Abstract
Organophosphates are highly toxic compounds as they are involved in irreversible inhibition of acetylcholinesterase, causing various neurotoxic effects via acetylcholine accumulation throughout the nervous system. Traditional treatments for organophosphate poisoning are not effective enough to overcome all the toxic effects. There is a need for alternate treatment of life threatening poisoning of organophosphates. For this purpose a biomimetic nanosponge of poly (lactic-co-glycolic acid) is prepared, characterized and analysed as an antidote for organophosphate poisoning. In this nanosponge red blood cell membranes are used for coating poly lactic co-glycolic acid nanoparticles. In vitro studies are conducted to investigate the retention of acetylcholinesterase activity on the prepared nanosponge as well as to assess the scavenging ability of prepared nanosponge for model organophosphate, chlorpyrifos. In vivo studies are conducted to evaluate the detoxification potential of nanosponge in rabbit model, poisoned with chlorpyrifos. Hepatotoxicity and renal toxicity of nanosponge/chlorpyrifos complex is also studied in survived rabbits and the data is analysed statistically.
Introduction
Poisoning by lethal chemical substances have become a potent healthcare issue worldwide. Moreover, a series of harmful toxicants do not possess specific antidotes. 1,2 Hence, the urgent need of development of safe and effective detoxifying agents has enhanced the application of nanoparticles to sponge and degrade toxic molecules. 3 Recently, nanoparticles have been synthesized and administered to scavenge the toxicant molecules by nonspecific absorption into porous nanostructures. 4,5 Moreover, nanoparticles are also designed so to specifically identify and bind to the toxic moieties through immune recognition, receptor binding or molecular imprinting. 6,7 Special nanoscavengers bearing prophylactic protection against toxicants are also under keen consideration. 8
Cell membrane coating aspect in nanotechnology opens new horizons to design biomimetic and biocompatible synthetic nanomaterials having biological cell-like functions and other diverse applications including broad spectrum chemical detoxification. 9 The cell membrane-camouflaged nanoparticles function as nanodecoys to capture and degrade toxicants or distract them away from their specified cellular target. 10,11 For instance, nanoparticles covered with red blood cell (RBC) membrane shells were able to adsorb bacterial toxins and autoantibodies. 12,13 Whereas, those having shells of macrophages, T-cells and neutrophils were able to fight against several pathological moieties and pathogenic organisms of complexed nature. 14
The sequestration mechanism of biomimetic nanoparticles is the stoichiometric binding between toxicants and the shells of cell membrane. However, the working mechanism of membrane alone may reduce overall detoxification potential, especially against deadly chemicals like organophosphate and other nerve agents. Therefore, additional detoxification modality may be impregnated into the plasma membrane formulations such as PLGA (poly (lactic-co-glycolic acid)). Keeping in mind of this perspective, we design RBC membrane cloaked PLGA nanosponge which consist of PLGA nanoparticles covered by RBC membrane shell.
To study the detoxification potential of PLGA-NS (poly (lactic-co-glycolic acid) nanosponge), organophosphate compound (chlorpyrifos) was considered as the model toxicant which is responsible for severe poisoning through irreversible inactivation of acetylcholinesterase (AChE) present on RBC membranes, blood plasma, cholinergic brain synapses and neuromuscular junctions. From the application perspective, these lethal organophosphates are commonly used as pesticides and also play major role as detrimental nerve agents. 15 The wide range of potency of OP causes difficulty to develop effective detoxification treatment strategies against their poisoning.
In this study, we applied RBC membranes and PLGA to formulate PLGA nanosponge (PLGA-NS). The extraction of OPs the PLGA nanoparticle cores was investigated by using chlorpyrifos (CPS) as model organophosphate. In rabbit model of OP poisoning, PLGA-NS reduced clinical signs of toxicity of organophosphate, lowered concentration of organophosphate in tissues, and boost rabbit survival. These results declare PLGA-NS functioned as a safe and effective nanomedicine and efficiently detoxify toxicant molecules of chlorpyrifos.
Materials and methods
Preparation of PLGA nanosponge and characterization
Red blood cell membrane coated poly (lactic-co-glycolic acid) nanoparticles were prepared by previously discussed protocol.
