Abstract
Increasing evidence supports the role of arsenic in dysregulated immune and inflammation responses, while, safe and effective treatments have not been fully examined. Rosa roxburghii Tratt (RRT), a traditional Chinese edible fruit with potential immunoregulatory activities, was considered as a dietary supplement to explore its protective effects and possible mechanism in arsenic-induced dysregulated inflammation responses. We enrolled 209 arsenicosis patients and 41 controls to obtain baseline data, including the degree of arsenic poisoning prior to the RRT juice (RRTJ) intervention. Then, based on criteria of inclusion and exclusion and the principle of voluntary participation, 106 arsenicosis patients who volunteered to receive treatment were divided into RRTJ (n = 53) and placebo (n = 53) groups randomly. After three months follow-up, 89 subjects (46 and 43 of the RRTJ and placebo groups, respectively) completed the study and were examined for the effects and possible mechanisms of RRTJ on the Th17 cells-related pro-inflammatory responses in peripheral blood mononuclear cells (PBMCs). The PBMCs had higher levels of Th17 and Th17-related inflammatory cytokines IL-17, IL-6, and RORγt. Furthermore, the gene expressions of STAT3 and SOCS3 in PBMCs increased and decreased, respectively. Conversely, RRTJ decreased the number of Th17 cells, secretion of IL-17, IL-6, RORγt, and relative mRNA levels of STAT3, and increased the transcript levels of SOCS3. This study provides limited evidence that possible immunomodulatory effects of RRTJ on the critical regulators, IL-6 and STAT3, of the Th17 cells in arsenicosis patients, which indicated that IL-6/STAT3 pathway might appear as a potential therapeutic target in arsenicosis.
Introduction
Exposure to arsenic is a major environmental and public health challenge for hundreds of millions of people worldwide. 1 Epidemiological studies have proved that long-term exposure to arsenic via contaminated drinking water, diet, or air can cause various adverse health effects in humans (respiratory, digestive, urinary, cardiovascular, and immune systems).2–6 Recent studies found that arsenic exposure can cause inflammatory damage to the body’s immune system,7–9 and T helper 17 (Th17) cells, a unique CD4+ T-cell subset characterized by the production of interleukin-17 (IL-17).10,11 IL-17 can effectively mediate neutrophil mobilization and pro-inflammatory responses that ultimately lead to inflammatory cell infiltration and tissue damage. 12 Retinoid-related orphan receptor-γt (RORγt) is the most specific transcription factor promoting Th17 cells, the gene expression profiles of Th17 cells indicate a high expression of RORc gene, which encodes RORγt in Th17 cells. Retroviral vector mediated transduction of RORγt into naive T cells promoted the development of Th17 cells, suggesting a clear association between RORγt and Th17 cells. 13 Patients with arsenic poisoning [0.23(0.10–0.57) μg/g hair] have higher levels of Th17 cells, protein level of IL-17A, and transcript level of RORγt, 14 however, the exact mechanism remains unclear. Current studies tend to assume that inflammation leads to injury through the interleukin-6 (IL-6)/signal transducer and activator of the transcription 3 (STAT3) signaling pathway, which is considered a major target for the treatment of inflammatory diseases.15–17 The suppressor of cytokine signaling 3 (SOCS3) is an inhibitory factor of the IL-6/STAT3 signaling pathway. IL-6 can induce STAT3 phosphorylation (p-STAT3), and p-STAT3 activates SOCS3, which in turn inhibits the activation of STAT3, thus negatively regulating the IL-6/STAT3 signal. 18 Nishihara M et al. reported a novel role of the IL-6/STAT3 signaling pathway in the Th17 response, where IL-6 acts directly to promote the development of Th17 by activating the T cell gp130-STAT3 pathway. 19 In addition, Li Y et al. revealed that the inhibition of IL-6/STAT3 pathway by triptolide could significantly reduce the production of IL-17. 20 To date, the underlying mechanisms of IL-6/STAT3 signaling in the Th17 response and IL-17 production on arsenicosis patients have not been elaborated. Study found that SOCS3 has a significant role in constraining the generation of Th17 cells, 21 a study demonstrated that an enhanced stimulation of macrophages and promotion of Th17 cell differentiation by down-regulation of the SOCS3-mediated IL-6/STAT3 pathway feedback inhibition. 22 Those studies suggest that SOCS3 might inhibit the Th17 cells-related pro-inflammatory responses via IL-6/STAT3 signaling. However, whether IL-6/STAT3 signaling is involved in Th17-induced pro-inflammatory responses in patients with arsenic poisoning remains unknown.
At present, there is still no effective treatment for arseniasis in biological systems, which should be of concern to the research of arsenic-induced diseases and their effective and safe treatment. Plants are used to treat diseases since ancient times. 23 Modulation of inflammation with the use of edible and medicinal plants poses an alternative to conventional therapeutic strategies for numerous ailments, including arseniasis. 24 RRT, the fruit of R. roxburghii Tratt.f. normalis Rehd. Et Wils., is a promising wild fruit crop growing in Southwest China and particularly the Guizhou Province, 25 and it is recognized for its nutritional and potential immunoregulatory activities; it has long been considered as a healthy edible fruit, and it is related to improved function of the immune system, reduced blood lipid and glucose levels, and improved antioxidant capacity.26–31 RRTJ retains the original nutrients and bioactive properties of RRT and can be used as a dietary supplement. 32 Some studies have found that RRTJ could effectively improve the immune function of rats-exposed to arsenic (RRTJ was used in 10 mL/kg for 45 d after cessation of arsenic exposure) and arseniasis patients (patients received 120 mL/d RRTJ orally for one month),33,34 however, the mechanism has not been extensively investigated.
