Abstract
Agmatine is an arginine metabolite that has neuroprotective capacity. Recently, it has been found to ameliorate atherosclerosis progression in rabbits. However, further molecular mechanisms of its anti-atherosclerotic properties remain unclear. High plasma levels of free fatty acids (FFAs) are an important risk factor for atherosclerosis due to their detrimental effects on vascular endothelial cells (ECs). Here, we used palmitate (PA), a kind of FFA, to induce endothelial dysfunction in human microvascular endothelial cells (HMECs) to determine the possible biological functions of agmatine. We found that PA caused ECs dysfunction in HMEC-1 cells, decreased cell viability, and elevated lactate dehydrogenase (LDH) release which could be reversed by agmatine treatment. Agmatine also improved the nitric oxide (NO) production and endothelial nitric oxide synthase (eNOS) activity in PA-induced HMEC-1 cells. The PA-caused mitochondrial dysfunction of HMEC-1 cells was diminished after agmatine treatment, as proven by the increased intracellular Adenosine Triphosphate (ATP) level, decreased mitochondrial reactive oxygen species (ROS) level, and increased mitochondrial oxygen consumption rate (OCR). Further, agmatine could alleviate PA-caused lipid accumulation with increased levels of Triglyceride (TG) and total cholesterol (TC) in HMEC-1 cells. Furthermore, Western blot analysis revealed that agmatine administration markedly decreased the expression levels of phosphorylated-AMP-activated protein kinase α (p-AMPKα), p-protein kinase B (p-AKT), and p-eNOS in PA-induced HMEC-1 cells. Inhibition of AMPK by compound C reversed the protective effects of agmatine on PA-induced HMEC-1 cells. Taken together, we hypothesize that agmatine mitigated PA-induced HMEC-1 cell dysfunction by alleviating mitochondrial and metabolic dysfunction via the AMPK/PI3K/Akt/eNOS signaling pathway.
Introduction
Free fatty acids (FFAs) are substrates that are physiologically formed by lipolysis. In obese patients, plasma concentrations of FFAs are increased, which may contribute to various metabolic disorders such as hepatic steatosis, type 2 diabetes mellitus (T2DM), and particularly diverse cardiovascular diseases (CVDs).1–3 The correlations between increased concentrations of FFAs in plasma and dysfunction of the cardiovascular system including stroke, coronary artery disease, and atherosclerosis have been reported.4–6
Vascular endothelium is an important structure that forms a functional boundary between the blood and nearby tissues. 7 Endothelial cells (ECs) are crucial components of vascular endothelium and regulate its normal function. 8 Growing evidence implies that elevated FFA levels mediate the onset of ECs dysfunction, due to impaired nitric oxide (NO) production, deregulated insulin signaling, and activated inflammation, renin-angiotensin system, and ECs apoptosis.9,10 These detrimental events result in an imbalanced ratio of vasodilation to vasoconstriction, disturbed regulation of hemostasis/thrombosis, angiogenesis, and fibrinolysis, leading to the pathogenesis of atherosclerotic progression.11,12 Therefore, targeting FFAs-mediated ECs dysfunction may serve as a preventive approach to the development of atherosclerosis.
Agmatine is an endogenous polyamine generated from arginine decarboxylase. It has been investigated as a neurotransmitter in the brain with various properties including anti-depressant, anti-anxiety, anti-convulsant, and anti-nociceptive activity.
13
In addition, several lines of evidence have demonstrated the role of several arginine metabolites in CVDs, such as hypertension and atherosclerosis, by reversing endothelial dysfunction.
14
However, the physiological roles of agmatine in CVDs are still under investigation. A recent study reported that agmatine exerts an inhibitory effect on atherosclerosis and liver steatosis in western diet-fed apoE-knockout mice through modulating fatty acid metabolism.
15
El-Awady et al.
16
reported that agmatine ameliorates atherosclerosis progression in rabbits with hypercholesterolemia, possibly by alleviating endothelial dysfunction. Further molecular mechanisms underlying its anti-atherosclerotic properties remain unclear. This study aimed to determine the possible protective effects of agmatine on cultured ECs in response to palmitate (PA), a kind of FFA, as well as to identify the dysregulated pathways that contribute to its biological functions
Materials and methods
The human microvascular endothelial cells (HMECs) culture
The HMEC-1 cells (ATCC) were cultured in the endothelial cell growth media (Sigma-Aldrich, USA), supplemented with endothelial cell growth supplement (Sigma-Aldrich, USA) and 5% fetal bovine serum (FBS) (Gibco, Grand Island, NY). The HMEC-1 cells were cultured under normal conditions, 37°C, and 5% CO2, and treated with 500 μM palmitate (PA) with or without the presence of agmatine (100 and 200 μM). To block the activation of AMPK, 10 μM compound C was added to the culture medium.