16
Briefly, PLGA cores were prepared by nanoprecipitation method. First, 100 mg of carboxy terminated PLGA (50:50) (Sigma-Aldrich Resomer® RG 502H, Poly (
The schematic illustration for designing of RBC-PLGA nanoparticles
The Particle size and polydispersity index and zeta potential of PLGA NP, PLGA nanosponge and PLGA nanoponge/CPS complex was evaluated via Zetasizer (ver. 7.11; Serial Number: MAL1127001, Malvern Zen 3600). The structure and morphology of the RBC-NP/CPS complex was observed with transmission electron microscope (TEM) (JEOL-JEM-1010). A drop of the 2 mg/ml of RBC-PLGA NP/CPS complex solution was placed over the carbon coated grid and waited for 5 min to let it dry. After 5 min the grid was rinsed with five drops of distilled water. For negative staining 1% uranyl acetate stain was used. The grid was then dried and imaged by using WI-TEM-36 OR microscope.
Acetylcholinesterase activity assay
The AChE activity was measured in the prepared RBC ghosts (Hollow RBC/RBC membrane) and PLGA nanosponge by using elabscience acetylcholinesterase colorimetric assay kit (Cat. No. E-BC-K174, Elabscience®, USA) based on an improved method. 17,18 The assay principle is an optimized version of Ellman method. The thiocholine produced by AChE enzyme from acetylcholine reacts with 5,5′-dithiobis (2-nitrobenzoic acid) to form a yellow-coloured product, 5-mercapto-nitrobenzoic acid (TNB). The intensity of colour of TNB was measured at 412 nm and was proportional to the acetylcholinesterase enzyme activity, calculated according to formulae and instructions on the kit insert. Unpaired Student’s t-test was performed to evaluate the significance (p Ë‚ 0.05).
Determination of chlorpyrifos removal capability of PLGA nanosponge
In vitro study was carried out to determine the CPS absorption and removal capability of PLGA nanosponge. For this purpose, the PLGA nanosponge (4 mg/ml) was added to PBS to prepare 100 µl solution and then the prepared solution was incubated for 30 min with different concentrations of CPS. Then centrifugation was done at 14,000 rpm by using HERMLE–Z 216 MK microcentrifuge for a duration of 10 min to pellet the nanoparticles. The free content of CPS in the supernatant was determined by using the Thermo Scientific Multiskan™ GO. The OP removal was calculated with the formula:
CPS removal (%) = (1 − CPS content in supernatant layer/total amount of CPS input) × 100%
All experiments were done in triplicate
To observe the effect of PLGA nanosponge dose concentration on CPS removal, 5 µg of CPS was added to 10 µl of PBS and incubated for 30 min with 100 µl of varied concentrations of PLGA nanosponge. After incubation, the process of centrifugation was done at 14,000 rounds per minute (rpm) in a HERMLE–Z 216 MK microcentrifuge for 10 min to settle down the nanoparticles resulting into a pellet formation and then CPS removal was calculated with the above-mentioned formula.
In vivo study for evaluation of CPS detoxification potential of PLGA nanosponge after intravenous administration of CPS
The rabbits were housed in a dedicated animal room of faculty of veterinary science and were given ad libitum feed and water. Firstly, 10% sucrose suspended PLGA nanosponge was prepared. All rabbits were randomized into three groups (n = 10). First group of rabbits was kept as control group. Second treatment group received intravenous injection of CPS (900 mg/kg) alone while rabbits of third treatment group were injected by 200 mg/kg of nanosponge formulation immediately after the administration of CPS injection (900 mg/kg). Survival rate after administration of CPS injection was recorded. Blood (50 μl) was collected instantaneously through jugular vein after death from the rabbits of treatment groups of CPS only. For the PLGA nanosponge group, blood (50 μl) was collected 1 h after CPS injection. From the collected blood, 10 µl of RBC ghosts were derived and the AChE activity on the RBC ghosts was investigated.