Although Th17 cells play a pivotal role in immune diseases, there is no data on RRTJ as a protective factor for Th17-related pro-inflammatory responses. Thus, we recruited a population exposed to arsenic via coal-burning to observe whether arsenic could increase the number of Th17 cells via activating IL-6/STAT3 signaling and explored the protective effects and possible mechanism of RRTJ in arsenic-induced pro-inflammatory responses. We determined the numbers of Th17 cells in PBMCs, the Th17-related levels of transcription factors and cytokines (protein levels of IL-17, IL-6, and the mRNA levels of RORγt, STAT3, and SOCS3). In addition, we examined the regulatory effects of RRTJ on the Th17 response. In present study, we aimed to explore the effects of RRTJ in the modulation of Th17 response in arsenic-exposed individuals.
Methods
Selection of population
The subjects were selected from Yuzhang Town, a coal-burning region in China that suffers from arsenic poisoning, and is located in Xingren County of Qianxinan Prefecture, Guizhou Province, Southwest China. To obtain baseline data, including the degree of arsenic poisoning in the population prior to the RRTJ intervention, 209 arsenicosis patients were enrolled and classified by trained biomedical personnel into mild (palmo plantar with skin keratosis of grade I or trunk with skin pigmentation of grade I and depigmentation of grade I simultaneously), intermediate (palmo plantar with skin keratosis or trunk with skin pigmentation of grade II or depigmentation of grade II), and severe (palmo plantar with skin keratosis or trunk with skin pigmentation of grade III or depigmentation of grade III)groups based on the different degree of arsenicosis skin damage symptoms using the Diagnosis of Endemic Arsenicosis WS/T 211-2015. 35 In addition, 41 participants from an arsenic-free area who shared a similar lifestyle, eating habits, nutritional status, and economic status were recruited as controls. The inclusion criteria included (1) participants have a history of arsenic exposure in Yuzhang Town, a typical coal-burning arsenic poisoning region in China; (2) aged 18–65 years; and (3) with hyperkeratosis, hyperpigmentation or depigmentation, and skin ulceration. Also, the exclusion criteria were defined as the following: (1) history of occupational arsenic poisoning; (2) long-term suffering from immunodeficiency or autoimmune-related diseases; (3) a recent history of infection, consuming drugs or intaking seafood that can affect immune function or the arsenic metabolism; (4) exposure to X-rays or other radioactive materials for a long time; and (5) unwillingness to join the study. Study materials and procedures were reviewed and approved by the Ethical Committee of Guizhou Medical University for international health research (No. 201403001), and written informed consent was obtained from all individuals.
Study design
The present study was designed as a double-blind, randomized, placebo-controlled trial to assess the potential value of RRTJ on arsenic-induced inflammatory processes. We enrolled 209 arsenicosis patients and 41 controls to obtain baseline data, including the degree of arsenic poisoning prior to the RRTJ intervention. Then, according to the criteria of inclusion and exclusion and the principle of voluntary participation, 106 arsenicosis patients were divided into different block groups according to age (an interval every 5 years) and gender; the same block group was distributed to the RRTJ (53) and placebo (53) groups randomly and were subjected to three months of oral administration of RRTJ (20 mL per day according to the recommended dosage in the health food instructions; RRTJ was purchased from Sinopharm Group Guizhou Healthcare Industry Development Co., Ltd; the health food permission number of National Health Commission of the People’s Republic of China is [2002]0004) or placebo tablets (20 mL per day, no RRTJ active ingredients, color and taste are consistent with those of RRTJ). Both participants and investigators were blind to the treatment conditions until the study was finished. At baseline and at the final follow-up, physical examinations were performed by medical professionals who worked in the former 44th Hospital of the Chinese People’s Liberation Army. The grouping and study-flow diagram are shown in Figure 1. Grouping and study-flow diagram. Grouping proceeded according to the Standard of Diagnosis for Endemic Arsenism (WS/T 211-2015, Ministry of Health of the People’s Republic of China), 41 normal controls and 209 arsenicosis patients were recruited to study the effects and possible mechanisms of RRTJ on the Th17 cells-related pro-inflammatory responses in PBMCs. According to the criteria of inclusion and exclusion and the principle of voluntary participation, arsenicosis patients were divided into a placebo (n = 53) and RRTJ (n = 53) groups randomly. After three months, 17 arsenicosis patients did not fully participate in the intervention, while the remaining subjects in the RRTJ (n = 46) and placebo (n = 43) groups were examined for the effects and possible mechanisms of RRTJ on the Th17 cells-related pro-inflammatory responses in PBMCs.
Sample collection
The hair of the subjects was collected within 3 cm of the hair root and stored in plastic zipper bags. Fasting venous blood samples were collected into tubes containing sodium heparin and non-anticoagulant tubes at the baseline and after RRTJ intervention. Serum was obtained after centrifugation at 400 × g for 20 min, and the blood samples were stored at −80°C until analysis. The PBMCs were isolated from heparinized blood by Ficoll density gradients 36 for flow cytometric analysis and extraction of total RNA.
