Abstract
Apigenin, a flavonoid, is reported to act as an estrogen receptor (ER) agonist and inhibit aromatase enzyme. However, amentoflavone, a biflavonoid bearing two apigenin molecules, has not been evaluated for its endocrine modulatory effects. Besides, it is highly consumed by young people to build muscles, enhance mood and lose weight. In the present study, apigenin was used as a reference molecule and ER mediated as well as ER-independent estrogenic/antiestrogenic activity of amentoflavone was investigated. Antitumor activity of amentoflavone was also investigated in both ER positive (MCF-7 BUS) and triple-negative (MDA-MB-231) breast cancer cells and its cytotoxicity was evaluated in human breast epithelial cells (MCF-10A). Our data confirmed ER agonist, aromatase inhibitory and cytotoxic effects of apigenin in breast cancer cells, where no ER mediated estrogenic effect and physiologically irrelevant, slight, aromatase inhibition was found for amentoflavone. Although selective cytotoxicity of amentoflavone was found in MCF-7 BUS cells, it does not seem to be an alternative to the present cytotoxic drugs. Therefore, neither an adverse effect, mediated by an estrogenic/antiestrogenic effect of amentoflavone nor a therapeutical benefit would be expected from amentoflavone. Further studies could be performed to investigate its in vivo effects.
Introduction
Endocrine disrupters are described by WHO as “an exogenous substance or mixture that alter function(s) of the endocrine system and causes adverse health effects in an intact organism or its progeny or (sub)populations.” 1 Although only synthetic/industrial chemicals have been blamed for endocrine disruption for long time, it is now known that endocrine disrupting potential of natural chemicals needs to be considered as well. 2
Phytoestrogens are an important group of naturally occurring hormone-like chemicals that human are exposed through dietary intake. The preventive effects of phytoestrogens in hormone-dependent breast cancers have been reported for many years.3,4 Epidemiological studies suggested controversial data on the relationship between the use of phytoestrogens and hormone-dependent breast cancers. 5 On the other hand, concerns about the adverse health effects of phytoestrogens has increased in recent years because of their estrogen-like structure and action.6,7 Among them soybean isoflavones (e.g. genistein, daidzein) gained traction with their endocrine modulating effects which indicate a possible similar role for other phytoestrogens as well.7,8
Apigenin, a flavonoid, is found in parsley, onions, oranges, tea, chamomile sp., red wine and beer.9,10 Amentoflavone (I3′, II8″ biapigenin), a biflavonoid, containing two apigenin molecules (Figure 1) is found in dietary supplements such as Hypericum sp. and Gingko Biloba. 11 Amentoflavone supplements are known to be consumed by sportsmen to build muscles, enhance mood and lose weight. The modulatory effect of apigenin on estrogen levels in various experimental models was reported,12,13 but to the best of our knowledge, no data related to the estrogenic/antiestrogenic potential of amentoflavone is reported so far.

Chemical structures of (a) apigenin (5,7-dihydroxy-2-(4-hydroxyphenyl) chromen-4-one) and (b) amentoflavone (8-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-hydroxyphenyl]-5,7-dihydroxy-2-(4-hydroxyphenyl) chromen-4-one).
Phytoestrogens can modulate endocrine system by two mechanisms; (1) acting as agonists or antagonists for ER, (2) altering the enzyme activities which are involved in estrogen synthesis (e.g. CYP19A1) or metabolism (e.g. Sulfotransferases). 14 Therefore, in the present study, our first aim is to investigate whether amentoflavone can cause hormone-dependent endocrine adverse effects when it is used as a dietary supplement. For this aim possible agonist/antagonist effect of amentoflavone for ER was evaluated via E-Screen analysis in ER (+) human breast cancer epithelial, MCF-7 BUS, cells. To evaluate its effect on estrogen synthesis, CYP19A1 (aromatase) inhibitory effect was investigated both by molecular modeling studies and by an in vitro assay where recombinant human CYP19A1 was used. Our second aim is to investigate whether amentoflavone can decrease proliferation of hormone-dependent breast cancer cells as a result of its possible antiestrogenic potential. To achieve this goal, antiproliferative effect of amentoflavone was explored in ER (+) MCF-7 BUS and human triple-negative breast cancer (MDA-MB-231) cell lines by MTT assay. Its cytotoxic potential was also investigated in normal human epithelial breast (MCF-10A) cells to evaluate its selectivity against cancer cells. All these effects of apigenin was also tested in the present study to compare its effects with amentoflavone, a biapigenin.
