Abstract
Methods
Urine samples were collected from 157 women with fetal malformations (case group) and 147 women with normal fetuses (control group). High-performance liquid chromatography-mass spectrometry (HPLC-MS) was used to detect the content of eight metabolites of phthalate compounds in urine, including monoethyl phthalate (MEP), mononbutyl phthalate (MBP), monoisobutyl phthalate (MiBP), mono-(2-ethylhexyl) phthalate (MEHP), mono-(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP), mono-(2-ethyl-5-carboxypentyl) phthalate (MECPP), mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP), and mono-benzyl phthalate (MBzP). Demographic data were collected from questionnaires administered in specimen collection.
Results
The exposure level of MEOHP and MEHP in the case group was higher than the others. And there were significant differences between structural malformations and chromosomal malformations in the levels of MEHHP and MEOHP. Pregnant women with low income, high body mass index (BMI), frequent plastic contact, and low nutrients intake were at risk of suffering from fetal malformation.
Conclusion
This study provides evidence for the correlation between the concentration of phthalates and fetal malformation. In addition, decreasing plastic exposure and supplementing nutrients may reduce the incidence of fetal malformations.
Introduction
Phthalate esters (also known as phthalates, phthalic acid esters, or PAEs) are synthetic compounds derived from esterification of phthalic acid. As a plasticizer, it has been widely used in plastic products, such as toys, packaging materials, decoration materials, personal care supplies, medical products, and so on. 1 It has been reported that PAEs are detected in air, dust, water sources, soil, plants, and even food, but their contents are extremely low.2,3 They exist and accumulate for a long time in the environment because they are difficult to be degraded. 4 PAEs have been detected in biological matrices from pregnant women and newborns including human blood, urine, saliva, amniotic fluid, and breast milk.5–7 Since phthalates are widely distributed in the environment, we are exposed to them frequently in our daily lives. The main ways of PAEs exposure in the general population are the respiratory tract (such as production workshop and atmospheric pollutants), digestive tract (such as food and drinking water), and skin (such as cosmetics and medical products). The most important route of phthalates exposure is through contaminating food. 8
Since the 1970s, the toxicity of PAEs has been noticed and studied. Extensive studies have found that these compounds are detrimental to organisms, and all systems may be involved. They may cause hormone secretion disorders, liver damage, cardiotoxicity, and it may result in respiratory diseases, including infections, allergies, and asthma, which are often mentioned.9,10 At the same time, it has been noted that it can pass through the placental barrier, so the exposure to phthalates in perinatal period may be related to fetal malformation. All kinds of deformities are visible. The most common discussed deformities are the reproductive system malformations, neural tube malformations, oral clefts, limb deficiency defects, and congenital heart defects.11–13 Besides, it has been proved that the abnormality is positively correlated with the concentration of these compounds in some animal studies. 12 Recent research indicated that prenatal phthalate exposure may also be associated with long-term effects on children’s development.14,15
However, there are few studies on the concentrations of phthalates exposure in pregnant women, especially those with fetal malformations. Therefore, urinary concentrations of phthalates metabolites were detected, and single factor and multiple regression analysis were performed in order to show the phthalate exposure level of different fetal malformation and try to find the risk factors.
Materials and Methods
Study populations
The cluster sampling method was used to select pregnant women who delivered in Maternal and Child Health Hospital of Hubei Province from March 1 to July 25, 2019, as the research objects. A total of 304 pregnant women were selected to participate in this study (Figure 1). And these cases were divided into two groups based on the presence or absence of fetal abnormalities. The case group (157 cases) consisted of pregnant women with fetal malformation confirmed by ultrasound or amniotic fluid test, and the control group (147 cases) was pregnant women with normal fetal ultrasound, neonatal physical examination, and chromosome examination. All fetuses were delivered naturally. Cases with other complications were excluded, such as cholestasis, hypertension, and diabetes. Structural malformations included hydrocephalus (18 cases), anencephaly (1 cases), meningocele (3 cases), tetralogy of Fallot (11 cases), endocardial cushion defect (4 cases), single ventricle (1 cases), double outlet of right ventricle (2 cases), transposition of great arteries (2 cases), pulmonary artery stenosis (2 cases), atrial and ventricular septum defects (2 cases), lip/cleft palate (12cases), diaphragmatic hernia (6 cases), pulmonary cystadenoma (5 cases), pulmonary sequestration (3 cases), bilateral renal dysplasia (2 cases), polycystic kidney (1cases), inborn micromelia (7cases), osteogenesis imperfecta (2cases), and multiple malformations (21 cases). Chromosome abnormalities include trisomy 21 syndrome (37 cases), trisomy 18 syndrome (8 cases), and deletion or duplication of chromosome fragments (7 cases). The ethics committees of Maternal and Child Health Hospital of Hubei Province have approved this research protocol. Flowchart of population included in our final analysis.
