Abstract
Ulcerative colitis (UC) is an inflammatory bowel disease (IBD) with rising prevalence in developing countries, and limited success of current therapies, natural products have immense potential for therapy due to their “disease modifying and side-effect neutralizing” potential. Myrica salicifolia is traditionally used for gastrointestinal diseases and have reported antiinflammatory activities, but its use in IBD has not yet been studied. Therefore, in the present study, the effects of the root extract of M. salicifolia (Ms.Cr) were investigated using the acetic acid-induced UC model in rats. For 6 days, the rats were given either vehicle (10 mL/kg), lower (200 mg/kg), and higher (400 mg/kg) doses of Ms.Cr, or the positive control drug (prednisolone; 2 mg/kg) orally. A single dosage of 5% acetic acid (1.0 mL) was administered intrarectally to rats on day 6 to induce UC. Disease activity index (DAI), histological observations, the biochemical parameters related to oxidative stress, and specific cytokines such as interleukin-6 (IL-6) and the tumor necrosis factor-α (TNF-α) were determined to assess the effect of Ms.Cr. In comparison to the AA-induced colitis rats, Ms.Cr’s pretreatment significantly decreased DAI, colonic ulceration, and inflammatory score. Total glutathione levels and catalase activity were considerably recovered in the colitis group treated with Ms.Cr, whereas enhanced lipid peroxidation in colon tissues was significantly decreased. Moreover, Ms.Cr pretreatment also caused inhibition of the activation of IL-6 and TNF-α in the colonic tissues of respective groups. Based on these findings, Ms.Cr might be developed to treat UC in the future.
Introduction
Amongst the chronic gastrointestinal diseases, Inflammatory bowel disease (IBD) is manifested mainly due to inflammation of the gastrointestinal tract (GIT). It is widely spread around the globe and is mainly of two types including ulcerative colitis (UC) and Crohn’s disease 1 Earlier, IBD was considered as the disease of the west but over the past 20 years, an increasing trend in disease prevalence is noted in developing countries attributed mainly to unhealthy lifestyles, which is a matter of serious concern. Amongst IBD, UC involves the inflammation in the colon and rectum which if not treated or managed appropriately could lead to colorectal cancer.2,3 Inflammation is integral in the manifestation of UC and presents with a rise in proinflammatory cytokines which are the consequence of both the innate and adaptive immunity with interleukin (IL)- 1β, IL-6, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ, as initial perpetrators of inflammation.4,5 In animal colitis models, the role of TNF-α has been more pronounced as it can trigger apoptosis. 6 Another simultaneous contributor of this inflammatory process is oxidative stress which is due to lipid peroxidation leading to elevated reactive oxygen species (ROS) and metabolites. The consequent increase in neutrophil infiltration leads to mucosal edema and further deterioration of mucosa 7 which subsequently disrupts the mucosal defense mechanism 8 and eventually prominent intestinal inflammation. This is further proven by the fact that catalase (CAT), an antioxidant enzyme, and glutathione (GSH), a non-enzymatic antioxidant are both depleted in IBD patients’ biopsies. Thus oxidative stress and innate immunity together play havoc in IBD and therefore are important targets of pharmacological therapies in IBD.
Understandably, anti-inflammatory drugs have shown to be clinically effective in the treatment of IBD, with antioxidants as a potential adjuvant combination. 9 Amongst the many traditionally used medicines in IBD, some have been pharmacologically validated for their use in these in both the animal models of colitis 10 and some in clinical studies such as Gingko biloba 11 fenugreek, 12 Curcumin, 9 Zingiber officinale, 13 and Camellia sinensis. 14 However, many traditional medicines have a lot of potential for this disease therapeutics but lack pharmacological validation. Amongst them, within the family of Myricaceae, “Myrica” has a promising potential for IBD therapeutics owing to their antioxidant and anti-inflammatory potential.15–18
Various species of “Myrica” have been used traditionally for GIT disorders including chronic dysentery, ulcers, and as a gut stimulant. 18 These species have also been used traditionally for hypertension, diabetes mellitus, 15 as well as airway disorders. 19 Myrica salicifolia Hochst, is an aromatic and resinous medicinal shrub 19 usually grown in humid lower highlands and is found in Southeast Africa, Ethiopia, and Saudi Arabia. It commonly belongs to the group of plant “Pedra-hume-ca’a” and is commonly known as “vegetable insulin” in general and “Shinet (Amharic)” in Ethiopia 20 It’s folkloric use worldwide include use in treatment of diabetes, 15 diarrhea, and gastroenteritis, 21 as well as in other gastrointestinal disorders.19,22 In addition, both the roots and bark of M. salicifolia Hochst have traditionally been used for the management of respiratory illnesses including chest congestion, and pneumonia indicating its antiinflammatory and antibacterial potential. It has also been used for varied disorders including hypertension, headache, pain, inflammation and mental disorders.17,19 Despite these strong folkloric claims for use of M. salicifolia Hochst in various ailments, the scientific study to support its traditional uses is limited. These activities indicate that the plant has gastroprotective potential along with antiinflammatory effect and therefore shall be explored for inflammatory diseases of the gut including IBD.
