Abstract
Objective
Sulforaphane, which exerts an effective anti-cancer ability, is a phytochemical converted from cruciferous plants. Here, we aimed to identify whether sulforaphane could suppress autophagy during the malignant progression of gastric carcinoma and to explore the underlying mechanisms.
Methods
SGC7901 cells were transfected with miR-4521 mimics, inhibitor, and pcDNA3.1-PIK3R3, and treated with sulforaphane or autophagy inhibitor. Cell proliferation, apoptosis, and miR-4521 or PIK3R3 expression were detected.
Results
MiR-4521 over-expression suppressed LC3-II/I ratio and Beclin-1 expression but induced p62 expression in SGC7901 cells. MiR-4521 also reduced gastric carcinoma cell proliferation and promoted apoptosis in vitro. In the mechanical observation, we identified that miR-4521 directly targeted PIK3R3 to repress its expression, and PIK3R3 up-regulation partly antagonized miR-4521-mediated autophagy, proliferation, and apoptosis in gastric carcinoma cells. In addition, sulforaphane exerted effective anti-cancer functions by repressing autophagy and growth in tumor cells at a concentration-dependent way. MiR-4521 inhibition or PIK3R3 over-expression weakened the anti-cancer functions of sulforaphane in gastric carcinoma cells.
Conclusion
Consequently, miR-4521 suppressed autophagy during the malignant progression of gastric carcinoma by targeting PIK3R3. Thus, miR-4521 may be applied as a therapeutic target for sulforaphane in gastric carcinoma.
Introduction
Gastric carcinoma, one of the most common malignant cancer, ranks second in causes of tumor-associated mortality worldwide. 1 Clinical follow-up investigation found that the overall 5-year survival rate of gastric carcinoma is only 20%, and it even present 50–90% risk of recurrence and death after resection operation.2,3 In spite of surgery, chemotherapy still has an important role in ameliorating overall survival of patients especially of those with advanced gastric carcinoma. 4 However, patients with gastric carcinoma develop resistance to chemotherapy drugs, which may lead to toxic side effect. In addition, up to now, there are still many aspects of the mechanisms underlying pathogenesis of gastric carcinoma remain to be elucidated, and the research of diagnosis and therapy of this tumor is still a long way to go.
Increasing insights demonstrated that high consumption of cruciferous vegetables can help to reduce the risk of cancer occurrence. 5 Sulforaphane (SFP), a dietary isothiocyanate, might be the most critical ingredient for the anti-cancer effects of cruciferous vegetables. 6 Moreover, the side effect of the monomer of traditional Chinese medicine is low. Previous observations have demonstrated that sulforaphane can suppress the proliferation and migration of various cancer cells by regulating multiple tumor-associated signaling, including MAPK pathway, the reactive oxygen species (ROS)-dependent axis, and microRNAs-pathway.5,7,8 Furthermore, sulforaphane also represses the proliferation of gastric cancer cells via changing the expression of CDX1, CDX2, miR-9, and miR-326. 8
MicroRNAs, a class of endogenous small noncoding RNAs with 19–25 nucleotides, modulate gene expression at the post-transcriptional level. 9 MicroRNAs mainly target the 3′-UTR of gene to repress its expression and change a variety of cellular signaling. 10 Increasing investigations have reported that dysregulated miRNAs exert vital roles in multiple types of human tumors, such as gastric carcinoma, acting as either oncogenes or cancer suppressor gene.11,12 Insights into the functions of microRNAs in tumors have made them attractive tools and targets for novel therapeutic strategies. 13 For example, miR-4521 has been found to under-express in advanced gastric carcinoma tissue, and suppress the progression and metastasis of cancer cells via directly regulating IGF2 and FOXM1 and inactivating AKT/GSK3β/Snai1 axis, 14 suggesting that miR-4521 may be a novel potential target for the treatment of gastric carcinoma. As several microRNAs were shown to modulate autophagy in cancer cells, 15 we explored the functions of miR-4521 in autophagy during gastric carcinoma progression. Furthermore, sulforaphane exerts the functions to regulate autophagy and exosome-mediated paracrine senescence in cancer therapy, 16 and inhibits the proliferation and migration of gastric carcinoma cells in vitro.17,18 Thus, we observed whether sulforaphane could suppress autophagy during the malignant progression of gastric carcinoma, and explore its regulatory effect on miR-4521/PIK3R3 axis in cancer cells.