Determination of arsenic levels in hair
The concentration of arsenic in the hair samples was tested as previously described. 37 Briefly, the hair samples were washed with deionized water, soaked in acetone, dehydrated with ether, dried in an oven at 60 °C, and cut into pieces. Before the determination, hair samples were digested with 6 mL of concentrated nitric acid (HNO3) using a microwave digestion instrument (Anton Paar, Multiwave GO, Sweden) for 1 h. Inductively coupled plasma mass spectrometry (Thermo Fisher, XSeries 2, USA) was used to analyze the total arsenic content in the hair samples. 38
Cell isolation and flow cytometric analysis
Cell isolation and flow cytometric analysis were performed as described in the literature. 14 Briefly, PBMCs were isolated from the fresh heparinized blood by Ficoll-Hypaque (Tian Jin Hao Yang Biological Manufacture Co., Tianjin, China) gradient centrifugation at 800 × g for 20 min, and distributed in 24-well plates at a density of 2 × 106 cells/mL coated with 4 μL Leukocyte Activation Cocktail with BD Golgi Plug™ (BD Biosciences, San Jose, CA, USA) at 37 °C with 5% CO2 for 5 h. Then, the cells were suspended in a FACS solution and incubated with cell surface antibodies FITC-labelled antihuman CD4 (BD Biosciences, USA) at 4 °C for 30 min in the dark. Fixation & permeabilization solution (BD Biosciences, USA) was used to permeabilize the membrane of the cell cytoplasm and nucleus at 4°C for 50 min for the intracellular staining, and the cells were incubated with cell intracellular and nuclear antibodies PE-cyanine7-labelled antihuman IL-17A (BD Biosciences, USA) at 4 °C for 40–50 min in the dark. The isotype controls were treated with the same procedures and then washed with a FACS solution. A stopping gate of 10,000 CD4+ events was set and the stained cells were analyzed with a FACS CaliburTM Flow Cytometer (BD Biosciences, San Jose, CA, USA). Data analysis was performed using the Cell Quest Pro software (BD Biosciences).
Measurement of serum cytokines via ELISA
The levels of IL-17, and IL-6 in the serum were measured following the manufacturer’s instructions. The Human IL-17A Platinum ELISA Kit (catalog number: BMS 2017) was purchased from eBioscience (San Diego, CA) and BD OptEIA™ Human IL-6 ELISA Kit II (catalog number: 550799) was purchased from BD Biosciences (BD Biosciences, USA). The concentrations were calculated from the absorbance, which was recorded at 450 nm in a microplate reader (Thermo Fisher). All samples were tested in parallel.
Real-time PCR analysis
Primer sequences for real-time PCR.

Statistical analysis
SPSS 22.0 software (SPSS, Inc., Chicago, IL, USA) was used for statistical analysis. The graphs were plotted using the GraphPad Prism software, version 8.3 (GraphPad Software). Prescriptive data are presented as mean ± standard deviations (SD). A chi-squared test, paired t-test and one-way analysis of variance (ANOVA) were used for comparison between the before and after variables and within groups, respectively. The Kruskal-Wallis H and Wilcoxon Signed-rank tests were used when data were presented as median (interquartile range). Spearman’s rank correlation coefficients analysis was applied. Differences were considered statistically significant at p < 0.05.
Results
Demographic data of the study population
Characteristics of the study population in baseline.

Arsenic induced pro-inflammation responses in peripheral blood of patients with arsenic poisoning
Arsenic disrupted the quantity of Th17 cells in PBMCs
