Abstract
Background
Periodontal dressing is used to cover the gum surface and protect the wound after periodontal surgery. Nanomaterials have been widely applied in dentistry in recent years. Zinc oxide (ZnO) is one of the main components of periodontal dressing.
Aim
This study aims to explore the toxicity ZnO nanoparticles (ZnO NPs) causes to human gingival fibroblast cells (HGF-1) and its effect on cell proliferation.
Methods
First, we identified and analyzed HGF-1, including cell morphology, growth curve, and immunohistochemistry staining. Then, we treated HGF-1 with ZnO NP. Cell viability, the integrity of the cell membrane, oxidative damage, and apoptosis were measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, lactate dehydrogenase (LDH) release assay, fluorescent probe, and flow cytometry. Furthermore, the expression of murine double minute 2 (MDM2) and p53 was determined by quantitative real-time polymerase chain reaction (qPCR) and Western blotting. We finally overexpressed MDM2 in HGF-1 to verify the relationship between MDM2 and cell proliferation.
Results
Our research indicated ZnO NPs did not affect cell proliferation at low concentrations. However, high-concentration ZnO NP inhibited cell proliferation, destroyed the integrity of cell membranes, and induced oxidative stress and apoptosis. In addition, high concentration of ZnO NPs inhibited the proliferation of HGF-1 by regulating the expression of MDM2 and p53.
Conclusion
High concentration of ZnO NP caused toxicity to HGF-1 cells and inhibited cell proliferation by regulating MDM2 and p53 expression.
Keywords
Introduction
Periodontal dressing is a special dressing for periodontal surgery, which covers the gum surface to protect the wound from mechanical trauma and keep the stability of the surgical site during the healing process.1,2 The proliferation of gingival cells is very important for wound healing after periodontal surgery. Therefore, the impact of periodontal dressing on proliferation and toxicity of gingival cells needed to be evaluated before selected as a suitable periodontal dressing. At present, zinc oxide is an important component of periodontal dressings. 3 Nanomaterials have been widely used in dentistry with the rapid development of nanotechnology. 4
Zinc oxide nanoparticles (ZnO NPs) had important advantages, such as antibacterial activity, low toxicity to the host, high stability, and good selectivity.5,6 ZnO NPs had gained recognition in biomedical fields. 7 Previous research has shown that ZnO NPs have a significant toxic effect on human periodontal ligament fibroblasts (hPDLF) and mouse dermal fibroblasts (mDF) at a concentration of about 50–100 μg/mL, and when it is lower than 50 μg/mL, there is no significant effect on cells. 8 However, ZnO NP significantly decreased cell viability and increased cell toxicity after exceeding 5 μg/mL. 9 Therefore, it was of great significance to understand the potential toxicity of ZnO NPs and their underlying molecular mechanisms for their application in oral clinics.
The p53 protein was closely related to cell cycle regulation, DNA repair, cell differentiation, and apoptosis. 10 Various cellular stresses triggered the induction of p53, such as DNA damage, oxidative stress, and so on. 11 As a negative regulator of p53, murine double minute 2 (MDM2) regulated the stability and activity of p53 protein.12,13 The previous research has shown that ZnO NPs inhibited the proliferation of mast cells through the regulation of MDM2 and p53 protein levels. 14 Therefore, we further explored whether ZnO NPs had the same regulation in gingival cells.
ZnO NPs are an effective antibacterial agent, also used in medicine for bioimaging and drug/gene delivery. Therefore, humans are exposed to ZnO NPs at high frequency. ZnO NPs could enter organisms through multiple routes such as the respiratory tract, digestive system, and injection routes, which have been proven in in vivo studies. ZnO NPs have the potential to reach any organ or tissue, posing a risk to human health. The potential molecular effects of ZnO NPs included decrease in cellular viability, loss of membrane integrity, and cell apoptosis.
Based on research results, we found that ZnO NPs did not affect cell proliferation at low concentrations. However, high concentration of ZnO NPs inhibited cell proliferation, destroyed the integrity of cell membranes, and induced oxidative stress and apoptosis. In addition, ZnO NP inhibited the proliferation of HGF-1 by regulating the expression of MDM2 and p53. As a common nanomaterial in the oral cavity, the toxicity research of ZnO NPs plays an indispensable and important role in its application and risk assessment.
Materials and methods
Cell culture and ZnO NP treatment
Physical and chemical properties of ZnO NPs.

