Abstract
Aim:
Hepatic ischemia/reperfusion (I/R) injury is a syndrome involved in allograft dysfunction. This work aimed to elucidate carvedilol (CAR) role in hepatic I/R injury.
Methods:
Male rats were allocated to Sham group, CAR group, I/R group and CAR plus I/R group. Rats subjected to hepatic ischemia for 30 minutes then reperfused for 60 minutes. Oxidative stress markers, inflammatory cytokines and nitric oxide synthases were measured in hepatic tissues.
Results:
Hepatocyte injury following I/R was confirmed by a marked increase in liver enzymes. Also, hepatic I/R increased the contents of malondialdehyde however decreased glutathione contents and activities of antioxidant enzymes. Furthermore, hepatic I/R caused elevation of toll-like receptor-4 (TLR-4) expression and inflammatory mediators levels such as tumor necrosis factor-α, interleukin-6 and cyclooxygenase-II. Hepatic I/R caused down-regulation of endothelial nitric oxide synthase and upregulation of inducible nitric oxide synthase expressions. CAR treatment before hepatic I/R resulted in the restoration of liver enzymes. Administration of CAR caused a significant correction of oxidative stress and inflammation markers as well as modulates the expression of endothelial and inducible nitric oxide synthase.
Conclusions:
CAR protects liver from I/R injury through reduction of the oxidative stress and inflammation, and modulates endothelial and inducible nitric oxide synthase expressions.
Introduction
Hepatic ischemia/reperfusion (I/R) is a devastating pathological syndrome associated with several conditions such as liver transplantation, trauma and liver resection. 1,2 The exact cellular and molecular mechanism for the development and prognosis of hepatic I/R injury is not clear enough to develop effective medicaments to be used in the prevention and treatment of I/R injury. 3 However, several studies reported the decisive role for reactive oxygen species (ROS) over-generation with subsequent activation of its downstream inflammatory mediators in the development and prognosis of hepatic I/R injury. 4,5 Once hepatic reperfusion occurred, Kupffer cells release several types of ROS, including superoxide anion and hydrogen peroxide (H2O2), with subsequent development of the oxidative stress condition. 6 This condition leads to several inflammatory signaling cascades that ultimately lead to hepatic injury. It is thought that the most common cascade is Toll-like receptor-4/nuclear factor-kappa-B (TLR-4/NF-κB) pathway which activates pro-inflammatory mediators including cytokines and chemokines as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) with subsequent leukocyte infiltration. 7 Leukocytes release myeloperoxidase enzyme (MPO) that converts H2O2 to hypochlorous acid which is more dangerous free radical. 8 Over production of hypochlorous acid in inflammation site can mediate protein, lipids, and DNA damage, which contributes to cellular dysfunction and tissue damage. 9
Nitric oxide (NO) biosynthesis is achieved through three isoforms of nitric oxide synthase (NOS). These isoforms are neuronal NOS (nNOS), inducible NOS (iNOS) and endothelial NOS (eNOS). Neuronal one is expressed in tissues of central nervous system. The iNOS is not expressed under physiological conditions, but expressed under inflammatory pathological conditions in several cell types, including hepatocytes, endothelial cells, biliary cells, Kupffer cells, neutrophils, and T lymphocytes. 10,11 The eNOS is regulated physiologically in several cell types, such as liver endothelial cells and hepatocytes. This isoform has a protective effect against several pathological conditions including hepatic I/R. 12
Carvedilol (CAR), (±)-1-carbazol-4-yloxy)-3-[[2-(omethoxyphenoxy)ethyl]-amino]-2-propanol, is a well-known drug acts as a competitive non-selective adrenergic antagonist. 13 It blocks α1, β1 and β2 adrenoceptors. It has a therapeutic role and clinical uses against several diseases as coronary artery diseases, hypertension, heart failure, ventricular remodeling and myocardial infarction. 14,15 CAR uses are based on several pharmacological actions such as anti-inflammatory, antioxidant, anti-apoptotic and anti-fibrotic properties. 16 The potent antioxidant and anti-inflammatory effects of CAR represent the major molecular mechanism, by which it can be used against different pathological conditions. 17,18
Based on these details, the present work focused on the role of CAR against hepatic I/R injury and exploring its effect on inflammation cytokines, oxidative stress and eNOS/iNOS.