The antibacterial effect has also been reported with chloroform extracts of M. salicifolia against Escherecia coli, Klebsiella penumoniae, Salmonella typhi and Pseudomonas aeruginosa 23 many of which have been implicated in pathogenesis of IBD. Recently, a blood glucose lowering effect, 24 antimalarial, 25 and antiplasmodial activities 26 have been reported for M. salicifolia. Earlier, the potent analgesic effect of M. salicifolia (100 mg/kg) extract has been reported. 27 Interestingly, the methanolic extract did not produce anti-inflammatory effect against carrageenan-induced rat paw edema but was a very potent analgesic. In contrast, reports of isolation of Adenodimerin A 19 from Myrica species indicate possibility of strong anti-inflammatory and antioxidant potential. Due to the folkloric use as an anti-inflammatory agent, we speculate that M. salicifolia may mediate anti-inflammatory effect through a different pathway such as through modulation for innate and adaptive immunity rather than the one used in carrageenan induced rat paw edema model which is predominantly a prostaglandin mediated pathway. It is important to assess possible mechanism for combating inflammation and oxidative stress in context of UC which is predominantly through cytokines and oxidative stress.23,28 Therefore, we hypothesized that M. salicifolia is effective in IBD through a combination of different activities including mucosal protective, anti-inflammatory and antioxidant activities. To prove this hypothesis, we determined the effectiveness of crude methanolic aqueous (70:30) extract of M. salicifolia against 5% AA-induced model of UC in Wistar albino rats by measuring their disease activity index, mucosal protective, antioxidant and anti-inflammatory effects. Mucosal protective effect was determined by macroscopic and microscopic damage scoring of Hematoxylin and Eosin and periodic acid-Schiff-stained colonic tissues. Antioxidant activity was determined invitro in colonic homogenates using total glutathione and catalase assays, whereas effect on oxidative stress was measured by using malondialdehyde assays. Anti-inflammatory effect was evaluated by measuring the levels of IL-6 and TNF-a in colonic tissue homogenates using tissue cytokine ELISA assay.Materials and methods
Study design
Rats were divided by random sampling into five groups, comprising of six animals in each group. The animals were first divided in the cages that were comparable after which they were randomized in each group moving horizontally through cages. Single blinding was done in histopathological scoring. Intraperitoneal (i.p.) route was adopted to administer the test material in order to avoid the first pass effect and any metabolic changes due to gastric environment since the initial idea was to screen the potential in plant.
Sham and colitis control groups were labeled as Groups 1 and 2, and they were only administered with the vehicle (IP) comprising of 3% Tween 80 in distilled water (v/v). Treatment groups (Group 3 and 4) were treated daily with IP injection of 200 and 400 mg/kg of Ms.Cr, respectively, for 7 days. However, it was made sure that the dose of the extract on the sixth day (which was the day for induction of colitis) was given 2 h before the induction of colitis. Group 5, which was considered as a positive control, was administered prednisolone (2 mg/kg; IP) for consecutive 7 days in a similar manner as Ms.Cr. Colitis was induced on the sixth day and 24 h after the induction of colitis, animals were sacrificed.
Chemicals and reagents
Acetic acid, Hematoxylin and Eosin, methanol, periodic acid, prednisolone, Schiff reagent, and Tween-80 were bought from the company of Sigma–Aldrich (St Louis, MO, USA). Pentobarbital was recruited from local Pharmacy. Analytical grade chemicals were used and distilled water was used for their solubilization.
Plant collection and extraction
Myrica salicifolia roots were gathered from Genfoquch located in the Amhara Region (Semien Gondar Zone) and north of Tana Lake on the Lesser Angereb River, Ethiopia in the last month of 2018. Abiyu Enyew (botanist), Department of Biology, College of Natural Sciences, University of Gondar, Ethiopia, authenticated the plant, and a specimen (voucher number. YK001) was deposited in the University of Gondar Herbarium for record purposes.
For the preparation of extract, the roots of M. salicifolia were washed in distilled water and shade dried. Then the pulverization of the dried plant material was done to ground it to powder. The powdered material (700 g) was then macerated for 72 h in 80% methanol (v/v) with periodic shaking (31). This procedure was repeated thrice to ensure maximum extraction of the active constituents and then subsequently filtered using Whatman filter paper (No. 1). This prepared our hydro-alcoholic filtrate which was then oven-dried ensuring that the temperature would not exceed 40°C. The resultant dried extract was refrigerated until further use.
Animals
Wistar albino rats of either sex (180–200 g) were used which were housed in the animal care unit of Prince Sattam bin Abdulaziz University (PSAU) in Saudi Arabia. The housing conditions were maintained as per standard recommendation for animal housing, including 23–25°C of room temperature with (55±5% relative humidity, and a 12:12 light:dark cycle, with lights being turned on at 08:00 and switched off at 20:00. All experimental group animals were placed in transparent plastic rectangular cages. Sawdust was used as bedding and refreshed every 48 h. The diet for the animals comprised of a standard pellet diet and water was provided ad libitum. During housing, the health status of the animals was also monitored and they were maintained as per Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council, guidelines 29 The study was approved by the Bio-Ethical Research Committee (BERC) at Prince Sattam Bin Abdulaziz University under the reference number BERC-001-12-19. Crude extract of M. salicifolia roots (Ms.Cr) and prednisolone was administered orally (PO). Rats were euthanized using the cervical dislocation method under anesthesia by using pentobarbital (50 mg/kg; IP).
Acute toxicity study
For assessing the toxicity effect at a higher dose in rats, OECD-423 guidelines were followed. 30 For this purpose, Wistar albino rats (n = 6 animals/group) who were fasted overnight were labeled into group I (test group) and were administered intraperitoneally (IP) plant extract of Ms.Cr (2000 mg/kg) whereas the animals of group II (control group) were injected the vehicle which comprised of Tween 80 (3% v/v) in distilled water, given in a dose of 10 mL/kg, IP. The observations were taken for the symptoms of toxicity and/or deaths for each animal as follows: in the first 4 h every 15 min, followed by every 30 min for the remaining 6 h and then for the successive 48 h. If the test group showed no signs of toxicity, it was further administered the plant extract (4000 mg/kg) and observed after 48 h for the signs of toxicity.