Cell viability assay
The HMEC-1 cell viability was analyzed using the cell counting kit-8 (CCK-8) assay (Dojindo Kumamoto, Japan). Briefly, the HMEC-1 cells were cultured with 10 μL of CCK-8 solution for 2 h. After that, the absorbance was read at 450 nm with a microplate reader (Tecan, Durham, NC).
Lactate dehydrogenase release assay
The LDH levels in HMEC-1 cells culture supernatant were determined using an LDH cytotoxicity detection kit (Roche, Basel, Switzerland). According to the instructions provided by the manufacturer, absorbance was measured at 490 nm using a microplate reader (Tecan).
Nitric oxide availability
The HMEC-1 cells were incubated with 5 μmol/L 4, 5-diaminofluorescein diacetate (DAF-2DA) (Abcam, USA), which is a fluorescent probe for detecting NO. After 20 min incubation, HMEC-1 cells were washed and observed under a laser confocal microscopy (Olympus, Japan) at the emission wavelength of 488 nm. The cell lysates of HMEC-1 cells were prepared for the determination of endothelial nitric oxide synthase (eNOS) activity with the colorimetric method using an eNOS activity assay kit (Sigma-Aldrich, USA).
Measurement of cellular Adenosine Triphosphate levels
Intracellular ATP levels in HMEC-1 cells were measured utilizing an ATP bioluminescence assay kit (Sigma-Aldrich, USA). Finally, the ATP concentration was measured referring to an ATP standard curve and then normalized to the cell number.
Mitochondrial reactive oxygen species detection
Mitochondrial ROS levels in HMEC-1 cells were determined using a MitoSOX-red staining assay. Briefly, after staining with 5 μM MitoSOX reagent for 10 min, HMEC-1 cells were visualized using a fluorescence microscope (ZEISS, Germany).
Mitochondrial oxygen consumption rate
Mitochondrial OCR of HMEC-1 cells was measured with a bioenergetic assay using the Seahorse Bioscience XFe24 Analyzer (Seahorse Bioscience, Billerica, MA), as described previously. 17 The HMEC-1 cells were placed in a 24-well plate containing Seahorse XF Assay medium and subjected to baseline measurements, followed by respective injections with 2 µmol/L oligomycin, 1 µmol/L carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP), and 5 µmol/L Antimycin. The OCR values were calculated and expressed as pmol/min/µg protein.
Assays of the triglyceride and total cholesterol contents
After exposure to the indicated agents, the HMEC-1 cells were collected for sonicated homogenization. The TG and TC contents in the prepared homogenate were directly measured using commercial kits (Jiancheng Bio., Nanjing, China).
Western blot
HMEC-1 cells were homogenized in ice-cold lysis buffer and centrifuged. After determination of protein concentration with Pierce bicinchoninic acid (BCA) protein assay (Thermo Fisher Scientific, USA), equal amounts of total proteins were resolved using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS‐PAGE). Afterward, the electrophoretic transfer of proteins onto polyvinylidene fluoride (PVDF) membranes was performed, followed by probing with antibodies against endothelin 1 (ET-1), endothelin A receptor (ET-A), p-AMPKα, AMPKα, p-Akt, Akt, p-eNOS, and eNOS (Thermo Fisher Scientific, USA) and secondary antibody conjugated with horseradish peroxidase (HRP) (Thermo Fisher Scientific, USA). Targeted bands were detected using an enhanced chemiluminescence detection system (Thermo Fisher Scientific, USA) and analyzed with Image J software.
Statistical methods
All experiments were repeated 3 times. Results were expressed as mean ± standard deviation (SD). All data were processed by SPSS software (version 13.0, SPSS Inc., Chicago, IL). A one‐way analysis of variance (ANOVA) with Bonferroni’s post-hoc test was applied for multiple comparisons. p < .05 was considered statistically significant.
