Abstract
Objective
How to reduce the neurodevelopmental toxicity of decabromodiphenyl ether
Methods
Pregnant Sprague–Dawley rats were treated with PBDE-209 and IGF-1, and the offspring were subjected to the Morris Water Maze test. Hippocampal neurons were cultured with PBDE-209 and IGF-1 or the PI3K inhibitor or MEK inhibitor for cell viability, apoptosis, immunofluorescence, and Western blot assays.
Results
Prenatal PBDE-209 exposure impaired the learning and memory ability of rats by delaying the mean latency to the platform compared, whereas prenatal treatment with IGF-1 treatment improved the learning and memory ability. In vitro, treatment of primary cultured hippocampal neural stem cells (H-NSCs) with PBDE-209 reduced cell proliferation and differentiation, but induced apoptosis. In contrast, IGF-1 treatment antagonized the cytotoxic effects of PBDE-209 in H-NSCs in vitro. At the gene level, IGF-1 inhibition of PBDE-209–induced cell cytotoxicity was through the activation of the PI3K/AKT and MEK/ERK signaling pathways in vitro because the effect of IGF-1 was blocked by the AKT inhibitor LY294002 and the ERK1/2 inhibitor PD98059.
Conclusion
Prenatal PBDE-209 exposure impaired the learning and memory ability of rats, whereas IGF-1 treatment was able to inhibit the neurodevelopmental toxicity of PBDE-209 by activation of the PI3K/AKT and ERK1/2 cell pathways.
Introduction
Decabromodiphenyl ether (PBDE-209), which belongs to a class of organobromine compounds, is frequently used as a flame retardant in building materials, electronics, furnishings, motor vehicles, airplanes, plastics, polyurethane foams, and textiles1,2 due to its excellent flame-retardant properties and low cost, especially in developing countries. 3 However, since the 1990s, numerous health and environmental concerns have been raised regarding PBDE-209, as it is widely distributed in the environment and poses a human health hazard.4–10 Exposure to PBDE-209 has been associated with developmental neurotoxicity, endocrine dysfunctions, reproductive disorders, and hepatotoxicity. 11 Evidence from epidemiological and animal studies has also indicated that PBDE-209 exposure impairs brain development and induces behavioral alterations; for example, PBDE-209 exposure impaired athletic and learning abilities in mouse pups, 12 as well as learning and memory abilities in rats, and had neurotoxic effects on hippocampal neurons. 13 Fortunately, governments around the world began banning the use of PBDE-209 in 2007. The US Washington state legislature passed a bill to ban PBDE-209 usage [https://www.seattlepi.com/local/article/Chemical-ban-puts-industry-on-the-defensive-1234423.php], and the US State of Maine also passed a bill to phase out PBDE-209 use [https://www.nrcm.org/news/]. However, a superior flame retardant capable of replacing PBDE-209 is still lacking [ref]; thus, further study of PBDE-209–induced neurotoxicity and human health risks as well as strategies to mitigate its toxicity can help improve human health.
Insulin-like growth factor-1 (IGF-1), a neurotrophic factor, plays an important role in neurodevelopment and in regulating the transformation of pluripotent stem cells and progenitor cells into mature neurons. 14 Thus, it plays an important role in childhood growth and has anabolic effects in adults. 15 Indeed, IGF-1 receptors are distributed throughout the brain, especially in the hippocampus. 14 Previous studies have reported that IGF-I might possess therapeutic potential for the control of many central nervous system diseases and may protect hippocampal neurons from certain types of cytotoxicity because it can alleviate amyloidosis-induced hippocampal neuronal toxicity 16 and doxorubicin-induced neuronal stem cell cytotoxicity. 17 Given that BDE-209 is widely used, the potential neurodevelopmental toxicity cannot be ignored, and there is also no other alternative to replace it, so it is necessary to study how to reduce the toxicity of BDE-209.
Insulin-like growth factor-1, a neurotrophic factor, plays an important role in neurodevelopment and in regulating the transformation of pluripotent stem cells and progenitor cells into mature neurons. 14 Thus, it plays an important role in childhood growth and has anabolic effects in adults. 15 Indeed, IGF-1 receptors are distributed throughout the brain, especially in the hippocampus. 14 Previous studies have reported that IGF-I might possess therapeutic potential for the control of many central nervous system diseases and may protect hippocampal neurons from certain types of cytotoxicity because it can alleviate amyloidosis-induced hippocampal neuronal toxicity 16 and doxorubicin-induced neuronal stem cell cytotoxicity. 17
In this study, we first established a rat model of prenatal PBDE-209 exposure to assess the learning and memory abilities of the offspring using the Morris Water Maze (MWM) in vivo, and then we investigated the neurotoxic effects of PBDE-209, and the protective effects and mechanism of IGF-1 on primary cultured hippocampal neurons in vitro. We expected to uncover novel information regarding the protective effects of IGF-1 on PBDE-209–induced neurotoxicity and potential uses for IGF-1 to improve learning and memory.
