Abstract
Inorganic arsenic (iAs) exposure is a serious health problem that affects more than 140 million individuals worldwide, mainly, through contaminated drinking water. Acute iAs poisoning produces several symptoms such as nausea, vomiting, abdominal pain, and severe diarrhea, whereas prolonged iAs exposure increased the risk of several malignant disorders such as lung, urinary tract, and skin tumors. Another sensitive endpoint less described of chronic iAs exposure are the non-malignant health effects in hepatic, endocrine, renal, neurological, hematological, immune, and cardiovascular systems. The present review outlines epidemiology evidence and possible molecular mechanisms associated with iAs-toxicity in several non-carcinogenic disorders.
Introduction
Inorganic arsenic (iAs) exists in nature as both a trivalent (arsenite: iAs III) and pentavalent forms (arsenate: iAs V) and is one of the most toxic metals derived from the natural environment due to natural contamination of groundwater, affecting millions of people around the world.1–3 iAs exposure through drinking water has been a serious public health problem in several countries such as Bangladesh, India, Taiwan, Mexico, Argentina, Chile, and the USA. 4 Since 2016, the US Environmental Protection Agency (EPA) and the World Health Organization (WHO) set the Maximum Contaminant Level for arsenic in drinking water as 10 ppb (parts per billion; 10 ppb = 10 μg/Liter). 5
The diagnosis of arsenic exposure depends on the type of arsenic being more toxic As III than AsV, route of exposure, and whether the exposure is acute, subacute, or chronic. The most specific overt sign of chronic inorganic arsenic ingestion is skin or dermal effects. Hyperpigmentation is the most common dermal effect, but hypopigmentation or alternating hyperpigmentation and hypopigmentation (raindrops on a dusty road) may occur. Hyperkeratosis with bilateral thickening of the palms and soles may also occur. Focal hyperkeratotic lesions or corns may occur on the feet, palms, face, or other parts of the body. Other effects of chronic arsenic exposure include peripheral neuropathy, gastrointestinal symptoms, diabetes, renal system effects, enlarged liver, non-cirrhotic portal hypertension, peripheral neuropathy, anemia, hypertension, and cardiovascular disease. 6 The determination of arsenic species could be evaluated in different biological matrices such hair, urine, plasma, and blood. Several techniques have successfully been used for oxidation-state specific speciation analysis of As in biological matrices, including high-performance liquid chromatography (HPLC)-HG-atomic fluorescence spectrometry (AFS), HPLC-inductively coupled argon plasma mass spectrometry (ICP-MS), and hydride-generation atomic absorption spectrometry using cryotrapping (HG-CT-AAS). 7
Chronic iAs exposure has been associated with the development of different cancers such as lung, skin, liver, bladder, and kidney.8–12 Also, iAs has been reported to cause other non-malignant disorders such as diabetes, cardiovascular diseases, pregnancy complications, neurological conditions, renal diseases, and neurobehavioral effects in exposed children to iAs.13–17
Arsenic undergoes in vivo several complex metabolic conversions, producing methylated metabolites such as monomethylarsonous acid (MMA-III) and dimethylarsonous acid (DMA-III), that are reactive and toxic, being more toxic methylated species. 18 Cytotoxic effects of trivalent and pentavalent arsenicals have been examined, and these have showed that pentavalent arsenicals are significantly less cytotoxic than their trivalent counterparts. Among trivalent arsenicals, MMA-III is the most cytotoxic species, followed by DMA-III and iAsIII. 19
Both iAs and its metabolites interact with extra- and intracellular macromolecules, in particular, those containing vicinal thiols affecting crucial enzymes such as pyruvate dehydrogenase, glutathione reductase, glutathione peroxidase, and thioredoxin reductase (TrxR).1,20–23 Furthermore, several molecular mechanisms associated with detrimental effects of arsenic exposure, including induction of oxidative stress, indirect genotoxicity, and epigenetic regulation of gene expression. 24
Regarding the treatment of arsenic exposure, the chelation therapy has been showed to improve outcomes whether started in minutes to hours after arsenic exposure. 25 The classic antidote against acute arsenic poisoning is BAL (British anti-Lewite or dimercaptopropanol). However, due to the rather high toxicity of BAL, and the necessity of frequent and inconvenient intramuscular administrations, the clinical use of this drug is currently restricted to only the initial treatment for few days after acute iAs intoxications.1,26 Nowadays, the treatment for acute intoxication uses sodium 2, 3-dimercaptopropane-1-sulfonate (DMPS) and meso-2, 3-dimercaptosuccinic acid (DMSA) as chelatings antidotes. No treatment of proven benefit is currently available to treat chronic arsenic toxicity. Treatment options advocated are vitamin and mineral supplements and antioxidant therapy. In cases of chronic arsenic poisoning, one should consider BAL as a chelator the signs of arsenicosis are severe or the patient is in complications. 27
A variety of methods of diverse complexity are available to remove arsenic from drinking water. The methodology, especially in developing countries, that is urgently required should be affordable, sustainable by the population, and cost effective. 28
The objective of this review was to describe the non-carcinogenic effects of arsenic on different organs and systems such as liver, pancreas, kidney, nervous, hematological, immunological, cardiovascular, and skin, focusing in their epidemiology and possible molecular mechanisms of iAs-related toxicity.
The literature search was performed employing keywords such as arsenic, liver, kidney, arsenic related toxicity mechanism, endocrine disorders, diabetes, immunosuppression, hematological, skin, and drinking water in EMBASE and MEDLINE databases accessed from Pubmed.
Arsenic and liver disorders
The liver is the major site of arsenic metabolism, showing a dose–response accumulation of this metalloid and its metabolites, possibly related with overexpression of iAs(III)-transporters, such as aquaglyceroporin-9 (AQP9) in response to iAs exposure.29,30
Epidemiology
Epidemiology data have showed an association between iAs exposure and non-malignant hepatic disorders such as hepatomegaly, non-cirrhotic intrahepatic portal hypertension (NCIPH), portal fibrosis, cirrhosis, hepatitis, and nonalcoholic fatty liver disease (NAFLD).
For instance, populations of endemic areas of arsenic contaminated drinking water, food, and air, showed a high prevalence of hepatomegaly accompanied in some cases by elevated serum levels of liver enzymes (eg., alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase) compared with control populations.31–37
A main consequence of chronic high-level arsenic exposure is NCIPH that is a result of chronic microangiopathy of portal vein branches, leading to intrahepatic portal vein occlusion. 38 In agreement with this, populations with elevated ingest of arsenic in drinking water have a high prevalence of NCIPH in presence of skin arsenicosis.32,33,35,39 In fact, a study performed in Chandigarh, India, showed that arsenic levels in liver biopsy of patients of NCIPH were significantly higher (0.887 ± 0.404 ppm) compared with cirrhosis patients (0.178 ± 0.171 ppm) and control individuals (0.071 ± 0.058 ppm); interestingly, some of the NCIPH patients consumed drinking water with high arsenic content (0.360 and 0.549 ppm). 40 Also, prolonged treatment of psoriasis patients with arsenical medications such as Fowler’s solution has been related with arsenical skin changes and NCIPH development, possibly because of iAs-direct damage of intrahepatic portal veins.41–44
In relation to more severe liver damage lesions, chronic hydroarsenism has been associated with portal fibrosis occurrence,35–37 increased risk, prevalence, and mortality of cirrhosis.12,45 Similarly to NCIPH, several cases of cirrhosis have been described as a consequence of occupational arsenic exposure or after prolonged use of Fowler’s solution for psoriasis treatment.42,44,46–48
In addition to iAs-related hepatotoxicity lesions, increased well water arsenic concentration (≥ 300.0 μg/L) associates with high prevalence of viral infections such as chronic hepatitis. 45 Specifically, high urinary arsenic levels associate with anti-HAV seroprevalence, HEV seroconversion during pregnancy, elevated risk of hepatitis B infection, and higher prevalence rates of hepatitis C in petrochemical complex area.49–52
Finally, increased urinary arsenic levels associates positively with elevated serum concentrations of biomarkers for NAFLD (ALT enzyme), suggesting a higher risk to this disease in Mexican Americans in the US. 53 Consistently, the presence of heavy metal contamination including iAs in soils increased the risk for NAFLD in men with a body mass index (BMI) <24, reinforced the proposal that arsenic exposure is a risk factor for NAFLD occurrence. 54
Molecular mechanisms
The liver constitutes a natural target of iAs-toxicity promoted by metalloid-induction of several hepatic damage mechanisms such as oxidative stress, inflammation, and cell death activation.
