, participate in airway inflammation and airway remodeling in asthma by strengthening pro-inflammatory activity and releasing growth factors. Therefore, finding the differences of airway epithelium between asthmatic patients and normal patients is helpful to find out new treatment strategies.
Mitogen-activated protein kinase (MAPK) pathway plays a critical role in the activation of inflammatory cells, which is activated by a three-tiered sequential phosphorylation of MAPK kinase kinase (MKKK), MAPK kinase (MEK), and MAPK. 9 In mammalian cells, there are three distinct MAPK pathways, p38 MAPK (MAPK14), extracellular signal regulated protein kinase (ERK or p42/44 MAPK pathway) and c-Jun NH2-terminal kinase (JNK). 10 The JNK and p38 MAPK are activated by inflammatory and environmental physical insults, whereas the ERK responds to mitogenic and proliferative stimuli. 11 The three protein kinases phosphorylate and activate one another. In asthma, the three kinases are activated and contribute the asthma exacerbation.11–15 In the treatment of asthma, targeting these kinases represents a novel approach.
Androgen receptor (AR), a member of the steroid hormone receptor family, plays important roles in the physiology and pathology of diverse tissues. 16 Recent studies have shown that high AR expression and high androgen levels in the bronchus are associated with better lung function, fewer symptoms and lower fractional exhaled nitric oxide in human asthma, and asthma prevalence is lower in men than in women.17,18 Indeed, the AR-deficient mice increased the severity of antigen-induced airway inflammation and bronchoconstriction. 18 Therefore, the roles and mechanisms of AR should be considered in the asthma management.
Gene expression microarrays have been generally applied in studying gene expression profiles, which offer broad application prospects for drug-based molecular targeting and permit a more profound study of molecular mechanisms. At present, gene expression microarrays are published on Gene Expression Omnibus (GEO). In this study, we downloaded three original microarray datasets (GSE89809, GSE41861, GSE104468) from the GEO dataset to assess the differentially expressed genes (DEGs) of epithelial tissue between patients with allergic asthma and healthy controls. Furthermore, TGF-β1 treated BEAS-2B cells and rats with ovalbumin induced allergic asthma were performed to confirm our assumption from gene expression analysis with microarrays.
Materials and methods
Select gene microarrays
In GEO database (https://www.ncbi.nlm.nih.gov/geo/), three keywords “Allergic asthma and Humo sapiens and Epithelial tissue” were used to select gene expression microarray datasets. Named GSE89809, GSE 41861 and GSE104468 were obtained. In GSE 89809, 110 samples (bronchoalvedar levage and epithelia burshings gene expression) were collected. In GSE 41861, 138 samples were collected. In GSE 104468, 48 samples (nasal epithelia and bronchial epithelia) were collected.
Screening differential genes
GEO2R online analysis software (Adjusted p < 0.05) was used to screen the differential genes of each GEO dataset between asthma group and control group.
Enrichment analysis
In this study, the Adjusted p < 0.05 was set thresholds to screen DEGs. The function and pathway enrichment of proteins encoded were analyzed using online tool Metascape (http://metascape.org). Gene Ontology (GO) function, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway were implemented. 19
Protein-protein interactions network construction and Hub gene selection
The protein-protein interaction (PPI) data were obtained from the screened DEGs by online tool STRING (http://string-db.org). Then the PPI network was visualized using the Cytoscape software (version 3.8.2). The top 10 Hub genes were screened in the PPI network using Cytohubba plug-in unit. 20
Cell culture
Bronchial epithelial cell line (BEAS-2B) was purchased (Article No. MZ-0677, http://www.mingzhoubio.com) and cultured in DMEM + 10% fetal bovine serum (FBS) at 37°C with 5% CO2.
Cell treatment
Experiment 1
The BEAS-2B cells were treated with TGF-β1 (10 ng/mL) for 24 h, then treated with vehicle or different concentrations (0, 0.5, 1 nM) of 5α-dihydrotestosterone (5α-DHT, AR agonist, D-073, Sigma) for 24 h.2,21 The 5α-DHT was dissolved in 0.1% DMSO.
