Abstract
Background
Curcumin has been reported to have many benefits, including anti-inflammatory, anti-cancer, and so on. In this research, we aimed to investigate the function of curcumin on lipopolysaccharide (LPS)-injured H9c2 cells.
Methods
H9c2 cells stimulated by LPS mimic the in vitro model of myocarditis injury. Comparative Toxicogenomics Database (CTD) was applied to detect the genes associated with curcumin. GEO database was used to analyze Intercellular Adhesion Molecule 1 (ICAM1) and CD40 expression in myocarditis patients. KEGG enrichment analysis was employed to investigate the meaningful pathways related to differentially expressed genes. Cell proliferation, apoptosis, expression of ICAM1/CD40/P65- NF-κB, and level of TNF-α, IL-6, and IL-10 were observed by cell counting kit-8, flow cytometry and western blotting assays, ELISA assay, respectively.
Results
After curcumin treatment, the decreased activity of H9c2 cells evoked by LPS was improved. ICAM1 and CD40, which highly expressed in myocarditis patients, were identified as targets of curcumin and negatively regulated by curcumin. Inhibition of ICAM1 or CD40 strengthened the protective effect of curcumin on LPS-evoked H9c2 cells damage, accompanied by increased cell viability and decreased cell apoptosis and inflammation. Additionally, addition of curcumin or depletion of ICAM1/CD40 suppressed p-P65 NF-κB expression.
Conclusions
Curcumin mitigated LPS-evoked H9c2 cells damage by suppression of ICAM1/CD40/NF-κB, providing a potential molecular mechanism for the clinical application of curcumin.
Introduction
Myocarditis refers to the clinical and histological manifestations of a broad range of pathological immune processes in the heart, occurring in the myocardium of both children and adults. 1 There are various causes of myocarditis and the most common type of myocarditis is viral myocarditis, which is caused by viral infection. 2 Previous studies have documented the occurrence of apoptosis in myocarditis infection of the heart, which plays an important role in the pathogenesis of myocarditis.3,4 However, if left untreated, myocarditis wound trigger arrhythmia, severe heart failure, and so on. 5 Although some chemical drugs have certain curative effects on myocarditis, they are often accompanied by more or less side effects. 6 However, several reports revealed that some herbal medicines can attenuate the symptoms of myocarditis, such as Astragaloside IV of Astragalus membranaceus (Fisch.) Bunge and Scutellaria baicalensis Georgi . 2 Therefore, it is of great significance to find effective herbs to improve myocarditis injury.
Curcumin is a polyphenol found in turmeric, which was first discovered by Vogel and Pelletier from the rhizomes of turmeric, Curcuma longa L. 7 Recently, curcumin has attracted wide attention because of its profound medicinal values. 8 In addition to its antioxidant and anti-inflammatory activities, curcumin has also been reported to have health benefits, such as improving brain function. 7 Multiple in vivo studies have also reported its potential against tumors, diabetes, obesity, neurodegenerative disease, and depression. 9 Because of curcumin’s magical properties, it has been marketed in various forms, such as tablets, capsules, ointments, energy drinks, soaps, and cosmetics in several countries. 10 However, how curcumin could present these multitudinous effects has been a clandestine over the years. Analysis from Comparative Toxicogenomics Database (CTD) discovered that Intercellular Adhesion Molecule 1 (ICAM1) and CD40 might be regulated by curcumin. However, the specific mechanism of curcumin action on ICAM1 and CD40 in myocarditis has not been reported.
Lipopolysaccharide (LPS), as the main component of the cell wall of Gram-negative bacteria, is one of the important regulator of shock and inflammation. LPS, which usually cause systemic inflammation, multiple organ failure, as well as cardiac function injury, was usually utilized to stimulate cardiomyocyte toxicity. For instance, LPS can lead to damage in rats and H9c2 cells. 11
Herein, we investigated the molecular mechanism of curcumin suppressing apoptosis evoked by LPS treatment in H9c2 cells, as well as the impact of curcumin on crucial pathway. These findings will afford a novel molecular biology base for the clinical application of curcumin in handling with myocarditis and promote the progression and application of curcumin.