Induction of acetic acid colitis
A 5% acetic acid (AA; 5%) model was used as a representative of ulcerative colitis, as this model is previously been used for the same purpose. 5% AAwas given intrarectally (IR) to Wistar albino rats. The colitis induced by this method was preferred in laboratory settings as it is easy and reproducible and the features presented in this model including apparent symptoms such as diarrhea, weight loss, and rectal bleed, as well as internal characteristics including ulcers and inflammation of the colon, resembles human features of UC. 31 Food cages of the rats were removed 16 h before the colitis induction, however, water was provided ad libitum. To minimize distress and ensure the homogenous spread of the colitic agent, light anesthesia was given to the rats before the colitis induction using diethyl ether.
Colitis was induced in all animals by using 1 mL of 5% AA (5% v/v, in 0.9% saline), except in the sham control group who was administered with 1 mL normal saline (IR). A polyethylene catheter was used for insertion of the drug by inserting the tip inside the anus up to 8 cm so that it reaches the rectum. They were then kept in the trendelenburg position for 45°s to ensure equal exposure and retention. This was followed by flushing with 0.5 mL saline in the same catheter to neutralize the acidic solution.
Assessment of disease activity index
Scoring of disease activity index.

Normal stools = well-formed pellets, loose stools = pasty and semi formed stools which do not stick to the anus, and diarrhea = liquid stools that stick to the anus.
Assessment of macroscopic damage
A semi-quantitative macroscopic damage scoring was used to assess the visual damage to the colonic mucosa. 33 Following parameters of the rats were assessed to gauge the extent of macroscopic damage:
Wet weight to length ratio
The ratio of both the colon and spleen’s wet weight to length (g/cm) was measured. The increase in this ratio is an indicator of inflammation because edema causes an increase in weight and reduction in length of these parts, which corresponds to inflammation.33,34
Mucosal damage score
To measure the extent of damage of colonic mucosa macroscopically, the colonic area from the mesenteric line was excised longitudinally. Different parameters were measured for this purpose as mentioned below:
Percentage area affected
Hyperemia with or without lesions was calculated in the 5 cm area proximal to the anus and quantified as follows, and previously described. 34 Briefly, the percentage of damage was categorized into seven scales ranging as 0%, 1–5%, 5–10%, 10–25%, 25–50%, 50–75% and 75–100% for scores 0, 1, 2, 3, 4, 5, and 6, respectively.
Ulcer Index (UI)
UI was the sum of lesion score and area of ulcer lesion.33,35 Ulcer lesion was scored from 0-3 with no macroscopic changes corresponding to score 0, mucosal erythema only to score 1, mild mucosal edema, slight bleeding or erosion, to score 2 and moderate edema, bleeding ulcers or erosions to score 3, whereas, severe ulceration, erosions, edema, and tissue necrosis corresponded to score 4. 35 The tissue was scored for ulcer lesion by measuring its area that had a lesion. For this purpose, the flattened tissue was spread on squared graph paper and the number of cells that covered the ulcer was counted by considering each box of the graph paper’s area as 1 mm2. This was followed by blinded histopathological analysis.
Microscopic assessment of UC
H & E and PAS staining
The colonic tissues were then subjected to histopathological analysis to assess the degree of inflammation and mucosal damage, by using the previously defined protocols 36 For this purpose, the 3–5 cm colonic tissues which were excised from the proximal anal area were stored in neutral buffered formalin. After 24 h they were treated with hematoxylin and eosin (H & E) for assessing intactness of mucosal architecture, as well as neutrophil and lymphocytic infiltration which corresponded to the degree of ulceration of mucosa and level of inflammation in colonic tissues, respectively. A microscope of model: Olympus BX 52 was used for assessing the histopathological changes. Blinded scoring was done by the histopathologist for assessing the microscopic damage using Neurath and Kiela scores37,38 Briefly Neurath’s maximum score for mucosal damage was 4 with a score of 0 corresponding to the absence of leukocyte infiltration and score 4 corresponding to four of these parameters including leukocyte infiltration across muscularis, loss of goblet cells, high blood vessel density/erythema, and thickening of the colon wall. Kiela score considered 3 parameters and therefore was scored maximum to 3 (Score 0-3: absence of inflammation; inflammation low coupled with low mononuclear cells infiltration (1–2 foci); inflammation of moderate-intensity coupled with multiple foci; inflammation of high level but with increased erythema and marked thickening of colonic mucosa).
PAS stain
The mucosal damage at goblet cells was quantified by using periodic acid and Schiff’s (PAS) stain. Mucin level was scored according to Dorofeyev et al. 39 with a maximum score of 3. It was scored 0 when the cells stained were <1%, score 1 if the stained cells were low, that is, 1–30%, scored 2 if staining was of medium level that is, 30–80%, and scored 3 if a high number of the cells were stained that is, up to 80%. For high-resolution snapshots, a camera (Olympus DP21) was used which was mounted above the microscope. Microsoft PowerPoint 2010 software was coupled with this software which was used to label the captured photographs.
Biochemical analysis
Preparation of tissue homogenate
For the biochemical analysis, frozen colon tissues were mixed with an ice-cooled solution of phosphate buffer (0.1 M; pH 7.4) and then homogenized with the help of a tissue homogenizer, while keeping the samples in ice. Further to this, the homogenized samples were centrifuged at a chilled temperature of 4°C for 10 min at 700×g, to separate nuclear debris. To isolate a post-mitochondrial supernatant (PMS), the separated supernatant was again centrifuged at 9000×g for 20 min at 4°C. 40 These supernatants were stored at −80°C for future biochemical assays.
