Abstract
Ischemic stroke is a leading cause of death and long-term disability worldwide. The aim of this study is to explore the potential function of ephedrine in ischemic stroke and the underlying molecular mechanism. A middle cerebral artery occlusion (MCAO) rat model was established. The potential effects of ephedrine on MCAO rats and LPS-stimulated BV2 microglial cells were evaluated. Ephedrine reduced the infarct volume, cell apoptosis, brain water content, neurological score, and proinflammatory cytokines (TNF-α and IL-1β) production in MCAO rats. Ephedrine treatment also suppressed TNF-α and IL-1β production and NOD-like receptor pyrin domain 3 (NLRP3) inflammasome activation in BV2 microglial cells. The expression of NLRP3, caspase-1, and IL-1β was suppressed by ephedrine. Moreover, ephedrine treatment increased the phosphorylation of Akt and GSK3β and nuclear NRF2 levels in LPS-treated BV2 microglial cells. Meanwhile, LY294002 attenuated the inhibitory effects of ephedrine on NLRP3 inflammasome activation and TNF-α and IL-1β production. In addition, the level of pAkt was increased, while NLRP3, caspase-1, and IL-1β were decreased by ephedrine treatment in MCAO rats. In conclusion, ephedrine ameliorated cerebral ischemia injury via inhibiting NLRP3 inflammasome activation through the Akt/GSK3β/NRF2 pathway. Our results revealed a potential role of ephedrine in ischemic stroke treatment.
Introduction
Stroke is the second leading cause of death and a major reason for long-term disability worldwide. 1 It is estimated that nearly one quarter of all adults will experience a stroke during their lifetime. 2 At present, there are more than 80 million of people surviving from a stroke all over the world. 2 These people have a high possibility to suffer from permanent disability. Moreover, occurrence of stroke is notably upregulated up to 40% in young populations over the past decades. 3 Ischemic stroke takes up for nearly 80% of all stroke cases, and focal ischemic stroke is the most common type. The main cause for ischemic stroke is blockage of cerebrovascular blood flow. Risk factors for ischemic stroke include hypertension, diabetes, smoking, and diet. 4 The primary treatments for ischemic stroke are thrombolytic therapies, which do provide great benefits for ischemic stroke patients. However, these treatments have negative effects, such as high risk of re-occlusion and intracerebral hemorrhage. 5 Besides, re-establishment of blood flow to a damaged brain tends to cause further injury in ischemic tissues due to infiltration of neutrophils, increasing of reactive oxygen species (ROS), dysregulation of ion homeostasis, and inflammatory response. 6 Therefore, there is an urgent need for developing new drugs for ischemic stroke treatment.
NOD-like receptor pyrin domain 3 (NLRP3) inflammasome is a vital factor that regulates caspase-1 activation and production of proinflammatory cytokines such as IL-1β and IL-18 in response to cellular damage. 7 NLRP3 inflammasome can be activated by various factors, such as accumulation of ROS, mitochondrial dysfunction, and lysosomal damage. Recently, NLRP3 inflammasome draws much attention for stroke treatment because it is an important regulator of inflammation in the ischemic region. 8 Microglia are macrophage-like glial cells in the brain. It can be activated and differentiated into the M1 or M2 phenotype, thus mediating inflammatory response during cerebral ischemia and reperfusion process. 9 There are increasing evidences proving that NLRP3 inflammasome is activated in microglia.10,11 Freeman et al. 11 report that NLRP3 inflammasome activation in microglia plays a central role in neuroinflammation. Thus, inhibition of NLRP3 inflammasome activation is a promising strategy for ischemic stroke treatment.
Ephedrine is a natural compound isolated from Ephedra genus. As a common Chinese herb medicine, ephedrine is firstly synthesized in 1920s. Ephedrine is a sympathomimetic amine with unique effects compared with other sympathomimetic agents such as pseudoephedrine and phenylephrine. 12 Ephedrine can bind directly to alpha and beta adrenoreceptors, but the effects of ephedrine are mostly due to inhibiting neuronal norepinephrine reuptake and displacing more norepinephrine from storage vesicles. 13 Increasing the norepinephrine level in the synapse results in a sustained or even increased heart rate and reflex bradycardia. Moreover, ephedrine has a profound influence on arterial blood pressure and venous system, thus elevating central venous pressure when the patient is fluid challenged.13,14 The pharmacological activity of ephedrine has been explored. Oral formulations of ephedrine have been used for treating asthma in the old times. The study by Diokno et al. in 1975 indicates that ephedrine has the activity against urinary incontinence. 15 Moreover, ephedrine is an FDA-approved drug to treat hypotension perioperatively. Dusitkasem et al. 16 report that ephedrine is a promising drug against hypotension induced by neuraxial blockade in high-risk pregnancies. Recent studies also suggest a role of ephedrine in ischemic brain injury, but the exact effect and underlying mechanism are still to be elucidated.17,18
Accumulated studies indicate that management of blood pressure is important for ischemic stroke treatment. 19 For instance, He et al. 20 report that maintaining appropriate blood pressure is associated with cerebral perfusion and neurologic function recovery. Kang et al. 21 find that phenylephrine-induced hypertension reduces ischemic brain injury during transient middle cerebral artery occlusion. As ephedrine can elevate central venous pressure when the patient is fluid challenged, we speculated that ephedrine might show some benefits for treating ischemic stroke. Thus, the effect of ephedrine was evaluated in middle cerebral artery occlusion (MCAO) rats and lipopolysaccharide (LPS)–stimulated BV2 microglial cells. We found that ephedrine alleviated brain injury and inflammatory response via inhibiting NLRP3 inflammasome activation through the Akt/GSK3β/NRF2 pathway. Our results suggested a potential role of ephedrine in treating ischemic stroke.