Materials and methods
Cell culture and treatment
The GES-1, AGS, SGC7901, and MKN-45 cells were provided by American Type Tissue Culture Collection. These cell lines were cultivated in RPMI-1640 medium containing 10% fetal bovine serum (FBS; Gibco, Capital, IN, USA, #16000-044) and 1% penicillin and streptomycin (Gibco) at 37°C in a humidified incubator with 5% CO2. Then the lentiviral plasmids carrying miR-4521 mimics/inhibitor and pcDNA3.1-PIK3R3 (Genscript, China) were transfected into SGC7901 cells. In addition, 0.1–100 μM of sulforaphane (Sigma-Aldrich, #4478-93-7) and 2 μM of autophagy inhibitor 3-MA were used to incubate SGC7901 cells.
Quantitative reverse transcription-PCR (qRT-PCR)
The total RNA was extracted from cancer cells by TRIZOL reagent (Invitrogen, #HY-2032-41) after treatment and reverse transcribed to cDNA using Super Script III kit (Sigma-Aldrich, #5230-21). Subsequently, the qRT-PCR of miRNAs and mRNAs was carried out by All-in-OneTM miRNA qRT-PCR Detection Kit (Sigma-Aldrich, #2525-24) and Bimake™ 2× SYBR Green qPCR master mix (Sigma-Aldrich, #4156-25-8), respectively. Then, the relative levels of miRNAs and mRNAs were normalized to U6 or β-actin. The results were calculated with 2−ΔΔCt method. All primers were as follows:
miR-4521: F 5′-AGCCUUGCCAAGCGUAGC-3′, R 5′-AUUCCGGAGAGCGUGCC-3′; PIK3R3: F 5′-ATTCGCGGGTTAAGCT-3′, R 5′-GGCCTAAGTGCTAGTGCC-3′; β-actin: F 5′-CATCAAGAAGGTGGTGAATC-3, R 5-TCAAAGGTGGAGGAGTGGG-3′.
CCK-8 assay
The cell viability was detected by CCK-8 assay in SGC7901 cells. In brief, after transfection, about 2 × 104 cells were grown into 96-well plates and further cultured for 12 h. After indicated treatment, 10 μL of CCK-8 solution was added into each well, followed by incubation for 1.5 h in a humidified chamber with 5% CO2 at 37°C. Finally, the optical density (OD value) of cells was analyzed by a microplate reader (Bio-Rad, Hercules, CA, USA) at 450 nm. Cell viability (%) was calculated as ODexperiment/ODcontrol × 100%. The assay was proceeded in triplicate at each concentration.
Colony formation assay
SGC7901 cells were transfected or treated as the indication, digested and suspended as single cells, seeded into 6-well plates with 1000 cells in each well. The cells were cultured in incubator for 14 days, and the medium was changed every 2 days till the visible clones formed. The formed clones were fixed with 4% paraformaldehyde and stained with crystal violet solution, captured and counted by a microscope (Bio-Rad, USA).
Enzymatic activity of caspase-3 or -9
The activity of caspase-3 or -9 was measured by commercial kits (Invitrogen). In brief, after transfection or treatment, cells were decomposed by lysis buffer on ice for 12 min. The cell homogenates were centrifuged at 14,000 g and 4°C for 10 min, and the collected supernatants were incubated at 37°C for 2 h with the associated reagent. The absorbance was measured at 405 nm.
Flow cytometry detection
Briefly, after transfection or treatment, cells were digested with trypsin, rinsed with phosphate buffer saline (PBS), and then resuspended in RPMI-1640 medium containing 2% FBS at a density of 1 × 106 cells/mL, and stained with 5 μg/mL FITC-Annexin V (Invitrogen, #BH-1522-35) at 37°C for 90 min. At the end of the incubation period, cells were counter stained with 2 μg/mL propidium iodide (PI; Invitrogen) at room temperature for 5 min. The cell apoptosis was analyzed by an FACS Ariall System (BD Biosciences, San Jose, CA, USA).