For the analysis of cytokines levels, the protocol for colonic tissue processing was different in a way that first the tissues collected were snap-frozen. At the time of analysis, they were thawed, weighed, and mixed with tissue extraction reagent (Invitrogen, Life Sciences) and a protease inhibitor cocktail (Sigma FAST). After that, they were homogenized and centrifuged to separate PMS, as previously stated.
Assessment of oxidative stress (MDA, CAT, GSH assays)
Malondialdehyde (MDA), which is an indicator of lipid peroxidation was measured to assess the impact of our treatment on oxidative stress. Effect on antioxidants was also assessed by measuring the levels of catalase (CAT), an antioxidant enzyme as well as levels of total glutathione (GSH), a nonenzymatic antioxidant.
For measuring MDA 41 colonic tissue homogenates were used, whereas, PMS of the colonic homogenate was used to measure the levels of GSH 42 and CAT, 43 according to the previously defined protocols. Briefly, Dithio-bis-2-nitrobenzoic acid (DTNB) was mixed with the supernatants mixtures and absorbance was taken within 5 min at a wavelength of 412 nm Standard protocol was used to estimate catalase enzyme activity.43,44
Assessment of tissue cytokines by ELISA
Two pro-inflammatory cytokines levels were measured comprising of IL-6 and TNF-α. This analysis was carried out on PMS obtained from colonic homogenates. Quantitative assays were used according to the manufacturer’s instruction using their respective Platinum ELISA kits.
Statistical analysis
To compare the effect between the control and different treatment groups, one-way analysis of variance (ANOVA) was used followed by post hoc Tukey’s test. Data were represented as Mean±SEM (Standard error of the mean). A less than 0.05 probability was considered as statistically significant. Software of GraphPad Prism program (version 4) (San Diego, CA, USA) was used for statistical comparisons of observed results.
Results
DAI
This index measures the disease’s severity in the form of the sum of three parameters, including weight loss, rectal bleed, and diarrhea severity. Expectedly, diseased controls, i.e. AA group exhibited a marked loss in body weight as evident in Figure 1(a). The body weight was significantly high in the AA group. Compared to this, weight reduction was significantly less in both the Ms.Cr treated groups as well as in the prednisolone-treated group (positive control). Other parameters i.e. diarrhea and rectal bleed were increased in the AA controls, whereas, treatment and positive control groups showed a significant reduction in diarrheal and rectal bleed scores, as evident in Figure 1(b). Comparing the overall DAI, the AA group showed a highest DAI (2.38±0.145), whereas, Ms.Cr showed a significant decrease in DAI, dose-dependently to 1.05±0.24 and 0.72±0.09 at 200 and 400 mg/kg doses (p < 0.01 and p < 0.001), respectively, which was comparable to prednisolone pre-treated group (0.55±0.13; p < 0.001). Pretreatment with Myrica salicifolia improved (a) percentage of weight loss and (b) disease activity index (DAI) in acetic acid-induced colitis rats. Data expressed as means ± SEM (n = 6). One way ANOVA was used for statistical analysis followed by Tukey’s post-test. ***p < 0.001 compared with sham control, #p < 0.05, ##p < 0.01, ###p < 0.001 compared with AA alone. Ms.Cr = Myrica salicifolia crude extract. AA = acetic acid; PO = orally; IR = intrarectally.
Macroscopic damage
Wet weight to the length ratio
The increase in wet weight is attributed to edema due to inflammation. Expectedly, in both the colon and spleen of the AA group, the average weight to length ratio was greater than the sham control. Compared to this, Ms.Cr (both the doses) and prednisolone-treated groups had a significantly lower ratio, as evident in Table 1. This indicates a possible reduction in edema that could be attributed to reduced inflammation as evident in Figure 2(a) and (b). Effect of Myrica salicifolia on (a) colon weight & length ratio and (b) spleen weight & length ratio against AA-induced colitis rats. Data expressed as means ± SEM (n = 6). One way ANOVA was used for statistical analysis followed by Tukey’s post-test. ***p < 0.001 compared with sham control, #p < 0.05, ##p < 0.01, ###p < 0.001 compared with AA alone. Ms.Cr = Myrica salicifolia crude extract. AA = acetic acid; PO = orally; IR = intrarectally.
Percentage area affected
The macroscopic inspection revealed a flaccid outlook with a marked thickness of colonic mucosa and a greater number of ulcers (3.66±0.21) in the AA group. Compared to this, Ms.Cr pre-treated groups showed a significant reduction in the percentage area affected with the number of ulcers being 0.66±0.4 at 200 mg/kg dose. This protective effect increased at a greater dose of 400 mg/kg as evident in Figure 3. Effect of Myrica salicifolia crude extract on gross anatomy of colons against acetic acid-induced colitis in Wistar albino rats. Ms.Cr = Myrica salicifolia crude extract. AA = acetic acid; PO = orally; IR = intrarectally.