In vivo study for determining the CPS detoxification potential of PLGA nanosponge after oral administration of CPS
First of all, 10% sucrose suspended PLGA nanosponge (25 mg/ml) were prepared. All experiments involving animals were carried out in accordance with guidelines for use and care of animals and were also approved by the Institutional Biosafety and Bioethics committee (IBC) of Agriculture, university Faisalabad. The rabbits were divided into three groups with each group having 10 rabbits. First group was control group. The rabbits of second group received treatment of CPS (150 mg/ml) through oral route. The treatment group received PLGA nanosponge formulation (200 mg/kg) intravenously immediately after oral administration of CPS (150 mg/kg). Survival rate was recorded. For rabbits suffering from acute death, blood (50 µl) was collected immediately after death. For surviving rabbits, same quantity of blood was collected 1 h after CPS oral administration and acetylcholinesterase enzyme activity was determined by measuring AChE activity of derived RBC ghosts (10 µl) from collected blood.
Determination of AChE activity recovery through PLGA nanosponge following CPS administration
Blood was collected from RBC-PLGA NPs treated rabbits challenged with oral CPS or intravenous CPS on 0 day, 2 day and 4 day. RBC ghosts were extracted from the collected blood and their AChE activity was determined by previously defined protocol to investigate the extent of recovery after PLGA nanosponge treatment and then statistically evaluated by graph pad prism version 8.0.1 using one-way ANOVA following Bonferroni test for group comparison.
Assessment of hepatotoxicity and nephrotoxicity potential of PLGA nanosponge
Two groups of rabbits were taken for the hepatotoxicity study. The animals of both the groups received an injection of PLGA nanosponge/OP complex. The rabbits of first group were sacrificed on the third day while the rabbits of second group were sacrificed on seventh day. Blood samples before sacrificing the rabbits were taken for biochemical investigation of both groups for liver function tests and renal function tests. The values of plasma ALP, total bilirubin, ALT, albumin, AST, direct bilirubin, total protein, fibrinogen, creatinine and urea were determined by using UV assay commercial kits (Quimica Clinica Aplicada S.A.). Statistical analysis of data was conducted by using graph pad prism software using two-way ANOVA (or mixed model) followed by Bonferroni test for comparison among groups.
For histological analysis, the liver and kidney tissues were preserved in 10% buffered formalin and processed through graded ethanol, sectioned, stained and observed microscopically. 19
Results
PLGA nanosponge characterization
The polymeric cores of poly (lactic-co-glycolic acid) were synthesized and analysed for polydispersity index, hydrodynamic diameter and surface zeta potential and the values were found to be 0.145, 100.09 nm and −22.5 mV respectively (Table 1).
Hydrodynamic diameters, zeta potential and polydispersity index of prepared formulations.

Transmission electron microscopy
In parallel, the vesicles of red blood cell membranes were fused with polymeric cores of PLGA to obtain PLGA nanosponge. The polydispersity index, hydrodynamic diameter and surface zeta potential of the resulting PLGA nanosponge were evaluated by using dynamic light scattering (DLS) technique and the values were found to be 0.231, 122.2 nm and −31.9 mV respectively (Table1).
Thirdly, RBC-NP-CPS complex formation was carried out and the values of polydispersity index and hydrodynamic diameter of the resulting PLGA nanosponge/CPS complex were found to be 0.260 and 124.4 nm respectively and the value of zeta potential was found to be −24.4 mV (Table 1).
TEM images were taken to examine the structure of the RBC-NP-OP complex. TEM images were obtained for organophosphate, CPS (Figure 1).