Phthalates measurements
Urine samples were collected and separated into phthalate-free tubes (about 5 mL), and they were frozen at −80°C before analysis. Eight metabolites of phthalates in the urine were measured, including monoethyl phthalate (MEP), mononbutyl phthalate (MBP), monoisobutyl phthalate (MiBP), mono-(2-ethylhexyl) phthalate (MEHP), mono-(2-ethyl-5-hydroxyhexyl) phthalate, (MEHHP), mono-(2-ethyl-5-carboxypentyl) phthalate (MECPP), mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP), and monobenzyl phthalate (MBzP), by high-performance liquid chromatography-mass spectrometry (HPLC-MS). The procedures for pretreatment and testing refer to Du et al. 16 with minor changes. In short, after adding 80 μL of 13C-labeled standards(400 ng mL−1, Cumbridge Isotope Laboratory, USA) and 10 μL βglucosidase (Sigma, USA), urine samples were incubated for 90 min at 37°C. Afterwards, phthalate metabolites were extracted using solid-phase extraction (Oasis HLB, Waters Co., USA). The collected eluent was dried in a 55°C nitrogen blower, and then analyzed with HPLC-MS (6400LC-MS, Agilent Technologies Co., USA). Each batch of samples was tested along with a blank and two quality controls. 1-mL water was used as the blank control, and one urine sample contained 5 μg/L of the target metabolites was used as quality control. Likewise, another control condition was urine sample contained 50 μg/L of the target metabolites. The limits of detection (LOD) of eight metabolites ranged from 0.01 to 0.05 μg/L.
Covariates
All the volunteers completed a survey under the guidance of two investigators who had passed the strict training. A face-to-face questionnaire survey was conducted. Information was collected including the following contents: (1) social demographic characteristics: age, gender, weight, marital status, education level, family income, occupation status, etc. (2) Medical history: disease history and medication history. The nutrients supplement included folic acid supplement alone and compound nutrients supplement (calcium supplement, multivitamin supplement, and iron supplement at the same time). (3) Living habits: smoking (smoking at least 1 cigarette per day on average), alcohol abuse (drink more than 100 mL a day), plastic contact frequency, etc. The information of all qualified questionnaires was checked and input by two people using SPSS software.
Statistical methods
The demographic characteristics and clinical data were analyzed by descriptive statistics. The measured concentrations were adjusted by the specific gravity (SG) to eliminate the effects of dilution. Correction formula:
To adjust the right-skewed distribution, the concentrations were based-10 logarithmic transformed before bringing them into statistical analysis. T-test was used to compare urinary concentrations of phthalates metabolites (μg/L) between two groups. And the chi-square test was used to compare interfering factors between two groups. Single factor and multiple regression analysis were utilized to analyze the correlation between covariates and fetal malformation. The data were analyzed by SPSS for Windows, version 23.0 (IBM Corp., NY, USA). It is considered statistically significant when p < 0.05. If OR > 1, it was considered as a risk factor, and if OR < 1, it was considered as a protective factor.
Results
General characteristics
157 pregnant women with fetal malformations were enrolled as the case group and 147 pregnant women with normal fetuses were enrolled as the control group in the study. The average age of the subjects was 29 years old (29.71 vs 29.52). They all denied smoking, alcohol abuse, and toxic exposure history. However, a small number of pregnant women (7/304) are exposed to second-hand smoke (non-smokers who have inhaled smoke exhaled by smokers for at least 15 min a day). All pregnant women also denied a family history. 127 out of 304 people were first pregnancies and 187 out of 304 were first births. The time of termination of pregnancy varies greatly (27.75 weeks vs 38.20 weeks) because most mothers decided to give up the fetus after the discovery of malformation. Most of the pregnant women are Wuhan natives (84.08% vs 89.02%), and others are transferred from the surrounding areas of Wuhan (all in Hubei Province).
Urinary concentrations of phthalate metabolites
Urinary concentrations of phthalates metabolites (μg/L) for all urine samples.

1: case group 2: control group.