Experiment 2
The BEAS-2B cells were divided into five groups, including control group, TGF-β1 treated group, TGF-β1 plus 5α-DHT treated group, TGF-β1 plus chicanine (a p38 MAPK and ERK1/2 inhibitor, #HY-N2270, MedChemExpresss) treated group, TGF-β1 plus 5α-DHT and C16-PAF (a MAPK and MEK/ERK activator, #HY-108635, MedChemExpresss) treated group. The treated concentration and time of drugs were as follows, 1 nM 5α-DHT for 24 h, 50 μM chicanine for 24 h, 22 10 nM C16-PAF 23 for 24 h. Chicanine and C16-PAF were dissolved in 2 % ethanol.
Cell counting kit-8
The cell viability was analyzed using Cell counting kit-8 (CCK-8) kit. In short, the treated BEAS-2B cells in each group (1 × 105 per well) were cultured at 37°C with 5% CO2. Then, 10 μL CCK-8 solution (KGA317, KeyGen Biotech, Jiangsu, China) was added into each well for 4 h. The absorbance of each well was measured at 450 nm by a microplate reader.
Enzyme-linked immunosorbent assays
Cell supernatants were collected by centrifugation, and measured using IL-25 (#EK0793, Boster, China) and IL-33 (#EK0929, Boster, China) Enzyme-linked immunosorbent assay (ELISA) kits, respectively. The absorbance of each well was measured at 450 nm by a microplate reader.
Animals and animal model
Thirty-six female Wistar rats (weighting 220 ± 30 g) were purchased from Jinan Pengyue experimental animal breeding Co., Ltd. (Number: SCXK (Lu)20190003, Shandong, China). All rats were housed with standard care in the specific pathogen free (SPF) level animal room with free access to food and water under a 12:12-h light-dark cycle.
Referencing to previous reports,24,25 rats were sensitized by ovalbumin (OVA, CAS: 9006-59-1, A800616, MACKLIN, China). Briefly, rats were intraperitoneally injected with 1 mg/kg OVA (dissolved in PBS) plus 100 mg Al(OH)3 (CAS: 21645-51-2, A800852, MACKLIN, China) on day 1, 2 and 3. A continuous 2% OVA aerosol challenge was treated for rats from day 4–21 in a 0.8 m3 chamber, with air flow of 8 lit/min for 20 min/day. In control rats, 0.9% saline was used instead of the OVA solution.
Animal treatment
The rats were randomly divided into six groups: control group, OVA model group (OVA), vehicle treated group (OVA + vehicle), 0.5 mg/kg 5α-DHT treated group (OVA + 5α-DHT), 15 mg/kg chicanine treated group (OVA + chicanine), 5α-DHT and 5 mg/kg C16-PAF treated group (OVA +5α-DHT + C16-PAF). Six rats in each group. The vehicle was 0.1% DMSO. During experiments, rats were orally administrated with vehicle or 5α-DHT or chicanine or 5α-DHT plus C16-PAF every 3 days.
Airway hyperreactivity
Rats were anesthetized with pentobarbital sodium (45 mg/kg), and placed a tracheostomy tube in the trachea of rats. Rats were mechanically ventilated through a Sci-Req FlexiVent machine with 150 breaths/min and a tidal volume of 2 mL. Airway resistance was determined at baseline and after administration of increasing doses (0–100 mg/mL) of nebulized methacholine (CAS: 62-51-1, Macklin, China).
Bronchioalveolar lavage and ELISA
Twenty-four hours after the last challenge, bronchioalveolar lavage (BAL) was performed through trachea. Bronchioalveolar lavage fluid (2 mL) was collected through injection of PBS into the rat lung. Total cells in the BAL fluid (BALF) were counted by trypan blue dye exclusion, and the counts of eosinophils, neutrophils and lymphocytes were analyzed using Wright stain solution (#G1040, Solarbio, China).
The levels of IL-4 (#EK0405, Boster, China), IL-5 (#EK0408, Boster, China), and IL-33 (#EK0930, Boster, China) in the BAL fluids were analyzed by ELISA kits, respectively.
Hematoxylin-eosin staining
Twenty-four hours after the last challenge, all rats were sacrificed using 100 mg/kg pentobarbital sodium. After fixing with 4% paraformaldehyde for 24 h, lung tissues were embedded in paraffin, and cut into slices (3 μm). After dewaxing with xylene, the sections were hydrated with different concentrations of ethanol (100%, 5 min; 95%, 2 min; 80%, 2 min; 70%, 2 min). After that, the sections were stained with hematoxylin-eosin (H&E) staining solution (G1120, Solarbio, China) for 30 min at 55°C. After washing, the slices were separated with 95% ethanol for 1 min, and dehydrated with 100% ethanol for 2 min. After clearing with the xylene, the slices were sealed with neutral gum to observe under an optical microscope.