Arsenic induces oxidative stress in the liver
Studies employing isolated rat hepatocytes showed that iAs(III) promotes reactive oxygen species (ROS) production and lipid peroxidation, possibly associated with a disruption of mitochondrial membrane potential.55,56 In a similar manner, mitochondria are the main ROS-source in rat liver cell line exposed to MMA(III); whereas in the same cellular model, DMA(III) treatment promotes ROS formation and stress in the endoplasmic reticulum (ER).21,57 Animal models exposed to iAs by drinking water showed increased ROS production, malondialdehyde (MDA) accumulation, elevated protein oxidation, low levels of glutathione (GSH), and decreased glutathione peroxidase (GSH-Px), glutathione reductase (GR), superoxide dismutase (SOD), and catalase (CAT) activities in the liver compared to control animals.58–62
Also, it has been reported that iAs therapeutic formulations, such as arsenic trioxide (ATO) promotes oxidative stress, increased lipid peroxidation, reduced GSH levels, and altered antioxidant enzymes activity (CAT, SOD, and GSH-Px) in hepatocellular carcinoma cell line, HepG2.63–69 In addition to iAs, the treatment with phenylarsine oxide, an organic arsenic compound, induces ROS generation in mitochondria and ER of HepG2 cells (Figure 1).
70
Molecular mechanisms of liver damage induced by arsenic. Inorganic arsenic (iAs) promotes hepatomegaly, ROS formation, and lipid peroxidation and reduces antioxidant enzymes (GSH-Px, glutathione peroxidase; GR, glutathione reductase; CAT, catalase; SOD, superoxide dismutase). Furthermore, it promotes pro-inflammatory cytokines (TNF-α and IL-1β), fibrosis markers (vimentin, and α-smooth muscle actin, α-SMA), mitochondria membrane potential (Ψm) disruption, changes in pro-apoptotic (Bax and Bad), anti-apoptotic (Bcl-xL and Bcl-2) proteins, death receptor-5 (DR5) expression, caspase-8, caspase-9, and caspase-3 activation, and DNA fragmentation. Liver images were obtained from Servier Medical Art.
Arsenic induces liver inflammation
Chronic environmentally arsenic-exposed populations with metalloid poisoning showed hepatic degenerative lesions characterized by chronic inflammation, vacuolation, focal necrosis, and collagen fibrosis in the periportal areas. 71 Animal models suggested that iAs(III)-induced inflammation and histopathological changes (e.g., dilatation of sinusoids, leukocyte infiltration, ballooning of hepatocytes, and fibrosis) in the liver might be related with elevated expression of pro-inflammatory molecules such as IL-1β, TNF-α, IL-6, IL-8 and nuclear factor kappa beta (NF-κB), increased IL-1β, TNF-α, IL-6, and IL-8 secretion, high tissue protein levels of TNF-α, IL-6, hypoxia-inducible factor-2α (HIF-2α), and several fibrosis markers, including PCNA, collagen I, vimentin, and α-smooth muscle actin (α-SMA) (Figure 1).58–62,72,73
Mechanistically, it has been described that iAs(III) treatment of human fetal hepatocytes (L-02 cells) induces hypoxia inducible factor-1α (HIF-1α)-dependent overexpression of monocarboxylate transporter-4 (MCT-4) and elevated glycolysis, favoring IL-6, IL-8, and TNF-α production. 74 Additionally, in L-02 cells, arsenite-induced inflammation can be activated through epigenetic mechanisms such as 1) overexpression of MALAT, a long non-coding RNA that promotes HIF-2α protein stabilization and 2) miR-379-5p downregulation, associated with high expression of its target gene Gasdermin and IL-1β release that participates in hepatic stellate cell line activation, critical for hepatic fibrosis72,73
Arsenic promotes liver cell death
Cytotoxic effect of iAs in hepatocytes of exposed animals has been related with oxidative stress–dependent apoptosis characterized by oxidative DNA damage, mitochondrial damage (disruption of membrane potential, mitochondrial swelling, and inhibition of mitochondria enzymes), changes of Bcl-2, Bcl-xL, Bad, Bax, and Bim proteins in association with increased level of Apaf-1, cytochrome c, activation of caspase-9, caspase-3, PARP cleavage, and DNA fragmentation (Figure 1).56,58,61,75–79 In addition to intrinsic apoptosis, iAs-toxicity in the liver can activate extrinsic and ER stress–dependent apoptosis pathways with caspase-8, and caspase-12 cleavage, respectively. 61 Recently, it has been reported that sodium arsenite activates ER-stress signaling pathways in L-02 cells, promoting elevated protein levels of transcription factor C/EBP homologous protein (CHOP) that induces Death Receptor 5 (DR5) expression, favoring extrinsic apoptosis activation through DISC assembly and caspase-8 cleavage. 80
This iAs-related hepatotoxicity involves activation of p53 and its targets genes (miR-34a and Bax), as well as NF-κB, pigment epithelium-derived factor (PEDF), PKCδ-JNK, p38, and MAPK signaling pathways activation.58,75,78,81 iAs(III) treatment reduces protein levels of critical regulators of protective routes such as ERK, PI3K-AKT, and Nrf2 in rat hepatocytes. 61 In contrast, it has also been described that iAs(III) activates Nrf2 pathway, even critical antioxidant molecules such as GSH is required for DMA-induced apoptosis in rat liver cell line.58,82 Interestingly, this capacity of iAs to modulate differentially cell survival pathways could be related with differentiation state of human hepatocytes. 83
Furthermore, iAs-treatment in hepatocellular carcinoma cell lines, induces pro-apoptotic mechanisms such as mitochondrial dysfunction, downregulation of Bcl-2 in parallel to upregulation of p53, Bax, caspase-9, and caspase-3 expression, decreased protein levels of Bcl-xL, Bcl-2, and XIAP, elevated p53 protein, activation of caspase-9 and caspase-3, phosphatidylserine exposure, and DNA damage.63,67–69,84–87
Arsenic and endocrine disorders
Endocrine glands are critical to control several processes essential to survival and reproduction. The main endocrine glands are the pituitary, thyroid, parathyroid, pancreas, adrenals, and gonads. Type 1 and 2 diabetes mellitus (T1DM and T2DM) constitute the most common endocrine disorder; however an other alterations including primary hypothyroidism, hyperthyroidism, polycystic ovary syndrome, and male hypogonadism. 88
Epidemiology of arsenic and diabetes