Materials and methods
Middle cerebral artery occlusion (MCAO) Model
The protocol of animal experiments was reviewed and approved by the Ethics Committee of Animal Experimentation of the Beijing University of Chinese Medicine. Male Sprague-Dawley rats (280–320 g) were purchased from the SLRC Laboratory Animals (Shanghai, China) and housed under a 12-h light/dark cycle, temperature controlling, and free diet environment. The rat MCAO model was established as previously reported.22,23 In brief, rats were subjected to fast for 12 h before the MCAO model was built, then anesthetized by pentobarbital sodium (50 mg/kg) before surgery. A silicon-coated 4-0 monofilament nylon suture was used to occlude the left middle cerebral artery of rats. At 90 min post-MCAO, the suture was withdrawn to allow reperfusion. Rats in the Sham group experienced the same surgical procedures without suture for left middle cerebral artery occlusion. Becasuse the rats were divided into four groups: Sham; I/R; IR+20 mg/kg ephedrine; IR+40 mg/kg ephedrine.. Ephedrine was administered at 90 min post-MCAO. Meanwhile, rats in the Sham group received equal volume of PBS as the vehicle control.
TTC staining
Mannitol (0.5
TUNEL staining
Cell apoptosis in the hippocampus region of brain of the rats was evaluated using a TUNEL assay Kit (Roche Biochemicals, Mannheim, Germany) according to the manufacturer’s protocol at 24 h post-MCAO. In brief, sections of the hippocampus region were deparaffinized by xylene and rehydrated by alcohol gradients. Sections were then stained with TUNEL staining buffer at 37°C for 1 h in a dark room. The nucleus of cells was stained by DAPI at room temperature for 15 min. Images were taken by microscope (Nikon, Tokyo, Japan). Each sample was done in triplicates.
Western blot
The brain tissues of each group at 24 h post-MCAO were frozen in liquid nitrogen and homogenized in tissue lysis buffer. Cell lysates from tissue samples or culture cells were extracted using RIPA buffer (Beyotime, China) with protease inhibitors (Sigma, USA). Protein concentration was determined by the BCA kit (Thermofisher, USA). A total of 25 μg protein was separated by 10% or 15% SDS-PAGE and transferred to PVDF membranes. The membranes were blocked with 5% skim milk at room temperature for 90 min and incubated with primary antibodies at 4°C overnight. The specific first antibodies were listed below. Cleave caspase-3 (#9661, CST, USA), cleave PARP (#5625, CST, USA), TNF-α (#8148, CST, USA), IL-1β (#12703, CST, USA), NLRP3 (#15101, CST, USA), AKT (#9272, CST, USA), p-AKT (#4060T, CST, USA), Nrf2 (#12721, CST, USA), GSK3β (#5676, CST, USA), p-GSK3β (#9322, CST, USA), and GAPDH (#5174, CST, USA) were purchased from cell-signaling technology (CST) and diluted at 1: 1000 for use. After washing for 4 times in Tris-buffered saline (0.1% Tween 20), the membranes were incubated with the corresponding horseradish peroxidase-conjugated secondary antibody (#7074, CST, 1: 5000) at room temperature for 1 h. The level of total protein was quantified by Image J software.
Determination of the brain water content
The brain water contents of rats in each group were measured to determine the brain edema. Briefly, the brains of rats in each group were collected at 24 h post-MCAO. Blood stains were absorbed and removed from the brain by neutral filter paper. Ipsilateral cortical segments of brains were dissected, and the wet weight of the tissue was measured. Then, the brains in each group were dried overnight at 105°C to measure the dry weight. The brain water content was calculated according to the formula: (wet weight − dry weight)/wet weight × 100%.
Neurological score
Rats were evaluated for neurologic deficits at 24 h post-MCAO according to Longa 5 scores blindly and randomly as previously reported. 25 The score was determined as follows: 0, no deficit; 1, failure to fully extend left forepaw, mild neurological deficit; 2, circling to the left, moderate neurological deficit; 3, failing to the left, severe neurological deficit; 4, unable to walk spontaneously, conscious loss.
ELISA assay
The expression levels of TNF-α and IL-1β in the brains of MCAO rats at 24 h post-MCAO or BV2 microglial cells were detected by the ELISA kit (TNF-α #ab236712 and IL-1β #ab255730, Abcam, USA) according to the manufacturer’s protocols. The expression levels of reactive oxygen species (ROS), malondialdehyde (MDA), and superoxide dismutase (SOD) in BV2 microglial cells were detected by the ELISA kit according to the manufacturer’s protocols (ROS #E004, MDA #A003, SOD #A001, Nanjing Jiancheng Bioengineering Institute, China). At the end of assay, the optical density at 450 nm was measured by a microplate reader (Bio-Rad, USA). Each sample was done in triplicates.
Immunohistochemistry (IHC) staining
The brains of rats from each group were collected for immunohistochemistry (IHC) staining at 24 h post-MCAO. The brains were fixed by 4% paraformaldehyde at 4°C overnight, embedded by paraffin, and sliced into 5-μm thick sections. The sections were deparaffinized and treated with 3% H2O2 methanol solution to eliminate endogenous peroxidase. Next, the sections were blocked by 5% goat serum for 10 min at room temperature and then incubated with specific primary antibodies at 4°C overnight and secondary antibodies at room temperature for 1 h. Then, the sections were incubated with streptomycin-labeled alkaline phosphatase at 37°C for 30 min. Then, sections were stained by DAB reagents, followed by hematoxylin staining. Images were acquired by a light microscope. Five fields were chosen randomly for each section. The degree of tissue damage was expressed by the denatured cell index (number of denatured cells/total number of cells). The specific primary antibodies were: Anti-TNF alpha antibody (Abcam #ab220210, 1: 300), Anti-AKT1 (phospho S473) antibody (Abcam #ab81283, 1: 300), and Anti-NLRP3 antibody (Abcam #ab263899, 1: 300).
CCK-8 assay
The cytotoxicity of ephedrine on BV2 microglial cells was measured by the cell counting kit-8 assay (CCK-8) (Beyotime, China) according to the manufacturer’s instruction. In brief, cells (3000/well) were seeded in 96-well plates and treated with ephedrine for 6 days. Then, 10 μL CCK-8 reagent was added to each well and re-incubated at 37°C for 30 min. The absorbance at 450 nm was measured by a microplate reader (Bio-Rad). Each sample was done in triplicates.