Periodic acid-Schiff
The lung sections (3 μm) were dewaxed as described in the H&E staining, then washed with water twice. After oxidation in an oxidizer for 6 min, the sections were washed with water and stained with periodic acid-schiff (PAS) solution (G1281, Solarbio, China) for 15 min. After staining with hematoxylin staining for 1 min, the sections were differentiated with acidic differentiation solution for 3 s. After washing for 15 min, the sections were dehydrated, cleared and sealed as described in the H&E staining.
Western blot
Proteins (30 μg) were separated with 12% SDS-PAGE (Bio-Rad, China) and transferred to PVDF membranes. After blocking with 5% milk, the membranes were incubated with appropriate primary antibodies diluted with 5% bovine serum albumin (BSA) for overnight at 4°C. The primary antibodies consisted of MAPK 1 (AMPK1 or ERK1, 1: 2000, ab109282, Abcam), MAPK14 (another name p38α MAPK, 1:2000, ab170099, Abcam), AKT serine/threonine kinase 2 (AKT2, 1: 1000, ab175354, Abcam), mitogen-activated protein kinase kinase 2 (MAP2K2 or MEK2, 1: 10000, ab32517, Abcam) and GAPDH (1:1000, BA2913, Boster, China). After washing with TBS-0.01% Tween 20 for 3 times, the second antibody HRP-Affinipure Goat anti-Rabbit lgG (H+L) (1:500, BM3894, Boster, China) was cultured with the membranes for 2 h at 25°C. After washing, the signals were visualized by enhanced chemiluminescence reagent (D085075, Bio-Rad, China). The intensity of each band was analyzed using Image J software.
Statistics
Data analysis was carried with SPSS 20.0 (National Institutes of Health) software. The analysis results were expressed as mean ± standard deviation, and the differences among groups were analyzed using one-way analysis of variance (ANOVA), followed by Tukey post-test. p< 0.05 was considered significant.
Results
Identification of DEGs
As shown in Figure 1, there was no significant difference between the asthma group and the control group in the GSE89809 (Figure 1(a)) and GSE104468 (Figure 1(b)) through GEO2R online software. In the GSE41861 (Figure 1(c)), a significant difference was found between the two groups (Adjusted p (Padj) < 0.05). Therefore, the GSE41861 was used to conduct subsequent analysis. Volcano plot of the differentially expressed genes (DEGs) in epithelial tissue between the asthma group and control group. (A) GSE89809; (B) GSE104468; (C) GSE41861. Black points represent the Adjusted p (Padj)> 0.05. Blue points represent the Padj< 0.05 and down-regulated genes. Red points represent the Padj< 0.05 and up-regulated genes.
Gene ontology term and KEGG pathway enrichment analysis
The GO term and KEGG pathway enrichment analyses of differentially expressed genes (330 genes) with Padj < 0.05 and absolute value of LogFC (fold change) > 1 were obtained through online tool Metascape, respectively. Please see the Figure 2. The differentially expressed genes of GO term were mainly enriched in endoplasmic reticulum protein-containing complex, protein acetyltransferase complex, protein domain specific binding, MAP kinase kinase activity, intracellular protein transport, regulation of peptidase activity, positive regulation of organelle organiza, immune system development, regulation of cellular localization. In the KEGG pathway, the differentially expressed genes mainly enriched in the salmonella infection and human T-cell leukemia virus 1 infection. GO and KEGG pathway enrichment analysis of GSE41861. (A) GO cellular components; (B) GO molecular functions; (C) GO biological processes; (D) KEGG pathway.
Screening of the key genes
The differentially expressed genes (330 genes) were constructed PPI network through SIRING database and Cytoscape software (Figure 3(a)). The top 10 hub genes were obtained by Cytohubba plug-in (Figure 3(b)). The 10 genes included mitogen-activated protein kinase kinase 2 (MAP2K2), LCK proto-oncogene (LCK), mitogen-activated protein kinase 14 (MAPK14), AKT serine/threonine kinase 2 (AKT2), mitogen-activated protein kinase 1 (MAPK1), AR, X-ray repair cross complementing 5 (XRCC5), mediator of DNA damage checkpoint 1 (MDC1), ERCC excision repair 1 (ERCC1), heterogeneous nuclear ribonucleoprotein M (HNRNPM). These genes were closely associated with the occurrence and development of allergic asthma. The differentially expressed genes (330 genes) in GSE41861 were construct PPI network through SIRING database and Cytoscape software (A). The top 10 hub genes were obtained by Cytohubba plug-in (B). Red, orange and yellow are related, and the degree of correlation decreases in turn.