Several epidemiological data have revealed a strong association between DM and arsenic exposure. A first observation was that residents of areas where chronic arseniasis is endemic showed a higher age- and gender-adjusted DM prevalence (odds ratio = 2.7), incidence and mortality rates compared with nonendemic areas.12,89–91 In this populations, environmental toxicant–disease relationship was reinforced because DM mortality decreased in females after elimination of arsenic exposure in drinking water. 92 In fact, areas with low, median and high inorganic arsenic water concentrations, for instance, in Serbia (<57 μg/L), USA (<100 μg/L), Bangladesh (<170 μg/L), Chile (>800 μg/L), and Cambodia (>900 μg/L) have a higher risk, incidence, and prevalence for T2DM in comparison to unexposed populations.93–98 Nevertheless, it has also been reported that T2DM risk exhibits a dose–response pattern as iAs levels increases in water and after long times of exposure.99–102 Meta-analysis data have proposed that T2DM risk increased by 13% for every 100 μg/L increment of iAs in drinking water. 103
A proper biomarker of iAs exposure is the concentration of metalloid levels in urine, some reports have identified a higher risk and prevalence of T2DM for drinking water exposed individuals with high levels of total iAs(III) and its metabolite DMA(III) in urine,104–106 and presence of iAs(III), MMA(III) and DMA(III) in exfoliated urothelial cells. 107 Other reports described an association between a lower proportion of monomethylated arsenicals in urine with increased insulin resistance (HOMA2-IR), and T2DM incidence.108,109 In contrast, higher proportions of MMA and DMA are associated with T1DM risk. 110
iAs poisoning might be related with pre-diabetic disturbances, for example, exposed populations showed a positive association between total water and urinary arsenic concentrations with risk and prevalence of prediabetes, respectively.111,112 Similarly, elevated urinary total arsenic levels were significantly associated with another glucose metabolism alterations, such as higher fasting and random glucose levels, increased glycated hemoglobin (HbA1c), insulin sensitivity diminution and reduced secretion capacity, possibly related with pancreatic β-cell dysfunction.113–116
In pregnant women, the presence of DMA in urine and iAs in blood, as well as, toenails of women postpartum and newborn meconium have been associated with a higher risk of gestational diabetes mellitus (GDM).117–120 An estimation considers that each 5 μg/L augment in water iAs concentration, is associated with nearly a 10% greater risk of GDM. 119
Molecular mechanisms of arsenic and diabetes
As previously described, epidemiological evidence supports that human populations exposed to iAs have a higher risk for diabetes development. Nevertheless, the molecular mechanisms through that arsenic plays a role in diabetes pathogenesis, nowadays still being evaluated.
The β-cell dysfunction followed by the changes in the insulin secretion is the hallmark of diabetes and can be generated by arsenic through a direct cell damage and functional deregulation of insulin secreting β-cells of pancreas.
Arsenic induces pancreas damage and dysfunction
Arsenic is taken up by pancreatic tissue and β-cells in a dose-dependent manner 121 and accumulated significantly in the pancreas121–130 Interestingly, it has been described in streptozotocin (STZ)-induced T1DM mice models, organ-specific overexpression, and high protein levels of AsV (PIT1 and PIT2) and AsIII (AQP7, AQP9, and GLUT1) uptake transporters in comparison with non-diabetic animals,131,132 suggesting that iAs transport might increase under diabetic conditions.
Animal models, subchronic or chronically exposed to iAs showed morphological changes, for instance, decreased islet cell number and size, pancreatic vasculature alterations, higher levels of oxidative markers (NO, MDA, and OH.), and reduced activities of antioxidant enzymes, such as, SOD, CAT, and TrxR in pancreas tissue, compared with control rats.122,133–137 Similarly, STZ-induced diabetic rats exposed to arsenic presented severe degeneration, vacuolation, necrosis, degranulation, and shrinkage in the islets of Langerhans
138
(Figure 2). Molecular mechanisms of pancreas damage induced by arsenic. Inorganic arsenic (iAs) promotes pancreas necrosis, inhibits glucose-stimulated insulin secretion (GSIS) through upregulation of UDP-glucuronosyltransferase 1 family, polypeptide a6a (Ugt1a6a), reduces insulin expression, and modulates insulin transcription factors (mafA and PDX1) via mir-149 and mir-2909 regulation. Oxidative stress induced by iAs produces apoptosis characterized by low protein levels of PPARγ-PINK1, high ASK1 levels, mitochondria membrane potential (Ψm) disruption, high pro-apoptotic Bax, and low anti-apoptotic Bcl-2 protein levels which contributes to caspase-3 activation. Furthermore, iAs induces ROS-dependent autophagy. Pancreas image was obtained from Servier Medical Art.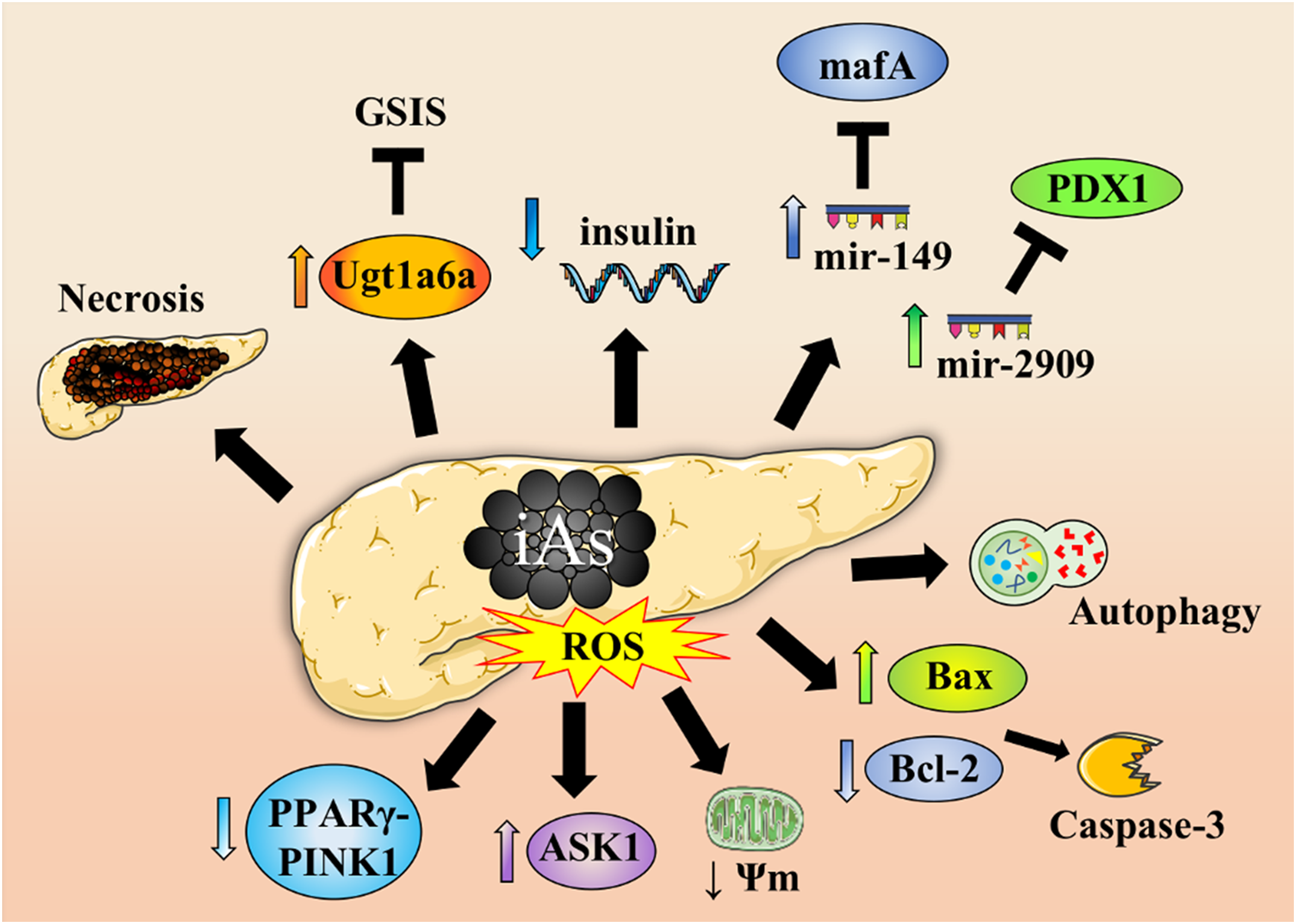
This iAs-associated diabetogenic toxicity might be related to molecular level with pro-oxidant properties of iAs that induces ROS overproduction, possibly through of elevated SIRT3 levels and decreased mitochondrial complex II activity in rats with deregulated glucose metabolism.139,140 This oxidative environment in pancreatic β-cells promotes low protein levels of PPARγ-PINK1 axis associated with mitophagy inhibition, TrxR inactivation, and high ASK1 levels, favoring apoptosis induction characterized by mitochondrial membrane potential disruption, cytochrome c release, increased pro-apoptotic protein Bax, reduced anti-apoptotic Bcl-2 protein levels, caspase-3 activation, and DNA fragmentation134,137,138,141,142 (Figure 2). In addition to apoptosis, in pancreas cells, iAs induces a ROS-dependent autophagic cell death accompanied with antioxidant Nrf2/TrxR pathway inhibition.135,143 This is important because Nrf2 activation is critical for pancreatic β-cell protection against iAs(III)-induced acute cytotoxicity. 144
Other critical alteration in T2DM pathogenesis is the deregulation of pancreatic β-cell function that include impairment of glucose-stimulated insulin secretion (GSIS) and defects in several cell types and tissues, such as adipose and liver which eventually leads to insulin resistance. 145
In vitro studies in β-cell lines showed a consistent reduction of GSIS associated with chronic, sub-toxic iAs exposure; however, there is some discrepancy in the literature about whether basal insulin secretion is altered by iAs exposure.146–150 Similarly, GSIS impairment has been observed in mouse pancreatic islets and insulinoma cells treated with iAs-metabolites, MMA(III) and DMA(III).151,152
To molecular level, it has been proposed that iAs affects several process that participate in GSIS, for instance inhibits mitochondrial metabolism, promotes metabolomic profile changes associated with impaired production of ATP in mitochondria, upregulates UDP-glucuronosyltransferase 1 family, polypeptide a6a (Ugt1a6a) expression favoring increased serotonin glucuronidation, and inhibits glucose-stimulated intracellular peroxide production through Nrf2-dependent expression of antioxidant enzymes146,147,152,153 (Figure 2).