LOD: below the limit of detection; MEP: monoethyl phthalate; MBP: monobutyl phthalate; MBzP: mono-benzyl phthalate; MEHP: Mono-2-ethylhexyl phthalate; MEHHP: mono (2-ethyl-5-hydroxyhexyl) phthalate; MECCP: mono-(2-ethyl-5-carboxypentyl) phthalate; MEOHP: mono(2-ethyl-5-oxohexyl) phthalate; MiBP: monoisobutyl phthalate.
Compare the two groups of the concentrations of phthalates metabolites. To adjust the right-skewed distribution, the concentrations were based-10 logarithmic transformed.
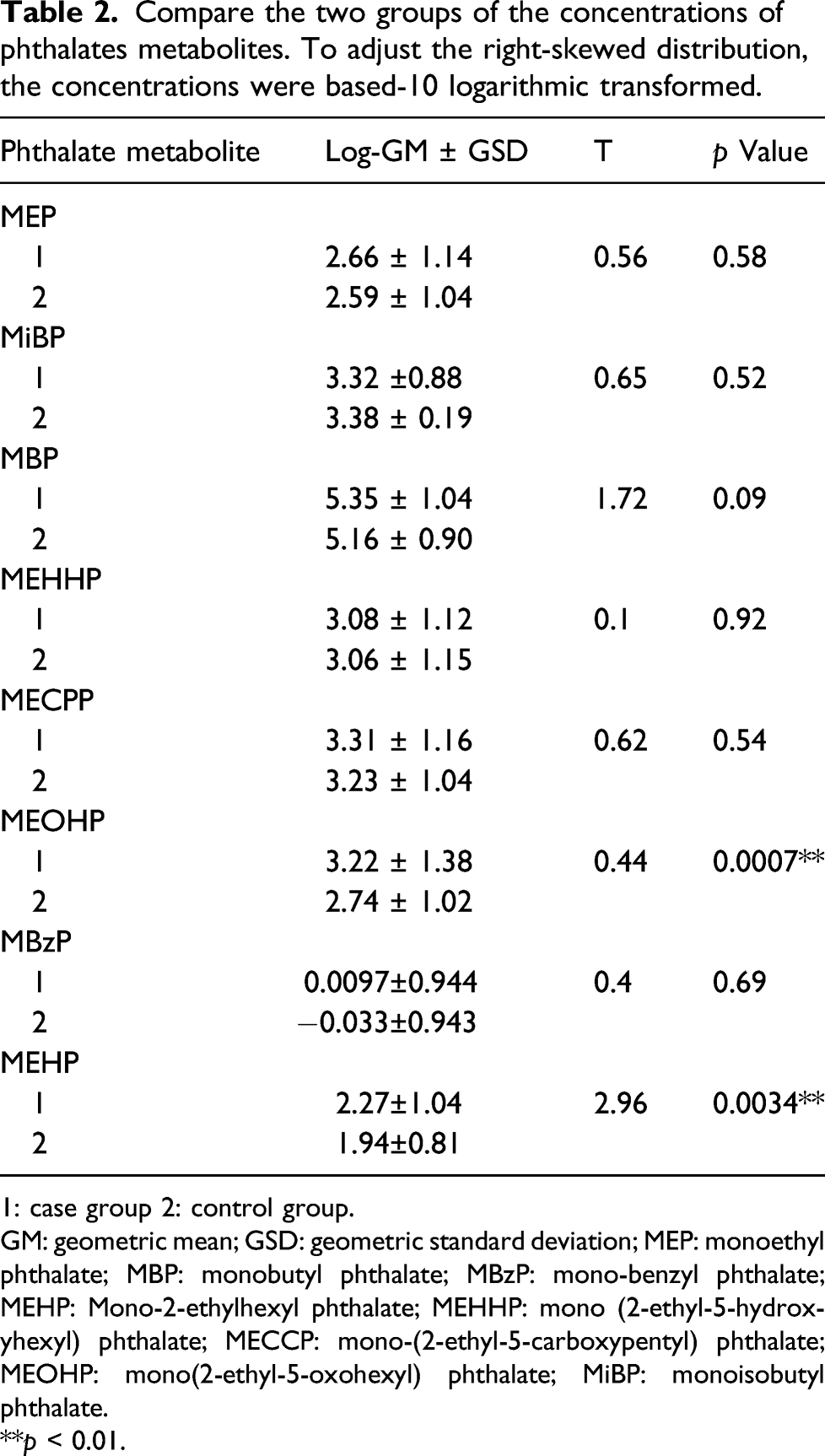
1: case group 2: control group.