Western blotting assay
Cell lysates were recovered and centrifuged at 14,000 g and 4°C for 12 min. Protein content in each sample was determined using the BCA Protein Detection Kit (Sigma-Aldrich, #4175-04). Protein was denatured for 7 min, and separated by 12% SDS-PAGE gel electrophoresis and transferred onto polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA). Membranes were blocked with 5% non-fat milk and then incubated sequentially with specific primary antibodies, including LC3-II (Abcam, #ab192890), LC3-I (Abcam, #ab185210), Beclin-1 (Abcam, #ab256743), p62 (Abcam, #ab207305), PIK3R3, (Abcam, #ab277882), and β-actin (Abcam, #ab7817). All antibody dilutions were 1:1000. Then, membranes were incubated with horseradish peroxidase-conjugated goat anti-rabbit antibody (diluted 1:3000) for 1.5 h at room temperature. The immune complexes were measured using enhanced chemiluminescence (Sigma-Aldrich).
Luciferase reporter gene assay
The potential binding sites of miR-4521 and the 3′UTR region of PIK3R3 were predicted by ENCORI website. The cells were transfected with PIK3R3-WT and -Mut along with miR-4521 mimics by Lipofectamine 2000 (Sigma-Aldrich). After 24 h culture, the cells were decomposed and the luciferase intensity was measured using a Dual Luciferase Assay Kit (Sigma-Aldrich). As control, the luciferase activity of Renilla was determined.
Statistical analysis
Data were shown as the mean ± standard deviation (SD). Statistical analysis was performed by unpaired Student’s t-test and one-way ANOVA using SPSS 20.0 (Chicago, IL). p < .05 was regarded as statistically significant.
Results
MiR-4251 suppresses autophagy in gastric carcinoma cells
We first explored the expression of miR-4521 in normal gastric mucosal epithelial GES-1 cells and gastric carcinoma cells, including AGS, SGC7901, and MKN-45 cells. We found that miR-4521 was significantly reduced in gastric carcinoma cells compared with GES-1 (Figure 1(a)). Then, we assessed the effects of miR-4521 over-expression on autophagy in SGC7901 cells by detecting the expressions of LC3, Beclin-1, and p62. After transfection with miR-4521 mimics, the expression of miR-4521 in SGC7901 cells was strongly up-regulated (Figure 1(b)). Furthermore, the exposure of miR-4521 mimics remarkably down-regulated LC3-II/I ratio and the expression of Beclin-1, but up-regulated p62 expression in SGC7901 cells (Figures 1(c)–(f)). Collectively, these findings suggest that miR-4521 suppresses autophagy in gastric carcinoma cells. MiR-4521 inhibits autophagy in gastric carcinoma cells. (a) The expression levels of miR-4521 in normal gastric mucosal epithelial and gastric carcinoma cells were measured by qRT-PCR. (b) The levels of miR-4521 in SGC7901 cells transfected with miR-4521 were detected by qRT-PCR. (c–f) The measurements of LC3-II/I ratio, Beclin-1, and p62 expression using Western blotting. *p < .05, compared with NC mimics.