Materials and methods
Cell cultures and chemicals
Apigenin and amentoflavone were purchasing from Sigma-Aldrich Co. with a purity of ≥97% and ≥99% respectively. Ketoconazole and 17β-estradiol (from Sigma-Aldrich Co.) were used as positive controls for the experiments. MCF-10A cell line was purchased from ATCC, MCF-7 BUS and MDA-MB-231 cell lines were kindly provided by Prof. Ana M Soto from Tufts University, USA and Prof. John Meerman from Leiden University, The Netherlands, respectively. MDA-MB-231 and MCF-7 BUS cells were grown in 10% FBS (Gibco, Thermo Fisher Sci.) containing DMEM (Gibco, Thermo Fisher Sci.) and MCF-10A cells were grown in DMEM: F12 (Gibco, Thermo Fisher Sci.) which is supplemented with 20 ng/ml hEGF (Peprotech Inc.), 1 mg/ml hydrocortisone (Sigma-Aldrich Co.), 10 mg/ml insulin (Sigma-Aldrich Co.) and 5% horse serum (Gibco, Thermo Fisher Sci.). Charcoal-Dextran FBS, phenol red free DMEM, MEM non-essential amino acid solutions, sulforhodamine B (from Gibco, Thermo Fischer Sci.) and MTT dye (Sigma-Aldrich Co.) were used for E-Screen and MTT analyses. The 4 × 10−3 M stock solution of 17β-estradiol was prepared in ethanol. Ketoconazole, apigenin and amentoflavone was dissolved in DMSO at 4 × 10−2 M concentration.
E-Screen analysis
E-Screen assay was performed as described in the study of Soto et al. 15 with minor modifications. Briefly, MCF-7 BUS cells were plated in 96 well plates with density of 6000 cells per well. The cells were incubated at 37°C in humidified atmosphere which was contained 5% CO2 for 24 hours. After the attachment period, cultivation media (DMEM with 10% heat inactivated FBS) was replaced with 150 µl hormone-free experimental media (phenol red free DMEM with 10% Charcoal-Dextran FBS, 1% MEM-NAA, 1% Sodium pyruvate) and 50 µl the test substances were added into wells for estrogenic activity evaluation. The cells were treated either with the 17β-estradiol (positive control), hormone-free experimental media (negative control), apigenin (reference molecule) or amentoflavone for 144 hours. 17β-estradiol is a natural ligand for estrogen receptor and was tested at 10−13 M, 4.7 × 10−13 M, 1.4 × 10−12 M, 4.2 × 10−12 M, 1.23 × 10−11 M, 3.7 × 10−11 M, 1.11 × 10−10 M, 3.33 × 10−10 M and 10−9 M concentrations. Apigenin and amentoflavone were tested at 4.7 × 10−9 M, 14 × 10−9 M, 4 × 10−8 M, 12.3 × 10−8 M, 3.7 × 10−7 M, 1.11 × 10−6 M, 3.33 × 10−6 M and 10−5 M doses which is the range of effective concentrations reported for phytoestrogens which have weaker estrogenic effects compared to 17β-estradiol. The final concentration of ethanol was 0.0000001% in 17β-estradiol studies and the final concentrations of DMSO was 0.1% in apigenin and amentoflavone studies. At the end of the treatment period, experimental media was removed. Cell proliferation was evaluated with sulforhodamine B assay. 16 The cells were fixed with 10% TCA solution at +4°C for 30 minutes and stained with 0.4% sulforhodamine B in 1% acetic acid solution. Excessive dye was washed with 1% acetic acid and plates were dried at room temperature for 24 hours. Stained cells were solubilized with 10−2 M tris base solution (pH = 10.7) and the optical densities were measured at 492 nm via Themo Scientific Varioscan Flash multiplate reader. The estrogenic activities of apigenin and amentoflavone were calculated as proliferative effect (PE). Proliferative effect was measured as the ratio between absorbances of tested substances and vehicle controls. Estrogen-dependent proliferative effect of amentoflavone was only investigated in MCF-7 BUS cells but not in MCF-10A and MDA-MB-231 cells since they both were not expressing ER alpha, shown by western blot analysis (data not shown).