GM: geometric mean; GSD: geometric standard deviation; MEP: monoethyl phthalate; MBP: monobutyl phthalate; MBzP: mono-benzyl phthalate; MEHP: Mono-2-ethylhexyl phthalate; MEHHP: mono (2-ethyl-5-hydroxyhexyl) phthalate; MECCP: mono-(2-ethyl-5-carboxypentyl) phthalate; MEOHP: mono(2-ethyl-5-oxohexyl) phthalate; MiBP: monoisobutyl phthalate.
p < 0.01.
Urinary concentrations of phthalates metabolites (μg/L) grouped by malformation type. To adjust the right-skewed distribution, the concentrations were based-10 logarithmic transformed.
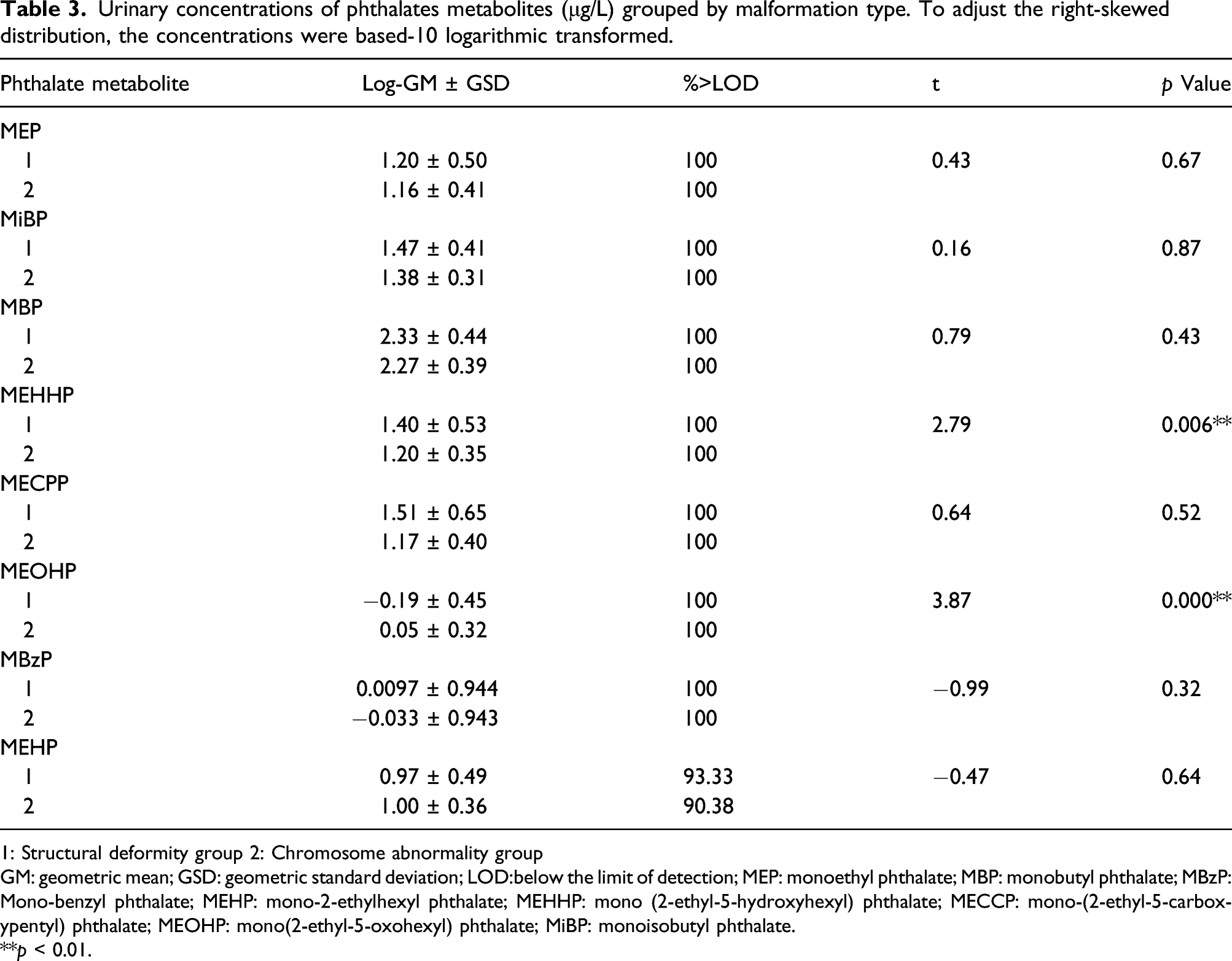
1: Structural deformity group 2: Chromosome abnormality group
GM: geometric mean; GSD: geometric standard deviation; LOD:below the limit of detection; MEP: monoethyl phthalate; MBP: monobutyl phthalate; MBzP: Mono-benzyl phthalate; MEHP: mono-2-ethylhexyl phthalate; MEHHP: mono (2-ethyl-5-hydroxyhexyl) phthalate; MECCP: mono-(2-ethyl-5-carboxypentyl) phthalate; MEOHP: mono(2-ethyl-5-oxohexyl) phthalate; MiBP: monoisobutyl phthalate.