Materials and methods
Drugs and chemicals
Carvedilol (MW: 406.47), a white to off white powder, was obtained from Marcyrl Pharmaceutical Industry, El-Obour City, Egypt. Ellman’s reagent, Thiobarbituric acid, 1,1′,3,3′-tetramethoxypropane and Pyrogallol were obtained from Sigma-Aldrich (St. Louis, MO, USA). All other chemicals were obtained from certified sources and were of analytical grade.
Animals and ethical approval
Adult male Wistar albino rats with 16–19 weeks age and weighing 200–250 g were purchased from the animal house of Faculty of Medicine, Assiut University, Egypt. Animals were housed in cages (2 rats per cage) at controlled environmental conditions (23 ± 2°C temperature, 60 ± 10% humidity, 12/12 h light/dark cycle) and kept free on a standard diet and tap water. Animal care followed the guidelines of the National Institute of Health Guide for the Care and Use of Laboratory Animals and was approved by the animal ethics committee (Approved NO: IRB0008719), Faculty of Medicine, Assiut University.
Experimental design
Forty rats were randomly allocated into four groups, each of 10 rats, as follows:
For hepatic I/R, rats were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg). Once adequate anesthesia was obtained, as determined by a negative foot pinch reflex, the animal was restrained in the supine position on the operating bed. The abdomen was shaved and sterilized with povidone-iodine (Betadine®, El-Nile, Egypt). A midline incision was made from the xiphoid process to the pubis. The portal circulation to the liver was carefully dissected. The portal vein, hepatic artery, and bile duct (portal triad) supplying liver were interrupted with the application of a microvascular clamp (ATICO Medical Pvt. Ltd., Egypt) for 30 minutes. After this time elapsed, the clamp was removed and liver was reperfused for 60 minutes. 19 The temperature was maintained by placing the animals under a heating lamp.
Sampling
At the end of reperfusion time, animals were sacrificed by cervical dislocation and blood samples were collected by direct heart puncture for blood collection. Rat sera were separated after centrifugation at 4000 rpm for 15 minutes at 4ºC using a cooling centrifuge (Beckman model L3-50, USA). Livers were dissected out and divided into three pieces; the first part was fixed in 10% formalin-buffered saline solution and used for histopathological and immunohistochemical analysis. Another part was rapidly stored at −80°C and then used for reverse transcriptase PCR and western blot analysis. The other one was homogenized in phosphate buffer saline and used for biochemical analysis.
Assessment of liver function markers
Serum levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were measured according to the method described by Reitman and Frankel. 20 Alkaline phosphatase (ALP) was assessed using method described by Belfield and Goldberg. 21
Assessment of liver oxidant/antioxidant status
Hepatic reduced glutathione (GSH) was determined using Ellman’s reagent. 22 Hepatic lipid peroxidation was evaluated as a level of malondialdehyde (MDA). MDA was determined in the form of thiobarbituric acid reacting substances. 23 The activity of superoxide dismutase (SOD) was assessed in liver tissue homogenate by determining the auto-oxidation of pyrogallol in the presence of SOD. 24 Catalase (CAT) activity was estimated in the hepatic tissues depending on the decomposition of H2O2 by CAT. 25
Measurement of TLR-4 by Western blot analysis
Following liver tissue homogenization in Tris lysis buffer (400 mM NaCl, 0.5% Triton X-100, 50 mM Tris pH 7.4) and a complete protease inhibitor cocktail, protein quantification was done by method of Lowry et al. 26 Equal amounts of protein samples were subjected to electrophoresis using SDS-polyacrylamide gel after which, proteins were transferred to polyvinylidene fluoride membrane. After blocking of the membrane with 5% fat free milk, the membrane was incubated with the primary antibodies (TLR-4 antibody, 1:500 or β-actin antibody, 1:1000; Santa Cruz, California, USA) overnight at 4ºC. Three membrane washing (10 minutes in each) was carried out and followed by membrane incubation with a secondary antibody (1: 5000) for 1 hour at 37ºC. The membrane was rewashed before immunodetection of blotting protein using a chemiluminescence detection kit. Densitometrical analysis was carried out for TLR-4 protein and β-actin using image J® software (National Institutes of Health, Bethesda, USA).
Evaluation of inflammatory cytokines, TNF-α and IL-6
Hepatic contents of TNF-α and interleukin-6 (IL-6) were determined in liver homogenates by using ELISA kits (Wkea med supplies Corp, Changchun Jilin, China) according to manufacturer’s instructions.
