Abstract
Objective
Long noncoding RNAs (lncRNAs) are involved in preeclampsia (PE), and apolipoprotein A-1 antisense RNA (APOA1-AS) and has been found to be associated with a number of diseases. Our study aims to understand the involvement of APOA1-AS in PE.
Methods
Clinically, APOA1-AS expression in early-onset severe PE (EOSPE) patients and healthy controls was detected by real-time quantitative polymerase chain reaction. In vitro experiments were divided into control [coculturing trophoblasts with human uterine microvascular endothelial cells (UtMVECs)], TNF-α [coculturing trophoblasts with UtMVECs treated with tumor necrosis factor-α (TNF-α)], and TNF-α + control siRNA/APOA1-AS siRNA groups (UtMVECs transfected with control siRNA/APOA1-AS siRNA were cocultured with trophoblasts in the presence of TNF-α). The animals were divided into normal group, PE group (PE model was established by administrating nitro-L-arginine methyl ester (L-NAME) in rats), PE + control siRNA group (PE rats were treated with control siRNA), and PE + APOA1-AS siRNA group (PE rats were treated with APOA1-AS siRNA).
Results
Increased APOA1-AS was found in the placental tissues of EOSPE patients. APOA1-AS siRNA abolished the decreased integration of trophoblasts into UtMVEC networks induced by TNF-α. Furthermore, APOA1-AS siRNA improved pregnancy outcomes in PE rats with increased expression of vascular endothelial growth factor, placental growth factor, and fms-like tyrosine kinase receptor (Flt-1) but decreased expression of E-cadherin, intercellular cell adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1).
Conclusion
Downregulation of APOA1-AS protected against TNF-α-induced inhibition of trophoblast integration into endothelial networks, thus exerting protective effects against PE rats.
Introduction
Hypertensive disorders in pregnancy are a spectrum of conditions specific to women, including preeclampsia (PE), and are the major contributors to maternal and perinatal morbidity and mortality worldwide. 1 PE generally develops after 20 weeks of gestation with elevated blood pressure and proteinuria, which occurs in 3–5% of pregnancies worldwide.2–4 At present, the exact pathogenesis of PE is still unknown, but studies have confirmed that trophoblastic infiltration and placental hypoplasia, placental ischemia, endothelial dysfunction, oxidative stress, and excessive maternal inflammatory response are thought to be risk factors in the development of PE. 5 Furthermore, noncoding RNAs have been found to play critical roles in the regulation of embryonic growth, opening up new avenues of investigation into placental differentiation and function. 5
To date, a variety of potential genes, including both coding and noncoding genes, have been reported to be linked to PE. 4 Long noncoding RNAs (lncRNAs), a class of tissue-specific RNAs, are defined as transcribed RNA molecules without protein-coding functions. 6 According to the literature, multiple lncRNAs, such as HEIPP, 7 INHBA-AS1, 8 SNHG14, 9 FAM99A, 10 GASAL1, 11 NEAT1, 12 and H19 13 have been shown to be involved in the biological activity of trophoblast cells in PE. LncRNA apolipoprotein A-1 antisense RNA (APOA1-AS, NR_126,362) has gained much interest as an endogenous regulator of ApoA1 biogenesis. At present, APOA1-AS has been found to be associated with a number of diseases, such as relapsing-remitting multiple sclerosis, 14 coronary artery disease, 14 and systemic lupus erythematosus. 15 In addition, APOA1-AS was found to be one of the top 20 candidate lncRNAs ranked with a human lncRNA microarray in 5 pairs of serum samples (5 PE patients and 5 matched controls). 16 However, the expression of APOA1-AS in placental tissues of PE patients and whether it affects the development of PE are still unknown. Therefore, qRT–PCR was first performed to detect the expression of APOA1-AS in patients with early-onset severe preeclampsia (EOSPE) and normal placental tissues. We next analyzed whether APOA1-AS exerts functions in the effect of tumor necrosis factor-α (TNF-α) in a human in vitro model of trophoblast invasion. Finally, a PE rat model was established by nitro-L-arginine methyl ester (L-NAME) and treated with APOA1-AS siRNA injection to explore its influence on pregnant rats.