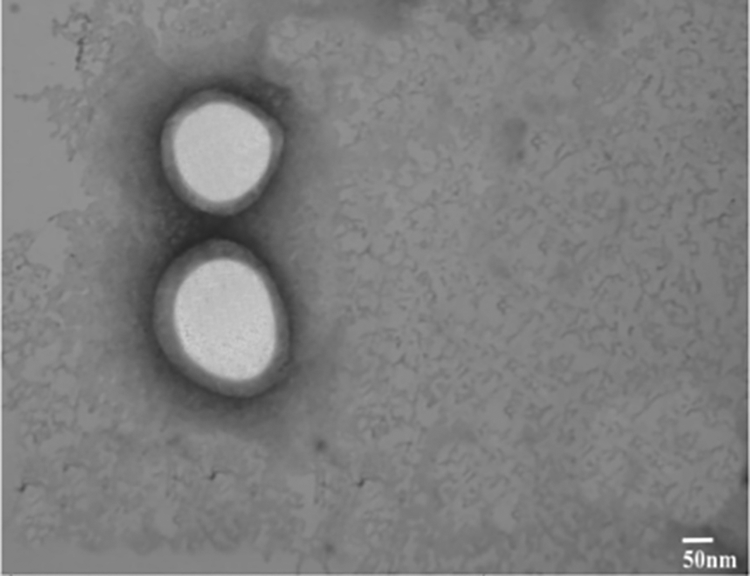
TEM images of PLGA nanosponge/CPS complex.
AChE activity retention in prepared RBC-PLGA nanoparticles
The AChE activity was examined in RBC ghost and prepared RBC-NP to confirm the retention of AChE activity. It was found that AChE activity was largely similar and there was nonsignificant difference among the AChE activity values of both RBC Ghost and prepared RBC-NP. Hence, indicating that there occurred a minimal loss of activity of membrane linked AChE enzyme during or after preparation of nanosponge (Figure 2).

AChE activity determined in RBC ghost and PLGA nanosponge.
In vitro analysis
In vitro analysis of CPS detoxification through PLGA nanosponge
Different concentrations of CPS were incubated with PLGA nanosponge for 30 mins and removal percentage was calculated.
As the concentration of CPS was increased, the removal capability of PLGA nanosponge was decreased. Because same quantity of PLGA nanosponge was used with increasing concentration of CPS. There was a minimum CPS removal percentage at 1000 µg/ml concentration of CPS (Figure 3(a)).
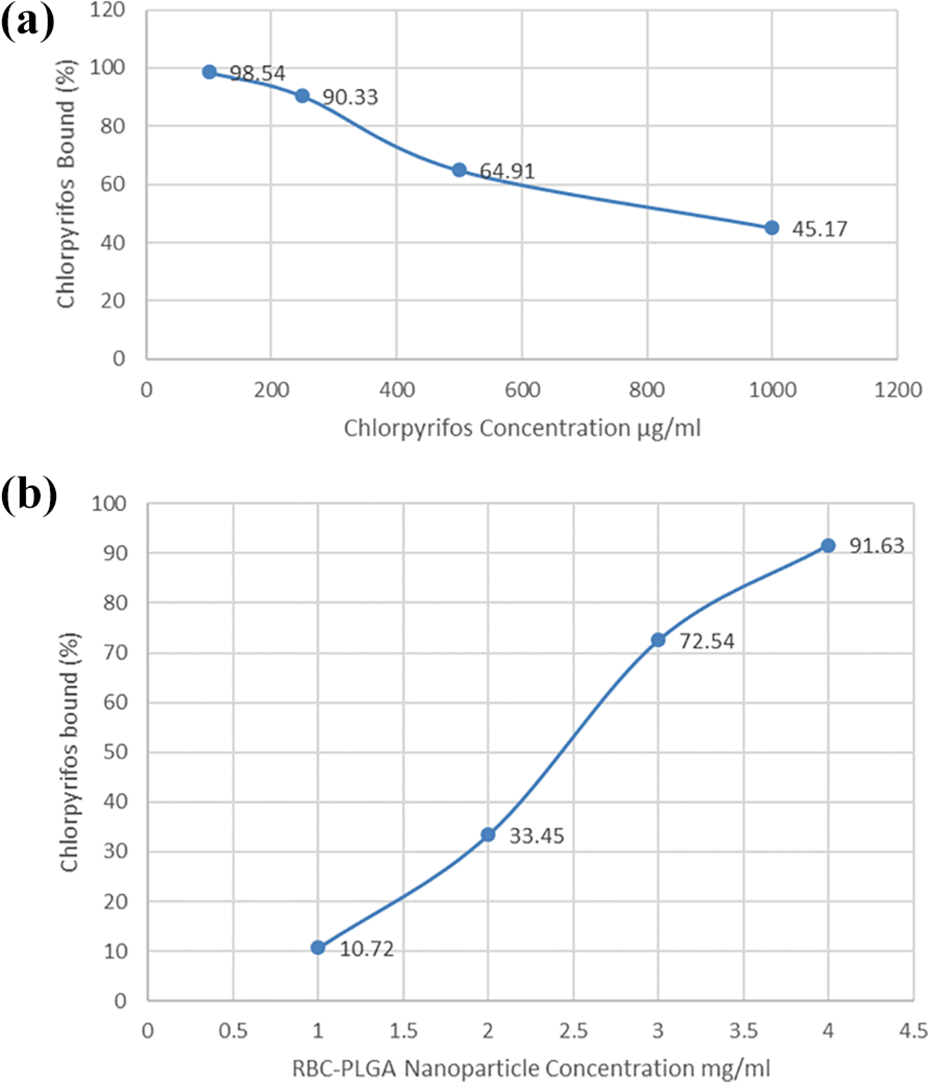
Measurement of absorption efficiency of PLGA nanosponge. (a) Percentage of chlorpyrifos bound to PLGA nanosponge when incubated at different concentrations of chlorpyrifos. (b) Percentage of chlorpyrifos bound to PLGA nanosponge when incubated at different concentration of PLGA nanosponge.
In vitro analysis of CPS removal through different concentration of PLGA nanosponge
As the concentration of the RBC membrane coated PLGA nanoparticles was increased, adsorption capability of RBC membrane coated PLGA nanoparticles was also increased (Figure 3(b)).
In vivo analysis
In vivo detoxification effect of PLGA nanosponge after oral CPS administration
Survival rate and AChE activity: First of all, survival rate was analysed. All the organophosphate (CPS) treated rabbits were died within minutes. Oral LD50 of CPS in rabbits is in the range of 1000–2000 mg/kg. Lethal dose 2000 mg/kg was given to all the rabbits of group A. They were died within 70–105 min. In treatment group B first CPS was given and then without any delay, 150 mg/kg of PLGA nanosponge formulation was administered, all rabbits were survived, showing 100% survival rate as shown in Figure 4(a).