Immunofluorescence staining
The protein expression of NRF2 in BV2 cells was viewed by immunofluorescence staining at 24 h post–LPS-stimulation. In brief, cells were fixed by 4% paraformaldehyde for 15 min and then penetrated with 0.2% TritonX-100 for 10 min at room temperature. After blocking with 0.5% BSA for 1 h at room temperature, the cells were incubated with the primary antibody (NRF2, CST #12721, 1: 200) at 4°C overnight and secondary antibody (Anti-rabbit IgG Alexa Fluor 594 Conjugate, CST #8889, 1: 500) for 1 h at room temperature in a dark room. Images were photographed using the LSM5Pa Laser Scanning Microscope (Zeiss Germany, Oberkochen, Germany). Five fields were chosen randomly for each section for statistical analysis.
Statistical analysis
SPSS 19.0 was used for statistical analysis. Two-tailed unpaired or paired Student’s t test was used to compare difference of two groups. One-way ANOVA (LSD post-hoc test) was used to compare the difference between multiple groups. Data were displayed as mean ± standard deviation (x ± SD). p ≤ .05 was considered statistically significant.
Results
Ephedrine ameliorates ischemia-induced brain damage, cerebral edema, and neurological dysfunction following MCAO in rats
To study the influence of ephedrine on ischemia-induced brain damage, a MCAO rat model was built in our study. Ephedrine (EH, 20 or 40 mg/kg) was administered at 90 min post-MCAO, and then infarct volume, brain histopathology, brain water content, and neurological score were evaluated at 24 h post-MCAO. The experimental design of the animal studies is shown in Figure 1(a). In the present study, the doses of 20 or 40 mg/kg EH were selected according to previous reports.26–28 According to these studies, a short time treatment (24 h) of 20 or 40 mg/kg EH would not cause serious impact on health of rats. Thus, we selected the doses of 20 and 40 mg/kg EH in our study by balancing the effect and toxicity of EH in SD rats. As shown in Figure 1(b) and (c), the infarct volume of the cerebral ischemia and reperfusion (I/R) group was evidently reduced by 20 or 40 mg/kg ephedrine (EH) treatment, indicating that ephedrine ameliorated ischemia-induced brain damage. The hippocampus region of infarct brains was also examined. The IR group exhibited more severe tissue damage compared with the Sham group, but this was diminished by 20 or 40 mg/kg ephedrine treatment (Figure 1(d) and (e)). Cell apoptosis in the hippocampus region of brain was determined by TUNEL staining. The TUNEL-positive cells were dramatically increased in the I/R group compared with the Sham group (Figure 1(f) and (g)). However, the number of apoptotic cells in the I/R group were reduced by 20 or 40 mg/kg ephedrine treatment (Figure 1(d) and (f)). Moreover, the levels of cleaved caspase-3 and cleaved PARP were apparently reduced by ephedrine treatment in the I/R group (Figure 1(h)), which was consistent with the results in TUNEL staining. We also measured the brain water content and neurological score in ischemic rats. The percentage of the brain water content in the I/R group was increased notably compared with that in the Sham group but reduced by ephedrine treatment (Figure 1(i)). Similarly, ephedrine administration apparently improved the neurological score in ischemic rats compared with that in