Materials and methods
Data collection
CTD was used to explore the key genes related to curcumin. Gene Expression Omnibus (GEO) database (GSE19496) covering 12 cases of myocarditis samples and 12 cases of normal samples were used to analyze ICAM1 and CD40 expression. Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis was employed to investigate the meaningful pathways related to differentially expressed genes in myocarditis samples.
Cell culture and treatment
Rat myocardial cells H9c2 were bought from American Type Culture Collection (ATCC), and grown in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS) and antibiotic in an incubator at 37°C of 5% CO2. In vitro model of myocarditis were constructed using H9c2 cells stimulated continuously by LPS for 12 h. SiICAM1 (5′-CGGCTGACGTGTGCAGTAATA-3′) and siCD40 (5′-GCAGGCACAAACAAGACTGATCTC-3′) were applied to knockdown ICAM1 and CD40 expression, respectively. Si-con (5′-CGAGAACTCACTGGTCTGACAC-3′) was served as a standard control. These small fragments were transfected into cells by Lipofectamine 2000 following the manufacture’s direction.
Cell viability assay
Cell viability of H9c2 cells was detected by cell counting kit-8 to evaluate the growth ability of H9c2 cells. Briefly, H9c2 cells were seeded in a 96-well plate with 1000 cells/well. After different stimulation, the CCK-8 reagent was put into the culture medium and cultivated for 1.5 h at 37°C. The optical density was read at 450 nm with a microplate reader. The experiment was repeated three times.
Apoptosis assay
The apoptosis rates of H9c2 cells undergoing different treatments were determined by using Annexin V-FITC and propidium iodide (PI) apoptosis detection kit by flow cytometry. After various stimulations, cells were rinsed with PBS and incubated with 10 μL Annexin V-FITC and 5 μL PI for 15 min away from the light at room temperature. Flow cytomery analysis was performed by a FACScan flow cytometry, and the data were processed by FlowJo software. The experiment was repeated three times.
qRT-PCR
TRIzol reagent was used to isolate the whole RNA from H9c2 cells following the supplier’s direction. Reverse transcription was carried out by using PrimeScriptTM RT reagent kit. For analysis of ICAM1 and CD40, a SYBR Green PCR kit was applied. GAPDH was amplified as a standard control. The relative expression levels of ICAM1 and CD40 were assessed by using 2−ΔΔCT method. The experiment was repeated three times.
Western blot assay
Total protein was isolated from H9c2 cells with the support of RIPA lysis buffer and quantified with the assistance of the bicinchoninic acid protein assay kit. Nuclear extracts from H9c2 cells were also prepared using a nuclear protein extraction kit (Beyotime Biotechnology, China). Thereafter, equal amount (20 μg) of protein was separated by SDS-PAGE and then blotted onto the PVDF membranes. The membranes were incubated with the primary antibodies against ICAM1 (1:1000, 4915, CST), CD40 (1:1000, 86,165, CST), p-P65 NF-κB (1:1000, 3033, CST), P65 NF-κB (1:1000, 8242, CST), p-IkBα (1:1000, 2859, CST), N-cadherin (1:1000, 13,116, CST), and GAPDH (1:1000, 5174, CST) at 4°C for 24 h. Then, the membranes were washed by PBS and incubated with secondary antibody for 2 h at ambient temperature. Finally, the bands were visualized by using ECL detection kit and quantified by using Image J software. The experiment was repeated three times.
Enzyme-linked immuno sorbent assay (ELISA)
The level of TNF-α, IL-6 and IL-10 in cellular supernatant were measured by ELISA kits according to the manufacturer’s instructions (R&D, Minneapolis, MN, USA). All measurements were performed in triplicate. TNF-α (catalog: RTA00), IL-6 (catalog: R6000 B) and IL-10 (catalog: R1000) ELISA kit was purchased from R&D (Minneapolis, MN, USA).
