Abstract
Thirteen female Wistar rats were divided into two groups: one treated with ethanol and the other of untreated. Four newborns from each mother were selected and weighed, measured, and evaluated for physical characteristics. From these neonates, chondrocytes were extracted from the articular cartilages of the femur and tibia, and cultivated in a chondrogenic medium at 37oC and 5% CO2. At 7, 14, and 21 days of cultivation, alkaline phosphatase activity tests, MTT conversion to formazan, and percentage area covered by cells per field were performed. At 21 days, the percentage of PAS+ areas in 3D cultures was performed, as well as the evaluation of gene transcript expression for aggrecan, SOX-9, collagen type II, collagen X, Runx-2, and VEGF by real-time RT-PCR. The means were compared by Student’s t-test. The weight of the ethanol group neonates was significantly lower than that of the controls. Chondrocyte cultures from the ethanol group showed significantly higher AP activity, MTT conversion, and cell percentage. There was higher expression of collagen type II and lower expression of SOX-9 in the ethanol group. There was no difference in the percentage of PAS+ areas in pellets and in expression of aggrecan, collagen X, Runx-2, or VEGF between groups. In conclusion, prenatal exposure to ethanol alters the phenotype and activity of offspring chondrocytes, which may be mechanisms by which endochondral bone formation is compromised by maternal ethanol consumption.
Introduction
The skeletal formation process is complex and dependent on genetic, hormonal, and nutritional factors. 1 Endochondral ossification is the process of prenatal bone formation of most bones in the skeleton, occurring from a cartilaginous mold. 2 The hypertrophic chondrocytes at the center of this mold induce vascular invasion through the synthesis of growth factors, including the vascular endothelial growth factor (VEGF).3,4 This event occurs in association with differentiation of mesenchymal stem cells (MSCs) from the perichondrium into osteoblasts, which secrete bone matrix over the cartilaginous mold, giving rise to ossification centers during the prenatal period. 5 This process of endochondral ossification is also directly dependent on the expression of transcription factors such as Runx2 and SOX-9.3,6 Endochondral bone formation can be altered by several factors or substances to which the mother is exposed during pregnancy. Among them, ethanol is notable for its harmful effects on the fetus.7–9 Ethanol, ingested by pregnant women through the consumption of alcoholic beverages, crosses the placental barrier and reaches fetal circulation.10,11 Studies have already shown that prenatal exposure to ethanol (PEE) is responsible for skeletal changes 12 such as low fetal growth and reduced cranial perimeter.13,14
However, studies that evaluate the effect of PEE on human progeny are limited by the difficulty in stating that the changes observed in the progeny are exclusively due to ethanol since its association with environmental factors and the use of other drugs is frequent. As a result, and for ethical reasons, murine models have been widely used to clarify the damage caused by consumption of ethanol during the prenatal period.15–18
In a murine model, PEE caused a delay in endochondral ossification caused by the discontinuity of the chondrocyte maturation process with consequent suppression of osteoclastogenesis, which is fundamental for the process of endochondral bone formation. 19 In another study, it was found that ethanol affects bone development in vivo in different ways depending on the bone site, with the ulna, radius, tibia, and sacrum of rats being more sensitive to the effect of ethanol. 1
Contrary to the hypothesis that endochondral formation in vivo may be affected by inhibition of stem cell differentiation into chondrocytes, some studies have shown the opposite since ethanol has increased in vitro chondrogenic differentiation of embryonic stem cells.20,21
However, despite the evidence that ethanol alters endochondral formation in vivo, this appears to be the first study that evaluates the phenotype and synthesis activity of joint chondrocytes in vitro extracted from neonatal rats with prenatal exposure to maternal consumption of ethanol. This study was conducted in order to understand the pathogenesis of changes in endochondral bone formation caused by maternal consumption of ethanol.
Material and methods
The experiment was performed at the Núcleo de Células Tronco e Terapia Celular Animal and in the histopathology laboratory, both at the Escola de Veterinária da Universidade Federal de Minas Gerais (UFMG). All experimental procedures were approved by the animal use ethics committee of the Universidade Federal de Minas Gerais (CEUA/UFMG) under protocol No. 119/16.