Immunohistochemistry (IHC)
First, round coverslips were prepared so that HGF-1 cells could grow normally on the round coverslip. The samples were incubated with PBS containing 10% goat serum and 0.2% Triton X-100 at room temperature for 2 h. Then, the samples were incubated with anti-vimentin (Abcam, Cambridge, UK) and anti-keratin (Abcam, Cambridge, UK) primary antibodies overnight at 4°C, which was further combined with corresponding secondary antibodies (Abcam, Cambridge, UK). Finally, the samples were counter-stained with hematoxylin, which were observed and photographed under a microscope.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
MTT assay was used to determine cell viability. In the growth curve experiment, the initial number of seeding cells were 2000, 5000, 8000, and 10,000, respectively. Finally, 8000 cells per well were selected for subsequent experiments. After treatment with or without ZnO NPs for 24 h, cells were rinsed with PBS and then incubated with 5 mg/mL MTT for 4 h. Dimethyl sulfoxide (DMSO) was used to dissolve the formed formazan, and the absorbance at 490 nm was measured. The absorbance value relative to the control was determined as relative cell viability. The data were obtained from at least three independent experiments.
Lactate dehydrogenase (LDH) release assay
LDH Toxicology Assay kit was used to determine LDH release (Sigma, Missouri, USA). After ZnO NP treatment for 24 h, the culture medium was centrifuged at 3000 rpm to obtain a supernatant without cells. LDH detection working solution was prepared, containing lactic acid, INT, and enzyme solution, which was mixed with cell supernatant and incubated for 30 min. The absorbance was measured at 490 nm and 600 nm for dual wavelength assay. The absorbance value relative to the control was determined as relative LDH release. In Supplementary Figure B, the difference between the negative control and the positive control group was detected. The control group was the normal cell culture supernatant, the negative control group was the culture medium, and the positive control group was the LDH-positive control solution. The data were obtained from at least three independent experiments.
Reactive oxygen species (ROS) detection
Intracellular ROS was determined by DCFH-DA (Invitrogen, California, USA). In short, cells were seeded in a 6-well plate at 4 ×105 cells/mL. HGF-1 cells were incubated with 5 μM DCFH-DA reagent for 30 min at 37°C after 24 h ZnO NP treatment. After being washed several times with PBS, cells were cultured in the DMEM medium and were imaged by a fluorescence microscope.
Apoptosis assay by flow cytometry
After HGF-1 cells were exposed to ZnO NPs for 12 h, the apoptosis level was detected using Annexin V-FITC apoptosis assay kit (Beyotime, Shanghai, China). 2 × 106 cells were collected by centrifugation, resuspended in 200 μL buffer, and placed in a 1.5-mL EP tube. Cells were stained with FITC-Annexin V and PI fluorescent dyes. After collection and PBS washing, cells were resuspended in Annexin-binding buffer and then incubated in the dark for 10 min. The apoptosis level was analyzed via flow cytometry.
Quantitative real-time polymerase chain reaction (qPCR)
After the 24-h ZnO NP treatment, HGF-1 cells were collected from 25-cm2 culture flasks using TRIzol reagent (Invitrogen, California, USA) to extract total RNA, and cDNA was synthesized using reverse transcriptase kits (Toyobo, Osaka, Japan). The cDNA was used for quantitative real-time polymerase chain reaction (qRT-PCR) with specific primers according to the SYBR kit (Toyobo, Osaka, Japan). The related mRNA level was analyzed by the 2−ΔΔCT method and normalized to the expression of the β-actin. The primers were listed as MDM2: Forward, 5’-CAGGCAAATGTGCAATACCAA-3’;Reverse, 5’-G GTTACAGCACCATCAGTAGGTACAG-3’; p53: Forward, 5’-GTGGAAGGAAATTTGCGTGT-3’; Reverse, 5’-AGCTGTTCCGTCCCAGTAGA-3’; β-actin: Forward, 5’-CATGAAGATCAAGATCATCGCC-3’; Reverse, 5’-ACATCTGCTGGAAGGTGGACA-3’.
Western blotting
After the 24-h ZnO NP treatment, the proteins were isolated from monolayer adherent cells by RIPA mixed with 1% protease inhibitor and phosphorylase inhibitor, and the concentrations of proteins were examined by a BCA Kit (Beyotime, Shanghai, China). Protein samples were separated using SDS-PAGE, which were further transferred to a PVDF membrane. The PVDF membrane was blocked for 1 h at room temperature, which further incubated overnight with antibodies against β-actin (Sigma, Missouri, USA), MDM2 (Abcam, Cambridge, UK), and p53 (Abcam, Cambridge, UK). After being washed with PBS-T, the PVDF membranes were incubated with secondary antibodies for 1 h at room temperature. The PVDF membranes were visualized and imaged by the GEL imaging system (Bio-Rad, California, USA). The quantification of proteins was analyzed by the software, Image J.
