Abstract
Type 2 diabetes mellitus (T2DM) is a metabolic disorder characterized by chronic hyperglycemia and insulin resistance. 4-hydroxyisoleucine (4-HIL) is a non-proteinogenic amino acid isolated from the fenugreek seeds and has enormous pharmacological activities. The present study was undertaken to investigate the antihyperglycemic effect of 4-HIL in streptozotocin (STZ)-induced diabetic rats. Moreover, its toxicity was evaluated in vitro and in vivo employing human embryonic kidney cells (HEK-293) and healthy rats, respectively. In experiment 1, STZ-induced diabetic male rats were subjected to an oral treatment of 4-HIL (100 mg/kg), while experiment 2 deals with the effects of 4-HIL on healthy male and female rats following oral administration. The treatment (experiment 1) declined the elevated blood glucose level, feed intake, and increased body weight(s). Additionally, blood glucose impairment was improved as observed by OGTT and IPGT tests. Pancreatic histopathology revealed mild changes in the 4-HIL group. Moreover, experiment 2 showed increased body weight, normal blood glucose levels (male—106.06 ± 7.49 mg/dl and female—100.06 ± 14.69 mg/dL), hematological parameters, and histopathological profiles in the treatment group. 4-HIL did not affect the viability of HEK-293 cells, and no signs of toxicity were observed in healthy rats. Therefore, the study concludes that 4-HIL has potential antihyperglycemic activity without any toxic effects.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder of the endocrine system characterized by insulin resistance and pancreatic β-cell dysfunction which leads to persistent hyperglycemia. It is associated with the development and progression of relatively specific long-term microvascular complications such as diabetic retinopathy, nephropathy, and neuropathy as well as an increased risk for cardiovascular diseases (CVD). These contribute to higher medical costs, poor quality of life, and increased mortality. 1 The global prevalence of T2DM and impaired glucose tolerance in 2019 is estimated to be 9.3% (463 million people) and 7.5% (374 million) which may be projected to 10.2% (578 million) and 8.0% (454 million) by 2030.2,3 In recent years, the use of various drugs for the management of T2DM has been increased, such as metformin and peroxisome proliferator activated receptor-γ (PPARγ) agonists which inhibits hepatic gluconeogenesis and maintains glucose homeostasis, respectively. α-glucosidases interfere with intestinal glucose absorption, and sulfonylureas stimulate insulin secretion from pancreatic β-cells. Insulin enhances the suppression of hepatic glucose production, and augments glucose utilization.4–8 These therapeutic drugs have limited efficacy, reduced tolerability, and mechanism-based toxicity with unavoidable side effects. 9 To overcome these problems, safe and cost-effective antidiabetic agents from plant sources have been found to be more effective than conventional drug compounds with no/fewer side effects. Therefore, identification of natural products that are helpful in ameliorating diabetes is a worthwhile endeavor. 10 The use of traditional medicinal plants in healthcare has increased over the last few decades due to their lower toxic effects. They serve as hot spots for valuable phytochemicals such as alkaloids, flavonoids, saponins, and terpenoids with bioactive constituents that possess several biological activities such as antioxidants, anti-inflammatory, antihyperglycemic, etc.11,12
Trigonella foenum-graecum Linn, commonly known as fenugreek, is a leguminous plant that has long been used for the treatment of diabetes. 4-HIL has been identified as one of the major bioactive compound present in fenugreek seeds, and its absolute stereo configuration was determined as (2S, 3R, 4S).13,14 Thus, discovery of a suitable bioactive agent for the management of T2DM with minimal toxicity poses a challenge in the search for a potential therapeutic agent. Therefore, in the present study, we checked the antidiabetic effect of 4-HIL by using STZ-induced diabetic rat model and its toxicity was evaluated on healthy rats.
Materials and methods
Collection and authentication of T. foenum graecum seeds
The fenugreek seeds were purchased from the local market of Lucknow (India), and authenticated by the Botany Division of CSIR-CDRI, Lucknow, India. The chemical structure of pure 4-HIL as given in Figure 1 was isolated from the fenugreek seeds as per the protocol previously described.
13
Chemical structure of 4-hydroxyisoleucine isolated from Trigonella foenum-graecum.