The percentages of Th17 cells in the PBMCs were detected based on their specific surface markers. There were differences in terms of Th17/PBMCs and Th17/CD4+T among control and arsenicosis groups (F = 3.973, 33.902, p = 0.009, p < 0.001, respectively). Compared with control group (0.28 ± 0.16 % and 1.03 ± 0.44 %), the frequencies of Th17 cells/PBMCs and Th17 cells/CD4+T cells increased by 0.16 % and 0.17 %, and by 1.06 %, and 1.12 % (p < 0.05) in the PBMCs of intermediate and severe arsenicosis patients, respectively. There were no significant differences in terms of Th17/PBMCs between control and mild group (p > 0.05) (Figure 2). These results suggested that sustained arsenic exposure promoted differentiation and significantly disturbed the expression of Th17 cells. Higher percentages of Th17 cells in peripheral blood caused by arsenic. To evaluate the effect of arsenic exposure on the immune response, we assessed the frequencies of Th17 cells in the PBMCs of arsenicosis patients using a FACS CaliburTM Flow Cytometer. Arsenic increases the level of Th17 in both PBMCs and CD4+ T cells, and triggers a response of Th17-related immunological dysregulation. PBMCs were obtained from control subjects and arsenicosis patients. (a, b and c) Representative flow cytometry (FCM) pictures of CD4+IL-17A+ Th17 cells in the PBMCs and in CD4+T cells of the subjects, FITC-labelled antihuman CD4, PE-cyanine7-labelled antihuman IL17A. A stopping gate of 10,000 CD4+ events was set and the stained cells were analyzed with a FACSCaliburTM Flow Cytometer. (d and e) The percentages of Th17 cells in PBMCs and in CD4+T cells were measured via FCM according to their specific surface markers. The Th17 cells were defined as CD4+IL-17+T cells, FITC-labelled antihuman CD4, PE-cyanine7-labelled antihuman IL17A. Data analysis was performed using the Cell Quest Pro software (BD Biosciences). Results are given as mean ± SD. *p< 0.05 compared with that in the control; #p<0.05 compared with that in the mild group.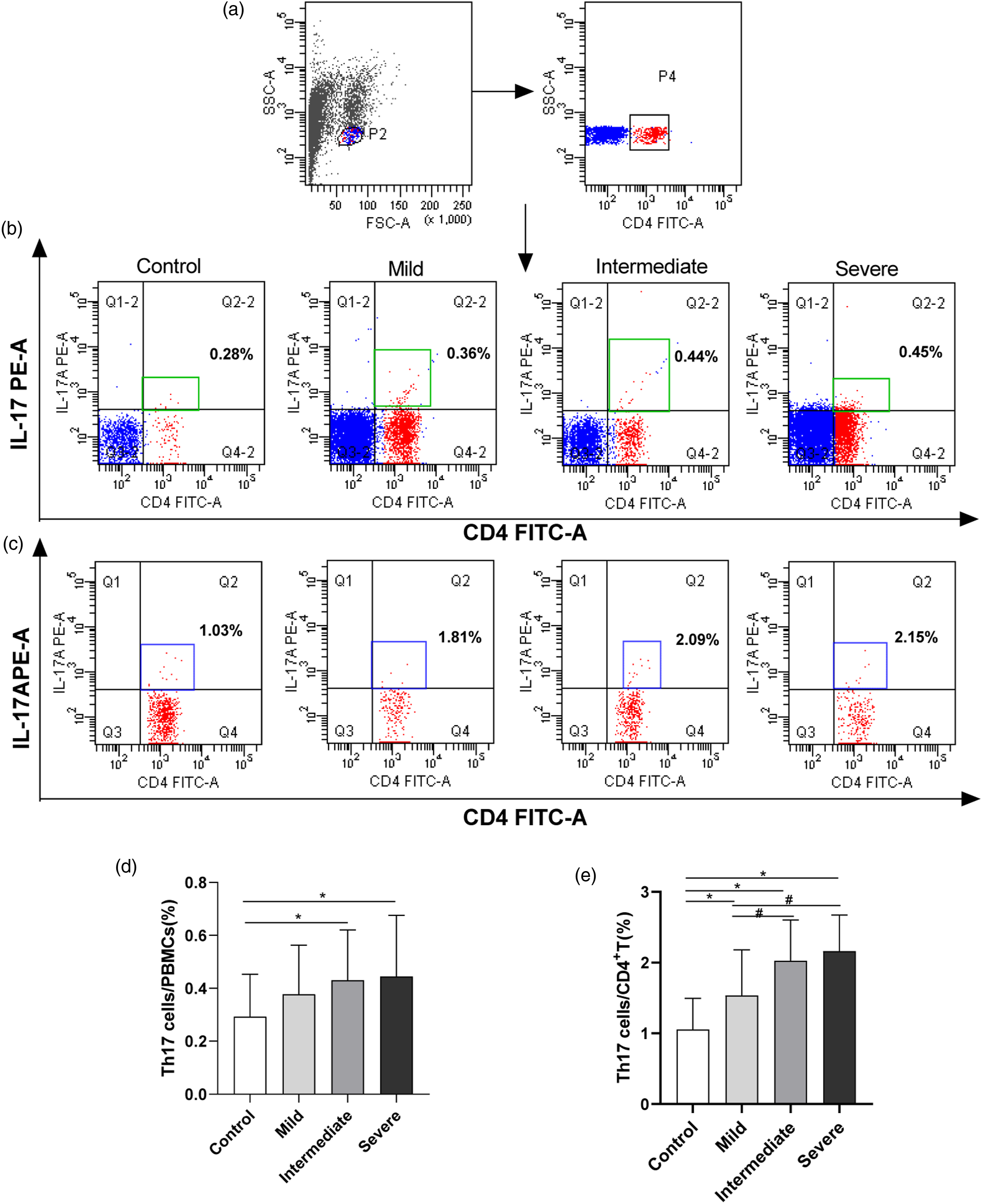
Arsenic increases the levels of IL-17 and IL-6 in serum
To determine the inflammatory response in the PBMCs, the levels of pro-inflammatory related cytokines IL-17 and IL-6 in the serum were determined using ELISA kits according to the manufacturer’s instructions. As presented in Figure 3, there were differences in the levels IL-17 and IL-6 among control and arsenicosis groups (F = 27.946 and 43.972, respectively; p < 0.001). Both the levels IL-17 and IL-6 in the arsenicosis groups [mild(11.86 ± 1.92 pg/mL and 9.95 ± 1.55 pg/mL), intermediate (12.87 ± 1.70 pg/mL and 10.45 ± 1.38 pg/mL), and severe (13.50 ± 1.72 pg/mL and 11.34 ± 1.77 pg/mL), respectively] were increased by 1.88 pg/mL and 2.62 pg/mL, 2.89 pg/mL and 3.12 pg/mL, 3.52 pg/mL and 4.01 pg/mL than in the control group (9.98 ± 1.66 pg/mL and 7.33 ± 1.52 pg/mL, p < 0.05). Furthermore, the levels of IL-6 and IL-17 in the severe group was higher than that in the mild group and were increased by 1.39 pg/mL and 1.64 pg/mL, respectively (p < 0.05). The level of IL-17 in the intermediate group was higher than in the mild group and was increased by 1.01 pg/mL (p < 0.05). These results revealed that higher levels of IL-17 and IL-6 in the serum might be involved in inflammatory responses in arsenicosis patients. Arsenic exposure leads to higher levels of IL-6 and IL-17 in serum. Arsenic exposure can increase the secretion of serum IL-6 and IL-17, which might be involved in inflammatory responses in arsenicosis patients. The levels of IL-6 (a) and IL-17 (b) in serum were measured using ELISA. Results are given as mean ± SD. *p < 0.05 compared with that in the control group; #p< 0.05 compared with that in the mild group.