In the key genes, we found that AR was associated with MAPK1 and MAPK14, suggesting that AR plays an important role in mediating MAPK1 and MAPK14 in epithelial cells of asthma. In allergic asthma, a large number of studies have reported that MAPK1 (ERK1),26,27 MAPK14 (p38 MAPK) 9 participated in the inflammation of epithelial cells. Recently, researchers also have found that AR could suppress allergic airway inflammation and improve lung function.17,18 However, whether AR regulates the MAPK1 and MAPK14 in the epithelial cells of asthma is unclear.
5α-dihydrotestosterone suppresses Th2 inflammation, MAPK1 and MAPK14 in TGF-β1 induced BEAS-2B cells
In this study, the 5α-DHT, AR agonist, was used to analyze the effects of AR on AMPK1 and MAPK14 in TGF-β1 induced BEAS-2B cells. Firstly, the effects of AR on normal BEAS-2B cells were observed, and the results showed that the cell viability and levels of Th2 related factors (IL-25 and IL-33) were not affected (Figure 4(a)). Secondly, the TGF-β1 (10 ng/mL) was used to induce BEAS-2B cells for 24 h, then 5α-DHT (0, 0.5, 1 nM) was treated for 24 h, respectively. The levels of IL-25 and IL-33 significantly upregulated after TGF-β1 stimulation, and treatment 5α-DHT clearly downregulated the levels of IL-25 and IL-33 in a dose-dependent fashion (Figure 4(b)). The high expression of MAPK1 and MAPK14 induced by TGF-β1 was also inhibited by 5α-DHT treatment in a dose-dependent fashion (Figure 4(c)). 5α-DHT suppress Th2 inflammation, MAPK1 and MAPK14 in TGF-β1 induced BEAS-2B cells. (A) The effects of 5α-DHT (0, 0.5, 1 nM) on cell viability and Th2 related factors (IL-25 and IL-33) were observed in normal BEAS-2B cells; (B) The effects of 5α-DHT (0, 0.5, 1 nM) on IL-25 and IL-33 were observed in TGF-β1 induced BEAS-2B cells; (C) The effects of 5α-DHT (0, 0.5, 1 nM) on MAPK1 and MAPK14 proteins expression were observed in TGF-β1 induced BEAS-2B cells. The 5α-DHT was dissolved in 0.1% DMSO. The cell viability was analyzed by CCK-8. The levels of IL-25 and IL-33 were analyzed by ELISA. The expression of MAPK1 and MAPK14 was analyzed by western blot. NS: no significance. Vs. control group, *p < 0.05, **p < 0.01; Vs. 5α-DHT (0 nM) group, ##p < 0.01; Vs. 5α-DHT (0.5 nM) group, &p < 0.01, &&p < 0.01.