Molecular docking studies
The crystal structure of human placental CYP19A1 (hCYP19A1) in complex with androstenedione was obtained from the Protein Data Bank (pdb entry: 3eqm, 2.9 Å). Hydrogens atoms were added using the protonate 3D tool of the MOE software package (v2015.10, Chemical Computing Group Inc., Montreal, Canada). All water and buffer molecules and the ligand were deleted, and the protein was saved as a mol2-file. Three-dimensional molecular structures were constructed for all ligands using the MOE software package. A steepest-descent energy minimization protocol was applied using the MMFF94x forcefield. Both ligands were docked into the hCYP19A1 structure using the P450 enzyme settings and the GoldScore scoring function in the GOLD suite package (v5.5, CCDC, Cambridge, UK). For each ligand 50 dockings were performed with default settings.
Aromatase inhibition assay
Aromatase inhibitory activity was evaluated by CYP19/MFC High-throughput Screening kit (BD Bioscience, Oxford, UK). The fluorometric analysis enables high-throughput screening in 96-well plate format with using 7-methoxy-4-trifluoromethyl coumarin (MFC) and human recombinant aromatase enzyme. This assay relies on CYP19 catalyzed reduction of (MFC) to its fluorescent metabolite 7-hydroxy-4-trifluoromethyl coumarin (HFC). Assay was performed according to manufacturer’s instructions. 17 Fluorescence was estimated at 409 nm excitation and 530 nm emission wavelength with Thermo Scientific VarioSkan Flash microplate reader. The assay was assessed by ketoconazole (KET) as positive control/reference inhibitor. Subsequently, apigenin, amentoflavone and KET solutions were prepared by serial dilutions (1:3) starting form 4 × 10−5 to 1.8 × 10−8 M and tested to obtain efficient dose-response curves. IC50 values were calculated for each test substance.
MTT analysis
MCF-10A, MCF-7 BUS and MDA-MB-231 cells were seeded in 96 well plates at a density of 5000 cells/well and incubated at 37°C in a humid atmosphere containing 5% CO2 for 24 hours for cell attachment. Cells were treated with 1.23 × 10−6 M, 3.7 × 10−6 M, 11.1 × 10−6 M, 33.3 × 10−6 M and 10−4 M concentrations of the apigenin and amentoflavone for 24 hours. Test materials were dissolved in DMSO (final concentration 1% in cell culture medium). Control (medium only), vehicle control (1% DMSO) and positive control (10% ethanol) were included in all experiments. At the end of the exposure period MTT assay protocol was conducted as our previous study. 18 Briefly, cells were stained with 1 mg/ml MTT solution in cell media for 4 hours at 37°C. Excessive MTT solution was aspirated and formazan crystals were dissolved in PBS. The optical densities were measured at 550 nm via Thermo Scientific Varioscan Flash multiplate reader. Cell viability of the tested samples was expressed as % viability calculated by taking the vehicle control as 100%.
The degree of selectivity of apigenin and amentoflavone to the cancer cell lines compared to the normal breast epithelial cells is expressed as selectivity index (SI) 19 :
Statistical analysis
Statistical calculations of estrogenic (EC50), aromatase inhibitor and cytotoxicity (IC50) values were performed with Graphpad Prism v5.0 and statistically significant differences were assessed with the Pasw Statistics v.18 by using student’s t test. All samples were investigated in triplicate for each run, and values were expressed as mean ± standard deviation.
Results
Estrogenic activities
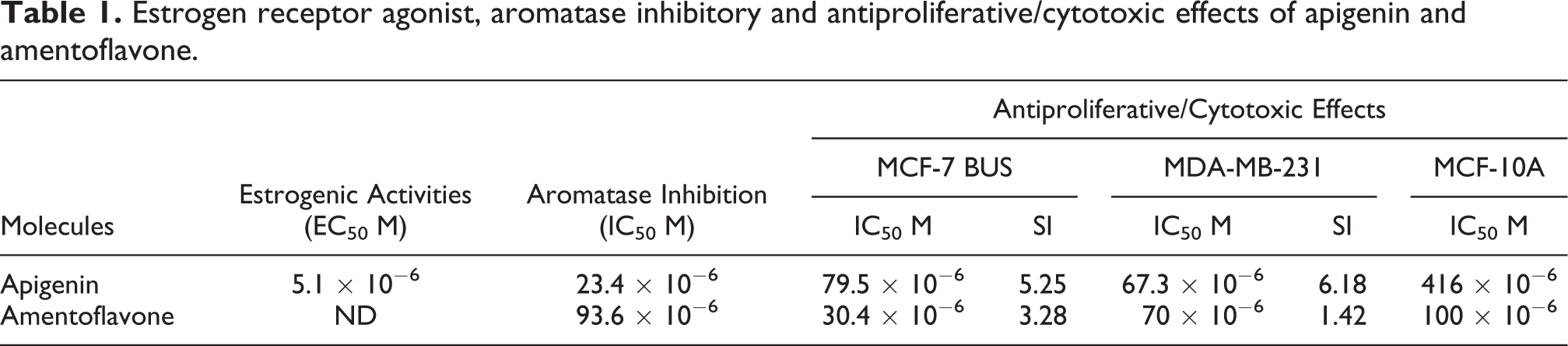
Estrogen receptor agonistic effects of of apigenin and amentoflavone were assessed by E-Screen assay. The ER agonistic effect of apigenin is confirmed in the present study with an EC50 value of 5.2 × 10−6 M (Table 1) which is a very weak effect relative to 17β-estradiol (EC50 value of 1.8 × 10−11 M). No ER agonistic effect of amentoflavone was detected in the E-Screen assay in the present study (Figure 2).