the I/R group (Figure 1(j)). Taken together, our results indicated that ephedrine ameliorated ischemia-induced brain damage, cerebral edema, and neurological dysfunction following MCAO in rats. Ephedrine ameliorates ischemia-induced brain damage, cerebral edema, and neurological dysfunction following middle cerebral artery occlusion (MCAO) in rats. (A) The experimental design of animal studies was shown. (B) Representative images of TTC staining in brain sections collected from rats receiving 20 or 40 mg/kg ephedrine (EH) treatment at 90 min post-MCAO were shown. (C) Relative infarct volumes in each group at 24 h post-MCAO were shown. D–E, IHC staining of the hippocampus region of the infarct brain at 24 h post-MCAO (D) was shown. The degree of tissue damage was expressed by the denatured cell index (E). Scale bar = 100 μm. F-G, TUNEL staining of apoptotic cells in the hippocampus region of brain in each group was conducted at 24 h post-MCAO. Representative images (F) and relative positive cells were shown (G). Scale bar = 100 μm. (H) Western blot analysis of cleave caspase-3 and cleave PARP in ischemic brain of each group at 24 h post-MCAO. (I), Percentages of brain water content in ipsilateral cortical segments were measured at 24 h post-MCAO. (J) Neurological scores of MCAO rats in each group were measured at 24 h post-MCAO. *p < 0.05 and **p < 0.01 vs I/R group. MCAO, middle cerebral artery occlusion; EH, ephedrine; TTC, 2,3,5-triphenyltetrazolium chloride; IHC, immunohistochemistry; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling.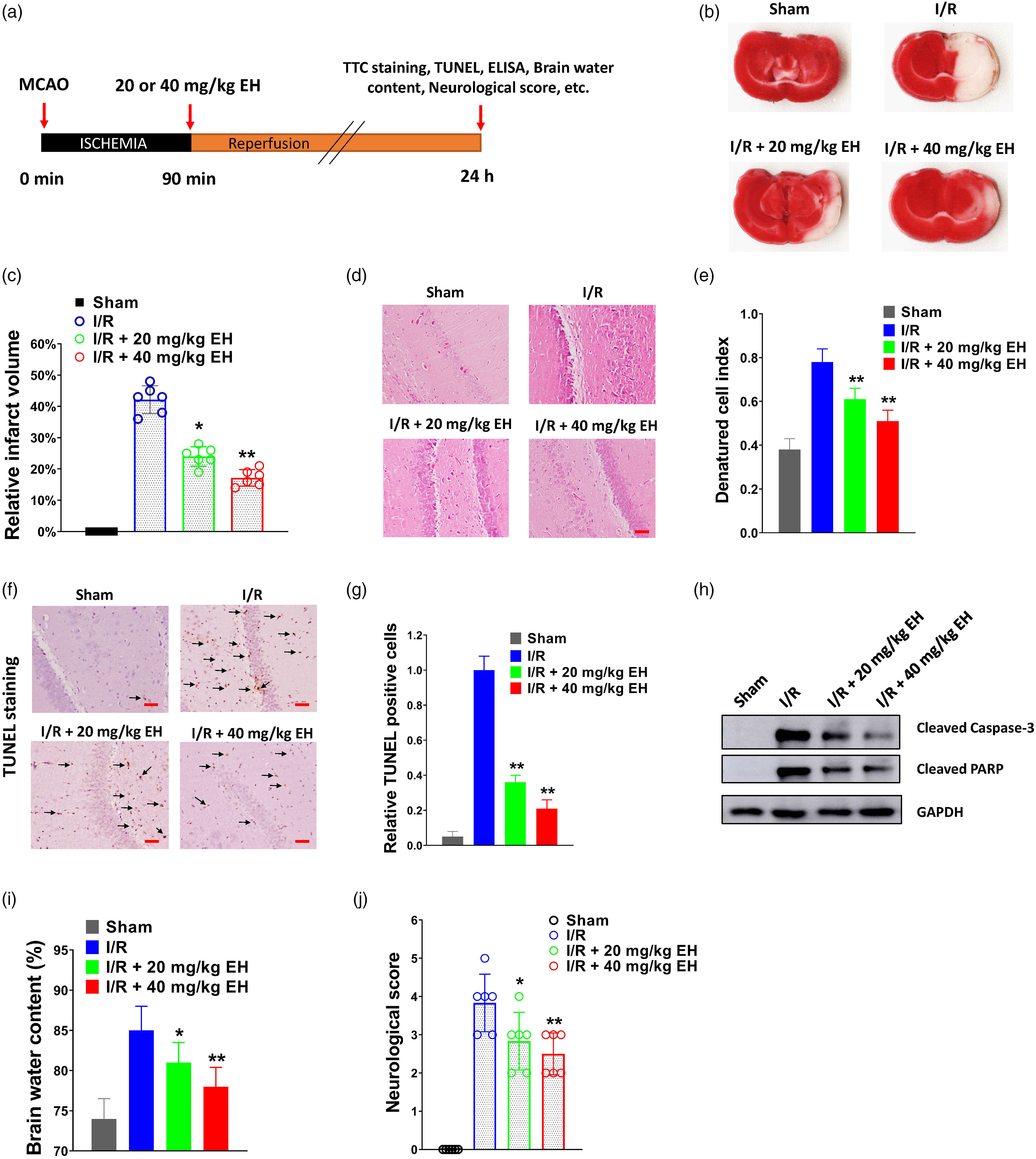
Ephedrine suppresses post-ischemic neuroinflammation following MCAO in vivo
The effect of ephedrine treatment on postischemic neuroinflammation following MCAO was evaluated. The levels of TNF-α and IL-1β in the brain tissues at 24 h post-MCAO were measured by ELISA. As shown in Figure 2 A, the levels of TNF-α and IL-1β in the I/R group were significantly increased compared with those in the Sham group but decreased by ephedrine treatment. In Western blot analysis, we found that the protein levels of TNF-α and IL-1β in the I/R group were notably reduced by ephedrine treatment (Figure 2(b) and (c)). Besides, IHC staining of TNF-α in brain tissues indicated that the number of TNF-α–positive cells were increased in the I/R group compared with that in the Sham group, but this was reduced by ephedrine treatment (Figure 2(d) and (e)). Above all, our results suggested that ephedrine suppressed post-ischemic neuroinflammation following MCAO in vivo. Ephedrine suppresses post-ischemic neuroinflammation following MCAO in vivo. (A) TNF-α and IL-1β levels in the brain tissues of each group were tested by ELISA at 24 h post-MCAO. (B–C) Western blot analysis of TNF-α and IL-1β in the brain tissues of each group at 24 h post-MCAO. Relative protein expression normalized to GAPDH was shown (C) D-E, representative images of IHC staining of TNF-α in hippocampus region of brain tissues of each group at 24 h post-MCAO were shown (D). The percentage of TNF-α-positive cells was exhibited (E). Scale bar = 50 μm. *p < 0.05 and **p < 0.01 vs I/R group. Each sample was done in triplicates. MCAO, middle cerebral artery occlusion; EH, ephedrine; IHC, immunohistochemistry; I/R, cerebral ischemia and reperfusion.