5α-dihydrotestosterone regulates MAPK1 and MAPK14 to suppress Th2 inflammation in TGF-β1 induced BEAS-2B cells
In order to confirm AR could regulate MAPK1 and MAPK14 to suppress Th2 inflammation in TGF-β1 induced BEAS-2B cells, the chicanine (a MAPK1 and MAPK14 inhibitor) and C16-PAF (a MAPK1 and MAPK14 activator) were chose to analyze the mechanism of AR. As shown in Figure 5, the levels of IL-25 and IL-33 were significantly decreased by 50 μM chicanine treatment, which were similar with the results of 1 nM 5α-DHT (Figure 5(a)). However, the effects of 1 nM 5α-DHT were attacked by 10 nM C16-PAF. Meanwhile, the high expression of MAPK1 and MAPK14 in TGF-β1 induced cells was declined after 50 μM chicanine treatment, consisting with the treatment of 1 nM 5α-DHT (Figure 5(b)). The effects of 1 nM 5α-DHT on MAPK1 and MAPK14 expression were blocked by 10 nM C16-PAF. 5α-DHT regulates MAPK1 and MAPK14 to suppress Th2 inflammation in TGF-β1 induced BEAS-2B cells. (A) The levels of IL-25 and IL-33 were observed in TGF-β1 induced BEAS-2B cells; (B) The proteins expression of MAPK1 and MAPK14 were observed in TGF-β1 induced BEAS-2B cells. Vs. control group, *p < 0.05, **p < 0.01; Vs. TGF-β1 group, #p < 0.05, ##p < 0.01; Vs. 5α-DHT (1 nM) group, &p < 0.05, &&p < 0.01.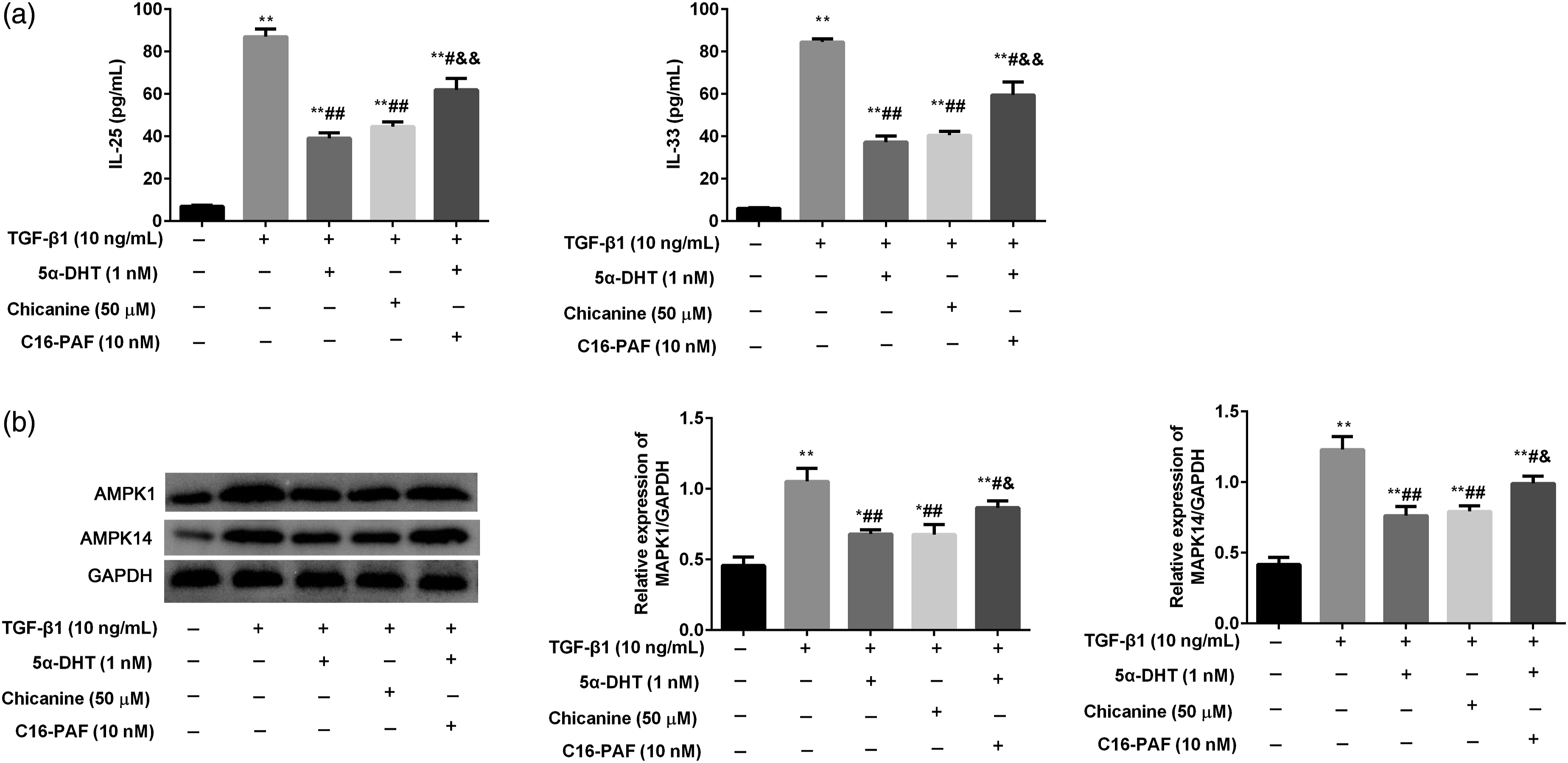
5α-dihydrotestosterone regulates MAPK1 and MAPK14 to suppress Th2 inflammation in OVA induced asthma rats
To further confirm these findings from BEAS-2B cells, OVA induced asthma rats were used to analyze the mechanism of AR in vivo. In OVA induced rats, the AHR was significant higher than the control group (Figure 6(a)). After treatment of 5α-DHT (0.5 mg/kg) or chicanine (15 mg/kg), the AHR was clearly decreased compared with the vehicle (0.1% DMSO) treatment rats (Figure 6(a)). However, the treatment of C16-PAF (5 mg/kg) notably restrained the effects of 5α-DHT (0.5 mg/kg). The numbers of eosinophils, lymphocytes and neutrophils in BALF were counted and found that 5α-DHT (0.5 mg/kg) or chicanine (15 mg/kg) treatment notably decreased the high numbers of eosinophils, lymphocytes and neutrophils induced by OVA stimulation (Figure 6(b)). The C16-PAF (5 mg/kg) treatment masked the roles of 5α-DHT (0.5 mg/kg). Furthermore, the Th2 inflammatory (IL-4, IL-5 and IL-33) in BALF was analyzed and found that 5α-DHT or chicanine treatment notably decreased the high levels of IL-4, IL-5 and IL-33 induced by OVA, and C16-PAF treatment masked the roles of 5α-DHT (Figure 6(c)). 