Arsenic exposure might activate the IL-6/STAT3 pathway in PBMCs
To explore the possible mechanisms underlying Th17-related pro-inflammatory responses in the PBMCs of arsenicosis patients, we measured the mRNA expression of RORγt, STAT3, and SOCS3 in PBMCs. As shown in Figure 4, there were differences in the levels of RORγt, STAT3 and SOCS3 among control and arsenicosis groups (F = 4.548, 20.301, and 12.112, respectively; P all<0.05). RT-PCR showed higher levels of RORγt and STAT3 but lower levels of SOCS3 transcripts in PBMCs in intermediate [2.31(0.42∼3.38), 2.41(1.61∼4.11) and 0.57(0.14∼0.94) fold changes, respectively] and severe [2.61(1.46∼3.81), 3.15(2.17∼4.76) and 0.50(0.18∼0.79) fold changes, respectively] group than that in the control group [1.08(0.62∼1.38), 1.00(0.76∼1.50) and 0.88 (0.49∼1.40) fold changes, respectively, p < 0.05]. There were no significant differences of the level of RORγt transcripts between mild group [1.85(0.02∼2.76) fold changes] and control group (p > 0.05). Combined with the abnormal inflammatory cytokines IL-6 and IL-17, these data suggested that arsenic exposure might increase the levels of IL-6 and STAT3, which might be involved in Th17-related pro-inflammatory responses in the PBMCs of arsenicosis patients. The current study revealed that the levels of Th17 cells, as well as the concentration of Th17 cell-related cytokines IL-17 and IL-6 in the PBMCs, increased significantly. To further explore the role of the IL-6/STAT3 signaling-related transcription factors in Th17-induced pro-inflammatory responses, we used Spearman’s correlation analysis to assay the hair arsenic, IL-6/STAT3 signaling-related transcription factors and pro-inflammatory changes. As shown in Table 3, we evaluated the correlations between hair arsenic and Th17/PBMCs (%), Th17/CD4+T (%), IL-17 and IL-6 in arsenicosis patients. The hair arsenic positively correlated with that Th17/PBMCs (%), Th17/CD4+T (%) as well as pro-inflammatory related cytokines IL-17 and IL-6 (rs = 0.225, 0.483, 0.439, and 0.450, respectively; P all<0.05). Moreover, We examined the correlations between STAT3/SOCS3 and Th17/PBMCs (%), Th17/CD4+T (%), IL-17 and IL-6 in arsenicosis patients, the gene level of STAT3 correlated positively (rs = 0.337, 0.305, and 0.370, respectively; P all<0.05) but the transcript level of SOCS3 negatively with Th17/CD4+T (%), IL-17 and IL-6 (rs = −0.274, −0.253, and −0.220, respectively; P all < 0.05); this indicated that IL-6/STAT3 signaling may be associated with Th17-related pro-inflammatory responses in the PBMCs of arsenicosis patients. Arsenic exposure increases the mRNA levels of RORγt and STAT3, but decreases those of SOCS3 in PBMCs. To explore the possible mechanisms underlying Th17-related pro-inflammatory responses in the PBMCs of arsenicosis patients, we measured the mRNA expressions of RORγt, STAT3, and SOCS3 in the PBMCs. RT-qPCR showed higher levels of RORγt and STAT3 but lower levels of SOCS3 transcripts in PBMCs based on the degree of arsenicosis. The mRNA levels of RORγt (a), STAT3 (b) and SOCS3 (c) were determined via qRT-PCR. Results are presented as median (interquartile range); M: median; P25, P75: interquartile range. *p < 0.05 compared with that in the control group; #p< 0.05 compared with that in the mild group. A one-way ANOVA test was performed, with the F-value as the statistical value. Relationship betweenTh17-induced pro-inflammatory responses and IL-6/STAT3 signaling in arsenicosis patients.

RRTJ ameliorated dysregulated immune and inflammation responses
RRTJ decreased the arsenic concentration in hair of arsenicosis patients
Hair arsenic in arsenicosis patients treated with RRTJ or placebo, M (P25∼P75).