Furthermore, Diaz-Villaseñor et al.
150
found that insulin mRNA expression decreases in primary rat pancreatic β-cells exposed to iAs(III) (5 μ
Besides pancreatic effects, Wauson et al.
155
evaluated iAs(III) exposure (6 μ
Taking together this information, it might be hypothesized that iAs exposure has been related to T2DM pathophysiology because of GSIS impairment and reduced insulin sensitivity in target cells such as adipocytes favoring insulin resistance development.
Arsenic and renal disorders
There are few studies addressing the effects of iAs on albuminuria, tubular injury, and biochemical variables as uric acid. However, there are important information about kidney disorders.
Epidemiology
Arsenic toxicity has been described since ancient times; however, iAs nephrotoxicity was just described 50 years ago when their exposure was associated with development of hemolysis and acute renal failure in industrial workers exposed to this metal; renal biopsies done in these patients showed acute tubular necrosis, renal cortical necrosis, diffuse interstitial fibrosis, and further progression to chronic kidney disease (CKD). 160
In a Chinese population exposed to arsenic was observed a significant increase of urinary levels of β2-microglobulin, albumin and N-acetyl-β2-glucosaminidase compared with unexposed population. These results showed that iAs could induce nephrotoxicity in both glomerular and tubular dysfunction. 161
Long-term arsenic exposure has also been reported to be associated with mortality attributed to renal disease, when the drinking-water supply system was improved, the mortality from renal disease declined gradually, then based on the reversibility criterion, the association between arsenic exposure and mortality attributed to renal disease is likely to be positively correlated. 162
On the other hand, Meliker et al. 163 realized a standardized mortality ratio (SMR) analysis in six counties of southeastern Michigan with low to moderate levels of arsenic (10–100 μg/L). They investigated the relationship between moderate arsenic levels and kidney disease and found an increase in SMR for both females and males, suggesting that low levels of arsenic also cause kidney disease. Besides, Zheng et al. 164 evaluated the association between iAs and albuminuria in American Indian adults living in rural areas of the United States with low to moderate exposure to iAs. This population had a high burden for diabetes (49.7%), obesity, and albuminuria (30%), and after statistical adjustments for diabetes, urinary Cd excretion, hypertensive medication, and systolic blood pressure, they found a higher prevalence ratio of albuminuria in subjects with higher iAs urinary excretion.
Also, in people among 30- to 49-year-old of Anfogasta in northern Chile exposed to arsenic concentrations (870 μg/L) was described an increase in the mortality by chronic renal disease. 165
Robles et al. 166 realized a cross-sectional and comparative study done in five communities localized close to Queretaro City. Subjects with no antecedents of renal disease, diabetes, hypertension, or industrial exposure to iAs were included, blood was taken for biochemical analysis, and a spot urine sample was collected for albumin, α1-microglobulin, and iAs measurements. They found an association between urinary iAs levels and α1-microglobulin urinary excretion (r2 = 0.07, p = 0.01) as marker of early renal injury, but not with albuminuria or other biochemical variables.
In order to evaluate the potential renal toxicity of As exposure in endemic area of chronic arsenicism was performed a cross-sectional study in 478 adults of Bangladesh recruited in two phases in 2001 and in 2003, whose renal function was evaluated measuring plasma cystatin C levels, and calculating the estimated glomerular filtration rate (eGFR). Consistent with renal toxicity of As, log-urinary As had a marginal inverse association with eGFR in the 2003 sample; however, this association was not significant in the 2001 sample. Adjustment for eGFR did not alter the associations between urinary Crn and the %urinary As metabolites, indicating that GFR does not explain these associations. 167
Finally, there is not enough information about the effects of arsenic exposure and renal injury in children; in the reported study done by de Burbure et al. 168 , there was no increase in markers of renal injury in 800 children across Europe with low iAs exposure. Also, there is information in children living in San Luis Potosi, Mexico, exposed to arsenic where the urinary arsenic levels were associated with higher urinary kidney injury molecule 1 (KIM-1). 169
Molecular mechanisms
The development of acute tubular necrosis with acute renal failure has been reported in patients with systemic toxicity occurring in severe acute arsenic poisoning, and some of these patients develop cortical necrosis and progression to CKD. Low-molecular-weight proteinuria, aminoaciduria, glycosuria, and phosphaturia (Fanconi syndrome) as well as progressive deterioration of renal function are clinical characteristics associated with chronic arsenic exposure. 160
Arsenic toxicity in proximal tubule cell (PTC) is initiated by depletion of the intracellular GSH stores, activation of the caspase-3 and -9 signaling pathway, increases in the expression of IL-6 and IL-8, and activation of the p53 apoptotic pathway. This leads to an increment in the production of ROS and other free radicals, inflammation, and apoptosis.170–172
iAs uncouples oxidative phosphorylation causing reductions in sodium, phosphate, and glucose transport, which is manifested clinically as Fanconi syndrome (phosphaturia, glucosuria, and low-molecular-weight proteinuria). 173
Albuminuria associated with iAs nephrotoxicity may be related to direct podocyte injury, or through a mechanism related with endothelial dysfunction as it has been showed that iAs increases the expression of the vascular cell adhesion molecule-1 (VCAM-1) and the angiotensin type I receptor. 174
Neurological disorders
Epidemiology
Epidemiological studies have described that exposure to iAs in drinking water has serious effect on the neurological system, affecting cognitive development in humans, especially children 4 causing deficits in intelligence and memory, 175 as well as a greater susceptibility to psychiatric diseases in adults, 4 since greater damage has been seen in both peripheral and central nervous systems, affecting body coordination, difficulty in solving problems, in clinical trials conducted by Sharma and Kumar 176 .