5α-DHT regulates MAPK1 and MAPK14 to suppress Th2 inflammation in OVA induced asthma rats. (A) The airway hyperreactivity (AHR) was determined by airway resistance to increasing concentrations of nebulized methacholine (0–100 mg/mL); (B) The counts of eosinophils, neutrophils and lymphocytes in bronchioalveolar lavage fluid (BALF) were analyzed using Wright stain solution; (C) The levels of IL-4, IL-5 and IL-33 in BALF were analyzed using ELISA; (D) Lung tissues inflammation was observed by H&E staining; (E) Lung tissues global cell metaplasia was observed by PAS staining. After treatment of 5α-DHT (0.5 mg/kg) or chicanine (15 mg/kg), the inflammation was clearly decreased compared with the vehicle (0.1% DMSO) treatment rats. However, the simultaneous treatment of C16-PAF (5 mg/kg) notably restrained the effects of 5α-DHT. Vs. control group, **p < 0.01; Vs. OVA group, #p < 0.05, ##p < 0.01; Vs. OVA + 5α-DHT group, &p < 0.05, &&p < 0.01.
Through observation of pathological change in lung tissue, the 5α-DHT or chicanine treatment reduced inflammatory infiltration, but C16-PAF treatment aggravated the inflammatory infiltration through inhibiting the effects of 5α-DHT (Figure 6(d)). Meanwhile, the numbers of goblet cell metaplasia were significantly increased after OVA stimulation, and 5α-DHT or chicanine treatment decreased goblet cell metaplasia (Figure 6(e)). The C16-PAF administration clearly inhibited the effects of 5α-DHT (Figure 6(d, e)).
5α-dihydrotestosterone regulates MAPK pathway in OVA induced asthma rats
To observe the effects of 5α-DHT on MAPK pathway in OVA induced asthma rats, the MAPK pathway related proteins finding in this study, MAPK1, MAPK14, AKT2 and MAP2K2, were analyzed in lung tissues by western blot (Figure 7). The results showed that OVA stimulation activated these proteins expression, and 5α-DHT (0.5 mg/kg) or chicanine (15 mg/kg) treatment notably downregulated these proteins expression. No significant difference was found in the simultaneous treatment of 5α-DHT (0.5 mg/kg) and C16-PAF (5 mg/kg) when compared with the OVA group. 5α-DHT regulates MAPK pathway in OVA induced asthma rats. (A) The proteins of MAPK1, MAPK14, AKT2 and MAP2K2 were analyzed by western blot; (B) The relative expression of MAPK1 was quantified, and GAPDH was internal reference; (C) The relative expression of MAPK14 was quantified, and GAPDH was internal reference; (D) The relative expression of AKT2 was quantified, and GAPDH was internal reference; (E) The relative expression of MAP2K2 was quantified, and GAPDH was internal reference. The results showed that OVA stimulation activated these proteins expression, and 5α-DHT (0.5 mg/kg) or chicanine (15 mg/kg) treatment notably downregulated these proteins expression. However, the simultaneous treatment of C16-PAF (5 mg/kg) notably restrained the effects of 5α-DHT. Vs. control group, **p < 0.01; Vs. OVA group, #p < 0.05, ##p < 0.01; Vs. OVA + 5α-DHT group, &p < 0.05, &&p < 0.01.