MiR-4521 inhibits cell proliferation and induces apoptosis in gastric carcinoma cells
We also observed the impacts of miR-4521 overexpression on the growth of gastric carcinoma cells in vitro. As shown in Figures 2(a) and (b), the cell viability and colony formation ability of SGC7901 cells were significantly decreased in cancer cells transfecting with miR-4521 mimics. In contrast, miR-4521 up-regulation remarkably induced the apoptosis and the activity of caspase-3 or -9 in cancer cells (Figures 2(c)–(f)). Taken together, our results suggest that miR-4521 represses cell proliferation and promotes apoptosis of gastric carcinoma cells. MiR-4521 inhibits the growth of gastric carcinoma cells. After transfecting with miR-4521 mimics, (a) cell viability was measured by using CCK-8 assay; (b) cell proliferation was assessed by using colony formation assay; (c and d) cell apoptosis was evaluated using flow cytometry; (e and f) the enzymatic activity of caspase-3 or -9 was detected by commercial kits. *p < .05, compared with NC mimics.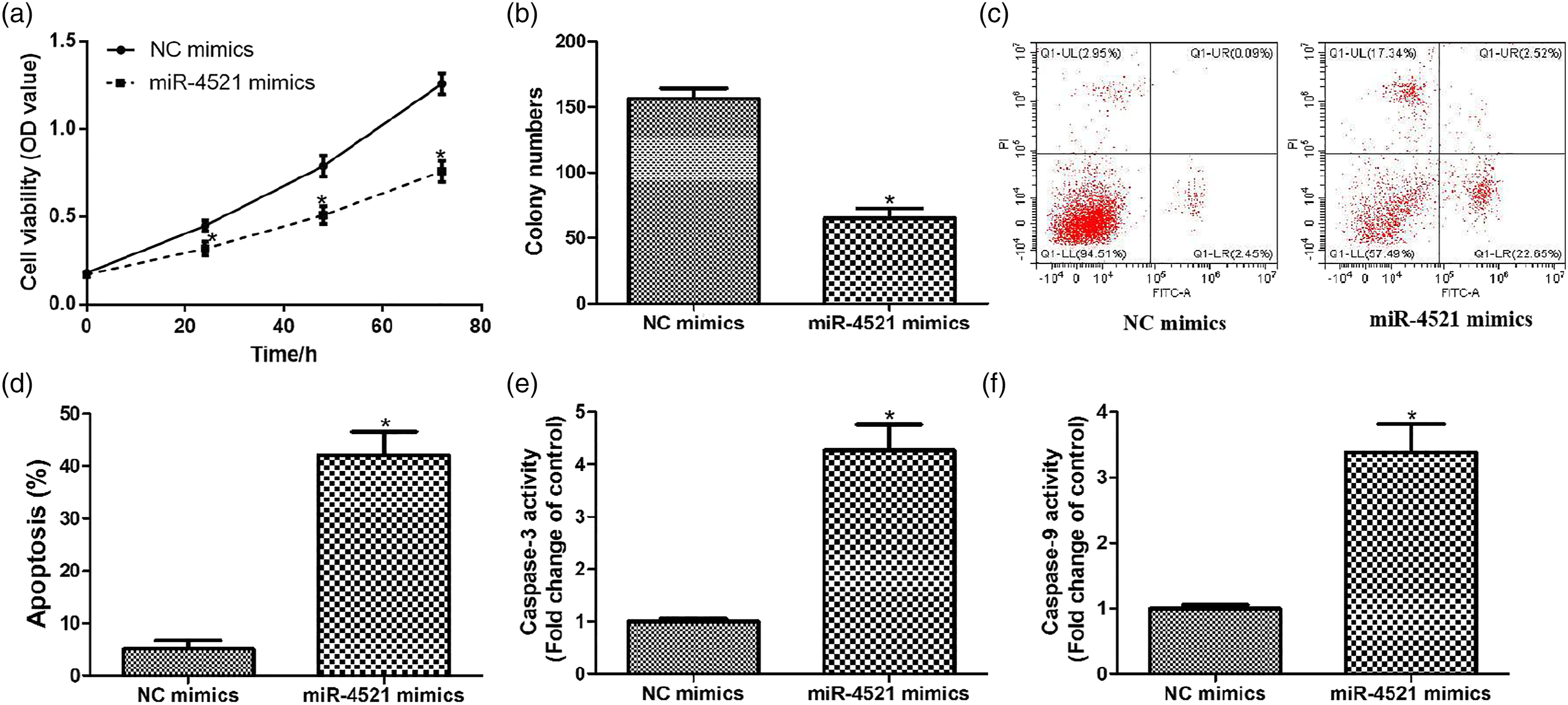
PIK3R3 is targeted by miR-4521 in gastric carcinoma cells
We then tried to study the underlying mechanism in miR-4521-mediated malignant progression of gastric carcinoma. The predicted analysis showed the potential binding of miR-4521 with PIK3R3 mRNA 3′UTR (Figure 3(a)). Additionally, miR-4521 mimics significantly suppressed the luciferase activity of PIK3R3 mRNA 3′UTR in SGC7901 cells (Figure 3(b)). The expression levels of PIK3R3 mRNA and protein were significantly down-regulated in cancer cells transfected with miR-4521 mimics (Figures 3(c)–(e)). These results indicate that PIK3R3 is directly targeted by miR-4521 in gastric carcinoma cells. PIK3R3 is targeted by miR-4521 in gastric carcinoma cells. (a) The interaction prediction analysis of miR-4521 with PIK3R3 mRNA 3′UTR using ENCORI online database. (b) The luciferase activity of PIK3R3 mRNA 3′UTR was detected by using luciferase reporter gene assay. (c) PIK3R3 mRNA expression was analyzed by using qRT-PCR. (d and e) PIK3R3 protein expression was analyzed by using Western blotting. *p < .05, compared with NC mimics.