On the other hand, studies in animal models suggest that chronic exposure to iAs damages brain and nervous, 176 prenatal and early postnatal exposure has showed a decrease in the number of neurons and glia, as well as impairments in neurotransmission. 175 Mice exposure to 50 ppb of iAs throughout all pregnancy has been associated with alterations in gene expression in the brain in adulthood, generating deficits in the neurogenesis of the hippocampus. 177
Recent study of Health Effects of Arsenic Longitudinal Study (HEALS) in Araihazar, Bangladesh, showed that adolescents with well-documented exposure data from the prenatal period to adolescence current blood and urine iAs concentrations were consistently associated with deficits in intellectual function. This work documents that exposure to iAs in childhood has an adverse impact on a wide range of intellectual functions, with implications for diminished performance likely to continue into adulthood. 178
Moreover, studies have showed that iAs exposure increases the risk for autism spectrum disorder (ASD), and this was observed in recent meta-analysis and found statistically significant higher iAs concentrations, in hair and in blood, for children diagnosed with ASD compared with controls across studies. 179
Furthermore, studies carried out in Japanese population have described that patients under chronic treatment with iAs have presented sensory alterations throughout the body, being more intense in the extremities, this is due to an alteration in the conduction between the proximal brachial plexus and the primary sensory cortex, and authors suggest that this sensory alteration is due to damage that patients present in the central nervous system, coupled with their peripheral neuropath. 180
Molecular mechanisms
There are several hypotheses on the mechanism of action of iAs, one of the most documented through the generation of oxidative stress, due to its ability to generate ROS, which through its binding with sulfhydryl groups of enzymes and proteins is capable of interfering in several cellular signaling pathways, which has been related to presence of neurodegenerative illnesses associated with exposure of this metalloid.181–184
Chandravanshi et al. 185 have suggested that among main mechanisms of neurotoxicity of iAs are alteration of mitochondrial activity and oxidative stress. Recent study evaluated the long term iAs exposure on mitochondrial activity of brain and memory in mice exposed to As (25 or 50 ppm) in drinking water for 16 weeks, the results indicated that the retention latency decreased after 12 weeks, and these toxic effects could be due to the mitochondria originated oxidative stress along with the depleted antioxidant capacity of the brain of mice. 186 In addition, studies by Chandravanshi et al. 187 have observed in brain of rat exposed to iAs an increase in ROS associated with lower activity in mitochondrial complexes I, II, III, and IV.
Keshavarz-Bahaghighat et al. 188 have showed that iAs exposure (NaAsO2, 20 mg/kg) generated oxidative stress in rats’ hippocampus, producing ROS, release of cytochrome c, and mitochondrial inflammation, the latter through activation of the microglia and NF-κB. Additionally, caspase 9 and 3 activities as well as Bcl-2/Bax expression ratio also were affected.
Lipid peroxidation generated by iAs-induced oxidative stress produces degeneration of the central nervous system (CNS) through DNA damage and consequent death of brain cells. 189 Previous studies in primary cultured rat neurons exposed to 1, 5, and 10 μmol/L ATO showed that the cell apoptosis rates, the mRNA expression levels of calpain 1, cdk5, and the protein expression levels of calpain 1, cdk5, and p25 in the 5 and 10 μmol/L ATO treated groups were significantly higher compared with untreated control group (p < 0.05). These findings suggest that ATO-generated neuronal cell apoptosis may be influenced by calpain 1-cdk5/p25 pathway. 190
On the other hand, in animal models, it has been described that iAs(III) causes a low expression of NF-L in sciatic nerve, 191 and of proteins in the neurofilament, generating alterations in function the cytoskeleton, and consequently axonal damage to the peripheral nerves. 192 In vitro studies conducted by Vahidnia et al. 193 suggested that neurotoxic effect of arsenic can be modulated by the induction of Ca2+ influx intracellular, which activates calpain, generating the cleavage of p35 to p25.
When iAs affects the Na+-dependent glutamate/aspartate transporter (GLAST), it generates an accumulation of glutamate (GLU) in the intrasynaptic space; this amino acid is considered neurotoxic in high concentrations, its uptake in the Bergmann glia being regulated by the transporter excitatory amino acid 1 (EAAT1/GLAST). In addition, EAAT1/GLAST activity through ³H-D-aspartate uptake was determined by exposure for 24 h to iAs(III) in chick primary Bergmann glial cell cultures, the results showed a lower chglast transcription coupled with a decrease in the Km and Vmax of PKC, PKA, and p38 MAPK. They observed an increase in Nrf-2 DNA-binding activity that correlated with a rise in total glutathione levels. Therefore, these findings suggest that glial cells are target of iAs toxicity through the EAAT1/GLAST transporter, thus regulating brain functions. 194
Moreover, a CD1 mouse model exposed on postnatal days 90, gestational exposure to iAs negatively modulated the NR2B subunit of the glutamate N-methyl-D-aspartate receptor (NMDAR) in the hippocampal region, altering the expression of cysteine/glutamate transporters in the hippocampus and cerebral cortex. 195 In addition, CD1 mice exposed for 6 months to 0.5 or 5 mg As/L in drinking water showed hypoactivity at the end of iAs exposure, as well as an inhibition in the mRNA expression of dopamine D2 receptor only in mice group exposed to 5 mg As/L, which suggests that dopaminergic system is a molecular target of iAs exposure. 196
In fact, there is uncertainty about neurotoxicity mechanisms which arsenic acts during sensitive periods of brain development; however, it has been suggested that tight junction proteins (TJs) that make up blood–brain barrier could have a relevant role in the neurological damage generated by the exposure to this metalloid. A previous work performed by Manthari et al. 197 in mice exposed to ATO in doses of 0.15/1.5/15 mg of ATO/L in drinking water, during pregnancy and lactation in the postnatal period on days 21, 28, 35, and 42 (PND) described the induction of autophagy in the hippocampus and cerebral cortex regions with a lower expression of the occludin protein and mRNA, as well as a low expression of the mRNA of the TJ proteins (ZO-1, ZO-2, and claudin). Moreover, an inverse relationship had been showed in As exposure between PI3K, Akt, mTOR, and p62 (decrease) and Beclin1, LC3I, LC3II, Atg5, and Agt12 (increase). Besides this, mTOR protein expression levels had been downregulated while Beclin1, LC3, and Atg12 showed a concomitant upregulation in every developmental age points. Histopathological analysis had reported a reduction in the number of pyramidal neurons in iAs-exposed mice hippocampus, besides neurons damage by degenerating axons, remarkable vacuolar degeneration in cytoplasm, karyolysis, and pyknosis in cerebral cortex; furthermore, it was found that mice exposed to high dose As at PND21 and 42 showed autophagosomes and vacuolated axons in the cerebral cortex and hippocampus when ultrastructural analysis was done by transmission electron microscopy. It is important to highlight that severities of changes were higher in cerebral cortex than in the hippocampus of iAs-exposed mice. All in all, iAs induces autophagy by inhibiting PI3K/Akt/mTOR signaling pathway through blood–brain barrier in cerebral cortex and hippocampus since it might be transferred, and this effect occurs in an age-dependent manner since PND21 animals were found to be more vulnerable to the iAs-induced neurotoxicity than the other three age points. 197
Arsenic and hematological disorders
Epidemiology
Epidemiological studies suggested an association between chronic arsenic exposed population and hematologic disorders, such as anemia, leukopenia, thrombocytopenia, aplastic anemia, leukemia, and lymphomas.198–207 Interestingly, in patients of arsenic exposed areas with different blood cancers (acute and chronic myeloid leukemia and lymphoid leukemia, Hodgkin’s and Non-Hodgkin’s lymphomas, multiple myeloma, and myelodysplastic syndrome), the increased presence of chromosomal aberrations is associated with highest iAs concentration in hair and nails, in comparison with unexposed healthy individuals. 205 Possibly, genotoxic capacity of iAs might be associated with hematological tumors incidence in exposed populations.