Plasmid constructs and transfection
The cDNA clone of MDM2 was inserted into the pcDNA3.1 vector to construct an overexpression MDM2 recombinant vector, which encoded MDM2. Then, cells were transfected with pcDNA-MDM2 or pcDNA-NC using Lipofectamine™ 3000 (Invitrogen, California, USA) at a density of 1×105 cell/well.
Statistical analysis
All data were expressed as mean ± standard deviation (SD). Statistical significance was analyzed by one-way analysis of variance (ANOVA) and Student t-tests. The p values less than .05 were considered significant.
Results
Identification of human gingival fibroblasts (HGF-1)
After 1- and 4-day culture, the morphology of human gingival fibroblasts (HGF-1) was observed by a microscope. As shown in Figure 1(a), the cells were spindle-shaped or star-shaped at 40× magnification. In addition, vimentin and keratin were detected to confirm gingival fibroblasts, which were the characteristic of HGF-1 cells. The cells were counter-stained with hematoxylin, and nuclei were stained blue. Vimentin staining was positive, showing cytoplasmic staining as brown, while keratin staining was negative, showing unstained cytoplasm (Figure 1(b) and (c)). Furthermore, we tested the growth curves of HGF-1 at different initial seeding numbers of cells. The cells entered the logarithmic growth phase at the 3rd and 4th days and then reached the plateau phase at the 5th and 6th days. HGF-1 cells got the best growth state when 8000 cells per well were seeded to a 96-well plate initially. (Figure 1(d)). Identification of human gingival fibroblasts (HGF-1). (A) Morphological observation of HGF-1 through a microscope (40×). (B and C) Detection of vimentin and cytokeratin in HGF-1 cells by IHC. (D) The growth curves of HGF-1 at different concentrations were determined by MTT assay. n = 3.
ZnO NPs significantly inhibited the proliferation of HGF-1 cells at high concentrations
The effects of ZnO NPs on cell proliferation were tested by MTT assay. ZnO NPs had less effect on cells proliferation at 5–25 μg/mL concentration, but cell proliferation was significantly inhibited when the concentration was greater than 50 μg/mL (Figure 2(a)). Then, the morphology of HGF-1 cells was observed by the microscope after ZnO NP treatment for 4 days. As shown in Figure 2(b), there was no significant change in cell number or morphology after treatment with 20 μg/mL ZnO NPs. After exposure to high concentrations of ZnO NPs, the number and morphology of HGF-1 cells changed significantly. Effects of ZnO NPs on the proliferation of HGF-1 cells at different concentrations. (A) The proliferation of HGF-1 cells was detected by MTT assay after ZnO NP treatment. (B) The changes of cell morphology were observed at 4 days through a microscope. n = 3. **p < .01, ***p < .001.
ZnO NPs caused significant toxicity to HGF-1 cells at high concentrations
We further tested three indicators to investigate the toxicity of ZnO NPs to HGF-1 cells, namely, lactate dehydrogenase (LDH) released, ROS level, and apoptosis level. First, the integrity of the cell membrane was detected by cytoplasmic LDH release. ZnO NP treatment had no significant effect on cytoplasmic LDH release at low concentrations, while LDH release significantly increased at high concentrations (Figure 3(a)). Then, intracellular ROS was detected after ZnO NP treatment. There was no significant effect on ROS level at low concentrations, which significantly increased after high concentrations of ZnO NP treatment (Figure 3(b) and (c)). The effect of ZnO NPs on cell apoptosis also presented a similar trend, showing no effect at low concentrations and increased apoptosis at high concentrations (Figure 3(d) and (e)). Effect of ZnO NPs on the toxicity of HGF-1 cells at different concentrations. (A) The integrity of the cell membrane was assessed by LDH release after ZnO NP treatment. (B and C) ROS was measured by DCFH-DA in HGF-1 cells after ZnO NP treatment. (D and E) Cell apoptosis was assessed via flow cytometric analysis after ZnO NP treatment. n = 3. **p < .01, ***p < .001.