Experimental design
Thirteen two-month-old Wistar rats were housed in plastic boxes and exposed to a 12-h light and 12-h dark regime receiving commercial feed and water ad libitum. After adaptation, vaginal cytology was performed in all rats to identify the estrous cycle phase. 22 Rats that were in the proestrus or estrus phase were placed with males during the dark period.
After confirmation of pregnancy, rats were separated into individual boxes constituting two experimental groups: (1) the control group (n = 6) and (2) the ethanol group (n = 7). Group 2 rats, from the ninth day of gestation, received 40% ethanol solution by daily gavage (12.5 ml/kg totaling 4 g of ethanol/kg) 19 2 h after the beginning of the light cycle. The control group received distilled water as a placebo.
All animals were weighed on days 0, 9, and 18 of gestation to readjust the volume of ethanol administered. The rats were evaluated for any behavioral changes, such as excitability, aggressiveness, or excessive lethargy during the entire experimental period. Daily feed consumption per female and weight gains were recorded throughout the experimental period. On the day of birth, all neonates were evaluated to identify any physical deformities or changes in behavior. Four neonates from each mother were selected at random and euthanized by overdose with thiopental (100 mg/kg) intraperitoneally, measured by the crown–rump–length method (frontal bone to the sacrum) and weighed. In addition, gestation time and litter size were analyzed.
Daily ration consumption
For 6 weeks, beginning on day zero of gestation, rats received 500 g of freely available feed. Every 7 days, leftovers were weighed to establish an average of the amount consumed per day, in grams per animal and per group in total. The mean and standard deviation were determined for each group.
Corporal weight gain
On days 0, 9, and 18 of gestation, rats were weighed to establish weight gain in grams for each rat per group. The mean and standard deviation were determined for each group.
Articular chondrocyte extraction and culture
Antisepsis of the skin of the newborns was performed with a solution of iodopolvidone and iodized alcohol to extract the pelvic limbs. The articular cartilages of the femur heads and the proximal ends of the tibiae were extracted.
Cartilage samples were washed with 0.15 M phosphate-buffered saline solution (PBS) and then fragmented and placed in low-glucose DMEM medium with gentamicin (60 mg/L), penicillin (100 UI/ml), streptomycin (100 mg/ml), amphotericin B (25 mg/L), 5% fetal bovine serum (SFB Somali, Brazil), and collagenase type I (1.5 mg/ml; Sigma-Aldrich, St. Louis, MO, USA). Cartilage fragments were incubated in an oven at 5% CO2 and 37°C for 12 h. Following this, the material was centrifuged for 10 min at 2000 g, washed with 0.15 M PBS, and centrifuged again for 10 min at 2000 g. Pellets were resuspended in chondrogenic medium containing low-glucose DMEM enriched with gentamicin (60 mg/L), penicillin (100 IU/ml), streptomycin (100 mg/ml), amphotericin B (25 mg/L), 1% fetal bovine serum, ascorbic acid (50 μg/ml), dexamethasone (10–17 M), 6.25 μg/ml insulin, 6.25 μg/ml transferrin, 1 mM pyruvate, and 1.25 μg/ml BSA, and then transferred to T-25 flasks and grown at 37°C and 5% CO2. At 80–90% confluence, cells were harvested and transferred to T-75 flasks and again harvested until the third passage. Change of medium was carried out twice a week.
At 7, 14, and 21 days of monolayer culture (2D) in chondrogenic medium, tests were performed to measure conversion of MTT into formazan crystals, alkaline phosphatase (AP) activity, and percentage of cells per field. At 21 days, the percentage of PAS+ areas in 3D culture (pellets) were measured, and quantification of gene transcripts for aggrecan, SOX-9, type II collagen, collagen X, Runx-2, and VEGF by real-time RT-PCR was performed.
Conversion test of MTT to formazan crystals
Cells were transferred to 24-well culture plates at a concentration of 2 × 104 cells per well. The conversion test of MTT [3- (4,5-dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide] into formazan crystals was performed at 7, 14, and 21 days of culture in chondrogenic medium. Further, 210 μl of chondrogenic medium and 170 μl of MTT (Invitrogen, Carlsbad, CA, USA) (5 mg/mL) were incubated at 37°C and 5% CO2 for 2 h, which allowed the formation of formazan crystals to be observed under an optical microscope. Then, 210 μl of SDS-10% HCl was added and incubated again at 37°C and 5% CO2 for 12 h. Then, 100 μl from each well was transferred to 96-well plates and read on a spectrophotometer at a wavelength of 595 nm. The mean and standard deviation were obtained from the results of six wells each for the ethanol group and the control group.