Materials and methods
Ethical statement
Sprague–Dawley timed-pregnant rats were kept at 21°C with a 12-h light/dark cycle and free access to food and tap water. All patients and their guardians were informed and signed an informed consent form. The study was approved by the ethics committee of Yantaishan Hospital (approval no. YTSH20180916).
Subjects
From January 2019 to December 2020, a total of 47 placental tissues from maternal patients in our hospital were randomly selected, consisting of 19 EOSPE patients and 28 normal placental tissues without pregnancy complications during the same period as the healthy control group. Subjects were excluded if they (1) had psychiatric diseases, (2) had a history of hormonal or antibiotic medication during pregnancy, or (3) had severe systemic immune system diseases. The diagnosis of EOSPE was based on the American College of Obstetricians and Gynecologists criteria. 17 Placental tissues were immediately collected after placental delivery to perform real-time quantitative polymerase chain reaction tests.
Cell culture
Human trophoblast HTR-8/SVneo cells (Catalog# CRL-3271) were purchased from American Type Culture Collection (ATCC, USA) and cultured in RPMI-1640 medium (Catalog# 30–2001, ATCC, USA) with 5% fetal bovine serum. In addition, human uterine microvascular endothelial cells (UtMVECs, Lonza, Basel, Switzerland) were grown in EGM-2MV medium when ∼80% confluent with culture conditions of 95% air and 5% CO2 at 37°C. Human recombinant tumor necrosis factor-α (TNF-α) was purchased from Abcam (Catalog# ab155699, USA).
In vitro experiment
An in vitro experiment was performed in the following four groups: a control group (HTR-8/SVneo cells were cocultured with human UtMVECs), a TNF-α group (HTR-8/SVneo cells were cocultured with UtMVECs and treated with TNF-α), a TNF-α + control siRNA group (UtMVECs transfected with control siRNA were cocultured with HTR-8/SVneo cells in the presence of TNF-α), and a TNF-α + APOA1-AS siRNA group (UtMVECs transfected with APOA1-AS siRNA were cocultured with HTR-8/SVneo cells in the presence of TNF-α). Control siRNA (Catalog# 4,390,843) and APOA1-AS siRNA (Catalog# 4,392,420) were purchased from Thermo Fisher Scientific (Shanghai, China). Transfection was performed using liposome-based methods with Lipofectamine 3000 transfection reagent (Catalog# L3000015, Invitrogen, USA) following the manufacturer’s instructions.
HTR-8/SVneo cells cocultured with UtMVEC
The 24-well plates were polymerized for 30 min at 37°C after being coated with undiluted Matrigel (300 μL/well, BD Biosciences). Each well was seeded with human UtMVECs (75,000 cells/well), and endothelial cell tubular structures were allowed to form (4 h incubation at 37°C). PKH67-labeled HTR-8/SVneo cells (75,000 cells/well) cocultured with UtMVECs were treated with human recombinant TNF-α (final concentration 0.1 ng/mL), and the visualization and quantification of cell integration were then performed based on a previous study. 18
Establishment of the preeclampsia rat model
The experiments were divided into a normal group, a PE group, a PE + control siRNA group, and a PE + APOA1-AS siRNA group, with 8 rats in each group. Except for the normal group, the PE rat model was established by administering nitro-L-arginine methyl ester (L-NAME, Sigma Aldrich Co., USA, 50 mg per kg body weight per day) through oral gavage from Days 14 to 19 of gestation.19,20 Rats in the PE + control siRNA and PE + APOA1-AS siRNA groups were injected with control siRNA and APOA1-AS siRNA in the tail vein from Days 12 to 19 of gestation, respectively.
Measurement of blood pressure
On day 20 of gestation, systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured by the tail-cuff method using a noninvasive blood pressure measurement system (BIOPAC System, Inc.; USA).21,22 In brief, prior to obtaining pressure measurements, the rats were placed in restrainers and kept in a scanner at 35°C for 10 min to warm the rats. Then, a cuff was placed on the base of the tail followed by placing a transducer close to the cuff. An average of at least three consecutive readings was taken for each rat.