Estrogen receptor agonist, aromatase inhibitory and antiproliferative/cytotoxic effects of apigenin and amentoflavone.

Proliferative effect (PE) curves of 17β-estradiol (as positive control), amentoflavone and apigenin in MCF-7 BUS cells from E-Screen analysis.
Molecular docking studies
In the present molecular modeling studies, apigenin and amentoflavone have been docked into the crystal structure of hCYP19A1 (pdb: 3eqm) (Figure 3). Apigenin is close to the haem group (Figure 3; Purple). The hydroxyl group at the para-position of the phenyl moiety forms a hydrogen bond to the side chain of Thr310. Additional hydrogen bonds are formed with the backbones of Met374 and Leu477. The ligand’s carbonyl group is located close to the side chain of Arg115 (distance < 3.0 Å) and electrostatic interactions may form.

Docked poses of apigenin (purple) and amentoflavone (turquoise) in the crystal structure of hCYP19A1. The haem group is indicated in CPK (Corey, Pauling, Koltun coloring scheme). Hydrogen bonds are indicated in red dashed lines.
Amentoflavone is too large to be docked near the haem group of the CYP19A1 structure. Instead, it may bind at the entrance of the active site and form hydrogen bonds with Lys243, Gln255 and His480 (Figure 3; turquiose). The side chain of Arg192 can form cation-π interactions with the ligand’s phenyl group.
Aromatase ınhibitory activities
Dose-dependent effects of apigenin, amentoflavone and ketoconazole (as a known aromatase inhibitor) on aromatase activity were evaluated (Figure 4) and IC50 values were determined (Table 1). Apigenin inhibited the enzyme activity with an IC50 value of 23.4 × 10−6 M where amentoflavone had IC50 of 93.6 × 10−6 M (Table 1). The inhibitory effect of the compounds was weaker than ketoconazole (IC50 = 2.1 × 10−6 M).

Aromatase inhibition curves of ketoconazole, apigenin and amentoflavone.
Cytotoxic effects
Cytotoxic effects of apigenin and amentoflavone on various human breast cell lines were investigated by MTT method. Decrease in cell viability induced by apigenin (Figure 5(a)) and amentoflavone (Figure 5(b)) showed a monotonic dose-response curve independent from the cell type. Apigenin and amentoflavone decreased cell viability slightly more in human mammary cancer cells (IC50 values were 7.9 × 10−5 M and 3.04 × 10−5 M in MCF-7 BUS; IC50 values were 6.7 × 10−5 M and 7 × 10−5 M in MDA-MB-231 cells respectively) than the normal human breast epithelial cells (IC50 values were 4.2 × 10−4 M and 9.9 × 10−5 M in MCF-10A respectively). Their selectivity for tumor cells was evaluated by calculation of selectivity index (SI) as shown in Table 1.

Dose-response curves of (a) apigenin and (b) amentoflavone on MCF-10A, MCF-7 BUS and MDA-MB-231 cells viability which were evaluated by MTT assay. *, #, ‡ symbols represent statistical significance (p < 0.05) for MCF-10A, MCF-7 BUS and MDA-MB-231, respectively, compared to their vehicle control group.