Ephedrine suppresses LPS-induced proinflammatory cytokines production and NLRP3 inflammasome activation in BV2 microglial cells
To evaluate the influence of ephedrine on neuroinflammation, BV2 microglial cells were stimulated with LPS and treated with a series of concentrations of ephedrine, and then proinflammatory cytokines production and NLRP3 inflammasome activation were determined via ELISA and Western blot. First, the cytotoxicity of ephedrine on BV2 microglial cells was measured by the CCK-8 assay. The relative viability of BV2 microglial cells treated with 200 μg/mL ephedrine was more than 90% compared with the 0 μg ephedrine group (vehicle control), which indicated that 0–200 μg/mL ephedrine had no obvious cytotoxicity on BV2 microglial cells (Figure 3(a)). Therefore, the concentrations of ephedrine in our study were all below 200 μg/mL to exclude the potential toxicity of ephedrine on BV2 microglial cells. The influence of ephedrine on proinflammatory cytokines (TNF-α and IL-1β) production in LPS-stimulated BV2 microglial cells was tested. We found that ephedrine suppressed TNF-α and IL-1β production in BV2 microglial cells at 15 min, 30 min, and 60 min post–LPS-stimulation, which indicated that ephedrine could inhibit inflammatory response in LPS-stimulated BV2 microglial cells dose-dependently (Figure 3(b) and (c)). We also tested this for a longer time course. As shown in Figure 3(d), ephedrine treatment also reduced TNF-α and IL-1β production in BV2 microglial cells at 24 h post–LPS-stimulation. NLRP3 inflammasome is reported to activate in microglial cells,10, 11, 29 and thus, we evaluated the protein expression of NLRP3, caspase-1, and IL-1β in BV2 microglial cells at 24 h post–LPS-stimulation by Western blot. We found that ephedrine treatment suppressed the expression of NLRP3, caspase-1, and IL-1β in LPS-stimulated BV2 microglial cells, suggesting that ephedrine restrained NLRP3 inflammasome activation (Figure 3(e) and (f)). Collectively, our results indicated that ephedrine suppressed LPS-induced proinflammatory cytokines production and NLRP3 inflammasome activation in BV2 microglial cells. Ephedrine reduces LPS-induced proinflammatory cytokines production and NLRP3 inflammasome activation in BV2 microglial cells. (A) BV2 microglial cells were treated with diverse concentrations of ephedrine for 6 days, and then cell viability was evaluated by the CCK-8 assay. B-C, BV2 microglial cells were treated with 25, 50, or 100 μg/mL ephedrine and stimulated with LPS (100 ng/mL), and then TNF-α (b) and IL-1β (c) levels at 0, 15, 30, and 60 min post-stimulating were evaluated by ELISA. (D), BV2 microglial cells were treated with 25, 50, or 100 μg/mL EH and stimulated with LPS (100 ng/mL), and then TNF-α and IL-1β levels at 24 h post-stimulating were evaluated by ELISA. E-F, BV2 microglial cells were treated with 25, 50 or 100 μg/mL ephedrine and stimulated with LPS (100 ng/mL), and then, cell lysates were collected for Western blot analysis of NLRP3, caspase-1, and IL-1β at 24 h post-stimulating (E). Relative protein expression normalized to GAPDH was shown (F). *p < 0.05 and **p < 0.01 vs LPS group. BV2 microglial cells treated with equal volume of PBS were used as the control group. Each sample was done in triplicates. EH, ephedrine; LPS, lipopolysaccharide; CCK-8, cell counting kit-8; NLRP3, NOD-like receptor pyrin domain 3.
Ephedrine inhibited NLRP3 inflammasome activation in LPS-treated BV2 microglial cells through activating Akt/GSK3β/NRF2 signaling
To elucidate the underlying molecular mechanism of ephedrine in LPS-treated BV2 microglial cells, the downstream signaling pathways were evaluated. In our study, we found that ephedrine treatment increased the phosphorylation of Akt and GSK3β and nuclear NRF2 level in LPS-treated BV2 microglial cells, indicating that Akt/GSK3β/NRF2 signaling might play a role in the effects caused by ephedrine treatment (Figure 4(a) and (b)). The nucleus accumulation of NRF2 was further demonstrated by immunofluorescence staining (Figure 4(c)). We could observe that ephedrine treatment enhanced the nuclear translocation of NRF2 in LPS-treated BV2 microglial cells. As NRF2 is predominantly involved in anti-oxidative defense,