Sample preparation
After blood pressure measurement, the rats were anesthetized using isoflurane inhalation and then underwent cesarean section, and the litter weight and number of litters were recorded. The placenta was weighed using an electronic balance (Ohaus, Parsippany, NJ, USA), after which it was immediately placed in liquid nitrogen and stored at −80°C for various biochemical and molecular analyses. The relative placental weight was expressed as [(placental weight/animal weight) × 100].
Enzyme-linked immunosorbent assay
Rat colorimetric sandwich enzyme-linked immunosorbent assay (ELISA) kits labeled with horseradish peroxidase were used to detect the protein levels of vascular endothelial growth factor (VEGF, Catalog# ab100787, Abcam, USA), placental growth factor (PLGF, Catalog# CSB-E07400r, CUSABIO, Wuhan, China) and fms-like tyrosine kinase receptor (Flt-1, Catalog# ER0087, Shanghai Fusheng Industrial Co., Ltd. Shanghai, China) in rat placental tissue in a 96-well plate. Plates were read on an EL800 microplate reader (Bio-Tek, Winooski, VT, USA) at a wavelength of 450 nm.
Real-time quantitative polymerase chain reaction
Primer sequences used in real-time quantitative polymerase chain reaction.

Statistical method
SPSS 21.0 statistical software (SPSS, Inc., Chicago, IL, USA) was used to analyze the data. The data are presented as the mean ± standard deviation (SD). The t-test was used for comparison of measurement data. Pearson analysis was used to find the correlation of APOA1-AS expression with SBP and DBP in placental tissues of EOSPE patients. One-way ANOVA was used for comparisons among multiple groups, and Tukey’s HSD test was used for comparisons within groups. The difference was significant when p < .05.
Results
Comparison of clinical data between early-onset severe preeclampsia patients and controls
Comparison of clinical data between early-onset severe PE patients and Controls.

SBP: systolic blood pressure; DBP: diastolic blood pressure.
APOA1-AS expression in the placental tissue of early-onset severe preeclampsia patients
To investigate whether abnormal APOA1-AS exists in EOSPE, its expression levels in placental tissues of EOSPE patients and healthy controls were detected by qRT–PCR. APOA1-AS expression in placental tissues of EOSPE patients was found to be significantly higher than that in the healthy controls (p < .001, Figure 1(a)), suggesting the pathogenic role of APOA1-AS in PE. Furthermore, positive correlations of APOA1-AS expression with both SBP (r = 0.853, p < .001, Figure 1(b)) and DBP (r = 0.620, p = .005, Figure 1(c)) were found in placental tissues of EOSPE patients according to the Pearson analysis, indicating that APOA1-AS inhibition may contribute to the decreased blood pressure. APOA1-AS expression in the placental tissue of early-onset severe PE patients. Notes: A: Comparison of APOA1-AS expression in placental tissues of early-onset severe PE patients (n = 19) and healthy controls (n = 28) by qRT–PCR; B-C: Significant positive correlations of APOA1-AS expression with both SBP (B) and DBP (C) in placental tissues of early-onset severe PE patients evaluated by Pearson analysis.
APOA1-AS siRNA protects against the deleterious effects of TNF-α on human trophoblast-endothelium cell interactions
Then, an in vitro experiment was performed to determine whether APOA1-AS inhibition protects against the inhibitory effect of trophoblast integration into endothelial networks. Compared with control cells, treatment with TNF-α reduced the integration of HTR-8/SVneo cells into human UtMVECs, which was abolished by APOA1-AS siRNA (all p < .05, Figure 2(a) and (b) shows fluorescent images of the integration of HTR-8/SVneo cells into endothelial networks after 20 h, suggesting that inhibition of APOA1-AS improved the deleterious effects of TNF-α on human trophoblast-endothelium cell interactions. APOA1-AS siRNA protects against the deleterious effects of TNF-α on human trophoblast-endothelial cell interactions. Notes: A: After 20 h of coculture, the proportion of tubular endothelial network with HTR-8/SVneo cell integration; n = 3. Data are presented as the mean ± SD. Comparisons among multiple groups were conducted with one-way ANOVA, and comparisons within groups were performed with Tukey’s HSD test. * compared with the control group, p < .05; # compared with the TNF-α group, p < .05; & compared with the TNF-α + control siRNA group, p < .05. B: After 20 h of coculture, representative fluorescence images of HTR-8/SVneo and UtMVECs are shown.