Similarly, clinical case reports described that arsenic acute intoxication produces several hematological disturbances, such as hemolytic anemia, megaloblastic anemia, karyorrhexis, and binucleated erythrocyte precursors in the bone marrow.208–210
Molecular mechanisms
The hematologic system is a direct target of arsenic toxicity because this metalloid affects bloodstream cellular components through hemolytic and apoptotic effects.
Hemolytic and apoptotic effects of arsenic
It has been described that incubation of human erythrocytes with 10 m
In addition to uncontrolled hemolysis, iAs promotes apoptosis of erythrocytes or eryptosis, increasing intracellular Ca2+ concentration, ceramide formation, phosphatidyl serine (PS) exposure, intracellular ATP depletion, and GSH decrease.212,216 Then, hemolysis and eryptosis might explain arsenic capacity to promote anemia.
Arsenic and immunosuppression
Epidemiology
The coordinated and collective response of immune system is critical to defend organisms against infectious agents and foreign substances capable of causing tissue injury and disease in some situations. 217 However, several environmental factors, such as metal and metalloid compounds exposition have detrimental effects in immune cells. 218 In this sense, epidemiological studies suggested that iAs exposure in human populations is associated with increased risk of several infective diseases, for instance acute respiratory infection, diarrhea, tuberculosis, Hepatitis E virus seroconversion, Hepatitis A, and Hepatitis B seroprevalence,49,50,219–221 suggesting that iAs induces an immunossupresive condition favoring a permissiveness to bacterial and viral infections.
Molecular mechanisms
Mechanistically, it has been described that arsenic significantly impacts both innate and adaptive immune defenses through mechanisms that involve deregulated expression of key immune regulators, apoptosis of immune cells, impaired macrophage function, and lymphocyte activation, affecting innate and humoral immunity. 222
Innate immunity and arsenic exposure
Several in vitro studies showed that iAs induces apoptosis in typical cells of innate immunity such as monocytes-macrophages and neutrophils.223–226 Particularly, iAs incubation in macrophages decreases phagocytosis activity, nitric oxide (NO) production, and promotes F-actin reorganization whereas in neutrophils it activates a caspase-dependent mechanism of cell death.223,224,226 Similarly, ex vivo monocyte-derived macrophages of chronic arsenic exposed individuals exhibit cell rounding, loss of cell adhesion, significant decrease in F-actin and expression of cell adhesion molecule, CD54, reduction of NO production, and phagocytic capacity in comparison with unexposed individuals.
227
This impairment of macrophage functions might be related with increased ROCK activity224,227 (Figure 3). Immunosuppressive effects of arsenic. Inorganic arsenic (iAs) reduces phagocytosis, nitric oxide (NO) production, cell adhesion, differentiation, and increased apoptosis of innate immune cells such as macrophages, neutrophils, and NK cells. In adaptative immune response, iAs reduces proliferation, expression or production of cytokines (IL-2, TNF-α, IFN-γ, IL-4, IL-5, and IL-10), NFAT binding, and increased apoptosis of T-lymphocytes. Similarly, iAs reduces proliferation, differentiation of plasma cells, IgG production, and cathepsin-L activity that promote apoptosis of B-lymphocytes. Innate and immune cells images were obtained from Servier Medical Art.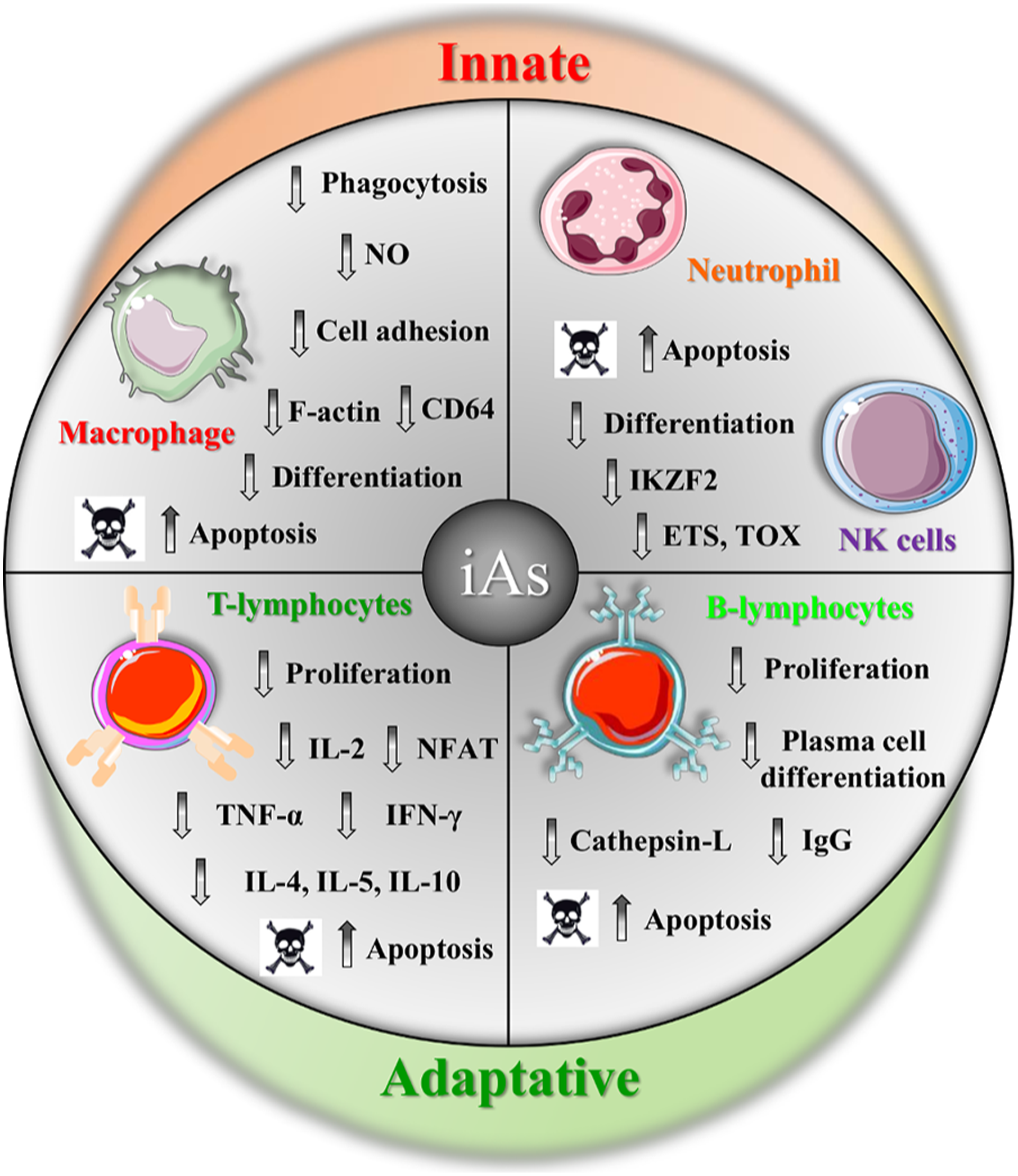
Additionally, iAs disrupts the differentiation of some cellular components of innate immunity. For example, induced marked apoptosis of human blood monocytes during differentiation into macrophages, involving caspase activities and inhibition of NF-κB pathway (Figure 3). 228 As well as, iAs(III) inhibits the CSF-induced in vitro maturation of monocytes into macrophages and induces morphological changes typical of dendritic cells (DCs) and expression of CD14, a monocyte marker. 229 In agreement with this, ATO-treated macrophages exhibit high levels of CD14 and could be differentiated into DC by GM-CSF and IL-4 incubation. 224 This interference of macrophage differentiation is accompanied by downregulation of genes critical for macrophagic functions such as immune response, inflammatory response, antigen processing, and presentation via MHC class I, suggesting that this repression of macrophage differentiation program induced by iAs might have detrimental effects in macrophage activity. 230 In a similar manner, iAs affects differentiation programs of another innate immunity cells, such as NK cells, whose differentiation from CD34 cells exhibit a delay after ATO-treatment, possibly associated with decrease in the expression of transcription factors (IKZF2, ETS1, and TOX) involved in NK cell differentiation and maturation 231 (Figure 3).