Survival rate of rabbits and RBC-AChE activity of rabbits. (a) Survival rate of group of rabbits administered with oral OP and PLGA nanosponge. (b) Survival rate of group of rabbits administered with intravenous CPS and PLGA nanosponge.
Assaying RBC AChE activity determined that PLGA nanosponge significantly preserved the RBC AChE activity as compared to chlorpyrifos alone treated group (p < 0.05). The AChE activity was quite low for the rabbits suffering from acute death through oral CPS administration whereas, PLGA nanosponge treated rabbits following CPS have relatively higher values of AChE activity (Figure 5(a)).

(a) RBC-AChE activity of group of rabbits administered with oral CPS and PLGA nanosponge. (b) RBC-AChE activity of group of rabbits administered with intravenous CPS and PLGA nanosponge. All error bars represent standard error of mean *p Ë‚ 0.05.
In vivo detoxification effect of PLGA nanosponge after intravenous OP administration
Survival rate and AChE activity: First of all, survival rate was analysed. All the organophosphate (CPS) treated rabbits were died within minutes. Intravenous LD50 value of CPS administered in experimental rabbits was 900 mg/kg, since reported value of oral LD50 of CPS is 1000–2000 mg/kg (Tomlin, 2006). Lethal dose 900 mg/kg was given to all the rabbits of group A. They were died within 50–60 min. In treatment group B first CPS was given and then without any delay, 150 mg/kg of PLGA nanosponge formulation was administered, all rabbits were survived (Figure 4(b)).
The PLGA nanosponge treatment following CPS showed significant retention of RBC AChE activity in blood circulation as compared to the chlorpyrifos alone treated group (p < 0.05). The statistical significance in AChE activity was observed between the chlorpyrifos alone treated group and the PLGA nanosponge plus CPS treatment group (Figure 5(b)).