APOA1-AS siRNA alters the gene expression of invasion markers in cocultured cells
The gene expression levels of adhesion markers (E-cadherin, ICAM-1, and VCAM-1) were examined using qRT–PCR to determine whether APOA1-AS siRNA alters the gene expression of invasion markers in cocultured cells. As illustrated in Figure 3, the expression levels of E-cadherin, ICAM-1 and VCAM-1 were increased in all TNF-α groups compared to the control group (all p < .05). No significant difference was found concerning the above genes between the TNF-α and TNF-α + control siRNA groups (all p > .05). However, the expression levels of E-cadherin, ICAM-1 and VCAM-1 were significantly lower in the TNF-α + APOA1-AS siRNA group than in the TNF-α + control siRNA group (all p < .05), implying that suppression of APOA1-AS improved the inhibitory effect of TNF-α on trophoblast invasion. Effect of APOA1-AS siRNA on the expression of adhesion markers (E-cadherin, ICAM-1, and VCAM-1) in cocultured cells determined by qRT–PCR. Notes: n = 3. Data are presented as the mean ± SD. One-way ANOVA was used for comparisons among multiple groups, and Tukey’s HSD test was used for comparisons within groups. * compared with the control group, p < .05; # compared with the TNF-α group, p < .05; & compared with the TNF-α + control siRNA group, p < .05.
APOA1-AS siRNA protects PE rats induced by L-NAME
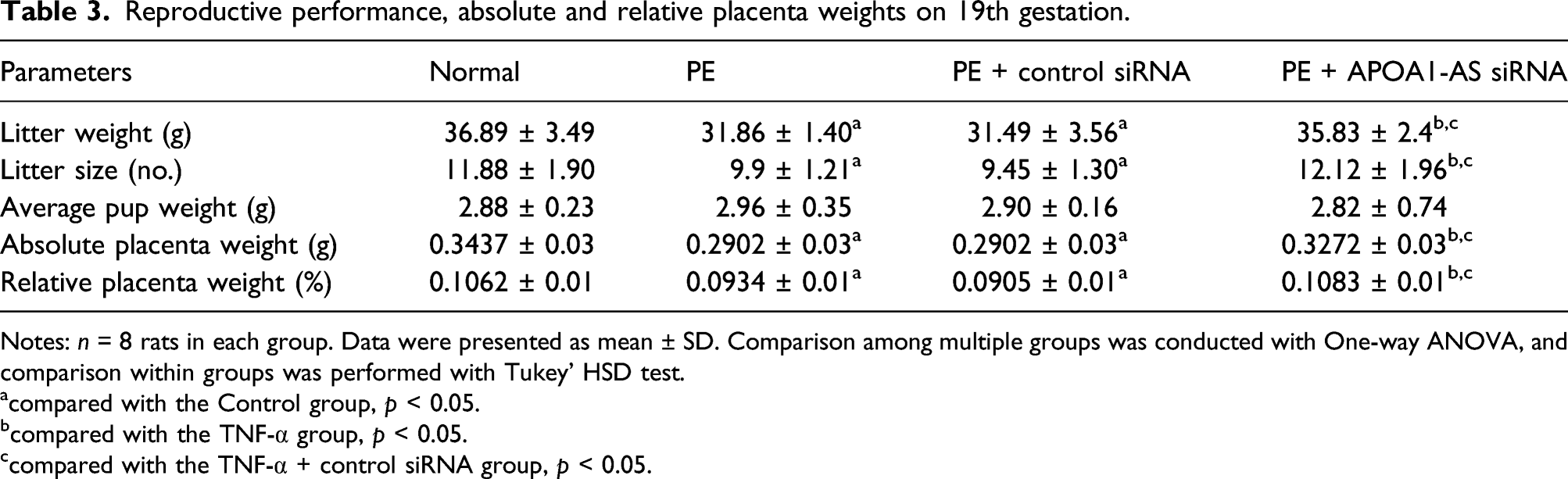
Furthermore, to determine the potential treatment role of APOA1-AS siRNA in PE, L-NAME-induced PE models were constructed in vivo. The results showed that APOA1-AS siRNA ameliorated the L-NAME-induced increases in SBP and DBP of PE rats (all p < 0.05, Figure 4(a) and (b)). In addition, significant decreases in litter weight, litter size, absolute placenta weight, and relative placenta weight were shown in PE rats (all p < 0.05). However, these parameters were significantly increased in PE rats after treatment with APOA1-AS siRNA (all p < .05, Table 3). Furthermore, APOA1-AS siRNA injection significantly increased the protein levels of VEGF, PLGF, and Flt-1 in the placental tissues of L-NAME-induced rats using ELISA (Figure 4(c) and (e)), with decreased expression levels of E-cadherin, ICAM-1, and VCAM-1 using qRT–PCR (all p < .05, Figure 5). All of the above results indicate that the inhibition of APOA1-AS may be beneficial for the treatment and prevention of PE by reducing blood pressure and promoting trophoblast invasion. Effects of APOA1-AS siRNA on L-NAME-induced blood pressure and protein expression of VEGF, PLGF and Flt-1 in the placental tissues of PE rats. Notes: A-B: Comparison of SBP and DBP in each group; C-F: Protein expression levels of VEGF, PLGF and Flt-1 in the placental tissue of each group; n = 8 rats in each group. Data are expressed as the mean ± SD. One-way ANOVA was used for comparisons among multiple groups, and Tukey’s HSD test was used for comparisons within groups. * compared with the control group, p < .05; # compared with the TNF-α group, p < .05; & compared with the TNF-α + control siRNA group, p < .05. VEGF: vascular endothelial growth factor; PLGF: placental growth factor. Reproductive performance, absolute and relative placenta weights on 19th gestation. Notes: n = 8 rats in each group. Data were presented as mean ± SD. Comparison among multiple groups was conducted with One-way ANOVA, and comparison within groups was performed with Tukey’ HSD test. acompared with the Control group, p < 0.05. bcompared with the TNF-α group, p < 0.05. ccompared with the TNF-α + control siRNA group, p < 0.05. mRNA expression of adhesion markers in placental tissues of rats detected by qRT–PCR. Note: A: E-cadherin; B: ICAM-1; C: VCAM-1; n = 8 rats in each group. Data are expressed as the mean ± SD. One-way ANOVA was used for comparisons among multiple groups, and Tukey’s HSD test was used for comparisons within groups. * compared with the control group, p < .05; # compared with the TNF-α group, p < 0.05; & compared with the TNF-α + control siRNA group, p < .05.

Discussion
In vitro and in vivo, APOA1-AS was shown to be transcribed from the DNA strand opposite to the APO gene cluster, which could function as a negative regulator of APOA1. 28 In addition, targeting APOA1-AS with a short antisense oligonucleotide also increased the expression of APOA1 in human hepatocytes. 23 Of note, a previous study showed that the serum level of APOA1 was significantly decreased in PE patients compared to controls, 29 indicating a potential association of APOA1-AS with PE. The current study discovered that APOA1-AS expression was significantly higher in placental tissues of EOSPE patients than in the normal population, and prior studies also found that APOA1-AS expression was similarly significantly upregulated in the serum of PE patients. 16 Mechanistically, EOSPE was diagnosed when at least one of the following criteria was present: SBP ≥160 mmHg and/or DBP ≥110 mmHg, thrombocytopenia, impaired liver function, renal insufficiency, pulmonary edema, new-onset headache unresponsive to medications, or visual disturbances.30,31 For the past few years, APOA1-AS expression has been found to be connected with blood pressure. For example, Abd-Elmawla MA et al. found that APOA1-AS expression could be used as a predictor of atherosclerosis in SLE, which was positively correlated with hypertension. 15 Similarly, in this study, APOA1-AS expression in the placental tissue of EOSPE patients was significantly positively correlated with both SBP and DBP, suggesting that inhibition of APOA1-AS may be a potential target for the treatment of PE.