Materials and methods
Materials
PBDE-209 for in vivo experiments was purchased from Beijing J&K Chemicals (Beijing, China), while PBDE-209 (>99% purity) for in vitro experiments was obtained from AccuStandard Corp (New Haven, CT, USA). IGF-1, epidermal growth factor (EGF), and basic fibroblast growth factor (bFGF) were acquired from Peprotech (Rocky Hill, NJ, USA). Dulbecco’s modified Eagle’s medium (DMEM)/F12 medium, fetal bovine serum (FBS), B-27 supplement, penicillin-streptomycin stock solution, and Annexin V-fluorescein isothiocyanate (FITC) kits were purchased from Invitrogen (Carlsbad, CA, USA). Poly-D-Lysine, LY294002 [a strong inhibitor of phosphoinositide 3-kinase (PI3K)], PD98059 [an extracellular signaling pathway (ERK) 1/2 inhibitor], dimethyl sulfoxide (DMSO), 4′,6-diamidino-2-phenylindole (DAPI), anti-protein kinase B (Akt) and pERK1/2 antibodies, FITC-conjugated goat anti-mouse immunoglobulin G (IgG), and rhodamine B isothiocyanate (RBITC)-conjugated goat anti-mouse IgG were from Sigma-Aldrich Co. (St Louis, MO, USA). The anti-ERK1/2 and pAkt antibodies were from Cell Signaling Technology (Danvers, MA, USA), the anti-glial fibrillary acidic protein antibody (GFAP clone GA5), and neuronal nuclei (NeuN) antibody (NeuN clone A60) were from obtained from Millipore (Billerica, MA, USA), and the anti-Nestin antibody was obtained from Arigo (Shanghai, China). Rabbit anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody was obtained from Proteintech Group (Hangzhou, China), and horseradish peroxidase (HRP)-conjugated anti-rabbit IgG was obtained from Sungene Biotech (Shanghai, China). In addition, the Cell Counting Kit-8 (CCK8) was obtained from Dojindo Laboratories (Kumamoto, Japan).
Animals and experiments
The animal protocol for this study was approved by the Institutional Animal Care and Use Committee (IACUC) of our university and followed the Guidelines of the Care and Use of Laboratory Animals issued by the Chinese Council on Animal Research. Specifically, 24 female and 11 male Sprague–Dawley rats aged 8 weeks with body weights of 160–180 g were obtained from the Experimental Animal Center of Guangdong Province. These animals were housed at our specific pathogen-free (SPF) animal facility at the Animal Center of our university. The housing conditions in the facility were 21 ± 1oC, 50 ± 5% relative humidity, and 12 h light/dark cycle with free access to food and water. Upon reaching 10–11 weeks of age and 200–230 g body weight, female and male rats were placed in the same cages at a 2:1 ratio overnight at 5:00 p.m. Successful mating was confirmed through vaginal smears at 7:00 a.m. the next day. The mating date was recorded as gestation day 1 (G1). Pregnant Sprague–Dawley rats were randomly divided into four groups (n = 6 per group), with similar initial mean body weights in the control and experimental groups. The groups were as follows: the rats received PBDE-209 (300 mg kg−1/day−1), an equal volume of arachis oil from G1 to G21 via gastric gavage, and/or IGF-1 (20 μg kg−1/day−1) via an intraperitoneal injection according to the group. After 21 days, the offspring were delivered, raised to postnatal day 28 (the 28th day after birth), and subjected to the MWM task test.
Morris Water Maze task test
To assess the learning and memory abilities of the offspring on postnatal day 28, we performed the MWM test as described in a previous study. 18 Briefly, a maze that was 120 cm in diameter and 60 cm in height was divided into four quadrants and a platform (12 cm in diameter and 30 cm in height) was placed in the southwestern quadrant. Each experimental group contained 16 rats, and the movement of the animals around the maze was recorded using a computer-connected camera. External stimuli were kept constant, that is, the water temperature was at 23–26oC and the room remained quiet with a low light during the experiments. The experiments consisted of four consecutive daily sessions lasting for 90 s per session with a 60 s interval. During the experiments, the rats began the maze from a different point every day. If the rats failed to reach the platform within 90 s, they were directed to the platform by an investigator and allowed to remain on the platform for 10 s. The escape latency was defined as the amount of time the rats took to reach the hidden platform, and the maximal escape latency was set to 90 s. The interval between two consecutive tests was no less than 60 s, and the mean escape latency for the 5-day trial was used to measure the learning ability of the rats. Twenty-four hours after the final acquisition trial, the rats were subjected to a probe trial without the platform (90 s duration). For this trial, the rats started at a position opposite the target quadrant where the platform was located during the training session, and their behavior was recorded with a camera. In the probe trial, we assessed the amount of time the rats spent in the target quadrant and the number of times they crossed the previous location of the platform to measure their memory ability. The values for each mouse in each daily session were averaged.