Chemicals and reagents
Streptozotocin (STZ) was purchased from Sigma Aldrich (Catalog No. S0130-1G, USA). Glucometer Accucheck active strips were procured from Roche Diagnostics (Catalog No. GB 13093001, Germany). Metformin was supplied by the Glyciphage plane. Sodium citrate and citric acid were purchased from Bio Basic (Catalog No. CB0035, Canada). Sucrose was procured from Sisco Research Laboratories Pvt. Ltd (Catalog No. 27580). Insulin (Catalog No. AF5109, USA) and GAPDH (Catalog No. AF7021, USA) polyclonal antibodies were purchased from Affinity biosciences. Goat antirabbit horseradish peroxidase (HRP)-conjugated secondary antibodies were supplied by Thermo Fisher scientific (Catalog No. 31460, USA). MTT {3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide} was purchased from MP biomedicals (Catalog No. 194592, USA).
Experimental animals
Healthy male Sprague Dawley (SD) rats (5–6 weeks old, body weight between 170 ± 30 g) were procured from the National Laboratory Animal Facility, CSIR—Central Drug Research Institute, Lucknow, India. The protocol for the animal studies was approved by the Institutional Animal Ethical Committee (IAEC) of the CSIR-CDRI, and experiments were carried out in accordance with CPCSEA guidelines. The IAEC number for antidiabetic in vivo study (experiment no. 1) was IAEC/2019/9; for the apparently healthy rat study (experiment no. 2), the no. was IAEC/2019/118, whereas the acute toxicity IAEC no. was IAEC/2021/5. The acute toxicity study was carried out at a National GLP Compliance Monitoring Authority (NGCMA)-accredited test facility following the guidelines of OECD and New Drugs and Clinical trials rules, 2019. Rats were housed in polypropylene plastic cages at 23 ± 2°C and 50 ± 20% humidity, and 12 h light/dark cycle throughout the experiment period. Animals were provided feed and water ad libitum.
Experiment 1: Study design for diabetic rats
In experiment 1, after 7 days quarantine and followed by 7 days acclimatization period, the rats (n = 24) weighing 250 ± 10 gm (7–8 weeks) were divided into four groups, each consisting of 6. The groups were control (I), STZ group (II) (35 mg/kg body weight
15
), 4-HIL group (III) (100 mg/kg body weight), and Metformin group (IV) (350 mg/kg body weight
16
) were administered. Rats were fasted 6 h prior to experiments and a single intraperitoneal injection of STZ (prepared in 0.1
Experiment 2: Study design for apparently healthy rats
Healthy SD rats (5–6 weeks of body weight 150-200 gm) of both sexes (n = 24) were divided into two groups (12 rats/group; six each sex): control group (I) and 4-HIL treatment group (II)—100 mg/kg body weight.
Routine cage side observation, body weight, blood glucose level, and feed consumption of rats were measured weekly. At the end of the experiment, blood samples were collected from orbital venous plexus for hematology and clinical biochemistry. The necropsy was performed, and all the organs were examined for gross pathological changes. Pancreas and kidney tissues were collected in 10% neutral buffered formalin, processed and embedded in paraffin. Tissue sections were cut into 4 μm thin sections and stained with hematoxylin and eosin (H&E). The histopathological changes were observed with a light microscope (Nikon, Eclipse TS2).
Oral glucose tolerance test and intraperitoneal glucose tolerance test
In experiment 1, an oral glucose tolerance test (OGTT) and intraperitoneal glucose tolerance test (IPGTT) were performed on the last day of treatment after overnight fasting of all the rats. 17 Blood samples were collected from the tail tip incision at 0, 15, 30, 60, 90, and 120 min after oral and intraperitoneal administration glucose at the dose of 2 gm/kg. Blood glucose was estimated using a glucometer. The area under the curve (AUC) was calculated using the trapezoidal method after constructing curves of blood glucose levels (mg/dl) vs time intervals (min).
Western blot analysis
The pancreatic tissue samples were lysed in RIPA (radioimmunoprecipitation assay) buffer. Protein concentration was determined using BCA (bicinchoninic acid) kit. Equal amount of proteins was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and blotted on to polyvinylidene difluoride (PVDF) membranes. Membranes were blocked with 5% BSA for 45 min and blots incubated with insulin antibody (1:1000) overnight at 4°C and subsequently incubated with HRP-conjugated secondary antibody for 2 h at room temperature. Enhanced chemiluminescence (ECL) solution was used to detect immunoreactivity and scanned using the Chemidoc XRS imaging system. Densitometry analysis of blots was performed by using Image J software. 18 To ensure equal loading in each lane, GAPDH (1:1000) was used as internal loading control.