Recovery of AChE activity following PLGA nanosponge
AChE activity recovery following administration of CPS was evaluated by using circulatory RBC AChE activity as marker. The investigation was done for both oral and IV route of CPS separately. It was found that for both experimental routes, the AChE activity of RBC became normal by the day 4 after administration of PLGA nanosponge following OP (Figure 6(a) and (b). Statistical significance was observed in the AchE activity at day 4 as compared to day 0 after administration of PLGA nanosponge following CPS. This shows the ultimate removal of CPS and the replenishment of AchE activity in the circulation.
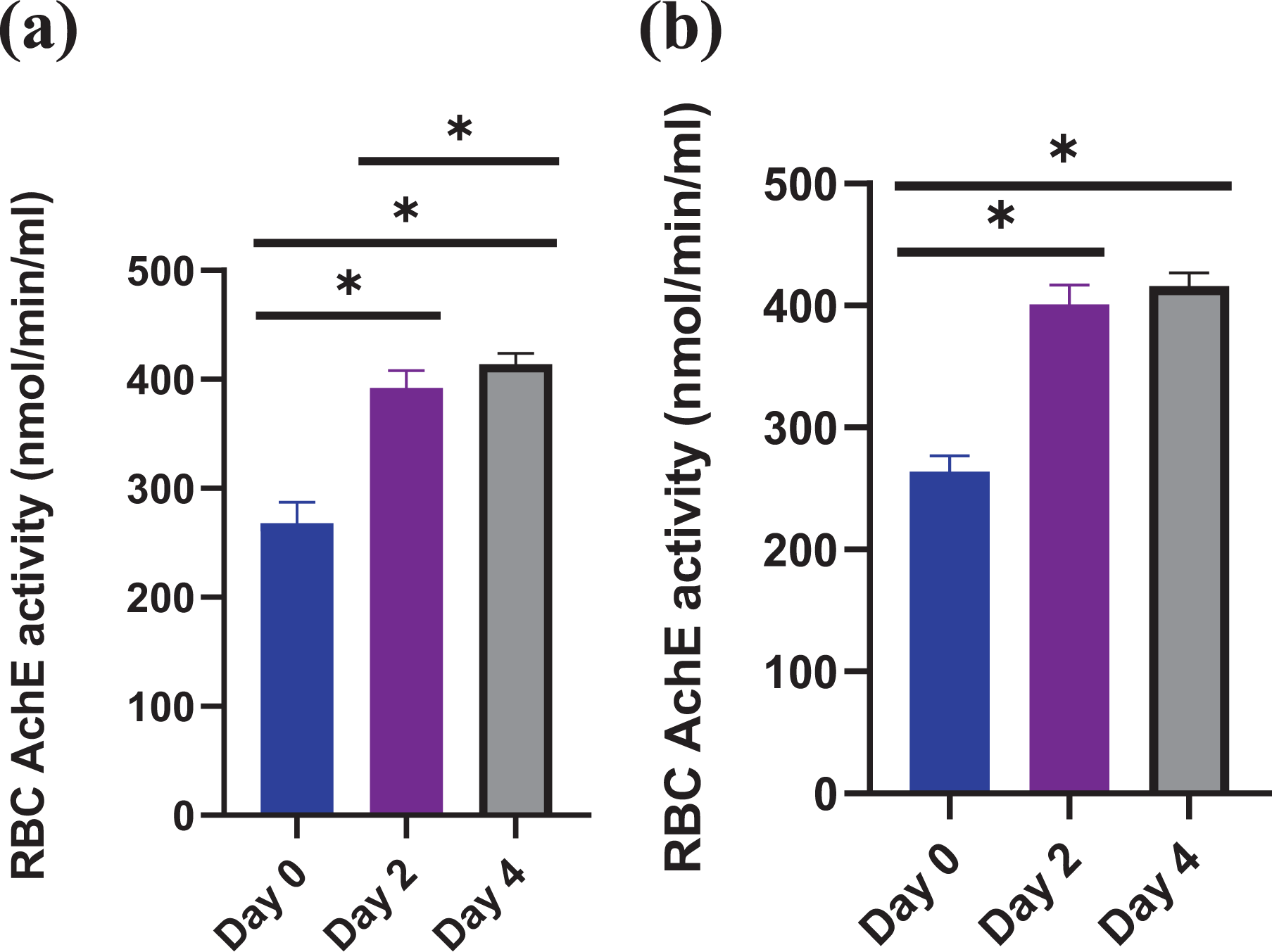
(a) RBC-AChE activity of survived rabbits at day 0, day2 and day 4 following oral CPS challenge. (b) RBC-AChE activity of survived rabbits at day 0, day2 and day 4 following I.V CPS challenge.