Discussion
A chemical can exert endocrine disruption by two mechanisms: by acting as agonist/antagonist on hormone receptor or by inducing/inhibiting synthesis of endogenous hormones. Phytoestrogens are natural agonists of estrogen receptors with lower affinities than 17β-estradiol 20 which led them to be used in hormone replacement therapy in postmenopausal women. 4 However, the first mechanism mentioned above for endocrine disruption suggests that this ER agonistic activity of phytoestrogens may also cause endocrine-related adverse effects.4,20 Previously, group of flavonoids, namely apigenin, genistein and kaempferol were investigated for their estrogen receptor binding affinities. 13 The authors reported that apigenin stimulated MCF-7 cell proliferation (EC50 = 0.41 × 10−6 M) and its binding affinity was found 0.04% compared to 17β-estradiol. Similarly, van Meeuwen et al. reported that apigenin increased cell growth with EC50 of 0.44 × 10−6 M. 21 In another study, estrogenic activity of apigenin was investigated by in vivo uterotrophic assay. 22 In this study apigenin caused an increase in uterus weight of exposed rats in a dose dependent manner which indicates ER agonist activity of apigenin. Recently, a research group was conducted a study to evaluate death-associated factor 6 related tumor inhibitory effects of phytoestrogens. 23 Consistent with previous studies, apigenin increased the proliferation of MCF-7 BUS cells instead of inhibiting the tumorigenic cell proliferation, therefore authors did not conduct further studies with apigenin. In the present study apigenin stimulated ER-dependent cellular proliferation in MCF-7 BUS cells. The EC50 value of apigenin was found to be higher in the present study (5.1 × 10−6 M) compared to the previous ones,13,21 this finding may be resulted from different subtypes of MCF-7 cells used in the studies. Unlike apigenin, amentoflavone, a biapigenin that is consumed via herbal dietary supplements, has not been investigated for its estrogenic or antiestrogenic effects so far. In one study molecular docking positions of amentoflavone to estrogen receptors have revealed that amentoflavone didn’t have any binding position for both ERα and Erβ. 24 However, amentoflavone mediated estrogen receptor modulation have not been demonstrated neither in in vitro nor in in vivo biological activity assays. In the present study, amentoflavone did not induce proliferation of estrogen-dependent MCF-7 BUS cells which indicates the lack of an estrogen receptor agonist activity of amentoflavone. This data of ours suggests that consumption of amentoflavone would not cause any estrogen receptor-mediated estrogenic adverse effects.
Aromatase is an enzyme catalyzing the rate-limiting step in estrogen synthesis where androgens are converted to estrogens. Aromatase inhibition can be a double-edged sword; it can be endocrine-related adverse effect of a pharmaceutical and/or dietary supplement (as mentioned above as the second mechanism of endocrine disruption) or it can be used as a therapeutical approach in hormone-dependent breast cancers. Therefore, the binding affinity of apigenin and amentoflavone to aromatase were elucidated with computer-based modeling in the present study. Apigenin, which is a small molecule, was suggested to bind to the active site of hCYP19A1. In contrast, amentoflavone, which is much larger, does not fit inside the active site. Instead, it may bind to the entrance of the active site and thereby preventing the substrates from binding. Effect of both compounds on aromatase activity was further investigated in an in vitro assay where a fluorescence ligand and human recombinant enzyme were used. Our results from in vitro aromatase inhibition assay confirmed the molecular modeling study. Apigenin inhibited the enzyme activity with an IC50 value of 23 × 10−6 M (Table 1). Previously two studies investigated the effect of apigenin on aromatase; apigenin was found to inhibit aromatase in a H295R cell-based assay with IC50 of 20 × 10−6 M 12 and in primary fibroblasts, with IC50 of 0.44 × 10−6 M. 21 The present study confirmed aromatase inhibitory effect of apigenin that was found in those studies, with slight to moderate differences in IC50 values probably because of different test systems used. Unlike apigenin, the effect of amentoflavone on aromatase has not been investigated yet. The present study demonstrated that amentoflavone has a weak aromatase inhibitory effect with IC50 of 93 × 10−6 M (Table 1). According to our findings apigenin and amentoflavone were approximately 10 and 44 times less active, respectively, than the reference aromatase inhibitor, ketoconazole. Thereby our data suggest that amentoflavone, at the daily supplementary doses, would not cause any endocrine modulating effect by decreasing estrogen synthesis. Besides, this data of ours also indicate that amentoflavone does not seem to be a potential candidate, as an aromatase inhibitor, for hormone-dependent breast cancer treatment.