Trophoblast integration into endothelial cellular networks in the uterus is interfered with by the proinflammatory cytokine TNF-α. 32 Previous studies also verified that APOA1 prevented the human trophoblast–endothelium cell interaction from the detrimental effects of TNF-α.18,33 Therefore, similar experiments were performed to further validate the effect of APOA1-AS siRNA on the invasive ability of trophoblast cells. The present results suggested that the integration of HTR-8/SVneo trophoblast cells into human UtMVEC networks was significantly reduced following TNF-α treatment and was abolished by APOA1-AS siRNA. PE is a pregnancy-specific disease, and the expression and localization of calmodulin are related to the pathogenesis of PE; for example, the expression of E-cadherin was significantly higher in PE than in the normal pregnancy population. 34 Vascular endothelial cell injury is also credited as the pathological basis for PE pathogenesis, which needs to be based on the premise that leukocytes and platelets adhere and migrate to endothelial cells, and this process requires the involvement of cell adhesion molecules, such as ICAM-1 and VCAM-1.35,36 Under normal conditions, these adhesion molecules are minimally or not expressed, but their expression levels increase rapidly upon stimulation by inflammatory factors, such as IL-1 and TNF-α,37,38 as similarly shown in the results of this research. APOA1-AS was negatively correlated with HDL-C, and importantly, TNF-α-induced VCAM-1 expression in endothelial cells could be inhibited by HDL.15,39 In the current study, TNF-α induction resulted in the significantly elevated expression of E-cadherin, ICAM-1 and VCAM-1 in cocultured HTR-8/SVneo and UtMVECs, while the expression levels of these genes were reduced by APOA1-AS siRNA, suggesting that downregulation of APOA1-AS may reduce inflammation via changes in associated gene expression, creating a more conducive environment for placental development.
Based on genetic, immunological, or pharmacological approaches, several animal models have been developed to study placental dysfunction and PE.40–42 The “L-NAME model,” first described in 1994, has been used since then according to chronic inhibition of nitric oxide synthase by continuous delivery of L-NAME. 43 L-NAME leads to altered blood pressure and vascular reactivity due to reduced nitric oxide bioavailability. 44 According to previous studies, non-invasive blood pressure measurements were used to detect blood pressure in L-NAME-induced hypertension in rats,21,45 though these measurements can lead to stress in animals. 46 In our study, prior to obtaining pressure measurements, the rats were placed in restrainers and kept in a scanner at 35°C for 10 min to warm the rats to try to avoid this issue. 22 However, other measurements for the evaluation of blood pressure should be further used to verify our results. In this study, L-NAME was used to create a PE rat model, and elevated blood pressure and poor pregnancy were observed in rats, consistent with the results of several previous studies.47–49 However, it was also found that APOA1-AS siRNA significantly ameliorated the L-NAME-induced increases in SBP and DBP in PE rats, suggesting that APOA1-AS downregulation could play a role in lowering blood pressure. In addition, APOA1-AS siRNA also reduced the expression levels of E-cadherin, ICAM-1 and VCAM-1 in placental tissues of PE rats, which further verified the in vitro experimental results. Notably, advances in some molecular alterations related to the VEGF family, which could act as predictors of maternal outcomes in the short term in PE, have clinical applications at present. 50 VEGF and Flt-1 (fms-like tyrosine kinase receptor or VEGFR1) are regulated by transcription factors such as Hif1α. 51 Placental VEGF and Flt-1 protein levels were reduced in L-NAME-induced PE rats compared to controls. 52 Additionally, decreased levels of PLGF have been detected in women with PE. 53 In this study, APOA1-AS siRNA injection significantly increased the litter weight, litter size, and absolute and relative placenta weights and elevated the levels of VEGF, PLGF, and Flt-1 in L-NAME-induced rat placental tissues, further demonstrating the therapeutic effect of APOA1-AS downregulation in PE rats. Although our study aimed to understand the potential involvement of APOA1-AS in PE from clinical, in vitro and animal experiment perspectives, the findings were still basic with limited results due to time and funding constraints, and more in-depth research should be pursued in the future.
In conclusion, APOA1-AS expression was elevated in placental tissues of EOSPE patients, demonstrating a positive correlation with blood pressure. However, downregulation of APOA1-AS protected against TNF-α-induced inhibition of trophoblast integration into endothelial networks, exerting a protective effect against L-NAME-induced PE in rats, accompanied by improved pregnancy outcome, decreased expression levels of E-cadherin, ICAM-1 and VCAM-1, and increased expression levels of VEGF, PLGF and Flt-1.
Footnotes
Acknowledgments
The authors appreciate the reviewers for their useful comments in this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