PIK3R3 up-regulation weakens the effects of miR-4521 on autophagy in gastric carcinoma cells
Next, we explore whether miR-4521 represses autophagy in gastric carcinoma cells through the inhibition of PIK3R3. As shown in Figures 4(a)–(d), miR-4521 over-expression suppressed LC3-II/I ratio and the expression of Beclin-1, but induced p62 level, in which the PIK3R3 up-regulation partly weakened these effects in SGC7901 cells. In parallel, PIK3R3 over-expression also partly rescued the cell viability and reduced cell apoptosis in cancer cells transfecting with miR-4521 mimics (Figures 4(e) and (f)), suggesting that miR-4521 inhibits autophagy in gastric carcinoma cells by repressing PIK3R3 expression. PIK3R3 is involved in miR-4521-mediated autophagy in gastric carcinoma cells. SGC7901 cells were transfected with miR-4521 mimics and pcDNA3.1-PIK3R3, (a–d) the LC3-II/I ratio and expression levels of Beclin-1 and p62 were detected by using Western blotting; (e) cell viability was determined by using CCK-8 assay; (f) cell apoptosis was analyzed using flow cytometry. *p<0.05, compared with NC mimics; # p < .05, compared with miR-4521 mimics.
Autophagy inhibitor 3-MA enhanced miR-4521-mediated gastric carcinoma progression
We further explored the effects of autophagy inhibitor 3-MA on miR-4521-mediated gastric carcinoma progression. As shown in Figure 5(a), miR-4521 over-expression reduced cell viability, in which autophagy inhibitor 3-MA treatment further enhanced these effects in SGC7901 cells. In parallel, 3-MA also promoted cell apoptosis in cancer cells transfecting with miR-4521 mimics by detecting apoptotic rates and enzymatic activity of caspase-3 or -9 (Figures 5(b)–(d)). Autophagy inhibitor 3-MA enhanced miR-4521-mediated gastric carcinoma progression. SGC7901 cells were pre-treated with 3-MA (2 μM) for 4 h, then transfected with miR-4521 mimics for 24, 48, and 72 h. (a) Cell viability was detected by using CCK-8 assay. (b) Cell apoptosis was analyzed using flow cytometry. (c and d) Enzymatic activity of caspase-3 or -9 was measured by using commercial kits. *p < .05, compared with NC mimics; # p < .05, compared with miR-4521 mimics. Sulforaphane suppresses autophagy in gastric carcinoma cells by inducing miR-4521/PIK3R3 axis.