Results
Agmatine improves PA-induced cytotoxicity in HMEC-1 cells
The viability of HMEC-1 cells viability was diminished in response to PA (500 μM) stimulation but could be recovered after treatment with 100 or 200 μM agmatine (Figure 1(A)). PA stimulation also caused an increased LDH release in HMEC-1 cells, whereas treatment with 100 or 200 μM agmatine alleviated the PA-induced LDH release (Figure 1(B)). Agmatine mitigates PA-induced cytotoxicity in HMEC-1 cells. (A) Cell viability; (B) LDH release (†††, p < .0001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5–6).
Agmatine mitigates PA-induced reduction in NO availability in HMEC-1 cells
HMEC-1 cells treated with PA (500 μM) exhibited reduced NO production. Intervention with 100 or 200 μM agmatine significantly elevated the ability of PA (500 μM)-induced HMEC-1 cells to produce NO (Figure 2(A)). Consistent with the trend in NO production, the eNOS activity in PA (500 μM)-induced HMEC-1 cells was significantly decreased, then reversed by 100 or 200 μM agmatine (Figure 2(B)). Agmatine alleviates PA-induced reduction in NO availability in HMEC-1 cells. (A) NO; (B) eNOS activity (†††, p < .0001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5).
Agmatine elevates PA-induced reduction in the expressions of ET-1 and ET-A in HMEC-1 cells
As shown in Figure 3, decreased expressions of ET-1 and ET-A at the protein level were observed in PA (500 μM)-induced HMEC-1 cells. However, treatment with 100 or 200 μM agmatine elevated the reduced ET-1 and ET-A expressions in HMEC-1 cells against PA (500 μM) stimulation. Agmatine attenuates PA-induced reduction in the expressions of ET-1 and ET-A in HMEC-1 cells. Western blot for expression of ET-1 and ET-A (†††, p < .0001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5).
Agmatine alleviates PA-induced lipid metabolism deregulation in HMEC-1 cells
In Figure 4, we found that the application of PA (500 μM) induced the accumulation of TC and TG in HMEC-1 cells. The PA (500 μM)-caused changes in TC and TG content were reversed by 100 or 200 μM agmatine pretreatment (Figure 4). Agmatine ameliorates PA-induced lipid metabolism deregulation in HMEC-1 cells. (A) Total TC; (B) Total TG (††, p < .001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5–6).
Agmatine reduces PA-induced mitochondrial dysfunction in HMEC-1 cells
The intracellular ATP levels in PA-induced HMEC-1 cells were dramatically reduced compared to the control HMEC-1 cells, which was found to be alleviated by 100 or 200 μM agmatine pretreatment (Figure 5(A)). The PA-induced increase in the mitochondrial ROS level was significantly inhibited by 100 or 200 μM agmatine (Figure 5(B)). We found a greatly decreased OCR in PA-induced HMEC-1 cells, whereas agmatine prevented the PA-induced change in OCR (Figure 5(C)). Agmatine mitigates PA-induced mitochondrial dysfunction in HMEC-1 cells. (A) ATP level; (B) Mitochondrial ROS level; (C) Mitochondrial oxygen consumption rate (OCR) (†††, p < .0001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5).