In vitro cytotoxicity study
For analyzing the cytotoxic effect of 4-HIL, the MTT assay was performed in the HEK-293 cell line. The cells were seeded (1 × 104 cells per well) in 96-well plates and treated with 1 μ
Cytotoxicity (%) = OD Sample (mean)/OD Control (mean) × 100%.
OD = optical density.
Acute oral toxicity
The acute toxicity study in rats was carried out as per New Drug and Clinical Trial Rules, 2019 (previously Schedule Y of Drugs and Cosmetic Act, 1940 and Rules, 1945, Government of India) and 420 test guidelines in the GLP-accredited test facility (OECD 420, 1992). Healthy adult rats (n = 48) were divided into four groups (each consisting of six male and six female). All treatments were done by oral gavage. In the present study, antidiabetic activity of 4-HIL was observed in rats at a dose of 100 mg/kg per oral. So, we assessed its toxicity at the 20X antidiabetic dose (2000 mg/kg as the maximum dose). Accordingly, rats were administered with control (vehicle, aqueous) and 4-HIL at 1000 mg/kg, 1500 mg/kg, and 2000 mg/kg body weight.
Statistical analysis
All results are expressed as mean ± SD. All results were analyzed by one-way ANOVA followed by Bonferroni’s multiple comparison tests used to compare the groups. Graph Pad Prism, Version 7.0 was used for statistical analysis. p value < 0.05 is regarded as statistically significant.
Results
Effect of 4-HIL on feed consumption and body weight
Effect of 4-HIL on body weight, feed consumption, and blood glucose level of diabetic rats (n = 6/group).

Values are expressed as mean ± SD.
###p < 0.001 compared to the control group.
**p < 0.01.
***p < 0.001 compared to the STZ group.

Experiment 1: (a) body weight, (b) feed intake, and (c) blood glucose analysis graph in different groups of rats (n = 24). ###p < 0.001 compared to the control group, **p < 0.01 ***p < 0.001 compared to the STZ group.
Effect of 4-HIL on body weight, feed, and blood glucose level of male and female healthy rats (n = 6 each sex).
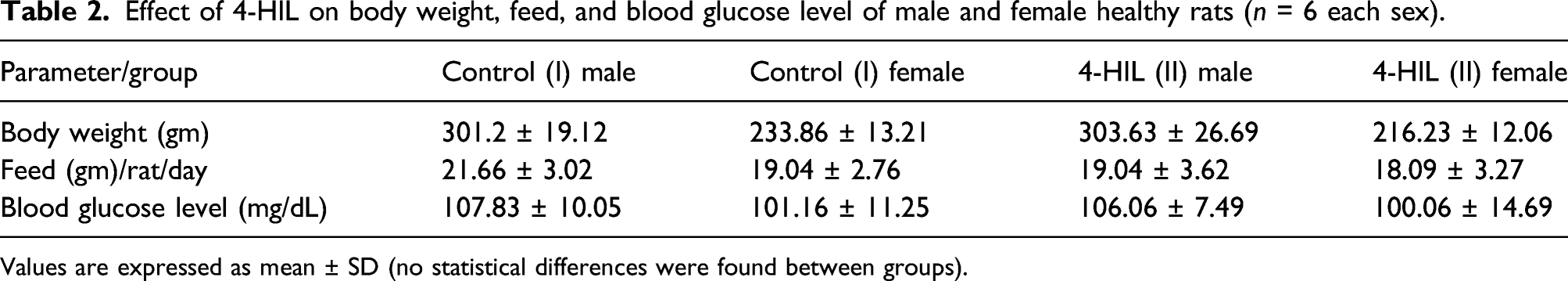
Values are expressed as mean ± SD (no statistical differences were found between groups).
Anti-hyperglycemic effect of 4-HIL in diabetic rats
There was a significant (p < 0.001) increase in blood glucose levels in the STZ group (II) (78.6%) as compared to control group (I), while treatment of 4-HIL and metformin significantly (p < 0.001 and p < 0.01) lowered the blood glucose level in group (III) (46.4%) and group (IV) (23.6%), respectively, as compared to the STZ group (II) in experiment 1 (Table 1; Figure 2).