Statistical analysis
Statistics were analyzed by using GraphPad Prism 8.0 (GraphPad Software, USA). Data used in this study were exhibited as mean ± standard deviation (SD) and statistically analyzed by ANOVA followed by Bonferroni’s post hoc test. Data were deemed as significant when p less than 0.05.
Results
The decreased viability and increased apoptosis of H9c2 cells triggered by LPS were suppressed by curcumin
To detect the impacts of curcumin on LPS-evoked cell damage, optimum dose of curcumin on H9c2 cells that has no remarkable inhibitory effect on cell proliferation was first elected. As exhibited in Figure 1(a), 0–20 µM curcumin had no evident effect on cell proliferation, but 50 μM curcumin notably reduced cell viability. Moreover, the data in Figure 1(b) revealed that curcumin attenuated LPS-evoked cell proliferation suppression in a dose-dependent mode. We observed that 20 or 50 μM curcumin significantly improved LPS-induced cell viability decline. Therefore, 20 μM curcumin was picked up in the next experiments. Subsequently, the data presented in Figure 1(c) and (d) exhibited that decreased cell viability and elevated cell apoptosis rate induced by LPS stimulation were inhibited by curcumin treatment. These results indicated that curcumin availably improved LPS-evoked cell damage. Curcumin reduced lipopolysaccharide (LPS)-induced H9c2 cells damage (a). Analysis from CCK-8 showed that the viability of H9c2 cells changed under the stimulation of different concentration of curcumin (b). The viability of H9c2 cells with LPS and curcumin treatment was detected by CCK-8 (c). The decrease in H9c2 cells viability induced by LPS was alleviated by curcumin (d). Curcumin attenuated the apoptosis of H9c2 cells induced by LPS. *p < 0.05 and **p < 0.01 vs. Control, #p < 0.05 and ##p < 0.01 vs. LPS.
The potential targets of curcumin were explored
Analysis from CTD showed that ICAM1 and CD40 were identified as targets of curcumin (supplementary figure). Importantly, KEGG enrichment analysis showed that ICAM1 and CD40 were closely related to NF-κB pathway (Figure 2(a)). Moreover, data from GEO database showed that ICAM1 (Figure 2(b)) and CD40 (Figure 2(c)) were both highly expressed in myocarditis patients (n = 12) compared with the normal samples (n = 12). Thereafter, we observed that LPS stimulation increased ICAM1, CD40, p-P65 NF-κB, p-IkBα and N-cadherin expression. However, the elevated expression of ICAM1, CD40, p-P65 NF-κB, p-IkBα, and N-cadherin