RRTJ treatment restored the quantity of Th17 cells
To investigate the possible effects of RRTJ on the number of Th17 cells, PBMCs were isolated, and flow cytometric analysis was performed to determine the quantity of the Th17 cells. The number of both Th17/PBMCs and Th17/CD4+T cells decreased after treatment with RRTJ (0.32 ± 0.17 % and 1.53 ± 0.36 %) compared with that before (0.42 ± 0.20 % and 2.04 ± 0.62 %, t = 2.718, and 5.097, respectively, p < 0.05). There were no significant differences in terms of Th17/PBMCs and Th17/CD4+T before (0.40 ± 0.22 % and 2.04 ± 0.56 %, respectively) and after the placebo treatment (0.41 ± 0.19 % and 1.97 ± 0.42 %, t = −0.302, and 1.037; p = 0.975, and 0.306, respectively). The results suggested that RRTJ could lighten the pro-inflammatory responses caused by imbalanced differentiation of Th17 cells in the PBMCs of arsenicosis patients (Figure 5). Effect of RRTJ on the number of Th17 cells in peripheral blood. PBMCs were obtained as described in the Methods section. The arsenicosis patients were treated with RRTJ (n = 46) or placebo (n = 43) for three months. (a, b and c) Representative flow cytometry (FCM) pictures of CD4+IL-17A+ Th17 cells in the PBMCs and in CD4+T cellsof the subjects, FITC-labelled antihuman CD4, PE-cyanine7-labelled antihuman IL17A. A stopping gate of 10,000 CD4+ events was set and the stained cells were analyzed with a FACSCaliburTM Flow Cytometer. (d and e) The percentage of Th17 cells in PBMCs and in CD4+T cells were measured via FCM according to their specific surface markersin both groups. The Th17 cells were defined as CD4+IL-17+T cells, FITC-labelled antihuman CD4, PE-cyanine7-labelled antihuman IL17A. Data analysis was performed using the Cell Quest Pro software (BD Biosciences). Results are presented as mean ±SD. A paired t-test was performed, with the t-value as the statistical value. **p<0.05 compared with that before treatment.
RRTJ decreases the levels of IL-17 and IL-6 in serum
To observe the role of RRTJ on the arsenic-induced pro-inflammatory response, we further focused on the concentration levels of the signature cytokines IL-17 and IL-6 of Th17 cells in the serum of arsenicosis patients who were treated with RRTJ or placebo. As shown in Figure 6, the levels of IL-17 and IL-6 decreased after treatment with RRTJ (10.63 ± 1.98 pg/mL and 8.86 ± 1.81 pg/mL) compared with that before (12.68 ± 1.80 pg/mL and 10.54 ± 1.75 pg/mL), their levels decreased by 2.05 pg/mL, and 1.68 pg/mL, respectively (t = 7.257, and 4.867, p < 0.001). However, there were no significant differences in the serum IL-17 and IL-6 before (12.68 ± 1.95 pg/mL and 11.03 ± 1.76 pg/mL, respectively) and after the placebotreatment (12.57 ± 2.08 pg/mL and 10.35 ± 1.85 pg/mL, respectively, t = 0.272, and 1.853; p > 0.05). These data indicated that RRTJ intervention suppressed the pro-inflammatory response by decreasing the secretion of pro-inflammatory cytokines corresponding to Th17 cells in the serum. RRTJ can decrease the expression of IL-6 and IL-17 in the serum of arsenicosis patients. RRTJ intervention can downregulate the expression level of IL-6 and IL-17 in the serum of arsenicosis patients, modulating the response of arsenic-induced immunological dysfunction. The serum levels of IL-6 (a) and IL-17 (b) in two groups were measured using ELISA. Results are given as mean ± SD. **p<0.05 compared with that before treatment. A paired t-test was performed, and the statistical value was the t-value.
RRTJ-mediated protective effects may be involved in IL-6/STAT3 signaling
To explore whether the IL-6/STAT3 signaling was involved in the protective effects of RRTJ, we performed RT-PCR to detect the mRNA levels of RORγt, STAT3, and SOCS3 in arsenicosis patients who were treated with RRTJ or placebo. As shown in Figure 7, compared to self control before intervention, the expression of RORγt mRNA decreased by 1.42-fold changes after RRTJ-treatment compared to that before the RRTJ-treatment (T = −3.203, p = 0.001). The expression pattern of STAT3 mRNA was similar, the expression level of STAT3 mRNA decreased by 0.87-fold changes after treatment with RRTJ (T = −2.623, p = 0.009). Conversely, the expression of SOCS3 mRNA increased by 0.27-fold changes in the RRTJ-treated group compared to that before the RRTJ-treatment (T = −2.373, p = 0.018). Furthermore, there were no significant differences in the mRNA expression of RORγt, STAT3 and SOCS3 in the PBMCs of the groups before and after the placebo treatment (T = −0.000, −0.305, and 0.000; p = 1.000, 0.760, and 1.000). These findings suggested that, in the PBMCs of arsenicosis patients, RRTJ could attenuate the Th17-related pro-inflammatory response, which could be associated with lower expression of RORγt and STAT3 but higher expression of SOCS3. The results indicated that RRTJ-mediated protective effects may be involved in the IL-6/STAT3 signaling. Changes in mRNA levels of RORγt, STAT3, and SOCS3 in arsenicosis patients treated with RRTJ or placebo. The mRNA levels of RORγt (a), STAT3 (b) and SOCS3 (c) were determined via qRT-PCR. Results are presented as median (interquartile range); M: median; P25, P75: interquartile range. **p< 0.05 compared with that before treatment. A wilcoxon Signed-rank test was performed, and the statistical value was the T-value.