Isolation of hippocampal neurons and primary culture
To isolate neurons from the hippocampus, we first sectioned neurons from normal neonatal pups, immediately dissected the neurons into small pieces, placed the sections in 0.25% trypsin (pH 7.8) solution, and incubated them for 25 min at room temperature. Thereafter, 10 mL of DMEM/F12 supplemented with 10% FBS was added to the tissue to stop digestion, and the mixture was centrifuged in a clinical centrifuge at 1000 rpm for 5 min to remove the supernatant. The resulting tissue debris containing neurons was resuspended in 10 mL of DMEM/F12 with 2% B-27 supplement, 20 ng ml−1 bFGF, 20 ng ml−1 EGF, and 100 unit/ml penicillin-streptomycin stock solution at a density of 5 ×10
5
ml−1 in a 25-cm2 flask and cultured in a humidified incubator with 5% CO2 at 37oC. The culture medium was refreshed every 3–4 days. The purity of the neurons was assessed 7 days later through immunofluorescence analysis of the neuronal nuclei and was found to be as high as 95%. Second passage neurospheres were harvested, resuspended, and cultured for 4 days, and then randomly divided into blank control, vehicle control, PBDE-209, PBDE-209 + IGF-1, PBDE-209 + IGF-1 + LY294002, PBDE-209 + IGF-1 + PD98059, and PBDE-209 + IGF-1 + LY294002 + PD98059 groups. PBDE-209 was dissolved in DMSO, and the PBDE-209 concentration used was 50 μg/ml; IGF-1 was used at 120 ng/mL; and both LY294002 and PD98059 were used at 50 μ
Cell viability assay
The change in cell viability was assessed using the CCK8 assay. In brief, the second passaged neurospheres were harvested, centrifuged in a clinical centrifuge at 1000 rpm for 5 min, and resuspended in a serum-free medium. The neurospheres were reseeded into a 96-well plate (100 μL/well) at a density of 1 × 105 ml−1, cultured for 4 days, and treated according to the conditions for each group (see the details in the previous section) for 24 h, 48 h, and 72 h. At the end of each experiment, the CCK8 reagent from the CCK8 (Dojindo Laboratories, Kumamoto, Japan) was added to the cells according to the manufacturer’s instructions, and the cells were further cultured for an additional 4 h. The optical density was measured using a spectrophotometer (Metash, Shanghai, China) at 490 nm. The experiments were in quintuplicate (n = 5) and repeated at least three times.
Flow cytometric Annexin V-FITC/propidium iodide (PI) apoptosis assay
To assay the neuron apoptosis rate, a flow cytometric Annexin V-FITC/PI apoptosis assay was performed. Briefly, the hippocampal neurons were cultured for 24 h and treated according to the conditions for each group (see the details in the cell culture section above). The second passage neurospheres were harvested and prepared for a single-cell suspension in 600 μL of the binding buffer with a density of 1 × 106 cells/ml. Afterward, 30 μL of Annexin V-FITC was added, followed by 6 μL of PI solution. The mixture of cells and kit reagents was incubated at room temperature in the dark for 15 min, and then subjected to flow cytometry as soon as possible with the fluorescence emission recorded at 530 nm and 575 nm (or equivalent), and excitation at 488 nm. The experiments were in quintuplicate (n = 5) and repeated at least three times.
Immunofluorescence
The second passage neurospheres were trypsinized and centrifuged in a clinical centrifuge at 1000 g for 5 min before being resuspended in DMEM/F12 supplemented with 2% B27, 20 ng ml−1 bFGF, 20 ng ml−1EGF, and 10% FBS. The neurospheres were seeded onto coverslips for each treatment group (see the details in the cell culture section above) in six-well plates for 7 days. Cell morphology was observed daily under an inverted microscope (Leica, Wetzlar, Germany). After 7 days, the neurospheres were immunostained with Nestin (Arigo ARG52345), a NeuN (Millipore SAB4300883) antigen, and the nuclei were counterstained with a DAPI solution. The proportion of NeuN-positive cells was assessed from six randomly selected microscopic fields at × 200 magnification under a fluorescent microscope (Leica) and summarized.
Western blot
Total cellular protein was extracted from the treatment groups in 10 mmol/L Tris homogenization buffer (pH 7.4) with protease inhibitors, and the protein concentration was measured using a Bicinchoninic Acid (BCA) Protein Assay Kit (CWBIO, Beijing, China). Equal amounts (25 μg each) of the protein lysates were separated in 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels and transferred to nitrocellulose membranes (Millipore, Billerica, MA, USA). For Western blotting, the membranes were incubated in 5% nonfat milk for 2 h at room temperature. Afterward, the membranes were incubated with rabbit monoclonal anti-GAPDH (ab8245) antibody at a dilution of 1:2000, rabbit monoclonal anti-ERK1/2 (SAB4301578) antibody at a dilution of 1:1000, rabbit monoclonal anti-pERK1/2 (RAB0349) antibody at a dilution of 1:1000, rabbit monoclonal anti-Akt (SAB4500800) antibody at a dilution of 1:1000, or rabbit monoclonal anti-pAkt (SAB4503853) antibody at a dilution of 1:1000 at 4oC overnight. The next morning, the membranes were washed three times for 10 min each time with Tris-based saline-Tween 20 (TBS-T). They were further incubated with HRP-conjugated goat anti-mouse IgG (SAB12-349) at a dilution of 1:3000 at room temperature for 1 h and the immunoreactive bands were visualized with an enhanced chemiluminescence (ECL) Western blotting detection kit (Beyotime, Shanghai, China) using a Fluor Chem E System (Protein Simple, CA, USA). The images were quantified and normalized to the GAPDH level using the ImageJ 1.39 software (National Institute of Heath, Bethesda, MD, USA).