The percentage of colon area affected was greater in the AA-control group with 86.33±2.40%. Ms.Cr pretreated rats dose-dependently showed a decrease in the area affected by the AA with 60.00±1.82% and 41.00±1.31% at 200 and 400 mg/kg doses respectively. Prednisolone pretreated rats showed maximum protection and the least colon affected area was noted (25.00±1.82%) as can be seen in Figure 4(a) Effect of Myrica salicifolia on (a) % area affected and (b) ulcer index in AA-induced colitis rats. Data expressed as means ± SEM (n = 6). One way ANOVA was used for statistical analysis followed by Tukey’s post-test. ***p < 0.001 compared with sham control, ##p < 0.01, ###p < 0.001 compared with AA alone. Ms.Cr = Myrica salicifolia crude extract. AA = acetic acid; PO = orally; IR = intrarectally.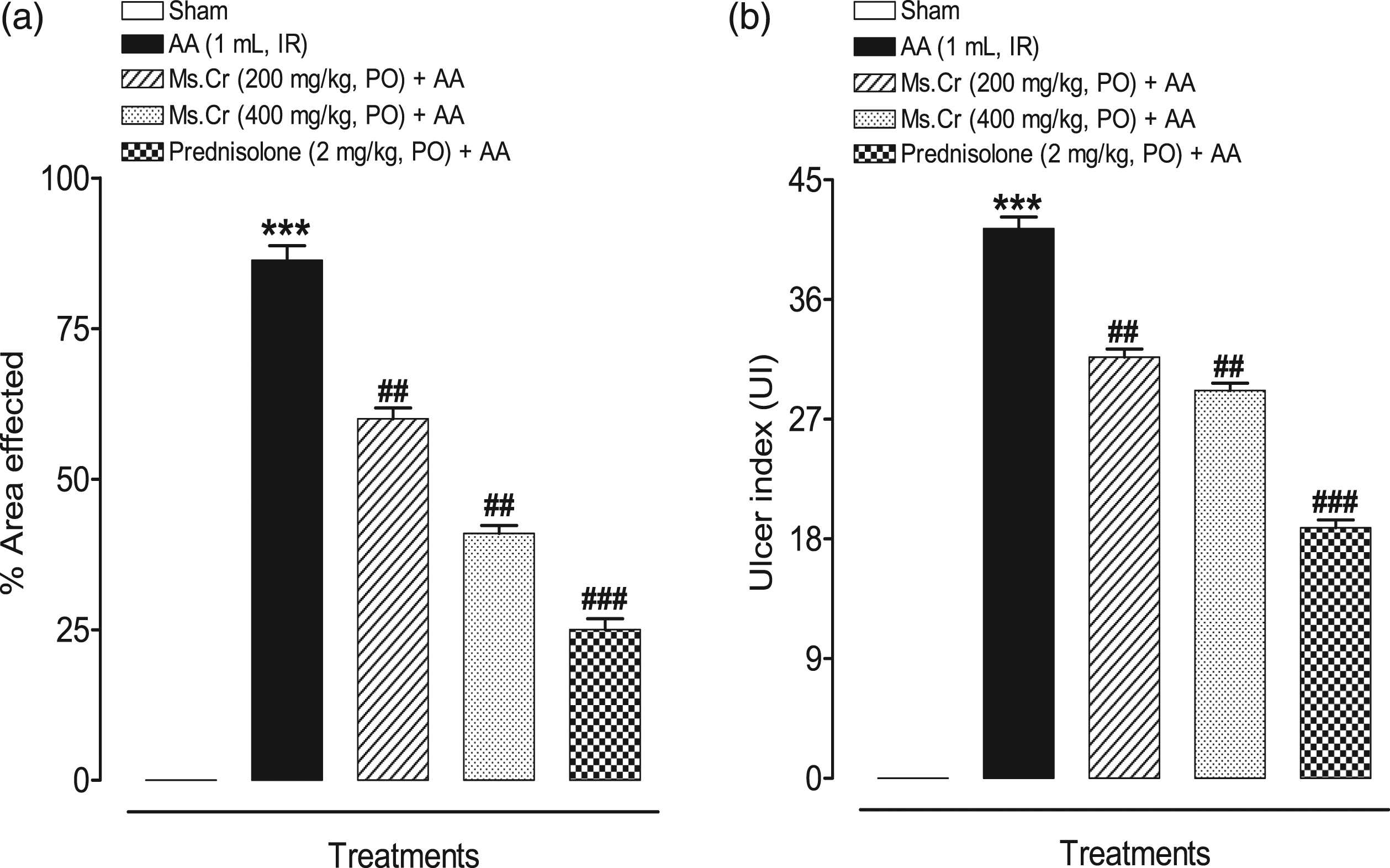
Mucosal damage
Ulcer index (UI) was used to determine the extent of mucosal damage. AA group exhibited the greatest UI (41.33±0.88) as evident in Figure 4(b). Compared to this, Ms.Cr treated groups at the dose of 200 and 400 mg/kg had significantly less UI with a score of 31.67±0.61 and 29.17±0.54 respectively, whereas, prednisolone-treated groups had a maximum reduction in UI (18.83±0.60).
Microscopic damage
H and E-PAS staining
To understand the ameliorating effect of Ms.Cr in the UC model of rats, the microscopic assessment was done to understand the level of inflammation and tissue damage. Figure 5(a) shows H and E stained colonic tissue sections of respective groups. Effect of Myrica salicifolia crude extract on histopathological changes in colon tissues in AA-induced colitis rats. Colon tissues were stained with (a) H&E, and (b) PAS for microscopic evaluation. Sham control rats showing normal morphological structure in both the stained photomicrographs. AA-induced colitis rats with H&E stain showing severe tissue damage of both mucosal and submucosal layers of intestine by showing areas of complete loss of goblet cells (L), remarkable necrosis (N), degeneration (D) as well as occlusion (O) of blood vessels and infiltration (I) of mucosa and submucosa by inflammatory cells. PAS staining shows severe loss of mucin and goblet cells. Pretreated rats with M. salicifolia crude extract show dose dependent improvement whereas prednisolone showed complete protection against AA-induced colon damage. Ms.Cr = Myrica salicifolia crude extract, AA: acetic acid; H&E: hematoxylin and eosin; PAS: periodic acid Schiff’s.