ZnO NPs at high concentration inhibited the cell proliferation, accompanied by the changes in MDM2 and p53 expression
MTT assay clearly showed that high concentration of ZnO NPs inhibited the proliferation of HGF-1 cells (Figure 4(a)). In addition, qPCR and Western blotting were used to determine the expression of MDM2 and p53 in HGF-1 cells after high concentration of ZnO NP treatment. In the qPCR results, MDM2 mRNA expression decreased, but p53 relative mRNA expression increased in HGF-1 cells (Figure 4(b) and (c)). Western blotting results were consistent with qPCR results, showing decreased expression of MDM2 protein and increased expression of p53 protein (Figure 4(d)). ZnO NPs at high concentration inhibited the cell proliferation, accompanied by changes in MDM2 and p53 expression. (A) The proliferation of HGF-1 cells was detected by MTT assay after high-concentration ZnO NP treatment. (B and C) The mRNA levels of MDM2 and p53 in HGF-1 cells were determined by qPCR. (D) The protein level of MDM2 and p53 in HGF-1 cells were detected by western blotting. n = 3. *p < .05, **p < .01, ***p < 0.001.
ZnO NPs at high concentration regulated p53 to affect the cell proliferation by inhibiting MDM2 expression
To explore the underlying molecular mechanism of ZnO NPs, the overexpression vector was constructed to overexpress MDM2 in HGF-1 cells for the following experiments. After high-concentration ZnO NP treatment, the expression of MDM2 and p53 were determined by qPCR and Western blotting in HGF-1 cells. Both in gene and protein results, MDM2 expression increased after overexpression of MDM2 in HGF-1 cells, while p53 expression decreased (Figure 5(a)–(c)). Furthermore, cell proliferation results showed that overexpression of MDM2 increased cell viability, which indicated that high concentration of ZnO NPs inhibited cell proliferation through the regulation of MDM2 and p53 levels. (Figure 5(d)). ZnO NPs of high concentration inhibited HGF-1 cells’ proliferation through the regulation of MDM2 and p53 expression. (A and B) The mRNA levels of MDM2 and p53 were detected by qPCR after overexpression of MDM2 in HGF-1 cells. (C) The protein level of MDM2 and p53 in HGF-1 cells were detected by western blotting after high-concentration ZnO NP treatment. (D) The proliferation of HGF-1 cells was detected by MTT assay after high-concentration ZnO NP treatment. n = 3. *p < .05, **p < 0.01, ***p < .001.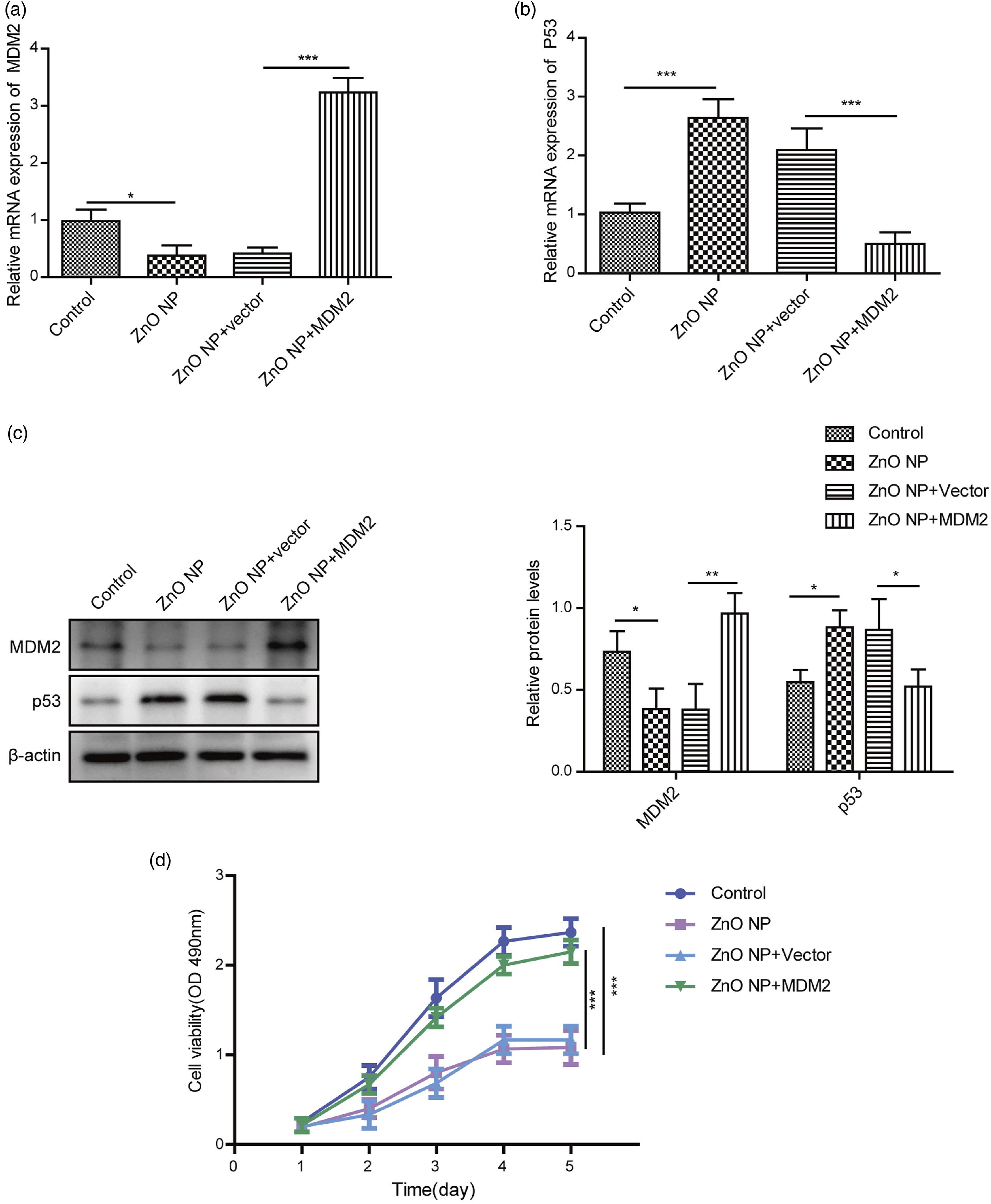
Discussion
This research found that high concentrations of ZnO NPs inhibited cell proliferation and caused toxicity to HGF-1 cells. We further observed that high-concentrations ZnO NPs affected HGF-1 cells through MDM2/p53 regulation. Our study explored the effects of ZnO NP on the proliferation and toxicity of HGF-1 cells for the first time.
In the last decade, nanotechnology had been extensively introduced into biomedical applications, including bioassay, drug delivery, diagnostic imaging and biotherapy, and so on. 15 Periodontal dressing is used to protect the wound from mechanical trauma and keep the surgical site stable to encourage the healing process after periodontal surgery. 16 Nanomaterials were also widely used in dentistry, such as antibacterial coatings, nanofillers, and dental implants.17,18 ZnO NPs, as a common nanomaterial, play important role in oral applications. 19