p < 0.01.
Associations between fetus malformation and socioeconomic factors
Analysis of influencing factors, T-test applied to mean comparison, chi-square test was used to compare the rates.

BMI: body mass index.
Compound nutrients supplement ncluding iron, calcium, and multivitamin supplements.
Plastic contact: Refers to daily contact with plastic containers and tableware.
Less contact: 0–3 times a week.
Frequent contact: more than 3 times a week.
p < 0.05
p < 0.01.
Multivariate analysis of the association between mothers’ risk factors and the occurrence of fetus malformations.

Discussion
In this study of 304 pregnant women, seven of eight targeted compounds were detected in urine samples of all pregnant women (100%), which further confirmed the widespread human exposure of PAEs in China. The detection rate is similar to that reported in the United States (98–100% of pregnant women). 17 Exposure of phthalates to pregnant women is a particular concern because previous studies have extensively demonstrated an association between phthalate exposures and malformations in the offspring. Our results show that MBP was the highest level, which is similar to that of Li’s report. 18 But the median value of our results (median level 196.89 μg/L) is much higher than that of his (median level 41.8 μg/L). Compared with other countries, many literatures showed that MEP was the predominant pollutant.17,19 It may due to the region, detection methods of urine, and sample size.
Results showed that the content of MEP, MiBP, MBP, MEHHP, MEOHP, and MEEHP in the deformity group were higher than that of the control group, of which MEOHP and MEHP were statistically significant. And multivariate analysis also substantiated that MEOHP and MEHP increased the risk of fetal malformation. According to previous reports, various animal experiments have confirmed that perinatal PAEs exposure can affect the development of the multiple organ system of offspring, may inhibit embryonic development, and may lead to fetal malformations, such as forebrain malformations, polydactyly, and ocular deformity.20–23 Its metabolites, including DEHP, MEHP, and DBP, have been reported to be teratogenic.12,23,24 Tang ’s study is the first one to prove that maternal exposure to DEHP may be an important factor leading to fetal cardiac malformations in mice. 25 Furthermore, Moore RW reported that DEHP exposure during pregnancy can lead to malformations such as eye, brain, vascular, and bone abnormalities in fetal rats. 23 A review evaluated data from 2003 through 2014, common congenital anomalies including hypospadias, cryptorchidism, anogenital distance, congenital heart defects, and oral clefts were well represented in the literature whereas other outcomes such as neural tube defects, limb deficiency defects, and gastroschisis were rarely described. 13 Our results are consistent with these researches, for MEHP and MEOHP are metabolites of DEHP. This study proved that pregnant women with fetal malformations do have high levels of phthalates exposure, although not all metabolites. The difference may be caused by different metabolic pathways.
After classification by deformity, we also found that the content of MEHHP and MEOHP in the structural deformity group were higher than that of the chromosome abnormality group. In the scope of our knowledge, our study was the first one to find this difference. The mechanism may be complex. Some studies reveal that environmental disturbances during prenatal period exert damages through estrogenic effect, oxidative stress damage, cell cycle arrest, apoptosis induction, and epigenetic modification at the gene level.26–28 Epigenetic modification, such as DNA methylation, has been hypothesized to be an important mechanism that mediates certain biological processes and pathogenic effects of in utero phthalate exposure. 29 It is unclear why only these two metabolites were higher, while others were not, which needs further study.
Data from this study suggest that the malformation is related to lower income. Income may affect lifestyle, resulting in frequent plastic exposure and increased levels of PAEs in the body. Many investigators have explored predictors of phthalate exposure, including income and consumer practices. Abby G. provides evidence that elevated phthalate concentrations were associated with several sociodemographic characteristics, including less educated, lower income, and higher body mass index. 17 Nonetheless, some scholars have different results that concentrations of phthalates were associated with increasing household income. They speculated that high income may lead to modern life, for example, contacting modern decoration materials, and these materials have proven to be related to high PAEs. 30 In conclusion, most studies support that income may affect the concentrations of phthalates, which may be associated with malformation.