Discussion
Arsenicosis has emerged as a global public health concern. As a well-known environmental toxicant and a potent carcinogen, arsenic has been epidemiologically proved related to the manifestation of multisystem toxicity.2–6 Studies have suggested that arsenic could cause abnormalities in inflammatory mechanisms and the immune system which negatively impact the body.8,40,41 Many studies have indicated that arsenic could decrease the levels of CD3+, CD4+, and the CD4+/CD8+ ratio in humans and rats, as well as disrupt the balance of Th17 and Treg cells in PBMCs of rats.33,42,43 However, the specific mechanisms of arsenic-induced immune system damage have not yet been fully elucidated.
Currently, there are no effective treatments of arseniasis in biological systems; treatment methods for arsenic-poisoning that are highly effective, lack side effects, and have low cost, should be developed. Some medicinal and edible plants were considered to attenuate the imbalance of pro-and anti-inflammatory processes. The modulation of inflammation using natural bioactive compounds poses an alternative to conventional therapeutic strategies for numerous diseases, including arseniasis. 24 RRTJ is a fruit crop rich in nutrients and contains bioactive molecules, including ascorbate, polysaccharides, polyphenols, and flavonoids,44–48 also, it can be used as a dietary supplement. RRTJ can inhibit oxidative modification of low-density lipoprotein and macrophage growth and cellular cholesteryl ester accumulation in vitro. 29 Furthermore, RRTJ could effectively improve the immune function of arsenicosis patients by increasing the levels of CD3+, CD4+, and the CD4+/CD8+ ratio in serum. After RRTJ intervention, the levels of CD3+, CD4+, and the CD4+/CD8+ratio were increased by 6.58%, 5.11%, and 0.17%, respectively (all p < 0.05) than those in before intervention. 33 However, the specific mechanisms of RRTJ protection against immune system damage in arsenicosis patients remains unknown. As a traditional Chinese medicine and edible fruit and as a dietary supplement with potential immunoregulatory activities, 49 RRTJ was considered to be a healthy edible fruit to explore its protective effects and potential mechanism in arsenic-induced dysregulated inflammation response.
Metals or metalloid elements can be detected in hair samples that have been exposed to metalloid elements over the past 2∼18 months. 50 Thus, hair arsenic has been used as a biomarker for the total arsenic burden. 51 Present data showed that the content of arsenic in the hair of arsenicosis patients was remarkably higher than that of the control group (Table2), which indicated that hair arsenic might be a biomarker of arsenicosis. RRTJ could decrease the accumulation of arsenic in the liver and hair of rats exposed to arsenic, 31 while arsenicosis patients had lower levels of arsenic in hair after treatment with RRTJ (Table 4); however, the specific mechanisms need to be fully elucidated.
Increasing evidence from animal and human studies supports the role of arsenic exposure in dysregulated immune and inflammation responses.8,40,41 The differentiation pathways and functional characteristics of T-cell subsets have been widely researched by immunologists. Th17 cells, a unique CD4+T-cell subset characterized by the production of IL-17. 10 Over expression of pro-inflammatory mediators of IL-6 was observed in individuals exposed to arsenic. 52 IL-6 is mainly produced in macrophages and lymphocytes, and it is a pleiotropic cytokine. 53 IL-6 is a key cytokine that initiates Th17 cell differentiation. 54 More importantly, IL-6 mediates chronic inflammation and generates a favorable cellular microenvironment for tumor growth. 55 RORγt is the most specific transcription factor promoting the production of Th17 cells. Current data suggested that the number of Th17 cells was higher in both PBMCs and CD4+T cells (Figure 2). Also, the expression of pro-inflammatory related cytokines IL-6 and IL-17 (Figures 3(a) and (b)) and Th17 cell-related transcription factor RORγt (Figure 4(a)) increased in the arsenic-poisoned population. This suggested that arsenic exposure can induce dysregulated immune and inflammation responses related to higher numbers of Th17 cells in PBMCs.
Dysregulation of immune and inflammation responses, which is a common mechanism of disease occurrence, was involved in arsenic-induced skin, 56 liver 43 and kidney 42 damage; RRTJ can improve immune function in arsenic-poisoned rats and patients.33,34 Therefore, it is necessary to study the role and possible mechanism of RRTJ in arsenic-induced Th17-related pro-inflammatory responses. Current data showed that RRTJ could decrease the number of Th17 cells in both PBMCs and CD4+T cells (Figure 5), as well as the expression of pro-inflammatory related cytokines IL-17 and IL-6 (Figure 6) and Th17 cells-related transcription factor RORγt (Figure 7(a)). Thus, RRTJ could decrease the higher concentration of Th17 cells and reduce the transcript level of RORγt, which is the specific transcription factor promoting Th17 cells, in the PBMCs of arsenicosis patients. Also, RRTJ could lower the levels of the inflammatory cytokines IL-17, which can effectively mediate pro-inflammatory responses that ultimately lead to inflammatory cell infiltration and tissue damage. 12 To our knowledge, RRTJ might be contributed to attenuate inflammation response related to higher levels of Th17 cells and their related cytokines caused by arsenic exposure on the body.