In experiment 2, administration of 4-HIL (group II) had no effect on the blood glucose level in healthy rats. There was no alteration in the blood glucose levels in the treated male group (II) (106.06 ± 7.49 mg/dl) and treated female group (II) (100.06 ± 14.69 mg/dl) as compared to those of the control male group (I) (107.83 ± 10.05 mg/dl) and the female group (I) (101.16 ± 11.25 mg/dl) (Experiment 2: Table—2).
4-HIL maintains glucose homeostasis
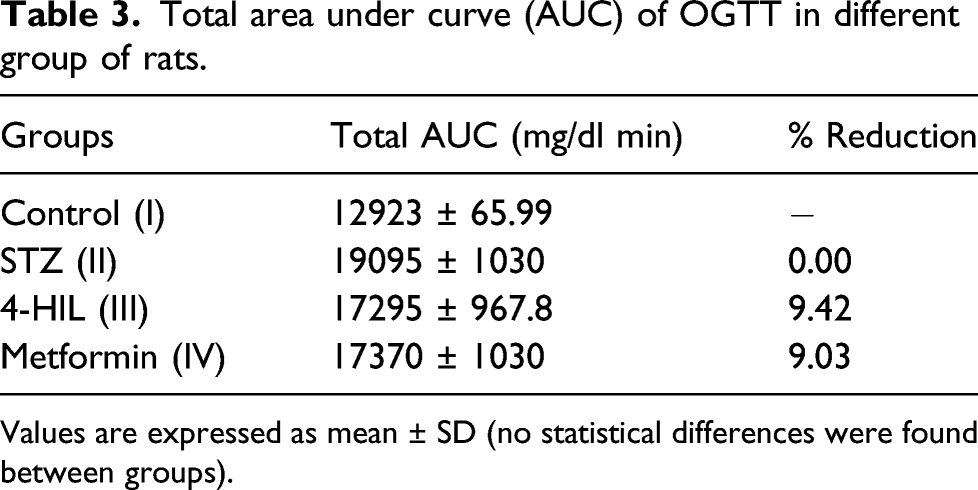
Oral administration of glucose (2 gm/kg) showed marked increase in blood glucose level in all the groups at 0, 15, 30, 60, and 120 min. The diabetic rats of the STZ group (II) showed higher blood glucose levels throughout the test (120 min) as compared to the normal control rats. Both the 4-HIL-treated group (III) and the metformin group (IV) showed decrease in blood glucose levels which was comparable to the glucose level of the control group (I) (Figure 3(a)). Moreover, 4-HIL (III) and the metformin group (IV) showed reduction in AUC relative to the diabetic control group by 9.42% and 9.03%, respectively (Table 3). Experiment 1: Effects of 4-HIL treatment on (a) OGTT and (b) IPGTT of diabetic rats (n = 24). ***p < 0.001 compared to the STZ group (II). Total area under curve (AUC) of OGTT in different group of rats. Values are expressed as mean ± SD (no statistical differences were found between groups).
Effect of 4-HIL on intraperitoneal glucose tolerance test (IPGTT)
Total area under curve (AUC) of IPGTT in different group of rats.
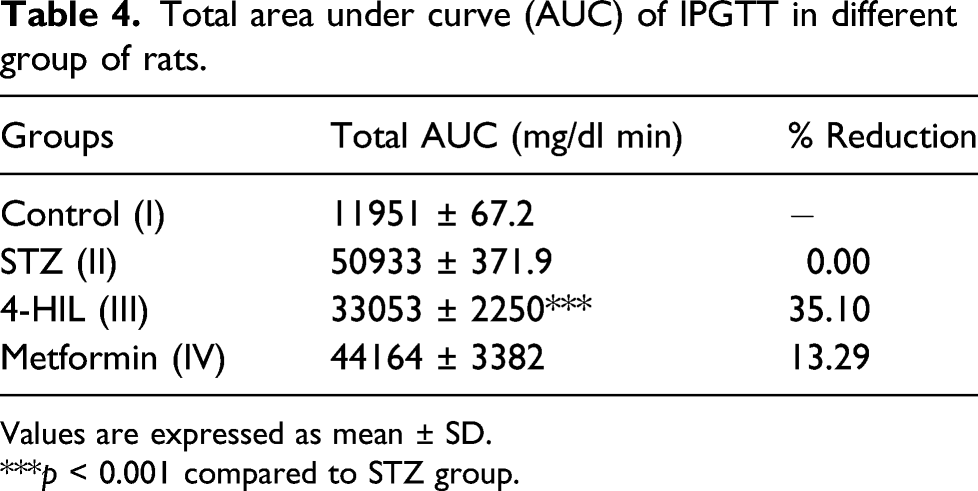
Values are expressed as mean ± SD.