Furthermore, we explored the effects of sulforaphane on cell growth, autophagy, and the signal transduction of miR-4521/PIK3R3 pathway in gastric carcinoma cells. As shown in Figure 6(a), cell viability fell significantly when sulforaphane concentration is more than 10 μM. Then, these doses (10, 20, and 50 μM) of sulforaphane were selected for subsequent assays. Sulforaphane treatment promoted cell apoptosis with a concentration-dependent manner, an effect associated with increasing of caspase-3 activity (Figures 6(b) and (c)). Meanwhile, sulforaphane also repressed autophagy in SGC7901 cells via reducing LC3-II/I ratio, Beclin-1 expression, and inducing p62 level at a dose-dependent way (Figures 6(d)–(g)). Additionally, sulforaphane significantly promoted miR-4521 and suppressed PIK3R3 expression in gastric carcinoma cells (Figures 6(h) and (i)). These findings indicate that sulforaphane may suppress autophagy in gastric carcinoma cells by inducing miR-4521/PIK3R3 signaling pathway. Sulforaphane represses autophagy in gastric carcinoma cells by regulating miR-4521/PIK3R3 pathway. (a) Cell viability of cancer cells was measured using CCK-8 assay after exposure to 0.1–100 μM of sulforaphane for 24 h. (b) Cell apoptosis and (c) caspase-3 activity in cancer cells were detected after treatment with sulforaphane (10, 20, and 50 μM) for 24 h. (d–g) The LC3-II/I ratio and expressions of Beclin-1 and p62 were determined by using Western blotting. (h and i) The expressions of miR-4521 and PIK3R3 mRNA were measured using qRT-PCR. *p < .05, compared to control group (CN). SFP = sulforaphane.
MiR-4521 low-expression antagonized the ability of sulforaphane to repress autophagy in gastric carcinoma cells
We further investigated whether sulforaphane represses autophagy in SGC7901 cells via the miR-4521 up-regulation. Thus, sulforaphane-treated SGC7901 cells were transfected with miR-4521 inhibitor. In cells exposed to sulforaphane, the cell viability, colony numbers, and PIK3R3 expression were remarkably increased, whereas cell apoptosis and caspase-3 activity were significantly increased, in which miR-4521 inhibitor treatment partly antagonized these effects (Figure 7). Our findings verify that sulforaphane could repress autophagy in gastric carcinoma cells via up-regulating miR-4521 expression. miR-4521 inhibition reduced the ability of sulforaphane to suppress autophagy in gastric carcinoma cells. (a) Cell viability was measured by using CCK-8 assay. (b) Cell proliferation was assessed by using colony formation assay. (c) Cell apoptosis was evaluated using flow cytometry. (d) The enzymatic activity of caspase-3 was detected by using commercial kits. (e and f) PIK3R3 expression was detected using Western blotting. *p < .05, compared to control group (CN); #p < .05, compared to SFP group. SFP = sulforaphane.
PIK3R3 over-expression reduced the ability of sulforaphane to repress the growth of gastric carcinoma cells
As shown in Figures 8(a) and (b), the viability and colony numbers of SGC7901 cells exposed to sulforaphane were strongly higher when cells were over-expressing PIK3R3. In contrast, PIK3R3 over-expression antagonized the inhibitory effects of sulforaphane on cell apoptosis and caspase-3 activity in gastric carcinoma cells (Figures 8(c) and (d)). These results verify that sulforaphane could inhibit the growth in gastric carcinoma cells via suppressing PIK3R3 expression. PIK3R3 up-regulation reduced the ability of sulforaphane to repress the growth of gastric carcinoma cells. (a) Cell viability was measured by using CCK-8 assay. (b) Cell proliferation was assessed by using colony formation assay. (c) Cell apoptosis was evaluated using flow cytometry. (d) The enzymatic activity of caspase-3 was detected by using commercial kits. *p < .05, compared to control group (CN); #p < .05, compared to SFP group. SFP = sulforaphane.