It is well known that National Cancer Institute and others have been exploring the vast resource of natural products for potent and selective anticancer agents but as yet, the success rate in discovery of such molecules has remained very low.25,26 Flavonoids are suggested to play beneficial role in hormone-dependent breast cancer because of their ER-dependent and/or -independent antiestrogenic activity. 4 Previously, bioactive compounds from propolis were investigated for their suppressive effect against both ER (+) MCF-7 and triple-negative MDA-MB-231 breast cancer cell proliferation. 27 According to the study, MCF-7 cell growth was suppressed at 39 × 10−6 M of apigenin in 24 hours treatment study and at 25 × 10−6 M of apigenin when the incubation time increased to 48 hours. Authors reported that apigenin inhibited MDA-MB-231 cell growth at 10−3 M and 88 × 10−6 M in 24 hours and 48 hours incubation, respectively. Similarly, Bai et al. investigated molecular mechanisms of apigenin cytotoxicity on MCF-7 cells. 28 Apigenin showed dramatic decrease in cell viability which was mediated by induction of apoptosis and necrosis. In accordance with previous findings, apigenin reduced number of MCF-7 BUS cells in a dose dependent manner in the present study. Selectivity for cancer cells is an important feature for a cytotoxic chemotherapeutic agent which can be expressed as SI; the higher SI value indicates better selectivity while low SI (generally <2) indicates the possibility of the compound to cause a cytotoxicity as a side effect. 19 Both apigenin and amentoflavone were found to have SI values higher than 2 in MCF-7 BUS cells. Apigenin seems to be more selective than amentoflavone in both ER (+) and ER (−) cancer cells where amentoflavone has lower selectivity (SI = 1.42) for triple-negative breast cancer cells (Table 1). Amentoflavone, with a moderate to low selectivity for cancer cells, does not seem to be a promising candidate for ER (+) breast cancer therapy. This finding is consistent with our data from E-Screen and aromatase inhibition assays which reported no ER-dependent or independent antiestrogenic effect for amentoflavone. Furthermore, amentoflavone was not selectively cytotoxic to triple-negative (MDA-MB-231) breast cancer cells indicating that it might result in general cytotoxicity at concentrations that is needed to cause a therapeutic effect.
As a conclusion, in the present study apigenin showed ER agonist and aromatase inhibitor effect at sub-cytotoxic concentrations in cell-based analyses. These effects of apigenin could balance each other; acting as a weak estrogen receptor agonist while having a weak antiestrogenic effect by inhibiting the endogenous estrogen synthesis. These two effects might result in a total non-modulation of estrogen action. Besides apigenin was found to have cytotoxic effect on both ER (+) and triple-negative breast cancer cells in the present study, which seems to be independent from its endocrine modulating effect. With the present data, it is not possible to draw a definite conclusion for the results of endocrine-related effects of apigenin, whether it will be beneficial or harmful. This conclusion of ours is in accordance with Rietjens et al. 4 who reported that the current evidence on potential health benefits of phytoestrogens is not apparent enough to clearly outweigh the possible health risks implying impossibility of making a certain conclusion on possible health effects of phytoestrogens.
To the best of our knowledge, for the first time, we investigated estrogenic or antiestrogenic actions of amentoflavone via in vitro experiments. Our data indicate no potential ER-dependent or -independent (aromatase inhibition) estrogenic/ antiestrogenic effects of amentoflavone which provokes thoughts about the dual result of this effect; no any unwanted/adverse effect while taken as dietary supplement by sportsperson as well as no beneficial effect in hormone-dependent pathologies, such as breast cancer. In recent studies amentoflavone is suggested to have anti-metastatic effect in human cancer cells 29 and antiviral effect against coronavirus.30,31 Therefore, if amentoflavone is going to become a pharmaceutical agent it must go through a profound toxicity testing which may include investigation of endocrine-related adverse effects. FDA, Center for Drug Evaluation and Research, reported a guideline for evaluating non-clinical endocrine-related drug toxicity which suggests that in vitro receptor binding and enzyme inhibition assays can serve as an initial screen. 32 From that point of view, the present data about ER-dependent and -independent estrogenic/antiestrogenic effects of amentoflavone might serve as base for assessing its potential to cause endocrine effects that are unintentional and adverse. Besides, there is insufficient data in the literature about the metabolism, half-life and metabolites of amentoflavone which are known to be very important in predicting possible toxicity as well as the biological activity of a chemical. Although the present data do not indicate either an adverse effect or a therapeutical benefit of amentoflavone, further in vivo studies could be performed to investigate its fate and kinetics in the living organisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific and Technological Research Council of Turkey under Grant No. 112S375 and Ege University Scientific Research Project under Grant No. 13ECZ008.