Photomicrographs of sham control animals showed normal morphological structure, whereas, animals treated with AA only showed severe tissue damage, degeneration, remarkable necrosis, occlusion of blood vessels in mucosal and submucosal both layers of the intestine with an enhanced level of mucosal edema, which correlated with enhanced neutrophil and lymphocytic infiltration till submucosa. The sections were scored using semi-quantitative scoring criteria with a score of 3.75±0.11 out of 4 for Neurath score and 2.83±0.10 out of 3 for Kiela score
Quantification of microscopic damage.

Data are expressed as mean ± SEM. Results for the Ms.Cr treatment group were compared against AA group with one way ANOVA followed by Tukey’s post-test. Ms.Cr: Myrica salicifolia crude extract; AA: acetic acid.
ap < 0.01 vs AA.
bp < 0.05 vs AA.
cp < 0.001 vs AA.
Biochemical assays
MDA assay
MDA was measured in colonic tissue homogenates of all the groups to represent the degree of oxidative stress. MDA levels were significantly elevated in the AA control group (13.65±1.15 nmol/mg protein) as compared to the negative control group (3.38±0.15 nmol/mg protein). Ms.Cr pretreated groups exhibited reduced levels of MDA at both doses of 200 and 500 mg/kg (6.47±0.52 and 5.58±1.05 nmol/mg of protein, respectively) as compared to the AA group. MDA levels were also significantly decreased in prednisolone-treated groups (4.37±0.11 nmol/mg of protein).
GSH and CAT assay
Antioxidant activity was assessed through measurement of total glutathione (GSH) and catalase (CAT) assays which showed an expected increase in Ms.Cr treated and prednisolone-treated groups, whereas, it was markedly reduced in the diseased control groups. In the AA group, GSH level was markedly reduced (4.40±0.39 nmol/mg protein) as compared to the negative control group (16.69±0.54 nmol/mg protein). As shown in Figure 6(b) and (c), Ms.Cr significantly increased the levels of GSH as compared to the AA controls at both the tested doses, whereas prednisolone also significantly enhanced GSH levels (13.71 ± 0.73 nmol/mg protein). Effect of Myrica salicifolia on (a) malondialdehyde (MDA), (b) total glutathione (GSH) and (c) catalase (CAT) in AA-induced colitis rats. Data expressed as means ± SEM (n = 6). One way ANOVA was used for statistical analysis followed by Tukey’s post-test. ***p < 0.001 compared with sham control, ##p < 0.01, ###p < 0.001 compared with AA alone. Ms.Cr = Myrica salicifolia crude extract. AA: acetic acid; PO: orally; IR: intrarectally.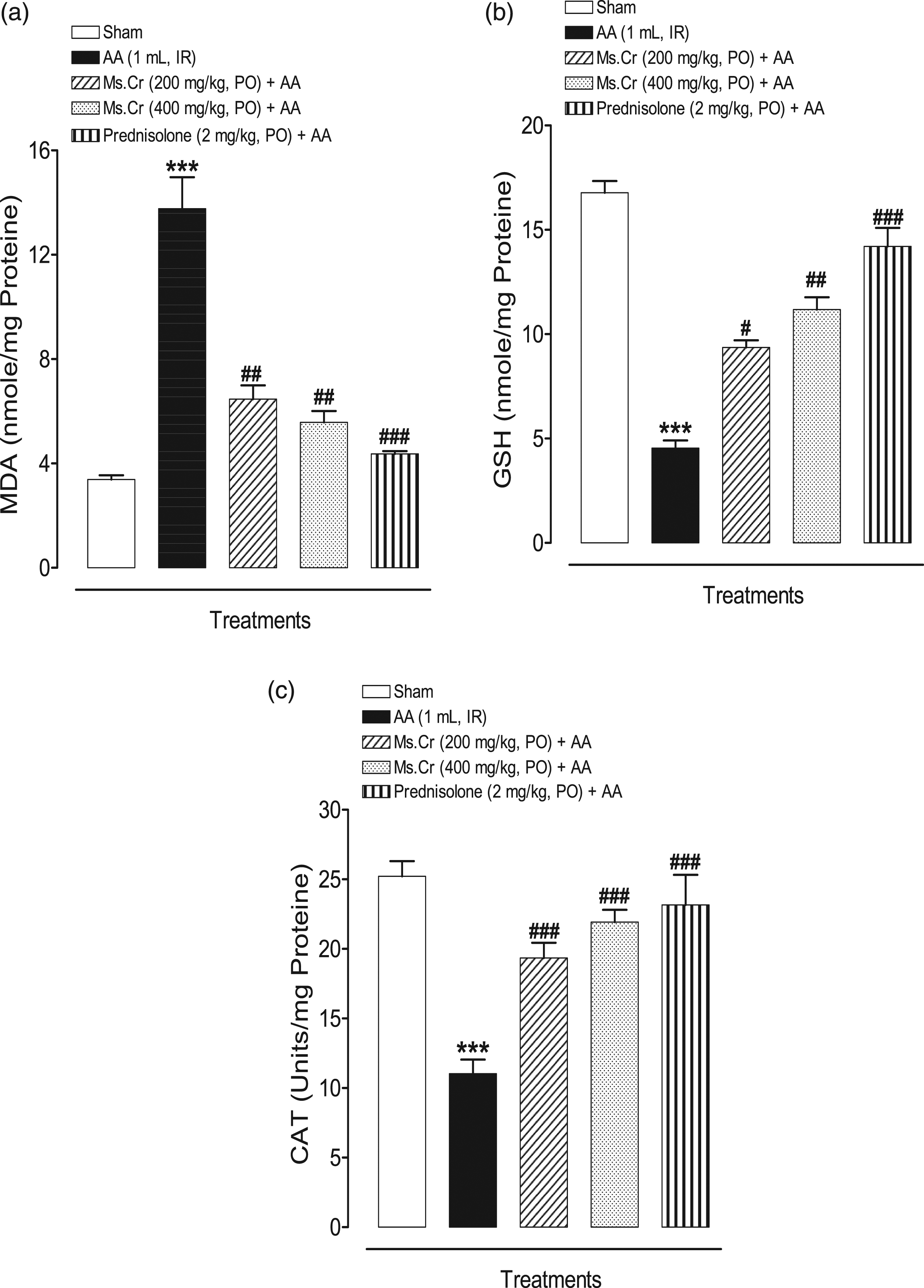
Similarly, CAT activity was lower in the AA group (10.53±1.06 units/mg proteins), as compared to the negative control groups (25.49±0.98 units/mg proteins). Pre-treated groups with Ms.Cr at both doses showed markedly elevated levels of CAT (19.35±1.09 Units/mg proteins and 21.93 ± 0.88 units/mg proteins for 200 and 400 mg/kg, respectively), which was similar to positive control group prednisolone, (22.86 ± 1.96 Units/mg proteins; p < 0.001).