30
we also evaluated the influence of ephedrine treatment on the levels of ROS and antioxidant enzymes MDA and SOD. We found that ephedrine treatment reduced the levels of ROS and MDA and increased the level of SOD in LPS-treated BV2 microglial cells, indicating that ephedrine alleviated oxidative stress (Figure 4(d) to 4(f)). To evaluate the influence of Akt/GSK3β/NRF2 signaling on NLRP3 inflammasome activation, the PI3K/Akt pathway inhibitor LY294002 was used in our study. As shown in Figure 5(a) and (b), the phosphorylation of Akt and GSK3β and nuclear NRF2 level in the LPS + EH group was significantly reduced by LY294002 treatment (LPS + EH + LY294002 group), suggesting that LY294002 inhibited the Akt/GSK3β/Nrf2 signaling in LPS-treated BV2 microglial cells. Moreover, the expression levels of NLRP3, caspase-1, and IL-1β were increased in the LPS +EH + LY294002 group compared with that in the LPS + EH group, indicating that LY294002 treatment abrogated the inhibitory effect of ephedrine on NLRP3 inflammasome activation (Figure 5(a) and (b)). The levels of TNF-α and IL-1β at 24 h post-LPS-stimulation were detected by ELISA. We found that LY294002 treatment attenuated the inhibitory effect of ephedrine on proinflammatory cytokines (TNF-α and IL-1β) production in LPS-stimulated BV2 microglial cells (Figure 5(c)). Taken together, our results indicated that ephedrine inhibited NLRP3 inflammasome activation in LPS-treated BV2 microglial cells through activating Akt/GSK3β/NRF2 signaling. Ephedrine activates Akt/GSK3β/NRF2 signaling in LPS-treated BV2 microglial cells. A–B, BV2 microglial cells were treated with 25, 50, or 100 μg/mL EH and stimulated with LPS (100 ng/mL), and then, cell lysates were collected for Western blot analysis at 24 h post-stimulating (A). Relative protein expression normalized to GAPDH was shown (B). (C) immunofluorescence staining of NRF2 in LPS (100 ng/mL), and/or 100 μg/mL EH-treated BV2 cells was shown. D–F, BV2 microglial cells were treated with 25, 50, or 100 μg/mL ephedrine and stimulated with LPS (100 ng/mL), and then, the levels of ROS (D), MDA (E), and SOD (F) were measured by ELISA. *p < 0.05 and **p < 0.01 vs. LPS group. BV2 microglial cells treated with equal volume of PBS were used as the control group. Each sample was done in triplicates. EH, ephedrine; LPS, lipopolysaccharide. Inhibiting Akt/GSK3β/NRF2 signaling by LY294002 partially abolishes the effect caused by ephedrine in LPS-stimulated BV2 microglial cells. A–B, BV2 microglial cells were treated with 100 ng/mL LPS, 100 μg/mL ephedrine, or 20 μM LY294002 as indicated, and then, cell lysates were collected for Western blot at 24 h post-drug administration (A). Relative protein expression normalized to GAPDH was shown (B). (C) TNF-α and IL-1β levels at 24 h post-drug administration were evaluated by ELISA. *p < 0.05 and **p < 0.01 vs LPS + EH group. BV2 microglial cells treated with equal volume of PBS were used as the control group. Each sample was done in triplicates. EH, ephedrine; LPS, lipopolysaccharide.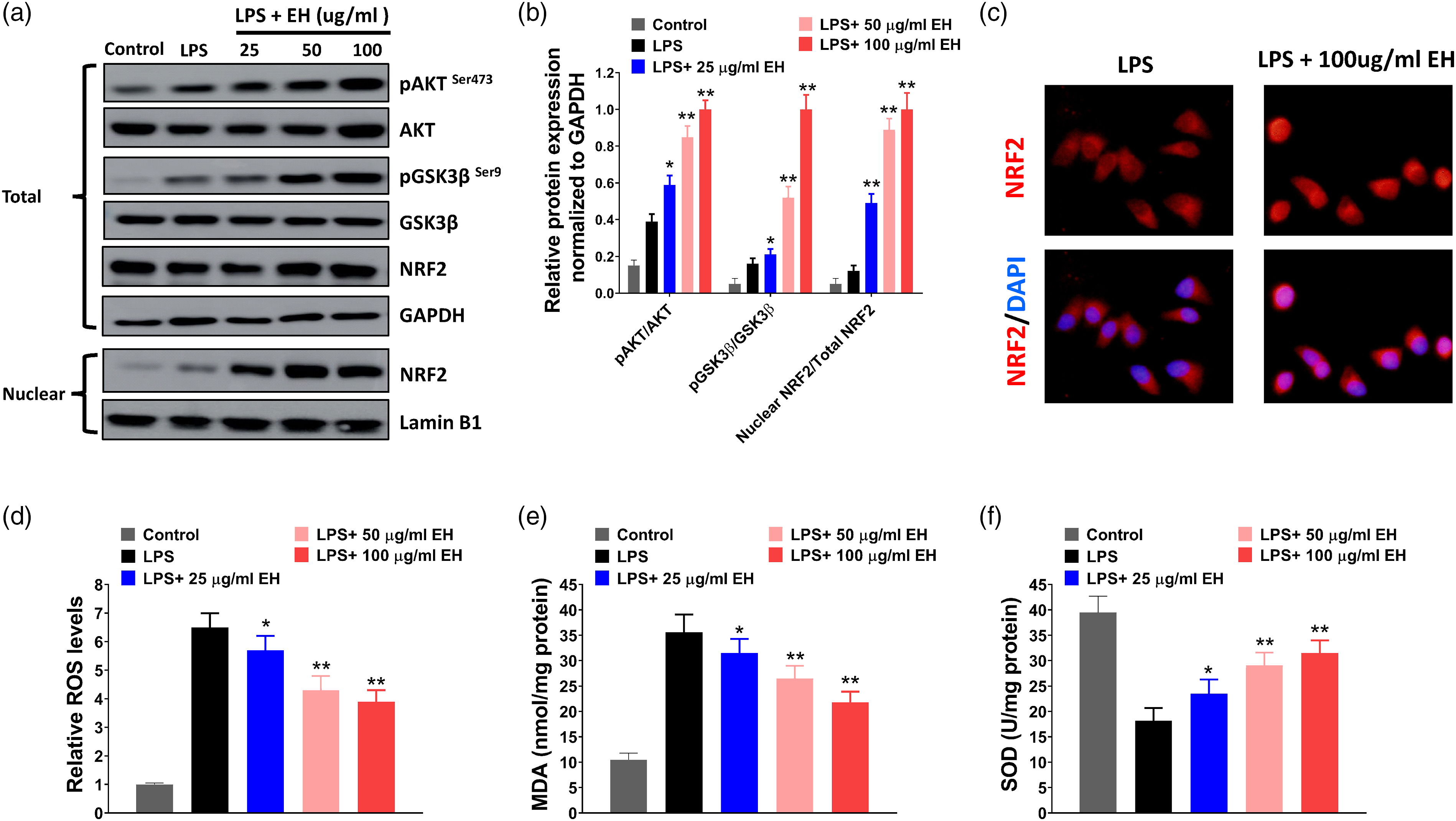

Ephedrine increases Akt phosphorylation and decreases NLRP3 inflammasome activation in ischemic brain following MCAO
The effects of ephedrine on Akt signaling and NLRP3 inflammasome activation in ischemic brain of rats at 24 h post-MCAO were evaluated by IHC staining and Western blot. In IHC staining, the number of pAkt-positive cells was increased by ephedrine treatment in the I/R group (Figure 6(a)). In comparison, the number of NLRP3 positive cells was reduced by ephedrine treatment (Figure 6(a)). The expression levels of pAkt, Akt, NLRP3, caspase-1, and IL-1β in each group were detected by Western blot. We found that the level of pAkt was increased, while the levels of NLRP3, caspase-1, and IL-1β were decreased by ephedrine treatment in the I/R group (Figure 6(b) and (c)). Our results indicated that ephedrine increased Akt phosphorylation and