***p < 0.001 compared to STZ group.
Effect of 4-HIL on serum biochemical parameters
Effect of 4-HIL on mean values of serum biochemistry of diabetic rats.
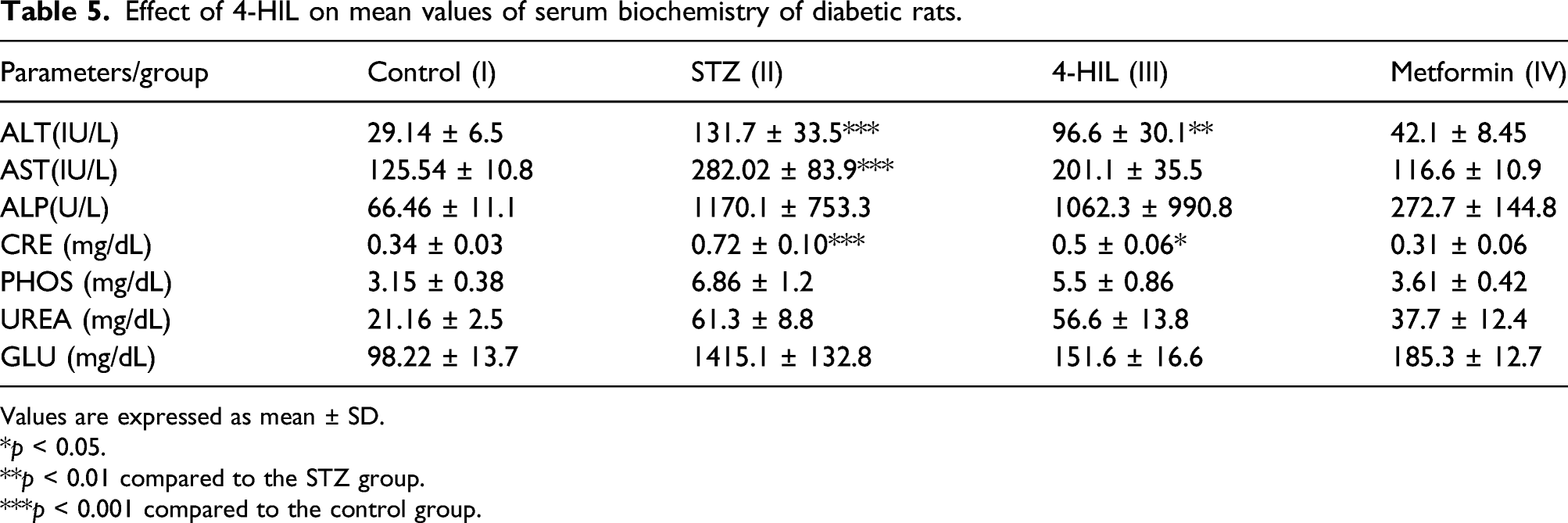
Values are expressed as mean ± SD.
*p < 0.05.
**p < 0.01 compared to the STZ group.
***p < 0.001 compared to the control group.

Experiment 1: Serum clinical biochemistry analysis of (a) ALP, (b) AST, (c)ALT, and (d) creatinine in different groups of rats (n = 24). *p < 0.05, **p < 0.01 compared to the STZ group (II), ***p < 0.001 compared to the control group (I).
4-HIL showed minimal hematological changes
The experiment 1 showed mild alterations in hematological parameters in the 4-HIL-treated group (III). These hematological changes were within the biological significance level as compared to the control group I (Supplementary Table- ST1). No noticeable changes were observed in WBC count, neutrophils, lymphocytes, and platelet counts between the 4-HIL group and the healthy rats (experiment 2).