IL-6 and TNFα levels
ELISA was performed to assess the levels of tissue cytokines in colonic homogenates. The mean IL-6 concentration of the negative control group was significantly low (10.83±1.53 pg/mL) as compared to the AA control group (66.67±3.8 pg/mL; p < 0.001). Expectedly, prednisolone-treated tissues had a significantly reduced IL-6 concentration (19.17±3.05 pg/mL; p < 0.001). Ms.Cr pretreated groups also showed marked reductions in the levels of IL-6 with similar effect at both doses with 36.67±2.78 pg/mL and 29.17±2.38 pg/mL at 200 and 400 mg/kg doses respectively (p < 0.001 for both doses) (Figure 7(a)). Effect of Myrica salicifolia on (a) interleukin-6 (IL-6), and (b) tumor necrosis factor (TNF-α) and in AA-induced colitis rats. Data expressed as means ± SEM (n = 6). One way ANOVA was used for statistical analysis followed by Tukey’s post-test. ***p < 0.001 compared with sham control, ##p < 0.01, ###p < 0.001 compared with AA alone. Ms.Cr = Myrica salicifolia crude extract. AA: acetic acid; PO: orally; IR: intrarectally.
TNF-α concentration was significantly increased in AA control groups (80±2.43 pg/mL; p < 0.001) as compared to the negative control rats (23.33±1.21 pg/mL). The TNF-a levels in the Ms.Cr pre-treated rats were significantly low (56.11±1.59 and 50.56 ± 1.59 pg/mL) at 200, and 400 mg/kg doses respectively. Prednisolone-treated rats also had a significantly reduced level of TNF-α (38.89±1.85 pg/mL) as evident in Figure 7(b).
Discussion
IBD which is a chronic inflammatory condition consists of UC and CD and its management requires therapies aimed at multiple targets, owing to its multifactorial etiologies. 45 That’s why a single therapeutic target is often not enough and leads to failure to remission. 46 Hence, a therapeutic option that has better compliance and manages multiple triggers is needed. With an inclination of population towards traditional medicine globally, 47 there is need for validation of traditional plants that have been used medicinally for this purpose.
Natural products have been evaluated and rationalised for their traditional use in IBD including Flaxseed, 10 G. biloba, 11 fenugreek, 12 Curcumin, 9 Z. officinale, 13 and C. sinensis. 14 Myrica species has also shown immense antiinflammatory potential with M. salicifolia, reported for its traditional use in inflammatory disorders. It has however, not been rationalized pharmacologically. Hence, to assess its effectiveness and understand the pharmacological rationale in IBD, we evaluated its effect against 5% acetic acid-induced colitis (AA) model in rats, a reasonable model for laboratory settings. 48 We were able to reproduce this model in our lab and it resembled salient features of UC in terms of increased DAI, severe colonic inflammation and ulceration, goblet cells, and subsequent mucin depletion, as well as, similar histopathological features including neutrophil and leukocytic infiltration till submucosal colonic tissues.
The improvement in DAI was our first evidence that Ms.Cr could be used for IBD therapy. To understand the pharmacological basis, we excised the colon of the rat and found that Ms.Cr had reduced the mucosal damage as evidenced by the reduced number of ulcers, as well as, ulcer index. This could be attributed to both the reduction in mediators of inflammation as well as due to reduced oxidative stress. One of the clues for us that Ms.Cr reduced mediators of inflammation was from the observation that spleen and wet colonic weight to length ratio was reduced in treated groups, as compared to the AA control group, indicating the reduction in mucosal edema, which corresponds to the reduction in infiltration of mediators of inflammation. 49
This finding was further validated through microscopic assessment that showed a reduction of infiltration of neutrophils and lymphocytes in the Ms.Cr treated rat’s colonic tissue sections, whereas this infiltration was till submucosal layer in AA control sections. Neutrophil infiltration is the sign of acute inflammation and the response of innate immunity. Preservation of goblet cells in the rats’ mucosal epithelium was another clue towards the protective role of Ms.Cr against UC. Goblet cells produce neutral mucin, which serves as a barrier against antigens present in the bowel lumen. 50 It is a possibility that Ms.Cr increased the goblet cells and subsequent mucin production which may have helped in the preservation of mucosal epithelium. This aspect is important for therapeutic purposes, as a leaky epithelial barrier has been postulated as a cause of exudative diarrhea, invasion of bacteria, and subsequent inflammation in IBD. 51 An in-depth mechanism needs to be probed in our future studies to understand which genes and peptides may have been upregulated to be responsible for this mucosal epithelial repair.