Discussion
Asthma is a heterogeneous disease, and allergic asthma is one of the important phenotypes, accounting for more than 50% of adult asthma and more than 80% of childhood asthma in China. 28 As the first barrier of the airway, airway epithelial cells participate in airway inflammation and airway remodeling in the allergic asthma. 29 In this study, we obtained top 10 hub significant difference genes from GSE41861, and found that AR was closely associated with MAPK pathway, especially MAPK1 and MAPK14, in airway epithelial cells of allergic asthma.
A sex bias exists in asthma already found that females have increased asthma prevalence compared with males after puberty, which coincides with increases in sex hormone levels during puberty.2,17 AR attenuates Th2 allergic airway inflammation to decrease asthma symptoms and improve lung function in males and females with asthma, 2 suggesting that AR might to be a potential therapeutic for allergic asthma. In our study, we used different dose of 5α-DHT, AR agonist, to observe its effects on TGF-β1 induced bronchial epithelial cells (BEAS-2B), and also found that 5α-DHT significantly decreased the Th2 inflammation (IL-25 and IL-33). Furthermore, we found that 5α-DHT decreased the MAPK1 and MAPK14 proteins expression, indicating that AR regulates MAPK1 and MAPK14 to suppress the Th2 inflammation in airway epithelial cells of allergic asthma.
As known, the MAPK pathway plays a critical role in the activation of inflammatory cells, which promotes the secretion of inflammatory proteins. 10 In allergic asthma, MAPK pathway promotes the inflammatory response of various cell types in lung and bronchus, including alveolar macrophages, lung lymphocytes, airway smooth muscle cells and bronchial epithelial cells.30–32 For the MAPK1 (ERK1) and MAPK14 (p38 MAPK), many studies have reported that they participate in the Th2 inflammation of epithelial cells in asthma.9,26,27,30 Targeting MAPK pathway is a therapeutic schedule for asthma treatment. In this study, we used chicanine (a p38 MAPK and ERK1/2 inhibitor) and C16-PAF (a MAPK and MEK/ERK activator) to confirm our hypothesis that AR inhibits Th2 inflammation through regulating MAPK1 and MAPK14 in airway epithelial cells of allergic asthma. The results showed that the roles of 5α-DHT were similar with the results of chicanine that Th2 inflammation, MAPK1 and MAP14 were suppressed in TGF-β treated BEAS-2B cells and rats with allergic asthma. However, the roles of 5α-DHT were masked by C16-PAF treatment.
Our results showed that AR limited secretion of Th2 inflammatory factors in airway epithelial cells of allergic asthma by downregulating MAPK1 and MAPK14, as a mechanism to decrease airway inflammation associated with allergic asthma. These findings expand our understanding of how AR decreased Th2 allergic airway inflammation, and are critical for the therapeutics of patients with asthma.
Conclusions
In this study, the original microarray dataset (GSE41861) from GEO datasets was used to assess the differentially expressed genes of epithelial tissue between patients with allergic asthma and healthy controls. We found that AR was closely associated with MAPK pathway, especially MAPK1 and MAPK14. In TGF-β treated BEAS-2B cells and OVA induced rats, we further confirmed that AR limited secretion of Th2 inflammatory factors by downregulating MAPK1 and MAPK14, playing a critical role for the therapeutics of patients with asthma.
Footnotes
Authors’ contributions
TX, JM, and YS designed the studies, performed experiments, analyzed data and wrote the manuscript. YS provided expertise, reviewed and reivised the manuscript. All authors read and agreed with the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The human data described in this manuscript, publicly available data from GEO database, has been reviewed by the Ethics Committee of Yantai Affiliated Hospital of Binzhou Medical College, and the ethics committee have waived the need for ethical approval. In this study, the animal experiments were approved by the Ethics Committee of Yantai Affiliated Hospital of Binzhou Medical College (Approval No. BMC-H-20210018).