Immunohistochemical assay of COX-II
Immunohistochemical staining was conducted according to the manufacturer’s protocols. Paraffin-embedded liver tissue sections (4–5 µm thickness) were prepared, deparaffinized and treated with 0.03% H2O2 for 20 minutes to inhibit endogenous peroxidase activity. For antigen retrieval, sections were boiled in citrate buffer for 10 min, then washed with Tris-buffered saline. Tissue sections incubated with primary COX-II antibody (Thermo Fisher Scientific, Massachusetts, USA) overnight at 4°C. Next, tissue sections were washed three times by PBS before exposure to HRP-conjugated secondary antibody for 30 minutes. After that, sections were washed and incubated with diaminobenzidine for 10 minutes. Sections were stained with hematoxylin as a counterstain. Six random non-overlapping fields were randomly selected in each section for the determination of the area percentage of COX-II expression levels in tissue sections. Measurements were analyzed, and micrographs were taken using Leica Application Suite attached to full HD microscopic imaging system (Leica Microsystems GmbH, Germany).
Assessment of MPO activity
The activity of MPO in liver tissue was measured according to the method of Krawisz et al. 27 In brief, MPO activity was assayed by measuring the H2O2 released following oxidation of O-dianisidine spectrophotometrically at 460 nm.
Determination of eNOS and iNOS by reverse transcriptase PCR
Total RNA was isolated from freshly collected liver tissues using TRIzol reagent (Invitrogen) following manufacturer’s instructions. cDNA was generated by using 1 µg of total RNA as a template and Reverse Aid First Strand cDNA Synthesis Kit® (Thermo-Scientific) according to manufacturer’s instructions. PCR amplification was performed using a conventional thermal cycler (Bio-Rad, USA) and Dream Taq PCR Master Mix® (Thermo-Scientific, USA). The amplification reaction includes one-time denaturation at first (95°C, 3 min), then repeated cycle which compromised denaturation (95°C, 30 sec), annealing 30 sec) and elongation (72°C, 30 sec). This is followed by one-time elongation (72°C, 5 min) at the end of PCR. The primers used in this study were purchased from Vivants Technologies (Malaysia), designed from PubMed, tested for annealing temperatures, and are listed in Table 1. Equal volumes of PCR products were loaded into 2% agarose gel and electrophoresed in Tris-acetate EDTA buffer at 75 volts for 50 minutes. DNA was visualized using ethidium bromide staining. Gel pictures were taken by UV trans-illumination using Gel-Doc imaging system (Bio-Rad, USA). Analysis of producing bands was done by Image J® software (National Institutes of Health, Bethesda, USA) using GAPDH as an internal control.
List of primers.

Histopathological examination
Tissue samples were flushed and fixed in 10% neutral buffered formalin for 72 hours. Tissues were washed in tap water for 12 hours followed by dehydration with serial grades of alcohol. Specimens were cleared with xylene and embedded in paraffin blocks. 5 µm sections were cut by rotatory microtome and stained with hematoxylin and eosin as a general examination stain method. The histopathological examination was done by light electric microscope. 28
Statistical analysis
Statistical analysis was performed by using GraphPad prism software program, version 6.0 (California, USA). Data were presented as means ± SE. comparisons among groups were done using one-way analysis of variance (ANOVA) followed by Tukey’s test as a post hoc test. Values of P < 0.05 were considered statistically significant.
Results
CAR inhibits I/R-induced liver dysfunction
Hepatic I/R induced a significant elevation in serum levels of ALT, AST and ALP as compared to sham control rats. Pretreatment with CAR significantly attenuated hepatic injury by decreasing serum levels of ALT, AST and ALP (Table 2) compared to I/R rats.
Effect of CAR on liver function biomarkers against I/R-induced liver injury.