Agmatine upregulates the AMPK/PI3K/Akt/eNOS signaling pathway in PA-induced HMEC-1 cells
As investigated using Western blot, after PA (500 μM) induction, the ratios of p-AMPKα/AMPKα, p-Akt/Akt, as well p-eNOS/eNOS were reduced in HMEC-1 cells. However, the suppressed activation of the AMPK/PI3K/Akt/eNOS signaling pathway was evoked by 100 or 200 μM agmatine (Figure 6). Agmatine upregulated the AMPK/PI3K/Akt/eNOS signaling pathway in PA-challenged HMEC-1 cells. Western blot for (A) p-AMPKα/AMPKα; (B) p-Akt/Akt; (C) p-eNOS/eNOS (†††, p < .0001 vs. vehicle group; ‡, ‡‡, p < .01, 0.001 vs. PA group, N = 5).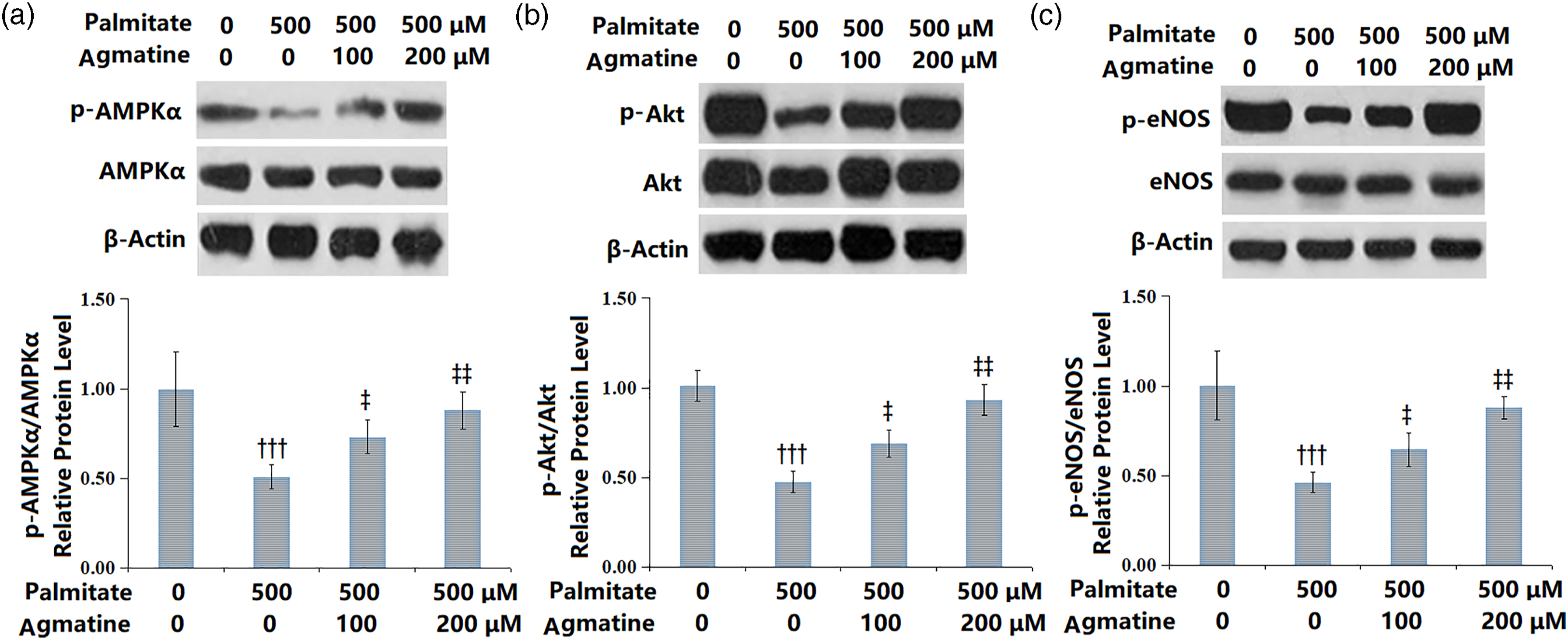
Inhibition of AMPK reverses the functional roles of agmatine in PA-induced HMEC-1 cells
As proved by Western blot, the expression levels of p-AMPKα, p-Akt, and p-eNOS in HMEC-1 cells were markedly decreased after treatment with 10 μM compound C, implying that compound C effectively suppressed the activation of AMPK/PI3K/Akt/eNOS (Figures 7(A)–(C)). The agmatine (200 μM)-caused elevated NO level and eNOS activity in PA-induced HMEC-1 cells were blocked by compound C (Figures 7(D)–(E)). The reduction of TC and TG accumulation in agmatine (200 μM)-treated HMEC-1 cells were diminished after treatment with compound C (Figures 7(F)–(G)). In addition, compound C also caused a significant decrease in the ATP levels in agmatine (200 μM)-treated HMEC-1 cells (Figure 7(H)). The effects of compound C alone on AMPK inhibition and metabolic function on HMEC-1 cells are shown in Supplementary Figure 1. As expected, the presence of compound C reduced the levels of NO, eNOS activity, and ATP, but increased the levels of