4-HIL restored morphological and histopathological alterations
At the end of the study, the animals were sacrificed and gross pathological changes of all the organs were noted and subjected to histopathological analyses. Except pancreas, all organs had normal morphology and consistency. In the STZ group (II) (experiment 1), the pancreas was found to be pale, inflamed and swollen from control group (I). However, minimal changes were observed in the pancreas of 4-HIL group (III) and metformin group (IV) (experiment 1). Pancreatic sections stained with H&E exhibit normal histological structure in control group (I) (experiment 1). While the parenchyma of diabetic rats showed fibrinous degeneration ((Supplementary FigureS1)), loss of islets/diminution in fibrosis (Figure 5(b)(i)), and prominent fibrosis band (Figure 5(b)(ii)). The pancreatic parenchyma of 4-HIL- (Figure 5(c)) and metformin- (Figure 5(d)) treated rats exhibited mild mononuclear cell infiltration and vacuolation compared to the control group. No histopathological alterations were observed in the sections of kidney tissues of different group(s) (Figure 5 (ii) a, b, c, and d) as compared with that of the control group I (experiment 1). There were no gross and histopathological changes observed in experiment 2. (i) Rat(s) experiment 1: Representative histopathological images of rat pancreas: (a) control group (I); (b) STZ group (II): (i) pancreas islets: diminution in fibrosis (red arrow) and (ii) pancreas: prominent fibrosis band (black arrow); (c) the 4-HIL group (III); and (d) the metformin group (IV) showed minimal changes. (ii) Rat(s) experiment 1: Kidney(s): (a) Control group (I), (b) STZ group (II), (c) 4-HIL group (III), and (d) metformin group (IV). No histopathological changes has been observed in the kidneys of all groups.
4-HIL upregulates insulin expression in pancreatic tissue
Western blot analysis showed treatment with 4-HIL significantly enhances the expression of insulin as compared to the diabetic group (II). However, the metformin group (IV) showed less expression as compared to group III in (Figure 6). 4-HIL regulates the expression of insulin. (a) Immunoblot analysis with antibody against insulin protein. (b) Densitometric quantification of insulin relative to GAPDH. **p < 0.01 compared to the STZ group.
Cytotoxic effect of 4-HIL in HEK-293 cell line
4-HIL treatment had no deleterious effect on the viability of the HEK-293 cell line at the concentration 1, 10, and 25 μ Graph depicts percent viability in HEK-293 cells in response to various doses of 4-HIL. ***p < 0.001 compared to the control group (I).
Effect of 4-HIL on acute oral toxicity
Single oral dosing of pure 4-HIL at the dose of 1000, 1500, 2000 mg/kg showed no mortality in rats. The 4-HIL was well tolerated up to 20X times of that effective dose (100 mg/Kg BW) in an acute toxicity study. All other parameters including clinical signs, body weight, feed consumption, and postmortem examination were comparable between the treated and control groups. There were no abnormal gross findings observed during the experimental period and the same were noted in the data recording sheet. Thus, 4-HIL was found to be safe in acute toxicity studies in rats.
Discussion
Diabetes has become a public health concern that has reached epidemic proportions globally. Decreased hyperglycemia may reduce the likelihood of developing microvascular diseases. 20 The traditional pharmacological therapies have a wide range of adverse and toxic effects. However, medicinal plants can have a similar degree of efficacy without any side effects. In this regard, the World Health Organization recommended the development of herbal medicines. 21
A growing amount of evidence indicates the glucose molecules cannot reach the cells of the satiety region in hyperglycemic condition which causes a low arterial venous difference that keeps the feeding center remains consistently active, leading to polyphagia. 22 Our results showed increased feed intake in STZ-induced rats but decrease in body weight. Whereas feed consumption was almost the same in the control group (I) and 4-HIL group (III). It has been shown that body weight of diabetic rats reduced due to improper glucose metabolism, leading to break down of fats and proteins.23,18 It is reported that 4-HIL regulates body weight effectively 12 which is in agreement with our results showing decrease in body weight of diabetic rats, while 4-HIL-treated rats have body weight close to that of non-diabetic rats.