Hepatotoxicity and nephrotoxicity potential of nanosponge
The photomicrograph of kidney of the control group showed the normal parenchyma. The capsule appeared normal in structure. The proximal and distal convoluted tubules were also normal. The group B rabbits also showed normal renal parenchyma at third day after PLGA nanosponge /CPS complex administration as the toxicological changes were not observed by the use of nanomedicine. In the group C, the microscopic toxicological changes were also not observed and glomerular texture was appeared normal, indicating the safe use of nanomedicine (Figure 7(a) to (c))

Histology sections of kidney and liver (20×). (a) Photomicrograph of control kidney. (b) Photomicrograph of kidney over day 3 after administration of PLGA nanosponge/CPS complex. (c) Photomicrograph of kidney over day 7 after administration of PLGA nanosponge/CPS complex. (d) Photomicrograph of control liver. (e) Photomicrograph of liver over day 3 after administration of PLGA nanosponge/CPS complex. (f) Photomicrograph of liver over day 7 after administration of PLGA nanosponge/CPS complex. (g) Comprehensive biochemical parameters study over day 3 and day 7 after administration of PLGA nanosponge/CPS complex (n = 10), mean ± SD values of ALT, AST, ALP, total bilirubin, direct bilirubin, total protein, albumin, bilirubin, creatinine and fibrinogen. Statistical analysis was performed by graph pad prism software using two-way ANOVA following Bonferroni test for determining the comparison among groups and the result showed nonsignificant values (p > 0.05).
The liver parenchyma of the control group appeared normal. The hepatic cells were intact with normal sheets of hepatic cords. Hepatic triad and central vein were also normal. Hepatic parenchyma also appeared normal at day 3 in the group B in which nanomedicine and pesticide were used in combination. Nanomedicine showed no toxic effect on hepatic tissues. In the group C in which nanomedicine-pesticide complex were used, the toxic changes were reduced indicating the safe administration and ameliorative potential of nanomedicine against pesticide (Figure 7(d) to (f)).
The results showed high safety profile of PLGA nanosponge. PLGA nanosponge is quite nontoxic to be administered as it had no deleterious effect on kidney and liver organ tissues.
Blood samples analysis indicated that the mean ± SD values of AST, ALT, total protein, albumin, bilirubin, creatinine, globulin remain statistically nonsignificant for the blood samples collected over day 3 and day 7 (Figure 7(g)).
Discussion
Nanoparticles have been prepared to detoxify organophosphate poisoning. Polymeric nanoparticles coated with cellular membranes were shown good functionality in reducing the organophosphate poisoning. The zeta size of the PLGA nanosponge was 122.2 nm which was found to be within required range. 20 Moreover, Zeta potential is necessary in determining the stability of the formulation. It tells us that how much the formulation value will be in negative or positive. Higher the negative or positive value, more will be the stability. Zeta potential of PLGA nanosponge was −31.9 mV which was considered to be stable.
The hydrodynamic diameter of the PLGA nanosponge/CPS complex was found to be 124.4 nm and zeta potential was found to be −24.4 mV. These values indicate that the organophosphate (CPS) reaction had minimal effect on the diameter, stability and structure of RBC-cloaked PLGA nanoparticles. The dynamic light scattering results of size measurements for PLGA nanosponge remained almost similar even after CPS challenge. However, CPS exposure, increased the ζ-potential value of PLGA nanosponge and it may likely, be due to the surface charge shielding effect produced by the binding of CPS molecules to the surface. This finding was similar to previous study which involved the change of zeta potential of nanoparticles due to dichlorvous, organophosphate molecules. 20
Transmission electron microscopy (TEM) images showed that CPS had little or no effect on the physicochemical properties of prepared biomimetic nanomaterial as the core shell structure of RBC-PLGA NP remained intact following CPS reaction, representing unilamellar membrane covering over the PLGA nanoparticles (Figure 1).