Statistical analysis
The data were summarized as the mean ± SD (n = 5 and three repeats) and statistically analyzed. Data on the latency to the platform in the MWM test were analyzed with the repeated measure analysis of variance (ANOVA) test, while other data were analyzed using the one-way ANOVA test. All statistical analyses were performed using the SPSS 16.0 software (SPSS, Chicago, IL, USA). p < 0.05 was considered statistically significant and indicated with an asterisk (*), whereas NS referred to non-statistically significant results (p > 0.05).
The flowchart of this research is shown as follows (Supplementary Figure 1)
Results
Effects of prenatal PBDE-209 exposure on rat learning and memory
In this study, we assessed the memory and learning impairments induced by PBDE-209 exposure in rats using the MWM test. The MWM test is used to assess the learning capacity of animals, including rats,
18
based on the latency to a platform (the duration to reach the platform). The probe trial is used to measure an animal’s memory ability by evaluating the number of the times it crosses the previous location of the platform or the amount of time it spends in the target quadrant of the pool. Our data showed that the mean latency to the platform in the PBDE-209–treated group was significantly longer than that of the blank and oil control groups on days 3–5 (p < 0.05 in the repeated-measures ANOVA test; Figure 1(a)). There was no difference between the blank and oil control groups (p > 0.05 in the repeated-measures ANOVA test; Figure 1(a)), indicating that prenatal PBDE-209 exposure led to significant rat learning impairments. In the probe trial on day 6, the data for the rats’ swim tracks, platform location crossings, and time in the target quadrant showed that rats in the PBDE-209 group spent more time moving along the wall of the pool and had a difficult time finding the previous location of the platform (Figure 1(b)). The success of the PBDE-209 group in finding the previous location of the platform (p < 0.05; Figure 1(c)) and target quadrant (p < 0.05; Figure 1(d)) was lower than that of the blank and oil control groups. Effect of prenatal decabromodiphenyl ether-209 (PBDE-209) exposure on learning and memory in a rat model assessed with the Morris Water Maze task test. (A) The learning ability was assessed in the offspring from day 1 to day 5. The mean escape latency data for the blank control, oil control, and PBDE-209 groups (300 mg kg−1 day−1) were collected and plotted. (B–D) The memory ability of the rats on day 6 was tested when the platform was removed. The graph in B shows the representative swim tracks of the rats. The graph in C summarizes the number of crossings at the target platform location. The graph in D summarizes the percentage of time spent in the target quadrant (n = 16 per group in all panels. The data are summarized as the mean ± SD; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; and NS, p > 0.05. Results were plotted (mean ± SD) with GraphPad Prism.
Protective effect of IGF-1 on rat learning and memory after prenatal PBDE-209 exposure
To assess the effect of IGF-1 treatment on inhibiting prenatal PBDE-209 exposure-induced neurodevelopmental toxicity in vivo, pregnant rats were treated with IGF-1 (see the Methods section for details) and later subjected the offspring to the MWM test. The test showed that the mean latency to the platform for the PBDE-209 + IGF-1 group on days 3–5 was significantly shorter than that for the PBDE-209 group (p < 0.05 using the repeated-measures ANOVA test; Figure 2(a)). In the probe trial on day 6, rats in the PBDE-209 + IGF-1 group spent more time moving around the location of the platform than those in the PBDE-209 group (Figure 2(b)). Moreover, rats in the PBDE-209 + IGF-1 group were more successful at finding the platform (Figure 2(c)) and spent more time in the target quadrant (p < 0.05; Figure 2(d)). These data suggest that IGF-1 reduced the neurodevelopmental toxicity induced by prenatal PBDE-209 exposure and improved the memory and learning abilities of the offspring. Protective effect of prenatal injection of insulin-like growth factor-1 (IGF-1) on learning and memory improvement in PBDE-209 prenatally exposed rats assessed with the Morris Water Maze task test. (A) The learning ability was assessed from day 1 to day 5. The mean escape latency data for the blank control, oil control, PBDE-209 (300 mg kg−1 day−1), and PBDE-209 + IGF-1 (20 μg kg−1 day−1) groups were collected and plotted. (B–D) The memory ability of the rats on day 6 was tested when the platform was removed. The graph in B shows the representative swim tracks of the rats. The graph in C summarizes the number of crossings at the target platform location. The graph in D summarizes the percentage of time spent in the target quadrant (n = 16 per group in all panels). The data are summarized as the mean ± SD; * p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; and NS, p > 0.05. Results were plotted (mean ± SD) with GraphPad Prism.