Majithiya et al. 24 investigated the treatment of non-diabetic rats with complex of curcumin (an active agent of Curcuma longa known to have hypoglycemic activity), and it had no effect on the body weight of normal rats which is consistent with our results showing no changes in the body weight and feed intake in the normal control group rats and 4-HIL-treated rats (experiment 2). Blood glucose level is an important parameter to detect the progression and status of diabetes. 18 In addition, it has been shown that ethanolic extract of Momordica cymbalaria reduced blood glucose level to 270 ± 6.9 mg/dl after 7 h of treatment in diabetic rats. 25 These findings are in accordance with our study in which blood glucose level was increased in STZ-induced diabetic group II (experiment 1) rats but reduced after oral administration of 4-HIL in diabetic rats. Unlike conventional therapies for the management of diabetes, 4-HIL has not been found to have any adverse/toxic effects, such as an increased risk of hypoglycemia or weight gain/any pathological changes. 26 When compared to control rats (experiment 2), 4-HIL had no influence on blood glucose levels in healthy male and female rats. To verify whether 4-HIL increased glucose utilization and tolerance, IPGTT and OGTT was performed in STZ-induced rats to evaluate whether the drug stimulated insulin secretion. The blood glucose level was measured in rats administered with 4-HIL (100 mg/kg) for 120 min and found that it was significantly lower than that in the STZ group (II). Researchers reported that 4-HIL pharmacologically regulates metabolic parameters such as blood glucose, lipid profile, and uric acid, and acts independently of insulin on the liver and muscle. 27 In experiment 1, biochemical serum parameters such as ALT, AST, and ALP were increased in the STZ group (II) as compared with that of the 4-HIL group (III) and metformin group (IV) rats. T2DM has been linked to several hematological abnormalities that affect the blood cells’ deformability. 28 In the present study, 4-HIL group (III) (experiment 1) restored the altered hematological parameters. In comparison to the control group (I) rats, 4-HIL showed normal hemato-biochemical parameters in healthy rats (experiment 2; group II).
It is well-known that hyperglycemia accelerates the production of free radicals. 29 These free radicals react with biomolecules such as lipids, proteins, and DNA which leads to the alterations in histological characteristics of organs. 30 A study reported degeneration and vacuolization in the endocrine pancreas cells as well as decrease in β-cell number and islets architecture of pancreatic tissue from diabetic rats. 31 The present findings showed histopathological alterations in parenchymal structure, fibrinous degeneration, and diminution/decrease islets of Langerhans were observed in STZ rats (group II, experiment 1). However, these typical changes were greatly reduced in the 4-HIL group (III) and metformin group (IV) as compared with the STZ group (II). STZ is a diabetogenic agent that causes necrosis by destroying the DNA of pancreatic beta cells. This remarkably lowers insulin production in rats which results in the development of a diabetic phenotype. 18 Insulin is involved in both protein and carbohydrate metabolism. Previous studies have shown that 4-HIL stimulates insulin secretion with the activation of insulin signaling pathways. 32 Interestingly, immunoblot analysis revealed the increased expression of insulin in the 4-HIL group (III) compared to the STZ group (II).
Herbal drugs are less toxic and free of side effects than synthetic molecules. It has been suggested that 4-HIL was found to be safe at a concentration of 10 μg mL−1 on the 3T3-L1 cell line. 33 The cytotoxicity results showed that it does not have any effect on the cell viability up to a concentration range of 25 μM. Moreover, 4-HIL was found to be safe and non-toxic in acute oral toxicity studies in rat up to 2000 mg. Kg, BW. Collectively, these results suggested the non-toxic effect and antidiabetic potential of 4-HIL in healthy rats and T2DM rats, respectively. We believe that this study can be used to design and synthesize more such effective and safer antidiabetic analogues.
However, our study has some limitations as well, such as lack of underlying mechanism of how glucose metabolism by 4-HIL in healthy rat preserved normoglycemia and altered metabolite profiles of diabetic and treated group rats.
In conclusion, our findings demonstrate the ameliorative effect of 4-HIL by lowering the altered blood glucose level in hyperglycemic rats as well as not changing the normal level of glucose in healthy rats without any toxic effects in vitro and in vivo. Our findings provide strong scientific evidence for antidiabetic potential of 4-HIL which intensely rationalize its use in the management of diabetes and associated secondary complications.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211061873 – Supplemental Material for Assessment of antidiabetic effect of 4-HIL in type 2 diabetic and healthy Sprague Dawley rats
Supplemental Material, sj-pdf-1-het-10.1177_09603271211061873 for Assessment of antidiabetic effect of 4-HIL in type 2 diabetic and healthy Sprague Dawley rats by Pragati Singh, Sharmeen Ishteyaque, Ramanand Prajapati, Karan S Yadav, Rupali Singh, Akhilesh Kumar, Sharad Sharma, Tadigoppula Narender and Madhav N Mugale in Human & Experimental Toxicology
Footnotes
Acknowledgment
The authors are thankful to the Director, CSIR-CDRI Lucknow, India, for providing excellent research facilities and histopathology department for their technical support. Research of all the authors’ laboratories was supported by CSIR-CDRI Institutional Fund (MLP0013). Institute manuscript number for this paper is 10308
Declaration of conflict of interest
The author(s) declare(s) that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