decreased NLRP3 inflammasome activation in the ischemic brain following MCAO in vivo. Ephedrine increases Akt phosphorylation and decreases NLRP3 inflammasome activation in the ischemic brain following MACO. (A), IHC staining of pAkt and NLRP3 in the hippocampus region of brain tissues of each group at 24 h post-MCAO were shown. Scale bar = 50 μm. B–C, Western blot analysis of pAkt, Akt, NLRP3, caspase-1, and IL-1β in each group (B). Relative protein expression normalized to GAPDH was shown (c). *p < 0.05 and **p < 0.01 vs. I/R group. Each sample was done in triplicates. MCAO, middle cerebral artery occlusion; EH, ephedrine; IHC, immunohistochemistry; NLRP3, NOD-like receptor pyrin domain 3; I/R, cerebral ischemia and reperfusion.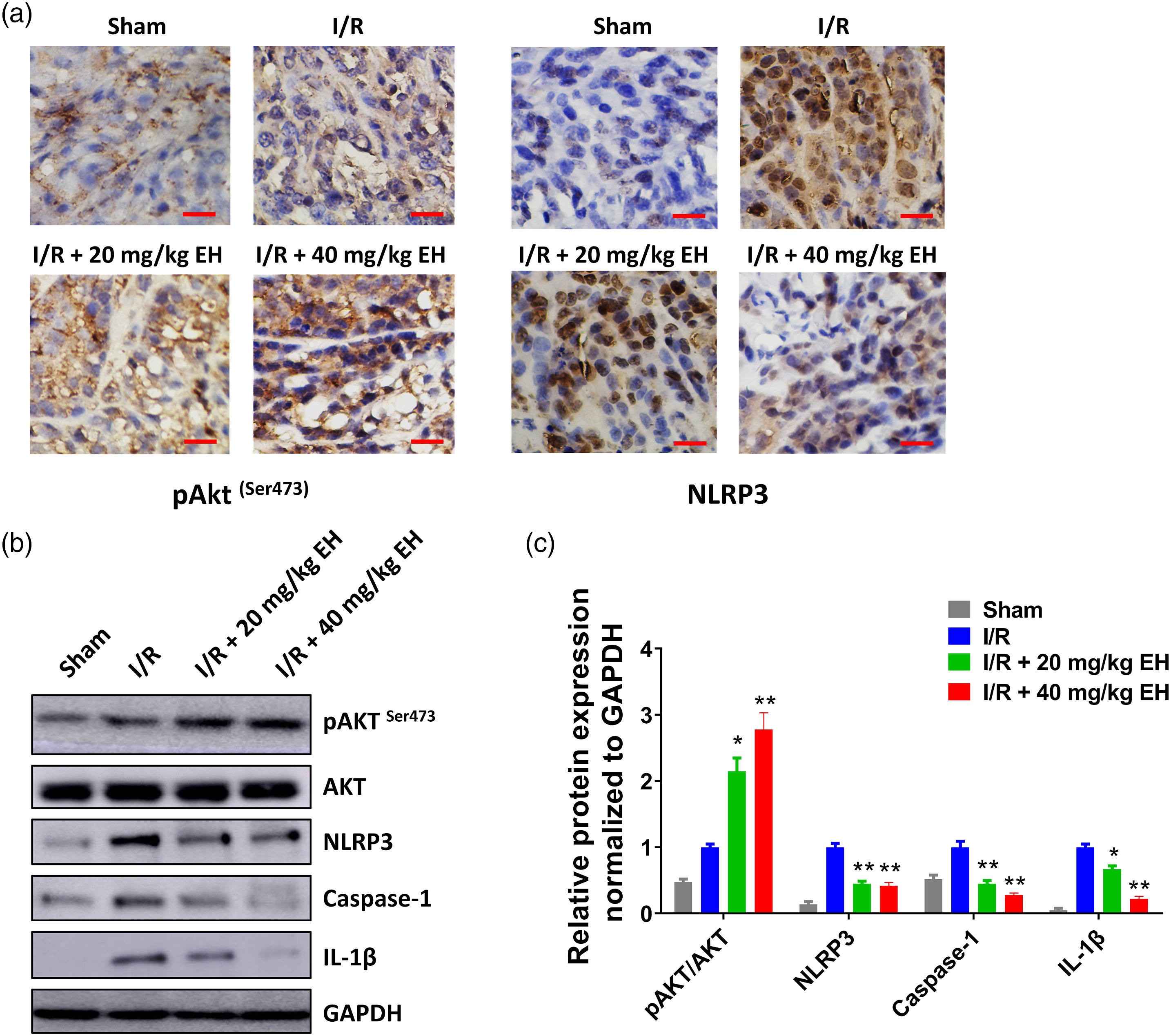
Discussion
Manipulation of blood pressure is important for the treatment of acute brain injury (AIS) such as ischemic stroke. There are increasing evidences that a U-shaped relationship exists between blood pressure and the outcome of acute brain injury. 19 In a retrospective study of 17,398 AIS patients, individuals with blood pressure at 140–179 mmHg have the lowest likelihood of death. 31 Thus, a mild increase in blood pressure may have benefit for ischemic stroke treatment. Indeed, mild induced hypertension is proved to improve blood flow and oxygen metabolism in mice suffered from transient focal cerebral ischemia. 32 Ishikawa et al. 33 prove that phenylephrine decreases the infarct volume in a rat model of complete unilateral carotid artery occlusion. Ephedrine is an FDA approved drug to treat hypotension perioperatively; thus, we reasoned that ephedrine might show some benefits for treating ischemic stroke. Virtually, the influence of ephedrine on cerebral oxygen consumption and blood flow has been evaluated in previous studies. In a randomized controlled trial for brain tumor patients, ephedrine is correlated with elevated cerebral blood flow and regional cerebral oxygen saturation compared with phenylephrine. 34 In a neonatal rat hypoxic-ischemic model, ephedrine increases the neuroprotective effect of hyperbaric oxygen treatment by suppressing caspase-3 and Nogo-A expression. 18 Moreover, Shi et al. find that ephedrine reduces cerebral ischemia/reperfusion injury by suppressing autophagy, apoptosis, and NF-κB activation. 17 Though this report and our study both prove that ephedrine attenuates ischemic brain damage, the underlying molecular mechanism is not the same. In our study, a rat MCAO model was established and the effect of ephedrine was evaluated in vivo and in vitro. We found that ephedrine ameliorated ischemia-induced brain damage, cerebral edema, and neurological dysfunction following MCAO in rats. Moreover, ephedrine suppressed post-ischemic neuroinflammation in ischemic brain and LPS-induced proinflammatory cytokines production and NLRP3 inflammasome activation in BV2 microglial cells. The underlying molecular mechanism of our study mainly focused on the impact of ephedrine on NLRP3 inflammasome activation in MCAO rats and BV2 microglial cells. Our study provided a novel insight into the protective role of ephedrine in cerebral ischemia injury. We speculated that ephedrine might influence not only one pathway, and this might explain the difference of our study and the previous report of Shi et al. 17 Potential limitations for our study include that we used LPS-stimulated BV2 microglial cells as an in vitro cell model to study the influence of ephedrine on neuroinflammation but not the more commonly used oxygen–glucose deprivation model. The second limitation for our study is that abused ephedrine use has been linked to increase risk of ischemic and hemorrhagic stroke, 35 and thus, the dosage and time window for ephedrine use should be carefully monitored; despite that, transient use of ephedrine alleviates cerebral ischemia injury in our study. Our data indicated that a single dose of ephedrine shortly after MACO ameliorated ischemia-induced brain damage, cerebral edema, and neurological dysfunction in rats, which might be safe and efficient.