Isolation and identification of hippocampal neural stem cells from rats
Hippocampal neural cells were isolated from the hippocampi of rats (1 day postnatal, with gestation between 19 and 21 days) and cultured in a serum-free medium for 7 days. Immunofluorescence was then performed to analyze the expression of the hippocampal neural stem cell (H-NSC) marker Nestin in the neurospheres. It shows that Nestin was highly expressed in over 95% of cells (Figure 3), indicating the high purity and self-renewing capacity of the multipotent H-NSCs. Isolation and identification of hippocampal neural stem cells (H-NSCs). The H-NSC purity was assessed by using immunofluorescence, that is, the H-NSCs were labeled with the H-NSC marker Nestin (green) and the nuclei were stained with DAPI (blue) (scale bar, 50 μM).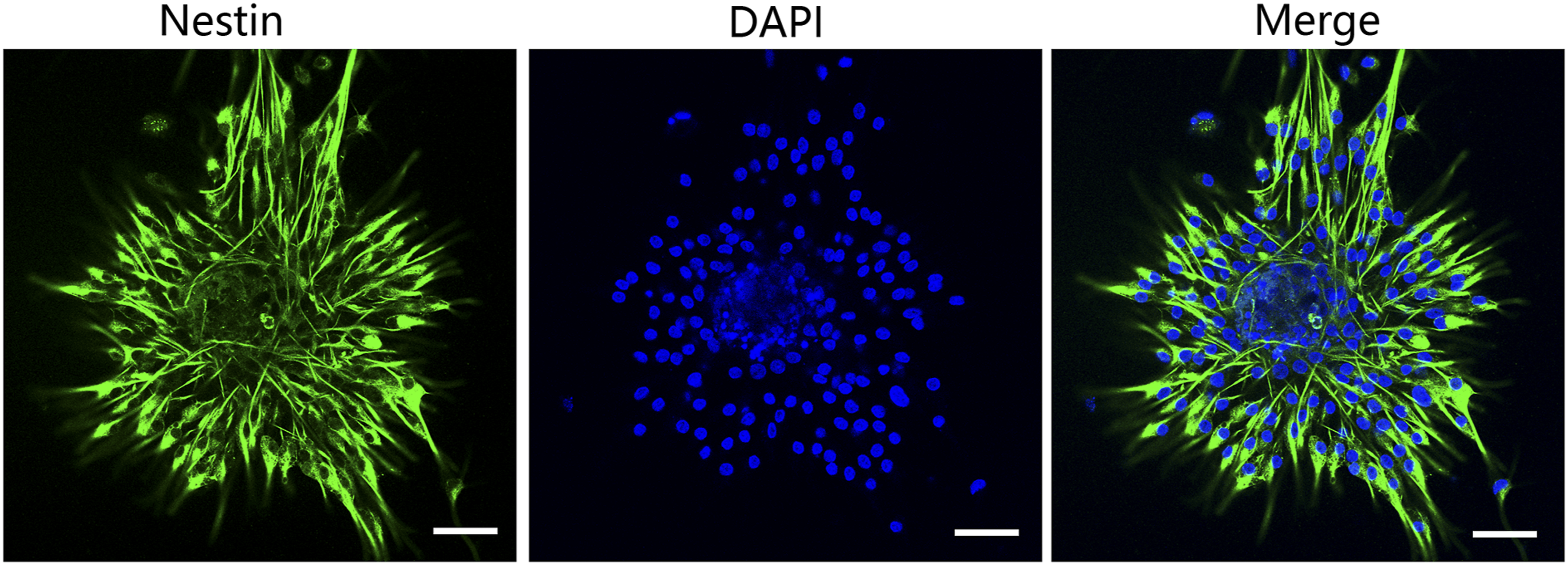
Effects of PBDE-209 exposure on cell proliferation, apoptosis, and differentiation of H-NSCs in vitro
To assess the effect of PBDE-209 exposure on the proliferation, apoptosis, and differentiation of H-NSCs in vitro, we separated primary cultured H-NSCs into blank control, vehicle control, PBDE-209, PBDE-209 + IGF-1, PBDE-209 + IGF-1 + LY294002, PBDE-209 + IGF-1 + PD98059, and PBDE-209 + IGF-1 + LY294002 + PD98059 groups and performed a cell viability assay. And it shows that the viability of the PBDE-209 (50 uµ/ml)-treated group was significantly lower than that of the blank and vehicle control groups (p < 0.05; Figure 4(a)). There was no difference between the vehicle (1% of DMSO) and blank control groups (p > 0.05; Figure 4(a)). Furthermore, PBDE-209 exposure significantly induced cell apoptosis (Figure 4(b)) but reduced cell differentiation, as indicated by the neuron marker NeuN, compared to the levels in the blank and vehicle control groups (p < 0.05; Figure 4(c)). Effects of PBDE-209 on regulation of viability, apoptosis, and differentiation of H-NSCs in vitro. (A) Cell viability CCK8 assay. The H-NSCs were grown, treated with PBDE-209 (50 μg/ml), DMSO, or nothing for up to 72 h, and then subjected to the CCK8 assay. (B) Flow cytometric Annexin V-FITC/PI assay. The H-NSCs were grown, treated with PBDE-209 (50 μg/ml), DMSO, or nothing for 72 h, and then subjected to the assay. The graph presents a summary of the assay results. (C) Immunofluorescence. H-NSCs were grown, induced to differentiate after treatment with PBDE-209 (50 μg/ml), DMSO, or nothing for 72 h, and then subjected to immunofluorescence using the neuronal marker NeuN (green). Blue color indicates the DAPI staining of the nuclei. The graph summarizes the results of the assay (scale bar, 20 μM, *p <0.05; **p < 0.01; ***p <0.001; ****p <0.0001; and NS, p > 0.05; scale bar, 50 μM). Results were plotted (mean ± SD) with GraphPad Prism.