TC and TG (Supplementary Figure 1). Inhibition of AMPK reversed the protective effects of Agmatine on PA-challenged HMEC-1 cells. Western blot for (A) p-AMPKα/AMPKα; (B) p-Akt/Akt; (C) p-eNOS/eNOS; (D) NO; (E) eNOS activity; (F) TC; (G) TG; (H) ATP level (††,†††, p < .001, 0.0001 vs. vehicle group; ‡‡, p < .001 vs. PA group; ※, p < .001 vs. PA+ Agmatine group, N = 5).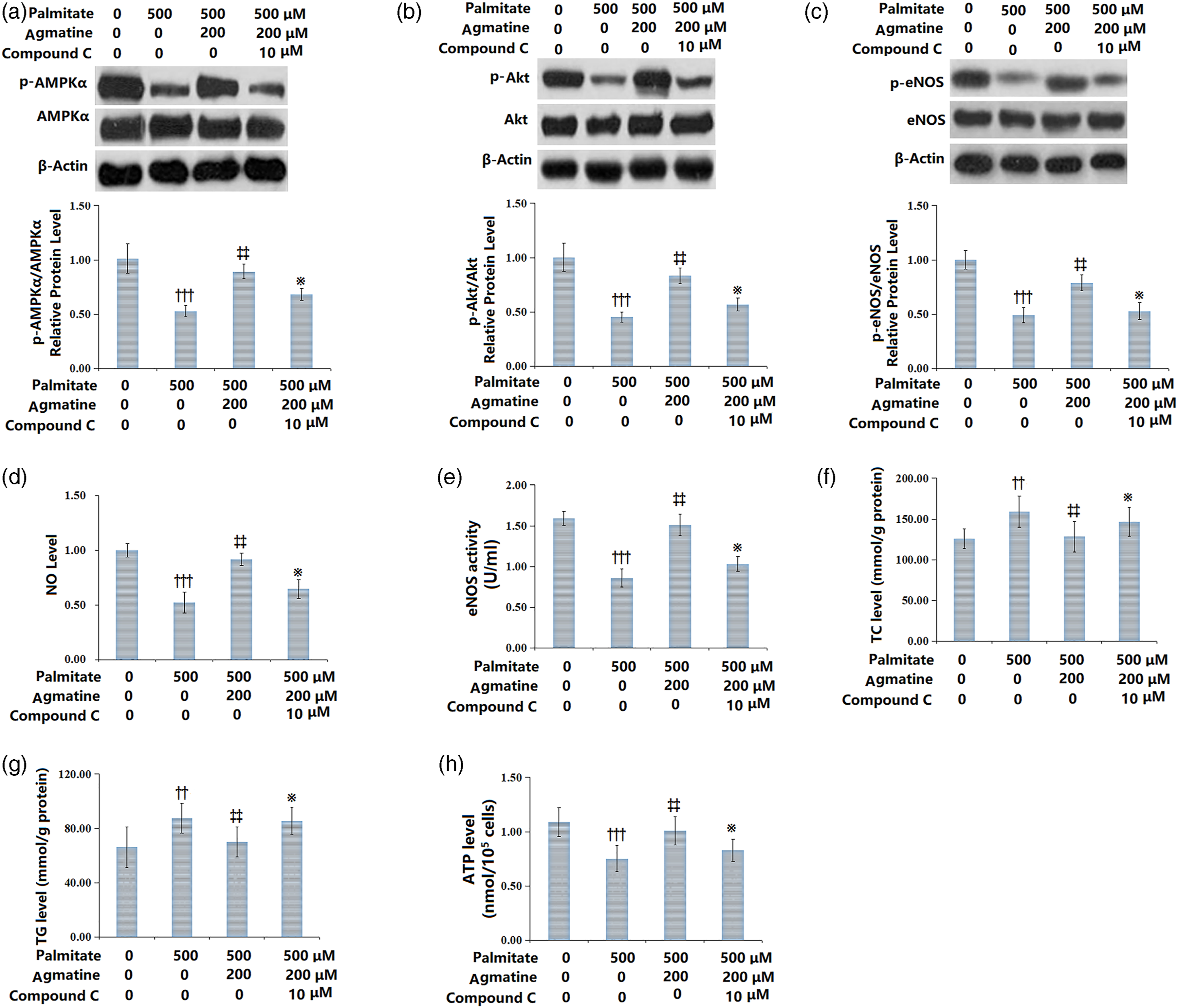
Discussion
Mitochondria are highly dynamic cytoplasmic organelles observed in almost all human cells. 18 Mitochondria play central roles in ATP synthesis, cellular metabolism, calcium handling, generation and control of ROS, regulation of programmed cell death, and biosynthetic processes. 19 Mitochondrial dysfunction is pathologically manifested as insufficient energy supply, elevated production of ROS, mitochondrial dynamics dysfunction, and mitochondrial oxidative stress damage. Although FFAs possess different physiological roles, such as their contribution to the membrane lipids in structure and their importance in acting as fuel molecules, they are also considered potent sources of ROS, and their abnormalities in metabolism may lead to ROS-mediated oxidative stress. 9 In fact, FFAs are found to easily attack mitochondria. In this study, the PA-caused mitochondrial dysfunction of HMEC-1 cells was diminished after agmatine treatment, as proven by the increased intracellular ATP level, decreased mitochondrial ROS level, and increased OCR. Moreover, we found that the PA-caused lipid accumulation with increased levels of TG and TC in HMEC-1 cells could be alleviated by agmatine, which might further mitigate mitochondrial and cellular damage.