by LPS treatment were inhibited by curcumin stimulation (Figure 2(d)–(k)). These data illustrated that ICAM1, CD40, and p-P65 NF-κB expression in LPS model could be affected by curcumin. Intercellular adhesion molecule 1 (ICAM1) and CD40 were identified as targets of Curcumin (a). KEGG enrichment analysis was used to detect the pathways related to differentially expressed genes in myocarditis (b). ICAM1 expression was obviously increased in myocarditis patients (n = 12) compared with the control (n = 12) (c). A marked augmentation of CD40 was happened in myocarditis patients (n = 12) than the control (n = 12) (d–k). Western blotting assay exhibited that the increased expression of ICAM1, CD40 and p-P65 NF-Κb, p-IkBα and N-cadherin induced by lipopolysaccharide were suppressed by curcumin treatment. **p < 0.01 and ***p < 0.001 vs. Control, ##p < 0.01 and ###p < 0.001 vs. LPS.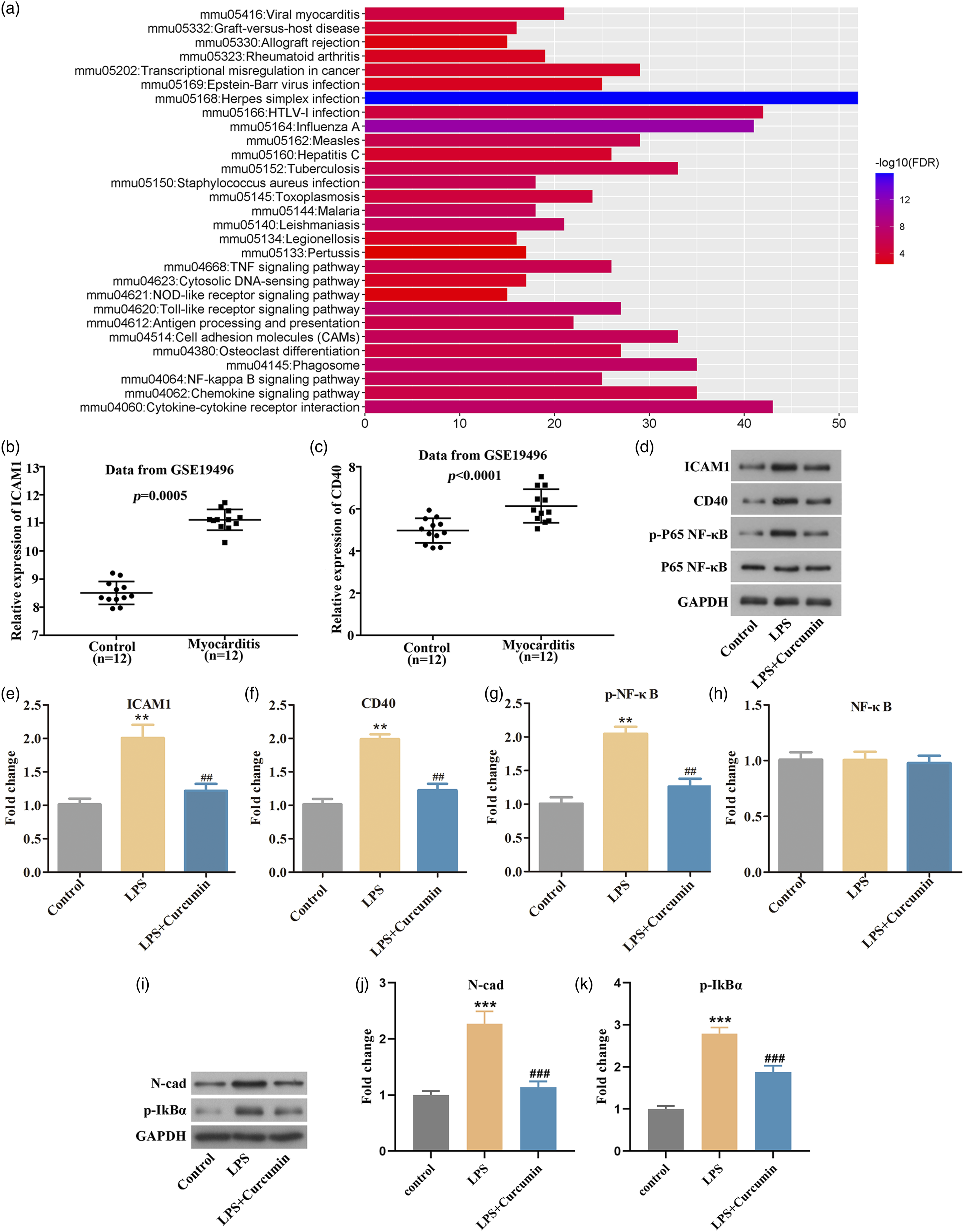
Curcumin alleviated the LPS-induced H9c2 damage by reducing ICAM1/CD40/NF-κB
To further study the effect of ICAM1 and CD40 on the protective action of curcumin in LPS-evoked H9c2 cells, siICAM1 and siCD40 were used. As presented in Figure 3(a), the expression of ICAM1 was decreased after H9c2 cells transfected with siICAM1. As shown in Figure 3(b), the pro-proliferative effect of curcumin on H9c2 cells was obviously enhanced by inhibiting ICAM1. Moreover, the suppressing impact of curcumin on LPS-evoked cell apoptosis was notably strengthened by depletion of ICAM1 (Figure 3(c)). As expected, the blocking effect of curcumin on p-P65 NF-κB, p-IkBα and N-cadherin expression was enhanced by depletion of ICAM1 (Figure 3(e)–(k)). Similarly, the expression of CD40 was decreased after H9c2 cells transfected with siCD40 (Figure 4(a)). In addition, we observed that knockdown of CD40 enhanced the promoting effect of curcumin on the proliferation of H9c2 (Figure 4(b)). Besides, knockdown of CD40 significantly strengthened the inhibitory effect of curcumin on LPS-evoked H9c2 cells apoptosis (Figure 4(c)). Additionally, knockdown of CD40 strengthened the inhibitory effect of curcumin on the expression of p-P65 NF-κB, p-IkBα, and N-cadherin (Figure 4(e)–(k)). These results suggested that curcumin alleviated LPS-evoked H9c2 cells damage by targeting ICAM1/CD40/NF-κB pathway. Curcumin alleviated H9c2 cells damage by regulation of Intercellular Adhesion Molecule 1 (ICAM1)/NF-κB (a). The expression of ICAM1 was detected after H9c2 cells transfected with