Effect of IGF-1 on PBDE-209–induced cell cytotoxicity in vitro
We further analyzed and confirmed the effect of IGF-1 on reducing PBDE-209–induced cell cytotoxicity in vitro by exposing the H-NSCs to PBDE-209 (50 μg/ml) alone or with IGF-1 (120 ng/mL) for up to 7 days. The viability of the PBDE-209 plus IGF-1–treated H-NSCs was significantly improved compared to that of the PBDE-209–only treated H-NSCs, which consistently declined over time (p < 0.05; Figure 5(a)). The apoptosis rate of the PBDE-209 + IGF-I–treated H-NSCs was lower than that of the PBDE-209–only treated H-NSCs (p < 0.05; Figure 5(b)). The rate of H-NSC differentiation into neurons in the PBDE-209 + IGF-I–treated H-NSCs was also better than that of the PBDE-209 group (p < 0.05; Figure 5(c)). IGF-1 inhibition of PBDE-209 exposure-induced cytotoxicity in H-NSCs. (A) Cell viability CCK8 assay. The H-NSCs were grown, treated with PBDE-209 (50 μg/ml) or IGF-1 (120 ng/mL) for up to 72 h, and then subjected to the CCK8 assay. (B) Flow cytometric Annexin V-FITC/PI assay. The H-NSCs were grown, treated with PBDE-209 (50 μg/ml or IGF-1 (120 ng/mL) for 72 h, and then subjected to the assay. The graph summarizes the results of the assay. (C) Immunofluorescence. H-NSCs were grown, induced to differentiate after treatment with PBDE-209 (50 μg/ml) or IGF-1 (120 ng/mL) for 72 h, and then subjected to immunofluorescence using the neuronal marker NeuN (green). The blue color indicates the DAPI staining of the nuclei. The graph summarizes the results of the assay (scale bar, 20 μM, *p <0.05; **p < 0.01; ***p <0.001; ****p <0.0001; and NS, p > 0.05; scale bar, 20 μM). Results were plotted (mean ± SD) with GraphPad Prism.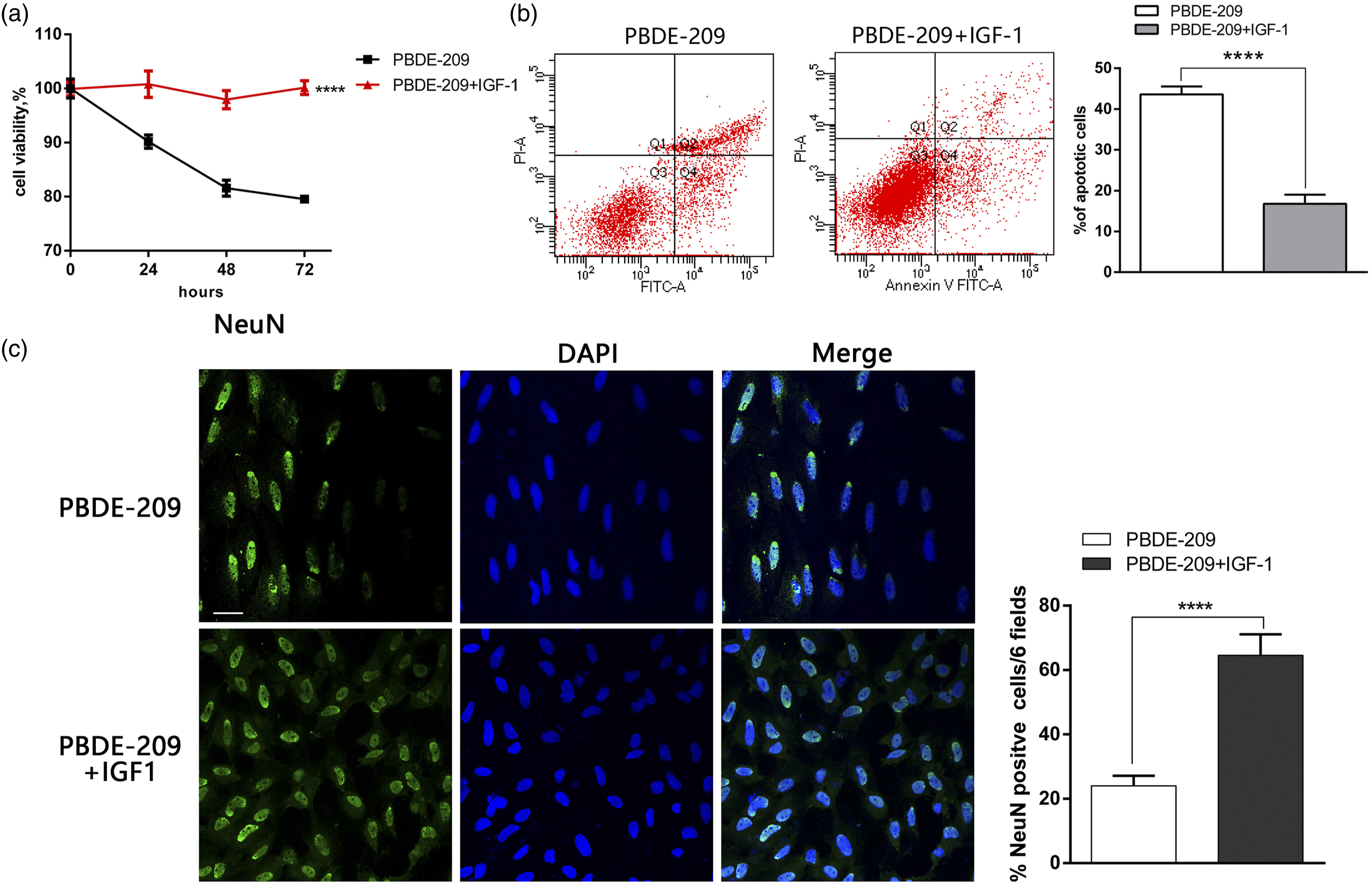
IGF-1 inhibition of PBDE-209–induced cell cytotoxicity through activation of the PI3K/Akt and MEK/ERK signaling pathways in vitro
To explore the underlying molecular events by which IGF-1 protects neurons against PBDE-209–induced cytotoxicity, the PI3K inhibitor LY294002 and the MEK inhibitor PD98059 were used. Our CCK8 assay showed that cell viability was reduced after treatment with LY294002 and PD98059 individually or in combination (p < 0.05; Figure 6(a)), whereas the apoptosis rate significantly increased (p < 0.05; Figure 6(b)). The rate of neuron differentiation was also lower in the PBDE-209 + IGF-1 + LY294002 and PBDE-209 + IGF-1 + LY294002 + PD98059 groups than that in the PBDE-209 + IGF-1 group (p < 0.05; Fig. 6CD), but there was no significant