It was investigated that the AChE enzyme retained over the surface of PLGA nanosponge as AChE activity was measured to be 421.07 nmol/min/ml in the RBC ghost and 411.98 nmol/min/ml in the prepared PLGA nanosponge, demonstrating largely similar AChE activity of RBC ghosts and PLGA nanosponge (Figure 2). This showed that the membrane proteins form effective coating/shell over nanoparticle moieties. Moreover, in statistical term, no significant difference was observed between the measured AChE activity of RBC ghosts and PLGA nanosponge indicating little loss as well as little alteration of membrane-associated AChE enzyme activity in prepared nanosponge. This finding was consistent to previous findings that showed retention of surface protein functionality over the membrane-camouflaged nanoparticles. 21 –23
The experiment for inspection of the capacity of PLGA nanosponge to capture OP showed that there occurred association between PLGA nanosponge and CPS in a concentration-dependent manner as the concentration of CPS was increased, the removal capability of PLGA nanosponge was decreased due to the saturation. It was evaluated that when different concentrations of CPS were used with 4 mg/ml of PLGA nanosponge, maximum removal capability was shown with minimum concentration of CPS (Figure 3(a)).
We next investigated the detoxification capability of PLGA nanosponge for capturing OP. It was found that as the concentration of PLGA-NS was increased, the concentration of CPS removal in the solution increased accordingly. When the PLGA nanosponge concentration reached to 4 mg/ml, the percentage of CPS removal in the solution was 91.6%. Hence, the PLGA nanosponge/CPS association may occur in a concentration-dependent manner (Figure 3(b)). Hence, it was concluded that concentration of PLGA nanosponge also play an important role in scavenging capability. This study suggests the capability and potential of PLGA nanosponge, as an innovative formulation to scavenge the CPS poison.
It was observed that the survival rate of rabbits was abruptly increased following treatment with PLGA nanosponge as compared to the rabbits of treatment group of organophosphate only, where the survival rate was approximately zero because no single rabbit was survived (Figure 4(a) and (b)). Therefore, approving the sole functionality of PLGA nanosponge as bio scavenger to detoxify the CPS. In comparison to other currently used anti-OP treatments which compete with the acetylcholine or cause blockage of receptors of Ach, the PLGA nanosponge formulation work as an OP remover/adsorber so it is thus less prone to cause anticholinergic side effects such as blurred vision, ventricular fibrillation, dry mouth, nausea, loss of balance, photophobia, dizziness, dilated pupils and extreme confusion. This entirely biocompatible and biodegradable platform also reduced the safety concerns accompanied with administration of nanomaterial as the mortality rate was abruptly decreased following PLGA nanosponge treatment. 24 It was also inferred that the AChE activity of rabbits suffering from acute death, was quite low whereas, AChE activity of PLGA nanosponge treated rabbits following CPS administration was relatively greater. It indicated that PLGA nanosponge act as source of AChE activity (Figure 5). AChE activity recovery with PLGA nanosponge following administration of CPS was determined. It was determined, the AChE activity of RBC approximately reached to normal by the day 4 (Figure 6(a) and (b)).
To investigate the safety of PLGA nanosponge, a comprehensive metabolic panel was performed at three day and seven-day duration, post administration of PLGA nanosponge/CPS complex into healthy rabbits. Compared to rabbits of control group, statistical differences evaluated in all parameters of rabbits of 3 day group and 7 day group, were found to be insignificant (Figure 7). Tissue sections of liver and kidney organs appeared normal, indicating safe use of nanosponge (Figure 7)
Conclusion
In conclusion, we report the development of biomimetic PLGA nanosponge formulation, which combine the competency of the natural RBC membranes and PLGA polymeric material for adsorbing CPS. Moreover, PLGA nanosponge helped to prevent the inhibition of AChE and acted in a dose dependant manner to improve rabbit survival rate. The treated rabbits showed complete AChE activity recovery and no observable liver or kidney injury was seen following the treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