siICAM1 by qRT-PCR (b). CCK-8 assay revealed that depletion of ICAM1 enhanced the promoting effect of curcumin on H9c2 cells viability (c-d). Flow cytometry assay suggested that knockdown of ICAM1 strengthened the inhibitory effect of curcumin on lipopolysaccharide-triggered H9c2 cells apoptosis (e–k). Western blotting assay indicated that ICAM1, p-P65 NF-κB, p-IkBα and N-cadherin expression was suppressed by curcumin, depletion of ICAM1 increased the inhibitory effect of curcumin on p-P65 NF-κB expression. Curcumin attenuated LPS-evoked H9c2 cells damage via targeting CD40/NF-κB (a). The expression of CD40 was detected after H9c2 cells transfected with siCD40 by qRT-PCR (b). Analysis from CCK-8 assay indicated that depletion of CD40 increased the promoting effect of curcumin on H9c2 cells viability (c-d). Data from flow cytometry exhibited that depletion of CD40 enhanced the inhibitory effect of curcumin on lipopolysaccharide-triggered H9c2 cells apoptosis (e–k). Western blotting assay indicated that CD40, p-P65 NF-κB, p-IkBα and N-cadherin expression was suppressed by curcumin, depletion of CD40 increased the inhibitory effect of curcumin on p-P65 NF-κB expression. 

Curcumin attenuated the inflammation of LPS-induced H9c2 cells via targeting ICAM1/CD40.
To explore the effect of curcumin on the inflammation of LPS-induced H9c2 cells, we examined the levels of inflammatory factors in LPS-induced H9c2 cells. The result showed that curcumin inhibited the TNF-α and IL-6 levels, while promoted the level of IL-10 in LPS-induced H9c2 cells; moreover, siICAM1 and siCD40 further promoted the effect of curcumin on the level inflammatory factors of LPS-induced H9c2 cells (Figure 5(a)–(f)). Curcumin attenuated the inflammation of LPS-induced H9c2 cells via targeting Intercellular Adhesion Molecule 1 (ICAM1)
Discussion
In this study, we discovered that curcumin alleviated the LPS-triggered damage in H9c2 cells. ICAM1 and CD40, which were regulated by curcumin, were highly expressed in myocarditis patients and LPS-induced H9c2 cells. By KEGG enrichment analysis, we discovered that ICAM1 and CD40 were closely related to NF-κB pathway. Under the treatment of curcumin, upregulation of ICAM1 and CD40 in LPS-induced H9c2 cells was suppressed. Moreover, the activated NF-κB pathway in the LPS-induced H9c2 cells was inhibited with curcumin treatment. Collectively, our data illustrated that curcumin mitigated LSP-evoked H9c2 cells damage by targeting ICAM1/CD40/NF-κB.