The serum levels of IL-6 were elevated in arsenicosis subjects. Typically, IL-6 binds to the IL-6 receptor subunit α IL-6R and the co-receptor gp130 to activate STAT3,
57
which consequently activates inflammatory cascades and oncogenic pathways.
58
STAT3 is a critical regulator of inflammation and the immune microenvironment, and its activation modulates the growth stimulation crosstalk of immune cells.
59
Nishihara M et al. showed a novel role of the IL-6/STAT3 signaling pathway in Th17 reaction and IL-17 production.
19
IL-6 is a key factor inducing Th17 cell differentiation by activating STAT3 and increasing the activation of RORγt.
54
Conversely, SOCS3 plays an important role in inhibiting the Th17 response.
21
As shown in present study data, qRT-PCR showed that the levels of STAT3 and SOCS3 transcription mRNA in the PBMCs were upregulated and downregulated with the degree of arsenicosis (Figures 4(b) and (c)). Combined with the higher expression of Th17 cells (Figure 2), RORγt and STAT3 (Figures 4(a) and (b)), lower level of SOCS3 (Figure 4(c)) and the abnormal levels of inflammatory cytokines IL-6 and IL-17 (Figure 3). Which suggested that arsenic exposure may induce inflammation processes related to higher Th17 cells in PBMCs. To our knowledge, the current data indicated that arsenic exposure might increase the levels of IL-6 and STAT3, implying that IL-6/STAT3 pathway may play an important role in the development of Th17-related pro-inflammatory response in the PBMCs of arsenicosis patients. As shown in Figures 7(b) and (c), after RRTJ treatment, the expression of SOCS3 increased while that of STAT3 was downregulated, which suggested that RRTJ-mediated protective effects may be involved in decreasing the levels of IL-6 and STAT3, increasing the level of SOCS3. Current study indicated that the self-healing ability (placebo treatment) for arsenic-induced Th17 cell-related pro-inflammatory response in the PBMCs of arsenicosis patients was poor (Figures 5, 6 and 7). RRTJ could attenuate Th17 cell-related inflammation response by decreasing the levels of IL-6 and STAT3, increasing the level of SOCS3 in peripheral blood of patients with arsenic poisoning (Figure 8); this indicates the application potential of RRTJ as a functional food or immunomodulator. Schematic diagram summarizing the possible mechanisms via which RRTJ protects against Th17-induced pro-inflammatory processes in the peripheral blood of patients with arsenic poisoning. Arsenic increases the level of Th17 cells, transcription factor RORγt, and Th17-related cytokines IL-17 and IL-6. RRTJ attenuates Th17-induced pro-inflammatory processes in the peripheral blood of patients with arsenic poisoning, which indicates that RRTJ decreased the stimulation of promotion of Th17-cell differentiation via up-regulation of the SOCS3-mediated IL-6/STAT3 pathway feedback inhibition.
Nonetheless, in our present study, we only detected transcriptional expression levels of STAT3, the STAT3 activity (the p-STAT3 level and nuclear translocation of STAT3) were not evaluated so that we could not obtain evidence about IL-6/STAT-3 pathway activation in PBMCs of arsenicosis individuals and its downregulation after RRTJ-treatment. Limited by our descriptive data, we aim to conduct more specific mechanism studies to elucidate the immune-protective effects and mechanism of RRTJ in vivo and in vitro in our follow-up study.
Conclusions
In summary, present study indicates that higher levels of Th17 cells as well as pro-inflammatory cytokines IL-17 and IL-6 in the peripheral blood of patients with arsenic poisoning are involved in the inflammation responses of arsenicosis. Possible immunomodulatory effects of RRTJ on the critical regulators, such as IL-6 and STAT3, of the Th17 cells in arsenicosis patients, which indicated that IL-6/STAT3 pathway might appear as a potential therapeutic target in arsenicosis.
Supplemental Material
Supplemental Material - Potential value and mechanism of Rosa roxburghii tratt juice on pro-inflammatory responses in peripheral blood of patients with arsenic poisoning
Supplemental Material for Potential value and mechanism of Rosa roxburghii tratt juice on pro-inflammatory responses in peripheral blood of patients with arsenic poisoning by Ling Dong, Shiqing Xia, Baofei Sun, Lu Ma, Xiong Chen, Shaofeng Wei, Zhonglan Zou and Aihua Zhang inHuman & Experimental Toxicology
Footnotes
Acknowledgements
We are very grateful to the medical professionals, who worked in the former 44th hospital of the Chinese People’s Liberation Army, for their invaluable medical guidance and assistance in the epidemiological investigation, physical examination, diagnosis and RRTJ intervention.
Author Contributions
Ling Dong: Methodology, Data Curation, Writing-Original Draft, Writing- Review & Editing. Shiqing Xia: Investigation, Methodology, Sample Determination, Data Curation, Writing-Original Draft, Writing-Review & Editing. Baofei Sun, Lu Ma, Xiong Chen, Shaofeng Wei and Zhonglan Zou: Investigation, Resources. Aihua Zhang: Conceptualization, Investigation, Resources, Methodology, Review & Editing, Validation, Supervision, Project administration, Funding acquisition. The manuscript was critically reviewed and approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Natural Science Foundations of China (81430077 and U1812403).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