One of the possibilities for promoting mucosal defense could be either reducing the oxidative stress and/or elevation in antioxidant activity in the epithelial layer. 8 In the AA-induced colitis model, oxidative stress is the key player to trigger colitis by intracellular acidification.52,53 This acidification is what activates the innate immunity and subsequent release of innate cytokines including IL-1, IL-6, and TNF-a. These cytokines after chemotaxis cause the release of cytotoxic oxidants. This subsequently ends in the trigger of reactive oxygen metabolic cascade which culminates in the lipid peroxidation and subsequent MDA and superoxides production. Simultaneously, antioxidants protect the tissue against oxidative damage by neutralizing these superoxides. This was evident in our model as there was evidence of oxidative stress and reduced antioxidant activities of GSH and catalase, which corresponds to previous reports on the AA colitis model. 54
Earlier, it has been shown that in AA colitis, GSH depletion was the consequence of neutralization by free oxygen radicals due to extensive intracellular acidification. The reduced GSH consequently leads to an increase in the MDA levels culminating in oxidative damage 55 which indicates the protective role of GSH against oxidative stress-mediated by free radicals. In the current study, GSH levels increased in the Ms.Cr-treated group, which was accompanied by MDA inhibition (a sign of lipid peroxidation), whereas vice versa was the case with the colitis control group. This is consistent with earlier research that found lower MDA levels and higher antioxidant activity of antioxidant-rich substances to compensate for the oxidative damage caused by colitis.56,57 Thus, Ms.Cr promoted mucosal healing in the AA model of rats by reducing the MDA levels, as well as by enhancing the antioxidant activities, as evident by an increase in CAT and GSH levels. Antioxidant activity is considered as one of the major elements in the prevention of inflammation 58 and hence, Ms.Cr antioxidant benefits in the prevention of acetic acid-induced UC may attribute to its anti-inflammatory and mucosal protective effects. There is limited research done on Ms.Cr to validate its traditional use, however, Myrica species have previously been reported to possess antioxidant activity 19 This is the first study to report the antioxidant activity of Ms.Cr in colitis models.
Our subsequent aim was to explore the anti-inflammatory activity in the experimental rats to further elucidate the mechanisms and downstream pathways of oxidative stress. Oxidative stress could result in the activation of the monocyte-macrophages system, which gives a positive feedback loop for inflammatory cytokines’ release, and subsequent chemotactic signals to cause neutrophil and lymphocytic infiltration. One of the cytokines released by activated mononuclear cells is TNF-a which further facilitates the release of other cytokines including IL-6 59 as well as, also assist in releasing chemotactic signals that recruit neutrophils in the AA colitis model 12 These pro-inflammatory cytokines have been attributed to the pathogenesis of UC as they disrupt mucosal integrity and aggravate colon injury in UC 60 In the present study, TNF-α, and IL-6 were elevated in the diseased control (AA) group, whereas, these cytokines were significantly reduced in the Ms.Cr treated groups, indicating an anti-inflammatory effect of the extract, possibly by regulating the innate immune arm. As TNF-α acts as a chemotactic signal for neutrophil infiltration, in our study we also simultaneously found the reduction in neutrophil infiltration in Ms.Cr pretreated rats. As evident in our histological assessment.
Hence, Ms.Cr ameliorated the severity of UC in our rat model as evident by reduced DAI, improved mucosal integrity as depicted by macroscopic and microscopic parameters. This coupled with the reduction in oxidative stress parameters and inflammatory markers of innate immunity including IL-6 and TNF-α indicates that Ms.Cr ameliorates IBD in the animal model of colitis by acting on multiple targets.
One of the limitations of the current study is that cytokines should have been measured at different time points because the kinetics of cytokines varies and therefore effect may also vary. Besides, IL-1 beta levels measureent would have completed the mechanistic involvement of innate immune activation. This aspect will be taken into consideration in our future studies.
With current findings, future elaborative molecular studies could be designed such as investigating the effect on mucosal repair genes involved in goblet cell and/or mucus production. Additional information can be achieved by studying elaborated cytokine mechanisms, such as the ones involved in modulating the adaptive immunity parameters including IFN-γ and IL-17, etc. 61 Further investigations are needed to evaluate the effect of pharmacologically active compounds from M. salicifolia extract with potential for use in UC and involvement on different targets of the disease.
Conclusions
The findings show that Ms.Cr ameliorates AA-induced UC in rats, which is evident by improvement in the disease activity index. The reduction in number and size of ulcers were evident in macroscopic assesment, whereas, microscopic assesment revelaed redcued neutrophil and lymphocytic infiltration and reduced gobelt cell depletion as comapred to the diseased control. Ms.Cr showed reduction in oxidative stress (reduced MDA activity), and increase in antioxidant activity as evident by increase in CAT and GSH activities in colonic tissue. Consequent antiinflammatory effect was evident by reduction in IL-6 and TNF-α levels. Hence M. salicifolia is effective in ameliorating the severity of ulcerative colitis in rat model of colitis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