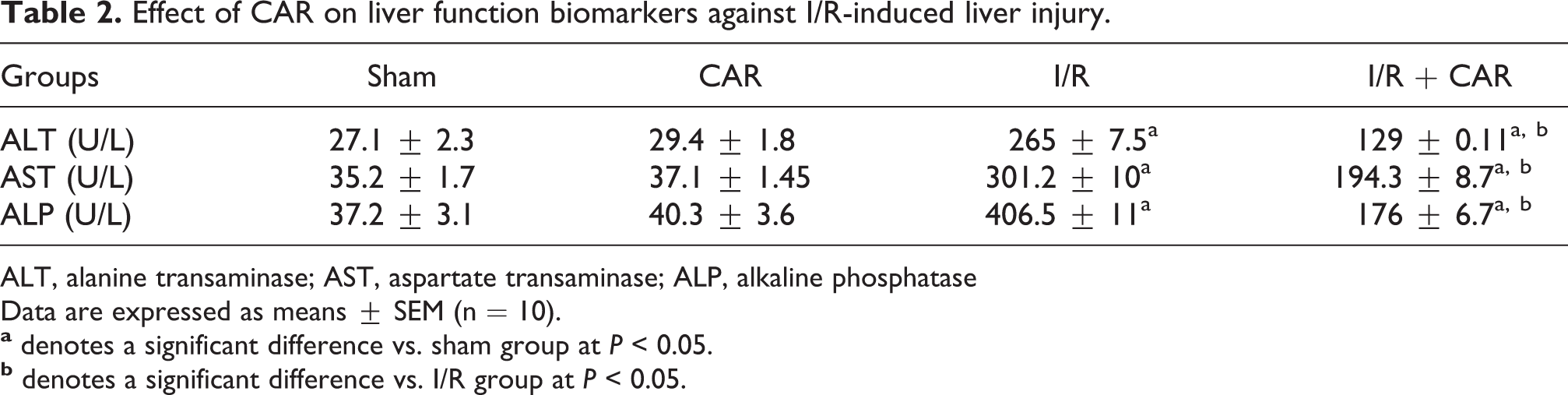
ALT, alanine transaminase; AST, aspartate transaminase; ALP, alkaline phosphatase
Data are expressed as means ± SEM (n = 10).
CAR attenuates I/R-induced oxidative stress injury
Hepatic GSH content was markedly depleted, while MDA content was notably increased in I/R rats as compared to sham control rats. Pre-administration of CAR resulted in a significant elevation in GSH and a remarkable reduction in MDA content as compared to animals challenged with I/R (Table 3). Moreover, enzymatic activities of SOD and CAT after I/R were markedly reduced regarding to sham control rats. Administration of CAR before hepatic I/R produced a significant increase in the activities of SOD and CAT enzymes as compared to I/R group (Table 3).
Effect of CAR on oxidative stress biomarkers against I/R-induced liver injury.

GSH, reduced glutathione; MDA, malondialdehyde; SOD, superoxide dismutase; CAT, catalase
Data are expressed as means ± SEM (n = 10).
CAR normalizes I/R-induced up-regulation of TLR-4 expression
Hepatic TLR-4 expression was significantly up-regulated in rats subjected to I/R injury as compared to sham control rats. Treatment of rats with CAR prior to I/R significantly reduced TLR-4 expression in comparison to non-treated rats (Figure 1).

Effect of CAR on hepatic TLR-4 expression against I/R-induced hepatic injury. Data are expressed as mean ± SEM (n = 10).
CAR attenuates I/R-induced cytokine levels changes
Hepatic levels of TNF-α and IL-6 were significantly elevated in I/R rats as compared to sham-operated rats. Pretreatment of rats with CAR significantly reduced TNF-α and IL-6 levels with respect to the animal paradigm (Figure 2). For further confirmation of the anti-inflammatory effects of CAR, we investigated COX-II expression by immunohistochemical analysis. As shown in Figure 3A and 3B, COX-II expression in hepatic sections obtained from sham and CAR groups was showed negative COX-II expression. On the other hand, a very extensive expression of COX-II was observed in I/R group as compared to sham control group (Figure 3C). However, COX-II expression was significantly decreased in rats treated with CAR before hepatic I/R (Figure 3D). The percentage of COX-II expression was represented in Figure 3E.

Effect of CAR on hepatic TNF-α (A) and IL-6 (B) levels against I/R-induced hepatic injury. Data are expressed as mean ± SEM (n = 10).

Effect of CAR on hepatic COX-II expression against I/R-induced hepatic injury estimated by immunohistochemical analysis. Data are expressed as mean ± SEM (n = 10).
CAR inhibits I/R-mediated elevation of MPO activity
Hepatic MPO activity was significantly elevated by I/R compared with sham control group. Rats exposed to I/R following CAR treatment showed a greater reduction in MPO activity with respect to the animal model (Figure 4).