Discussion
It is well known that autophagy is an important cellular process, and the regulators of autophagy are thought to be underlying therapeutic targets for gastric carcinoma. 19 Several microRNAs control autophagy and metabolism to modulate chemoresistance of gastric carcinoma. 20 Autophagy regulates cell proliferation and invasion in gastric carcinoma. 21 Circular RNA circNRIP1 promotes gastric carcinoma progression via inducing AKT1/mTOR pathway through autophagy-mediated miR-149-5P. 22 Furthermore, several microRNAs have been found to modulate autophagy in gastric carcinoma cells. MiR-582-5p activates cell autophagy and induces resistance in gastric adenocarcinoma. 23 MiR-30 suppression promotes autophagy-related gastric carcinoma cell death by up-regulating Beclin-1. 24 Additionally, it has been demonstrated that miR-4521 up-regulation suppresses gastric carcinoma progression via targeting IGF2 and FOXM1. 14 mIR-4521 targets FAM129A and further reduces the migration and invasion abilities of renal cancer cells through the TIMP-1/MMP2/MMP9 pathway. 25 In this study, we found that miR-4521 reduced autophagy-associated phenotypes in gastric carcinoma cells. MiR-4521 suppressed the proliferation and promoted apoptosis of gastric carcinoma cells in vitro. These findings suggest a novel function of miR-4521 in the autophagy modulation and gastric carcinoma progression, providing insights into the roles of miR-4521 in malignant tumors.
In our research, we further assessed the functions of miR-4521 as an example in the regulation of autophagy in gastric carcinoma. And, whether or not have other microRNAs as well as miR-4521 to repress autophagy in gastric carcinoma progression and their synergic effect should be further observed. Several factors influence the outcome of miR-4521-mediated autophagy and tumor progression. Based on the previous investigation, these upstream factors of miR-4521, such as hypoxia 14 and STAT3 signaling, 26 affect cancer progression, including gastric carcinoma and colorectal cancer. Moreover, whether these factors regulate miR-4521-mediated autophagy in gastric carcinoma should be confirmed in further observations.
PIK3R3 acts as an oncogene in a variety of tumors, including glioma, lung cancer, and gastric cancer.27–29 Some observations have reported that several microRNAs can repress the growth of cancer cells by targeting PIK3R3.30,31 In this investigation, we found that PIK3R3 is targeted by miR-4521 and involved in miR-4521-mediated autophagy in gastric carcinoma cells. These results indicate that miR-4521 directly targets PIK3R3 to repress its expression and reduce autophagy in gastric carcinoma cells. However, in the present study, we have not repeated the experiment with PI3KR3 inhibitor to assess its effect on the ability of sulforaphane to repress the growth of gastric carcinoma cells.
Sulforaphane was reported to modulate several microRNAs expression through direct or indirect mechanism.7,32 For example, sulforaphane increased the expression of miR-124 in gastric carcinoma cells. 19 However, the association among sulforaphane and miR-4521 has not been reported. Here, we validated that sulforaphane up-regulated miR-4521 expression and down-regulated PIK3R3 level to repress autophagy in gastric carcinoma cells. Previous study also found that resveratrol could induce autophagy to inhibit the proliferation of non-small-cell lung cancer cells. 33 Both miR-4521 low-expression and PIK3R3 up-regulation attenuated the functions of sulforaphane on gastric carcinoma progression, indicating that the activation of miR-4521/PIK3R3 pathway was required for sulforaphane to repress gastric carcinoma cell growth. Similar to previous studies, sulforaphane was reported to induce cell apoptosis through the inhibition of autophagy in esophageal squamous cell carcinoma or human breast cancer.6,34 Consequently, miR-4521 suppressed autophagy during the malignant progression of gastric carcinoma by targeting PIK3R3.
Conclusions
In summary, miR-4521 might be an important mediator in the anti-cancer effects of sulforaphane. To our best knowledge, this is the first observation that miR-4521/PIK3R3 axis is implicated in the effects of sulforaphane on autophagy in cancer cells, suggesting that miR-4521 may be applied as a therapeutic target for sulforaphane in gastric carcinoma. Our conclusion would help to better explore the novel chemotherapeutic drugs derived from sulforaphane for gastric carcinoma.
Footnotes
Acknowledgment
The authors thank the Hubei Key Laboratory of Kidney Disease Pathogenesis and Intervention.
Author' contributions
Z-TP and PG conceived and designed the research. PG conducted the experiments. Z-TP analyzed and interpreted the data. Z-TP drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