change in neuron differentiation in the PBDE-209 + IGF-1 + PD98059 group (p < 0.05; Fig. 6CD). Our Western blot data showed that LY294002 and PD98059 blocked the IGF-1–induced increase in the expression of p-Akt and p-ERK separately (Figure 6(e)). IGF-1 reduced PBDE-209 exposure-induced cytotoxicity in H-NSCs through activating of the PI3K/Akt and MEK/ERK pathways. (A) Cell viability CCK8 assay. The H-NSCs were grown, treated with PBDE-209 (50 μg/ml), IGF-1 (120 ng/mL), LY294002 (a PI3K inhibitor; 50 μ
Discussion
In the current study, a rat model of prenatal PBDE-209 exposure was established to assess the learning and memory abilities of the offspring by using the MWM test in vivo. Compared with the blank and oil control groups, the mean latency to the platform in the PBDE-209 group was significantly longer. The number of platform crossings and the amount of time the PBDE-209 group spent in the target quadrant were also lower, indicating that the learning and memory abilities of these rats were impaired. Our current data are consistent with a previous report by Wu et al., indicating that exposure to a certain dose of PBDE-209 during pregnancy impaired the spatial learning and memory abilities of the offspring 19 as well as to the study by Du et al., 13 showing that PBDE-209 exposure damaged the learning and memory abilities of rats. Our data also showed that the latency to the platform for the PBDE-209 + IGF-I group was significantly shorter than that to the platform for the PBDE-209 group, and the number of platform crossings and the time spent in the target quadrant were higher. These results indicate that IGF-I can protect against PBDE-209 impairment of spatial learning and memory in rats. Previous studies showed that estradiol interacts with IGF-I signaling to improve brain functions. 20 Trejo et al. 21 also reported that IGF-1 injection (50 μg/kg/day for a month) significantly increased hippocampal neurogenesis and improved the spatial learning ability of IGF-I–deficient C57/BL6 mice. Growth hormone intervention increased the serum IGF-1 level in elderly Fisher 344 rats, effectively improving their spatial learning ability. 22 Our study confirmed the protective effects of IGF-I on brain activity and demonstrated the protective effects on PBDE-209–induced neurodevelopmental toxicity. Our finding provides further evidence that IGF-I can alleviate brain injury induced by different risk factors and also plays an important role in protecting the nervous system.