Our research suggested that ZnO NP did not affect cell viability at low concentrations and significantly inhibited cell proliferation at high concentrations in HGF-1 cells. Similar phenomena were also found in primary human nasal mucosa cells and LoVo cells.23,24 In addition, ZnO NP also caused toxicity to HGF-1 cells at high concentrations, manifested by the destruction of cell membrane integrity and increased oxidative stress and apoptosis levels. LDH leakage was also induced in a concentration-dependent manner in HGF-1 cells after exposure to different concentrations of ZnO NPs. Metal oxide NPs could promote generation of reactive oxygen species and cause oxidative stress by interfering with cell metabolism, damaging DNA, and interactions to impair antioxidant mechanisms. Previous studies also suggested that ZnO NPs induced cell apoptosis mainly through the mitochondrial-independent pathways.
p53 protein was closely related to a series of activities such as DNA damage repair, cell cycle regulation, cell differentiation, and apoptosis. 20 As an important inhibitor of p53, MDM2, bound to p53 protein to form a negative feedback regulatory loop, had p53-dependent activity.21,22 Our study demonstrated ZnO NPs inhibited the cell proliferation through the regulation of MDM2 and p53 expression at high concentrations. Previous studies also showed ZnO NPs inhibited the proliferation of mast cells by decreasing the expression of MDM2. 14
Our study proved that low concentration of Zn NP was safe for oral clinical applications. The study also showed the potential toxicity effects of ZnO NPs and their underlying molecular mechanisms, providing a theoretical basis for the practical application and risk assessment of ZnO NPs in oral clinics. It is necessary to explore the relationship between the cytotoxicity of ZnO NPs and the surface modification, size, and shape of nanoparticles. In addition, whether toxic zinc ions are released during ZnO NP exposure and their effects on cytotoxicity need to be tested. Considering the above information, it is important to further study the toxicity mechanism of ZnO NPs and establish rules for the safe use.
Supplemental Material
sj-tiff-1-het-10.1177_09603271221080237 – Supplemental Material for Effect of nano zinc oxide on proliferation and toxicity of human gingival cells
Supplemental Material, sj-tiff-1-het-10.1177_09603271221080237 for Effect of nano zinc oxide on proliferation and toxicity of human gingival cells by Fang-Chuan Chen, Cong-Ming Huang, Xiao-Wan Yu and Ya-Yu Chen in Human & Experimental Toxicology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their gratitude to for his help for directing our article. This work was supported by Quanzhou Science and Technology Plan Project (Grant number:2019N102S).
Supplemental Material
Supplemental material for this article is available online.
Appendix
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