Effect of CAR on hepatic MPO activity against I/R-induced hepatic injury. Data are expressed as mean ± SEM (n = 10).
CAR mitigates I/R-induced alteration in nitric oxide synthase expression
Rats exposed to I/R exhibited a significant down-regulation of eNOS, while up-regulation of iNOS expressions as compared to sham control rats. However, CAR before I/R resulted in a significant increase in eNOS expression concomitant with a marked decrease in iNOS expression as compared to the animal paradigm (Figure 5 and 6)

Effect of CAR on hepatic eNOS expression against I/R-induced hepatic injury estimated by reverse transcriptase PCR. Data are expressed as mean ± SEM (n = 10).

Effect of CAR on hepatic iNOS expression against I/R-induced hepatic injury estimated by reverse transcriptase PCR. Data are expressed as mean ± SEM (n = 10).
CAR diminishes histopathological changes evoked by I/R injury
As shown in Figure 7, microscopic examination of different group samples revealed that sham and CAR groups were showed a normal histological structure of hepatic parenchyma without detected abnormal changes. Rats subjected to hepatic I/R showed severe diffuse vacuolar degeneration of hepatocytes with dilation and congestion of hepatic blood vessels. Also, showed sever congested sinusoids and periportal inflammatory cells infiltration. Rats that received CAR before hepatic I/R showed an apparent normal hepatic architecture with moderate periportal inflammatory cell infiltration, activation of Kupffer cells and sinusoidal congestion.

Histopathological investigation using H&E staining (200X): Sham and CAR groups showing normal hepatic structure (A and B, respectively). C and D are sections obtained from I/R group showing vacuolar degeneration of hepatocytes, congested hepatic blood vessels and sinusoids (stars) and periportal inflammatory cells infiltration (arrow). CAR pretreated group showing mild vacuolization of hepatocytes (arrow), mild sinusoidal congestion (star) and activated Kupffer cells (arrowhead).
Discussion
Hepatic I/R injury is the main factor attributed to allograft dysfunction following liver transplantation. Excessive ROS production and inflammatory response activation represent the solid foundation for initiation and progression of hepatic I/R injury. 29 CAR is an adrenergic blocker with potent anti-inflammatory and antioxidant effects. 10,11 In this study, hepatic I/R injury was determined and characterized by using a biochemical and molecular markers as well as histopathological examination.
Following hepatic I/R, serum levels of ALT, AST and ALP, which are indicators of liver cell injury, are remarkably increased. These results could be attributed to excessive ROS generation and activation of inflammatory cascades following reperfusion, with subsequent ROS diffusion into liver cells and mitochondrial dysfunction with calcium accumulation leading to formation of permeable pores and cell death. 8 CAR pre-treatment greatly decreased serum levels of ALT, AST and ALP indicating a protective role against hepatic I/R. This effect could be attributed to CAR ability, depending on its anti-inflammatory and antioxidant effect, to enhance mitochondrial function via elevating mitochondrial membrane potential, by which CAR prevents the formation of permeable pores and cell death. 30,31 Histopathological examination revealed that rats exposed to hepatic I/R showed severe diffuse vacuolar degeneration of hepatocytes, dilatation and congestion of hepatic blood vessels and congested sinusoids with periportal inflammatory cells infiltration. Pretreatment with CAR before I/R exhibited apparent normal hepatic architecture with mild periportal inflammatory cells infiltration, activated Kupffer cells and mild sinusoidal congestion.