We further investigated the neurotoxic effects of PBDE-209 on primary cultured hippocampal neurons in vitro; data showed that PBDE-209 inhibited neuron viability and differentiation and induced apoptosis. These findings are similar to those in previous studies in which hippocampal neuron exposure to PBDE-209 resulted in oxidative stress and apoptosis, 23 PBDE-209 exhibited in vitro cytotoxic effects on human red blood cells, 24 and PBDE-209 inhibited neurite outgrowth and NSC differentiation into neurons in a concentration-dependent manner. 25 However, our current data are inconsistent with a previous study of PBDE-209 in different cancer cells, which showed that PBDE-209 promoted the proliferation of various cancer and normal ovarian cell lines, and reduced tamoxifen, protein kinase C α (PKCα), and ERK inhibition–induced cell apoptosis. 26 To figure out the protective effect of IGF-1, we found that IGF-I induced the proliferation and differentiation of H-NSCs and inhibited apoptosis. Both the PI3K inhibitor LY294002 and the MEK inhibitor PD98059 blocked the protective effects of IGF-1 on these neurons, although the MEK inhibitor had no effect on the differentiation of neurons. IGF-I treatment significantly increased the levels of p-Akt and p-ERK1/2. Indeed, a previous study showed that IGF-I upregulated p-ERK1/2 and p-Akt proteins in dorsal root ganglion neurons, suggesting that the PI3K/Akt and ERK1/2 pathways might be involve in the IGF-1 neuroprotective effects. 27 Another previous study also showed that IGF-I activated both the PI3K/Akt and MEK/ERK pathways, but only the activation of the PI3K/Akt pathway induced the expression of a transcription factor, brain transcription factor 4 (Brn-4), and promoted the differentiation of H-NSCs into neurons. 28 IGF-I also regulated cell proliferation, apoptosis, and differentiation through activation of the PI3K/Akt and mitogen-activated protein kinase (MAPK) signaling pathways.29–31 Taken together, our current study supports the existing data, and our research finds the protective role of IGF-1 in antagonizing PBDE-209–induced neurotoxicity in vivo and in vitro.
A previous study demonstrated that IGF-1 overexpression in aged mice could help to increase the level of p-ERK and upregulate the expression of neuronal proteins (NeuN) in hippocampal neurons, suggesting that these changes could improve spatial learning and memory abilities. This effect was suppressed by the MEK inhibitor U0126, and the authors suggested that the MEK/ERK pathway played an important role in the protection of the nervous system. 32 Another study reported that IGF-I induced Brn-4 expression through activation of the PI3K/Akt pathway to promote neuronal differentiation and restore de-nerved hippocampal functions, but the MEK/ERK pathway was not involved in this process. 32 However, our current data suggest that IGF-I promotes neuronal cell viability and differentiation through both the PI3K/Akt and MEK/ERK pathways. Nonetheless, our study is preliminary, and more studies are needed to investigate and identify the precise mechanism of IGF-1 action on these H-NSCs in vitro. For example, Raman-enhanced spectroscopy (RESpect) probe provides a forward-looking strategy for the diagnosis of neurodevelopmental abnormalities. 33 In addition, multiple models are needed to verify the neurodevelopmental toxicity of PBDE-209 and the function of IGF-1, such as the zebrafish model. 34 It is also necessary to expand the study of neurodevelopmental toxicity induced by PBDE-209, such as whether it is related to the increasing autism spectrum disorders (ASD). 35 In the future study, new drugs like Phyllanthus albizzioides which shows great effective biological activities in antioxidant, antidiabetic, cytotoxicity, antimicrobial, antitumor, and NO inhibition activities should be considered to study whether it can reduced neurodevelopmental toxicity. 36 These unknown issues are worth exploring.
Conclusion
Prenatal PBDE-209 exposure impaired the learning and memory ability of rats, whereas IGF-1 treatment was able to inhibit this neurodevelopmental toxicity of PBDE-209 by activation of the PI3K/AKT and ERK1/2 pathways.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211045959 – Supplemental Material for Insulin-like growth factor reduced against decabromodiphenyl ether-209–induced neurodevelopmental toxicity in vivo and in vitro
Supplemental Material, sj-pdf-1-het-10.1177_09603271211045959 for Insulin-like growth factor reduced against decabromodiphenyl ether-209–induced neurodevelopmental toxicity in vivo and in vitro by Yuanxiang Yang, Qianyun He, Zhengyu Zhang, Chunli Qi, Lina Ding, Tingting Yuan, Yanhong Chen and Zhihua Li in Human & Experimental Toxicology
Footnotes
Acknowledgments
We thank Medjaden Bioscience Limited (Hong Kong,China) for assisting in the preparation of this manuscript.
Declaration of conflicting interests
The authors declared that there is no conflict of interest in this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article; this study was supported by a grant from the Guangdong Province Nature Science Foundation (2018A0303130298).
Supplementary material
Supplementary material for this article is available online.
Ethic Approval
The animal protocol for this study was approved by the Institutional Animal Care and Use Committee (IACUC) of Guangzhou Medical University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