Adaptative immunity and arsenic exposure
Several in vitro studies have showed that different arsenic compounds decrease lymphocyte proliferation that could be related with G1 phase arrest, inhibition of IL-2 secretion, and a delay in the kinetics of IL-2 production, a critical growth factor for T-cells.232–236
Human population studies suggested that arsenic exposure might affect cell immunity since early development during pregnancy promoting a reduced thymic size and function, as well as, decreased cell mediated immunity in children, and reduction of T lymphocytes of environmental and occupational iAs-exposed groups.219,237–239 Similarly, individuals exposed chronically to iAs via drinking water (390 μg/L and 412 ± 16 μg/L) showed a lymphocyte proliferation kinetic slower compared with low exposed (60 μg/L) or unexposed individuals (37.2 ± 18 μg/L) that might be related with a delayed progression from S to M phase.240,241 Children exposed to iAs exhibit a decrease in Th cells proportion and CD4+/CD8+ T-cells ratio, and reduced secretion of IL-2 that correlated with high levels of total arsenic in urine. 242 At molecular level, defective IL-2 activation might be explained because T-cells derived of coal-burning arsenic poisoned population showed a decrease of intracellular calcium levels, reduced levels of PKCθ and p-PKCθ and reduced DNA binding of NFAT, an important transcription factor in active T-cells that regulates IL-2 expression 243 (Figure 3).
Another cytokines with reduced secreted levels in iAs-exposed populations, including TNF-α, IFN-γ, IL-10, IL-5, and IL-4238,244 (Figure 3). Consistently, transcriptome and methylome analysis in CD4+ T-cells of high-arsenic exposure individuals (∼300 μg/L in urine) showed a downregulation of cytokine genes (TNF, IFNG, IL10, and IL12 A), several genes related to MHC class 2 (HLA-DRA, HLA-DQA2, and HLA-DPB1), and NF-κB pathway (REL, RELA, and RELB), as well as hypermethylation of genes of interleukins, members of the interferon regulatory factor (IRF), and the cluster of differentiation (CD) families. 245 Possibly, arsenic causes immunosuppression in T-cells through modifications in whole gene expression patterns that impaired its normal functions.
Another immunosuppressive mechanism includes arsenic related immunotoxicity in lymphocytes. 225 Specifically, sodium arsenite promotes extrinsic apoptosis with high levels of TNFR1, TRADD, FADD, and PARP cleavage in T helper cells; whereas arsenic trioxide induces intrinsic apoptosis in both CD4+ and CD8+ T-cells promoting oxidative stress, depolarization of mitochondrial membrane potential, cytochrome c release, and differential regulation of Bcl-2 and Bax protein levels246,247 (Figure 3).
Arsenic has immunosuppressive effects in humoral immunity, in murine models, sodium arsenite exposure suppresses B cell proliferation increasing parasitic susceptibility, inhibits IgM and IgG-producing cells in mouse splenocytes, decreases cathepsin L activity, and induces apoptosis of murine antigen-presenting B cell line (TA3).248–250 Similarly, in vitro studies showed that arsenic promotes apoptosis in human B lymphocytes, plasma cells, and inhibits plasma cell differentiation.225,251 Finally, in subjects chronically exposed to drinking-water contaminated with iAs, has been detected high levels of serum IgG and IgE related with arsenical skin lesions and a reduced production of diphteria and mumps-specific IgG antibodies during pregnancy and childhood, respectively252–254 (Figure 3).
In summary, several studies support that arsenic has deleterious effects in normal functions of cellular components of innate and humoral immunity, so is necessary analyze with more detail how immunosuppressive effects of arsenic in populations exposed might contribute to prevalence of another infective and chronic diseases.
Cardiovascular disorders
Epidemiology
Several epidemiological studies have showed the link between chronic arsenic ingestion and various cardiovascular disorders, including peripheral vascular disease, ischemic heart disease, aterosclerosis, 255 and hypertension.256,257
A study evaluated the association of mean arsenic concentrations (<1 and 118 μg/L) in drinking water with cardiovascular mortality in the Spanish population. Compared with the total Spanish population, cardiovascular mortality rates increased in municipalities with arsenic drinking water concentrations >10 μg/L. 258 A case–control study conducted in a hospital in China showed that exposures to arsenic concentrations in the 10–40 range carry an increased risk of cardiovascular disease. 259 A retrospective analysis in the US population showed that low arsenic exposure (2.6 μg/L in household water) could increase mortality from ischemic heart disease among long-term smokers, suggesting a synergistic relationship between exposure to arsenic and smoking due to ischemic heart disease. 260 Previously, a study showed an elevated risk of carotid atherosclerosis associated with a higher arsenic concentration (> 50 μg/L) in Taiwanese subjects carrying the purine nucleoside phosphorylase (PNP-AT) haplotype and at least the arsenic-3-methyltransferase (As3MT) risk polymorphism or the glutathione S-transferaseomega-1 (GSTO-1) risk haplotypes (OR, 6.43; 95% CI, 1.79–23.19).255The impact of chronic exposure to arsenic is evidenced in a retrospective study where they observed an association between mortality from cardiovascular diseases in a population exposed to well water contaminated with arsenic (up to > 300 μg/L) for 10–20 years in Mongolia, China. 261
A recent cross-sectional study conducted in selected patients from different Bangladesh areas who underwent open-heart surgery showed higher iAs concentrations in cardiac tissue, reflecting greater histological injury. They also showed a significant correlation between the accumulation of iAs and As3MT, nitric oxide synthase (NOS3), intercellular adhesion molecule-1 (ICAM1), and superoxide dismutase (SOD2) genotypes. 256 A retrospective study in a Chilean population found for the medium exposure categories (60–623 μg/L) and higher (>623 μg/L), a hypertension OR of 1.49 (95% CI: 1.09; 2.05) and 1.65 (95% CI 1.18, 2.32), respectively. Similar results were observed when analyzing cumulative lifetime exposure. 257
Molecular mechanisms
Arsenite at concentrations of 5 μ
Also, has been described that iAs is able to induce new blood vessels formation or angiogenesis. In vitro studies, showed that low doses of arsenite in human microvascular endothelial cells (HMVECs) induced heme oxygenase (HO-1) and VEGF gene expression and increased protein levels, favoring cell migration and tube formation. 269 To transcriptional level, arsenite promotes HO-1 expression, inducing Bach1 repressor dissociation from the enhancers of HO-1 gene and increasing Nrf2 activator binding to these elements. 269 In a similar manner, in human umbilical vein endothelial cells (HUVECs), vascular smooth muscle cells (SMC), and ovarian cancer cells, iAs(III) promotes VEGF expression consistent with its pro-angiogenic capacity.270–272 iAs(III) also promotes HIF-1α protein accumulation, increased DAG levels, and PKCδ activation, suggesting a role of this signaling pathways in the induction of VEGF expression.271,272
Consistently, in vivo models showed that trivalent arsenic increases significantly the blood vessel density in a chicken chorioallantoic-membrane (CAM) assay and mouse matrigel model. 273 Furthermore, lower dose of iAs(III) increases the size, number of blood vessels, and lung metastasis of tumors induced by B16-F10 mouse melanoma cells inoculation in nude mice.273,274 In blood vessels of isolated tumor melanoma cells, HIF-1α and VEGF levels were increased, suggesting that these two proteins might be involved in the in vivo angiogenic effect of chronic low-dose of iAs. 274 Altogether, this information suggests that arsenic might promote angiogenesis in vivo, possibly cooperating with other factors promoting cardiovascular alterations.