Meanwhile, we found that women in the case group had a higher BMI. It may be because the dietary intake of environmental phthalates is higher, and high level of phthalates is a cause of fetal malformations. Previous studies through various statistical methods have found associations between obesity and many environmental chemicals such as MEHHP, MEOHP, MEHP, MBZP, and MEP in adults. 31 Moreover, prior studies have indicated that phthalate exposures may play an important role in the development of obesity. 32 So these two influence each other, leading to a vicious circle. Thereby, both high BMI and high PAEs levels increased the incidence of deformities.
Contaminating food is a major source of exposure to many deleterious environmental elements. Ample evidence indicates that plastic constituents can migrate into foodstuffs. Consumption of plastic packaged food has been related to higher urinary levels of phthalate metabolites. 33 Our results complied with this view. It is shown daily contact with plastic containers and tableware increases risk of deformity, and OR is 2.24. As Sterrett, M.E showed using plastics for food storage, microwaving in plastic containers, and using hard plastic water bottles was associated with urinary phthalate concentrations, especially DEHP metabolites. 33 And another article has demonstrated that food with limited packaging can substantially reduce phthalate exposures. 34 So, in the future, reducing the deformity rate may be achieved by reducing plastic contact.
Inadvertently, it was found that the supplement of compound nutrients in case group was less, and it was a protective factor of fetal malformation. In general, previous data indicated that micronutrients may affect the process of pregnancy, including fertility, embryogenesis, and placentation. And it is beneficial that prophylactic use of micronutrients in prevention of certain adverse pregnancyoutcomes. 35 Folic acid has been proved to be able to prevent neural tube defects. 36 One animal experiment suggested that folic acid supplementation before and after pregnancy can reduce the toxic damage of pregnant mice and embryos caused by DEHP. 37 Foods and dietary patterns associated with healthier food choices (e.g., folic acid supplements) were generally associated with lower urinary phthalate metabolite concentrations. 38 The possible reason is that folic acid deficiency can affect cell biological function like differentiation, proliferation, and function. But unfortunately, there was no statistical difference in folic acid use alone in this study. A review proposed there is currently no evidence that, compared with iron and folic acid supplementation, routine maternal antenatal multiple micronutrient supplementation improves short-term and long-term outcomes of newborns. 39 In conclusion, this study supported that proper nutrient supplement may be more beneficial.
Our research has some limitations. First, urine samples were collected only at one time point, which could not reflect the trend of exposure. Second, the data were from a cross-sectional survey. The measurement of metabolites could only reflect exposures at that time. And lastly, other potentially important factors may not have been taken into consideration, such as race. Because the majority of the residents are Han nationality. Future work will incorporate more information and additional urine sampling.
Conclusions
As a type of plasticizer, PAEs have become an important component of endocrine disruptors distributed in the global environment. This study has confirmed that a high level of phthalates (MEOHP and MEHP) may be related to fetal malformations. And different kinds of metabolites exposure may be related to different types of deformities. But the specific reasons need to be further studied. This study demonstrated the importance of decreasing plastic exposure to reduce fetal malformations. In addition, increasing the intake of multiple nutrients may provide a feasible solution for clinical prevention of fetal malformation in the future.
Footnotes
Acknowledgments
The authors are grateful to the patient who gave his informed consent for publication. Special thanks to Dr Yufeng Li for the contribution to the detection of PAEs.
Author' contributions
All authors read and approved the final manuscript. Conceptualization: Ling Wang, Ying Gao, Yun Zhao; methodology: Ling Wang; software: Ling Wang and Wei Duan; validation: Ling Wang, Wei Duan, and Ying Gao; investigation: Ling Wang, Ying Lin, and Wei Duan; resources: Ling Wang, Ying Lin, and Guoqiang Sun; data curation: Ying Gao and Guoqiang Sun; writing–original draft preparation: Ling Wang; writing–review and editing: Ling Wang, Wei Duan, and Ying Gao; supervision: Yun Zhao and Guoqiang Sun; project administration; Ling Wang and Ying Gao.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Foundation of Hubei Province Health and Family Planning Scientific Research Project, China (NO:WJ2018H0154).
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the ethics committees of Maternal and Child Health Hospital of Hubei Province.
Informed consent
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patients to publish this paper.