NLRP3 inflammasome is considered as an important factor that promotes the pathological progression of ischemic stroke. 36 The expression of NLRP3 inflammasome proteins such as NLRP3, caspase-1, and IL-1β are dramatically increased in the brain tissues of stroke patients and I/R animals.37,38 Thus, NLRP3 inflammasome serves as a promising target for ischemic stroke treatment. Accumulated studies indicate that inhibiting of NLRP3 inflammasome activation attenuates the inflammatory injury in ischemia and the reperfusion process. For instance, Yang et al. 39 find that NLRP3 deficiency reduces cerebral injury and neurovascular damage in MCAO rats. Peng et al. 40 report that idebenone suppresses cerebral inflammatory injury in I/R rats by inhibiting NLRP3 inflammasome activation. Han et al. 41 find that vinpocetine reduces the infarct size and improves behavior recover of MCAO rats by decreasing NLRP3 inflammasome expression. In our study, we found that ephedrine suppressed NLRP3 inflammasome activation in the ischemic brain of MCAO rats. This might explain the effect of ephedrine treatment on reducing proinflammatory cytokines production in ischemic brain tissues. NLRP3 inflammasome is also critical for the inflammatory response of microglial cells. For example, in Parkinson’s disease, NLRP3 inflammasome is activated by stimuli from impaired mitochondria in LPS-primed microglial cells. 42 Feng et al. 43 report that NLRP3 inflammasome activated by glucocorticoid in microglial cells is involved in the stress-mediated depressive-like behaviors in rat model. In our study, we found that NLRP3 inflammasome was activated in LPS-primed microglial cells, with increasing levels of NLRP3, caspase-1, and IL-1β. Ephedrine treatment suppressed the activation of NLRP3 inflammasome in LPS-primed microglial cells through Akt/GSK3β/NRF2 signaling. These results indicated that ephedrine might reduce cerebral inflammatory injury by inhibiting NLRP3 inflammasome activation in microglial cells of the brain.
It is reported that the Akt/GSK3β signaling is involved in the regulation of NRF2 activation,44,45 and GSK3β is a typical downstream target of Akt. 46 In our study, we found that ephedrine activated Akt/GSK3β/NRF2 signaling in LPS-treated BV2 microglial cells, and inhibiting of Akt/GSK3β/NRF2 signaling by LY294002 partially abolished the inhibitory effect caused by ephedrine treatment. Thus, we speculated that ephedrine might increase Akt phosphorylation, thus inactivating GSK3β by phosphorylating at Ser9 and subsequently activated NRF2 in BV2 microglial cells and promoted NLRP3 inflammasome activation. Indeed, we found that ephedrine treatment increased the phosphorylation of Akt in the ischemic brain of MCAO rats. There are previous studies reporting that ephedrine regulates inflammatory response through Akt signaling. For example, Zheng et al. 47 find that ephedrine restrains inflammatory response of PGN-stimulated primary peritoneal macrophages and Raw264.7 cells via activating the PI3K/Akt/GSK3β pathway. In Staphylococcus aureus–induced peritonitis, ephedrine treatment increases IL-10 expression and reduces TNF-α expression in dendritic cells through regulating PI3K/Akt and p38 MAPK signaling pathways. 48 Moreover, NLRP3 inflammasome is also reported to be activated by Akt/GSK3β/NRF2 signaling. In hypoxic-ischemic encephalopathy, N-acetylserotonin derivative is proved to reduce infarct volume, cerebral edema, and neuronal apoptosis by activating PI3K/Akt/NRF2 signaling and inhibiting NLRP3 inflammasome activation. 49 Zhai et al. 50 find that Notoginsenoside R1 exhibits neuroprotective effects in diabetic encephalopathy by suppressing NLRP3 inflammasome activation through the Akt/NRF2 axis. In our study, we also found that ephedrine inhibited NLRP3 inflammasome activation in LPS-treated BV2 microglial cells and MCAO rats through activating Akt/GSK3β/NRF2 signaling, corresponding with previous studies.
Conclusion
In summary, we evaluated the effects of ephedrine treatment in MCAO rats and LPS-treated BV2 microglial cells. We found that ephedrine ameliorated ischemia-induced brain damage, cerebral edema, neurological dysfunction, and post-ischemic neuroinflammation in MCAO rats in vivo. Ephedrine also suppressed LPS-induced proinflammatory cytokines production and NLRP3 inflammasome activation in BV2 microglial cells in vitro. In addition, ephedrine inhibited NLRP3 inflammasome activation through activating Akt/GSK3β/NRF2 signaling. Our results provided a novel insight into the effect of ephedrine in cerebral ischemia injury and revealed a potential role of ephedrine in ischemic stroke treatment.
Footnotes
Authors contributions
Jing Wu guaranteed the integrity of the entire study. The experiments were conducted by Qunxian Li, Jing Wu, and Lixian Huang. Data were analyzed by Bo Zhao and Qingbin Li. The manuscript was prepared and reviewed by Jing Wu. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the third batch of “tongzhou district high-level talent development support plan” leading talent project (No. YHLD2019019).
Ethical approval
The protocol for animal studies were reviewed and approved by the Ethics Committee of Animal Experimentation of the Beijing University of Chinese Medicine.