Skin lesions
Epidemiology
Prolonged arsenic ingestion with exposures 1–2 orders of magnitude above the current WHO’s guidelines of 10 μg/L 275 has been correlated with skin damage, being documented in several studies in humans populations.276–279 Humans are at risks to develop keratosis (diffuse or blotchy lesion characterized by raised, rough, hard skin seen on the palm or back of the hands and/or the sole of the foot), hyperpigmentation (often with hypopigmented spots) of chest, neck, and trunk, dermatological problems as melanosis (diffuse or blotchy lesion characterized by dark pigmentation on the face, oral cavity, neck, upper and lower limbs, chest, or back), and leukomelanosis (depigmentation characterized by black and white spots present anywhere on the body), even gangrene can be generated by consuming high iAs concentration through drinking water. 4 Also, hyperhidrosis and hyperkeratosis (extensively thickened keratosis presented on the palm or dorsum of the hands and/or the sole or plantar of the foot), as well as melanoderma and vascular disturbances such as dark abrasions on the feet (black foot disease) are skin manifestations or signs by chronic iAs exposure.280–283
A follow-up study of 500 people with arsenic skin lesions carried out in Bangladesh in 2001–2003 showed that a reduction of 41% in the concentration of arsenic in drinking water was associated with a 22% improvement in skin lesions, where 11.8% of the individuals who had lesions at the beginning of the study, these were not detectable at the end of follow-up, suggesting a full recovery. Therefore, the presence of premalignant skin lesions such as melanosis, leukomelanosis, keratosis, and hyperkeratosis can be reversed with the reduction of exposure to arsenic, this being a public health priority in endemic areas for this metalloid. 284
Molecular mechanisms
Hyperkeratosis on the palms or soles are non-malignant lesions typical of iAs exposure, which progress to malignant lesions, including basal cell carcinoma and squamous cell carcinoma. In vitro studies carried out by Al-Eryani et al. 285 in isolated keratinocytes from individuals chronically exposed to 59 to 172 ppb of iAs in drinking water, to evaluate miRNA expression, concluding that miR-433 and miR-425-5p expression are associated with the malignancy of skin lesions, while miR-576-3p and miR-184 induction were associated with the stage of metastasis of malignant cells. Also, Weinmuellner et al. 286 proposed a 3D model from immortalized human keratinocytes (NHEK/SVTERT3-5) to evaluate iAs-induced skin disorders, where by exposures of ATO (0.05–0.25 μM) for 6 months, generated an increase in cells in the G2 phase of the cell cycle, a larger size of the nucleus and alterations in tubulin, observing in the cells a dedifferentiation and regulation of various epithelial to mesenchymal transition markers, generating squamous cell carcinoma in situ (Bowen’s disease). In vitro studies carried out in immortalized human keratinocytes (HaCaT) exposed for 18 weeks to 0.05 ppm of sodium arsenite showed a higher growth rate in the G2/M and S phases of the cell cycle, associated with a significant increase in the colony formation efficiency (CFE) and the expression of CD44v6, coupled with a decrease in the expression of the activating genes of the transcription factor NF-κB and p53. These results suggest that arsenic promotes malignant transformation in human skin lesions through the expression of CD44v6 stimulated by the NF-κB signaling pathway. 287 On the other hand, mice chronically exposed to 0.5 and 5 ppm of arsenic through drinking water for 6 months showed an increase in the expression of the p62 protein in the epidermis, which induces the levels of Nrf2 that is associated with a greater proliferation in mouse epidermis and less activity of p21. These results suggest that modulation of p62 protein may help to prevent arsenic-induced skin lesions. 288
The HEALS conducted in adult population of Araihazar, Bangladesh, exposed to iAs in drinking water, showed that the risk of skin lesions from this metalloid is independent of age, gender, and arsenic exposure, being caused by the presence of genomic deletions in copy number of APBA2, GALNTL5, GOLGA6L7P, OR5J2, PHKG1P2, SGCZ, VN1R31P, and ZNF658 genes. 289
Also, studies have found an association between aberrant DNA methylation and iAs-induced skin lesions. Recent studies suggest alterations in DNA methylation patterns throughout the genome due to iAs exposure by studying three generations (grandparents, parents, and grandchildren) in nine control families, and 18 families exposed at different stages of their life: adulthood, childhood, and in utero. Exposure across generations shared common methylated DNA loci and regions: 744 differentially methylated loci (DML) 417 were hypermethylated and 330 hypomethylated, as well as 15 differentially methylated genomic regions (DMR) despite the distinctive exposure time in each generation. In addition, a series of DMLs and 25 affected molecular pathways (neuropathic pain signaling in neurons, the arsenate I detoxification pathways, and chondroitin and dermatan biosynthesis) were detected that were significantly distinguished between controls, exposed populations, and patients with skin lesions. The authors suggest that some of these DMLs altered by iAs-exposure can be inherited, also affecting later generations; therefore, they could serve as biomarkers to identify people at risk of suffering skin lesions due to chronic iAs-exposure. 290 This interindividual susceptibility could be explained by differential arsenic methylation capacity,276,291,292 variation in genome as a single nucleotide polymorphism, haplotypes or telomere length,293–295 and individual epigenetic pattern (DNA methylation, histone modification, and miRNA expression) in population chronically exposed to iAs. 296
Conclusion
Worldwide an estimated 200 million people are exposed to iAs in drinking water above the WHO recommended guideline of 10 μg/L, causing different effects on human health. Chronic arsenic exposure activates different molecular mechanisms such as oxidative stress, inflammation, and cytotoxicity that affect structures and functions of different organs and systems producing several non-carcinogenic disorders. The serious and diverse health effects produced by chronic arsenic exposure in several populations highlight the need to implement urgent preventive polices in order to reduce the iAs exposure to the levels recommend by WHO, mainly in developing countries.
Footnotes
Acknowledgments
MMC acknowledged their membership of the National System of Researchers (SNI-CONACYT).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Secretaría de Investigación y Posgrado of the IPN, Mexico, for grants that supported this work (20200717).