Currently, the therapeutic effects of herbal medicine on viral myocarditis have been more and more identified by researchers. 12 For example, Baicalin mitigated the symptoms of myocarditis by inhibiting NF-κB and PDCD4/JNK pathways. 2 Astragaloside IV protected myocarditis against injury by suppressing NF-κB pathway. 13 Previously, curcumin has been discovered to protect mice from coxsackievirus B3-induced myocarditis by blocking the PI3K/Akt/NF-κB pathway. 14 However, the detailed relationship between curcumin and NF-κB pathway remains unknown. In our study, we not only observed that curcumin can alleviate H9c2 cells damage, but also found that curcumin worked by targeting ICAM1/CD40 to inactivate the NF-κB pathway. It has been reported that curcumin inhibits cigarette smoke-induced inflammation via modulating the PPARγ-NF-κB signaling pathway 15 ; Curcumin reduces inflammation in knee osteoarthritis rats through blocking TLR4/MyD88/NF-κB signal pathway. 16 Similar to these studies, we found that curcumin inhibits inflammation of LPS-induced H9c2 cells by targeting ICAM1/CD40 to regulate NF-κB pathway
ICAM1 is a cell surface glycoprotein and an adhesion receptor expressed in immune cells and endothelial cells, which regulated leukocyte recruitment from circulation to sites of inflammation.17,18 ICAM1 concentrations have been found to be elevated in patients with cardiovascular disease and inflammation. 19 In our study, ICAM1 was also found to be highly expressed in patients with myocarditis. Additionally, ICAM1 was verified as a target of curcumin. Soveyd and colleagues reported that curcumin regulated the gene expression and serum levels of ICAM1 by influencing transcription factors, thereby improving the symptoms of migraine patients. 20 Significantly, we observed that depletion of ICAM1 enhanced the mitigative effect of curcumin on H9c2 cells damage, which confirmed the relationship between curcumin and ICAM1.
CD40 is a co-stimulatory molecule belonging to the tumor necrosis factor super-family. 21 CD40 is a marker of pro-inflammatory cytokines and plays an important role in the occurrence and maintenance of inflammatory responses. 22 CD40, which has been found to be widely overexpressed in myocarditis, plays an important role in the pathogenesis of myocarditis and may become a potential target for the treatment of myocarditis.23,24 Similarly, we discovered that CD40 was highly expressed in myocarditis patients, and silencing CD40 could inhibit the pro-inflammatory factors (TNF-α and IL-6) level and promoted the anti-inflammatory factor (IL-10) level. Importantly, CD40 was identified as a target of curcumin. A previous study revealed that curcumin alleviated angiotensin-induced endothelial cells injury by suppressing CD40 expression. 25 Herein, we observed that curcumin mitigated H9c2 cells damage by reducing CD40 expression, revealing the association between curcumin and CD40.
NF-κB pathway is implicated in the pathogenesis of myocarditis by regulating immune response, and the exaggerated inflammatory response induced by NF-κB pathway in myocarditis is the primary cause chronic heart failure and cardiac hypertrophy. 26 Previous reports have indicated that drug that targets NF-κB pathway can be used to treat myocarditis. 27 Curcumin, as a NF-κB suppressor, has been illustrated to exert protective function in several diseases by inactivating NF-κB pathway, such as osteoarthritis, airway inflammation and cancers, and of course myocarditis.14,28–30 In this study, expression level of p-P65 NF-κB increased in LPS-induced H9c2 cells. However, with curcumin treatment, p-P65 NF-κB expression significantly reduced. Additionally, we also observed that ICAM1 and CD40 were closely related to NF-κB pathway. Moreover, knockdown of ICAM1 or CD40, p-P65 NF-κB expression decreased significantly again. All these data indicated that curcumin alleviated H9c2 cells damage by inactivating NF-κB pathway through targeting ICAM1 and CD40.
Taken together, suppression of ICAM1 and CD40 enhanced curcumin’s alleviating effect on H9c2 cells damage triggered by LPS. A reduction of p-P65 NF-κB expression was observed in the LPS-induced H9c2 cells after inhibiting ICAM1/CD40 or adding curcumin, indicating that curcumin alleviated H9c2 cells damage by blocking ICAM1/CD40/NF-κB signaling.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