The conducted work revealed that hepatic I/R significantly increased and decreased MDA and GSH respectively. Furthermore, it markedly decreased the activity of antioxidant enzymes including SOD and CAT. The present results confirmed with an earlier studies. 32,33 These effects could be ascribed to over-production of ROS in hepatocytes in a rate that overwhelms the capacity of endogenous ROS scavengers. This leads to activation of Kupffer cells and producing more amounts and several types of ROS. 2,34 CAR, owing to its antioxidant effect, markedly decreased MDA and greatly increased GSH contents of liver tissues. Additionally, it significantly induced the activity of antioxidant enzymes including SOD and CAT. In accordance, Mohammed et al. showed the antioxidant effect of CAR in hepatic I/R model. 35 CAR reduces oxidant stress depending on free radical scavenging activity due to presence carbazole ring in its structure. 36 The antioxidant effect of CAR in agreement with the study of Wang et al. could be ascribed to CAR activation of nuclear factor erythroid-2 related factor 2/antioxidant response element (Nrf2/ARE) signaling pathways. 17
Free radicals generated from mitochondrial and cytosolic enzymes of liver cells activate ischemic hepatocytes to release nuclear protein high-mobility group box 1 (HMGB1). 37 This protein is a danger-associated molecular patterns and endogenous ligand for TLR4, migrates from liver cells and binds to TLR-4 that located on several cell types of liver cells. 38 These molecular events resulted in induction of TLR4 signaling pathway, immune response activation and further ROS generation. TLR-4 signaling activates NF-κB leading to up-regulation of its downstream components including several cytokines such as TNF-α, COX-II and IL-6. 37 TNF-α is a classical downstream target for NF-κB and represents the most important and common inflammatory mediator for I/R injury. 39,40 TNF-α induces the expression of chemokines and adhesion molecules such as intracellular adhesion molecules (ICAM) and VCAM, which recruit the leukocytes at the site of inflammatory injury. Sequestered leukocytes release MPO enzyme that converts H2O2 into a more toxic radical, hypochlorous acid, which initiates additional inflammatory cascades with subsequent augmentation of the inflammatory responses. 8
In this work, hepatic I/R caused a remarkable induction of TLR-4 protein expression. Furthermore, I/R produced a significant increase in the protein level of inflammatory mediators including TNF-α, IL-6 and COX-II as well as MPO enzyme activity. Administration of CAR before hepatic I/R resulted in significant modulation in the level of TLR-4 protein expression and inflammatory cytokines as well as enzymatic activity of MPO. These results indicated CAR anti-inflammatory effect might be mediated by CAR ability to inhibit TLR-4 signaling which plays a crucial role in the development of inflammatory response and I/R injury. 18 CAR, via its adrenoceptors blocking effect, enhances microvascular perfusion. 15,41 Therefore CAR improved perfusion rate and leads to a decrease in leukocyte infiltration, reduction of the inflammatory responses and amelioration of hepatic I/R injury. 42
The protective effect of eNOS-derived NO against hepatic I/R injury is approved by several works. Peralta et al. displayed that eNOS knockout mice showed a significant increase in liver injury following I/R than wild-type. 43 Also, Rassaf et al. proclaimed that during I/R, genetic deletion of eNOS mitigates the cardio-protection and aggravates the cardiac injury while eNOS induction augments cardio-protection and inhibits cardiac deleterious effect. 44 Furthermore, eNOS protects the hepatocytes by reducing of portal pressure, a decrease of storage/reperfusion damage and enhancement of sinusoids microcirculation. 45 The molecular mechanism for eNOS protective roles in liver I/R might compromise down-regulation of TNF-α, inhibition of macrophage activation, diminishment of leukocytes infiltration and minimize of ROS production. 46 iNOS is a common downstream component of TNF-α. 47 So, NO derived from iNOS has inflammatory pathological role by reacting with superoxide anion to form peroxynitrite which can react directly with all cellular molecules including lipid, protein and DNA resulting in cellular function impairment and cellular death. 48 In the present work, rats challenged with I/R showed a significant decrease in eNOS but a notable increase in iNOS expression. CAR prior administration markedly increased eNOS and greatly reduced iNOS expression. This effect of CAR is confirmed by the results of Akbas et al. which explained the protective effect of CAV in a rat model of hid limb I/R Injury. 49 CAR effect on eNOS expression might be due to activation of phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/protein kinase B (AKT) signaling pathway. 17 AKT is considered the classical upstream regulator for eNOS expression. 50 CAR downregulation of iNOS could be attributed to its inhibitory effect on TLR-4/IL-6 signaling pathway leading to a decrease in the level of inflammatory mediators including TNF-α.
Conclusion
In summary, the current work indicated that CAR pre-treatment before liver I/R confer a protective effect. This effect is founded at least partially on CAR ability to interfere with oxidative stress and TLR-4/IL-6/TNF-α and COX-II signaling pathway as well as modulation of eNOS/iNOS expression, thus CAR is a promising therapy against hepatic I/R injury and there is an urgent need for clinical trials during human liver allograft surgery.
Footnotes
Acknowledgement
The authors are thankful to Prof. Dr. Kawkab A. Ahmed, Professor of Pathology, Faculty of Veterinary Medicine, Cairo University, Egypt, for her kind help in performing the histopathological and immunohistochemical examinations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