Previous studies indicate that mitochondrial dysfunction of ECs is involved in the pathogenesis and progression of atherosclerosis.20,21 That is because vascular ECs are responsible for maintaining vascular homeostasis through regulating vasodilation, smooth muscle cell function, leukocyte adhesion, and platelet activation. It has been documented that ECs dysfunction is characterized by reduced NO synthesis and secretion, thereby contributing to the initiation of atherosclerosis.9,22 In ECs, NO is synthesized by eNOS through the conversion of L-arginine into L-citrulline. Thus, the changes in NO production are commonly accompanied by impaired eNOS activity. 23 ET-1 is a potent endogenous vasoconstrictor that has been implicated in a wide variety of events through two types of receptors, ET-A and ET-B. 24 Dysregulation of ET-1 and its receptors has been implicated in ECs dysfunction. Here, we found that PA caused ECs dysfunction in HMEC-1 cells with decreased cell viability, elevated LDH release, decreased NO production and eNOS activity, as well as decreased expression levels of ET-1 and ET-A, which could be reversed by agmatine treatment. The protective effect of agmatine on ECs dysfunction might be attributed to its beneficial effects on mitochondria and lipid metabolism.
AMPK is a sensor of cellular energy status and sustains intracellular ATP levels in response to cellular stress. Emerging research has shown the crosstalk between AMPK and mitochondria. AMPK is crucial for mitochondrial homeostasis, and in turn, mitochondrial dysfunction may cause the abnormal activation of AMPK. 25 Accumulating studies have demonstrated that AMPK plays pivotal roles in regulating cardiovascular processes and becomes an inhibitor of CVDs progression. Multiple pharmacological reagents used for CVDs have appeared to exert their therapeutic effects partially through the modulation of AMPK signaling. 26 To deliver its protective/therapeutic effects, AMPK is in charge of numerous signaling pathways. In cultured ECs, activation of AMPK is able to stimulate the phosphorylation of eNOS at Ser1177 and Ser633 and stimulate the NO production. 27 Further studies have shown that the regulatory effect of AMPK on eNOS phosphorylation is mediated by the PI3K-Akt pathway. 28 A previous study reported that high-fat diet treatment in rats induces endothelial dysfunction, which is linked to a deregulated AMPK/PI3K/Akt/eNOS pathway. 29 As expected, our results also show that PA induced inactivation of AMPK/PI3K/Akt/eNOS in HMEC-1 cells, which was evoked by agmatine. Moreover, inhibition of AMPK by compound C reversed the functions of agmatine on PA-induced HMEC-1 cells, illustrating the critical role of AMPK/PI3K/Akt/eNOS in the beneficial effects of agmatine.
This study discussed the biological functions of agmatine on PA-induced HMEC-1 cell dysfunction, which might be attributed to its beneficial effects on mitochondria function and lipid metabolism. We also found the critical role of AMPK/PI3K/Akt/eNOS in the biological functions of agmatine. In summary, these current findings provide details about the therapeutic potential of agmatine in atherosclerosis treatment.
Supplemental Material
Supplemental Material - Agmatine mitigates palmitate (PA)-induced mitochondrial and metabolic dysfunction in microvascular endothelial cells
Supplemental Material for Agmatine mitigates palmitate (PA)-induced mitochondrial and metabolic dysfunction in microvascular endothelial cells by Dan Zhang, Jinzhao Li and Tianzhu Li in Human and Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by “The First Affiliated Hospital of Jinzhou Medical University”.
Data availability
Data will be made available on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