Alkaline phosphatase activity test by BCIP/NBT method
Cells were transferred to 24-well plates at a concentration of 2 × 104 cells per well. After 7, 14, and 21 days of culture, culture media were removed, and each well was washed with sterile 0.15 M PBS solution and 200 μl of BCIP/NBT solution (Invitrogen, Carlsbad, CA, USA).
Plates were incubated at 37°C and 5% CO2 for 2 h. After this period, 210 μl of SDS-10% HCl was added and incubated again for 12 h. Further, 100 μl was transferred from each well to 96-well plates for reading on a spectrophotometer at 595 nm wavelength. The mean and standard deviation were obtained from the results of six wells each for the ethanol group and the control group.
Percentage of area covered by cells in monolayer (2D culture)
Cells were transferred to 24-well plates containing 13-mm diameter plastic coverslips at a concentration of 2 × 104 cells per well. After 7, 14, and 21 days of culture, the coverslips were washed twice with 0.15-M PBS solution and fixed with 4% paraformaldehyde solution for 24 h. Subsequently, they were washed with distilled water and stained with periodic acid Schiff (PAS) staining.
The percentage of area covered by cells was determined in eight fields under the 10× objective, using the 121-point graticule. The mean and standard deviation were obtained from the results of six wells each for the ethanol group and the control group.
Percentage of PAS+ areas in 3D culture
The cells were transferred to 15-mL Falcon tubes, at a concentration of 1.5 × 106 cells per tube, and centrifuged at 2000 g for 10 min. Cells were grown at 37°C and 5% CO2 for 21 days. After this period, the medium was removed, and the pellets were washed with 0.15 M PBS solution twice and then fixed in 4% paraformaldehyde for 24 h.
After this period, the pellets were dehydrated in ethanol and imbedded in paraffin. Pellets were cut with a microtome into sections 4-µm thick and stained by PAS. The PAS+ areas were determined using an optical microscope and a 121-point graticule, over two fields, viewed with a 40× objective. The mean and standard deviation were obtained from the results of six wells each for the ethanol group and the control group.
Gene transcript expression for aggrecan, SOX-9, collagen type II, collagen X, Runx-2, and VEGF by real-time RT-PCR
Cells were transferred to T-25 flasks at a concentration of 2.5 × 105 cells per flask and kept in chondrogenic medium for a period of 21 days. After this period, cells were harvested, and q-RT-PCR was performed.
Primers of gene transcripts delineated based on mRNA sequence Rattus norvegicus.

Statistical analysis
The design was carried out entirely at random, and the mean and standard deviation were determined for each variable. Comparisons of averages were performed using the Student t-test using the GraphpadInstat 3.05 program (GraphPad Software Inc., San Diego, USA). Differences were considered significant if p < .05.
Results
Daily feed intake and corporal weight gain
The rats of the ethanol group did not show any behavioral changes when compared to the control group. The group of rats that received ethanol showed significantly lower daily consumption of food than the control group. However, there was no significant difference in the animals’ weight gain. In addition, gestation time and litter size did not differ significantly between groups (data not shown).
Weight and measurements of neonates
Mean and standard deviation of weight (g) and length (cm) of rat neonates born to untreated rats (control group) and to rats that consumed ethanol during pregnancy (ethanol group) from the ninth day of gestation.

Means followed by different letters differ (p ≤ .05).
Conversion of MTT to formazan crystals and alkaline phosphatase activity
Chondrocytes from the ethanol group showed greater conversion of MTT to formazan crystals than those of the control group at 7, 14, and 21 days of culture (Figure 1(a)). The same result was observed in the activity of AP: the ethanol group showed greater activity than the control group at 7, 14, and 21 days of culture (Figure 1(b)). Chondrocyte cultures of neonates, born to untreated rats (control group) and to rats that consumed ethanol during pregnancy (ethanol group), at 7, 14, and 21 days of culture in chondrogenic medium. (a) Mean and standard deviation of MTT reduction in formazan crystals. (b) Average and standard deviation of alkaline phosphatase activity using the BCIP/NBT method. * p < .05.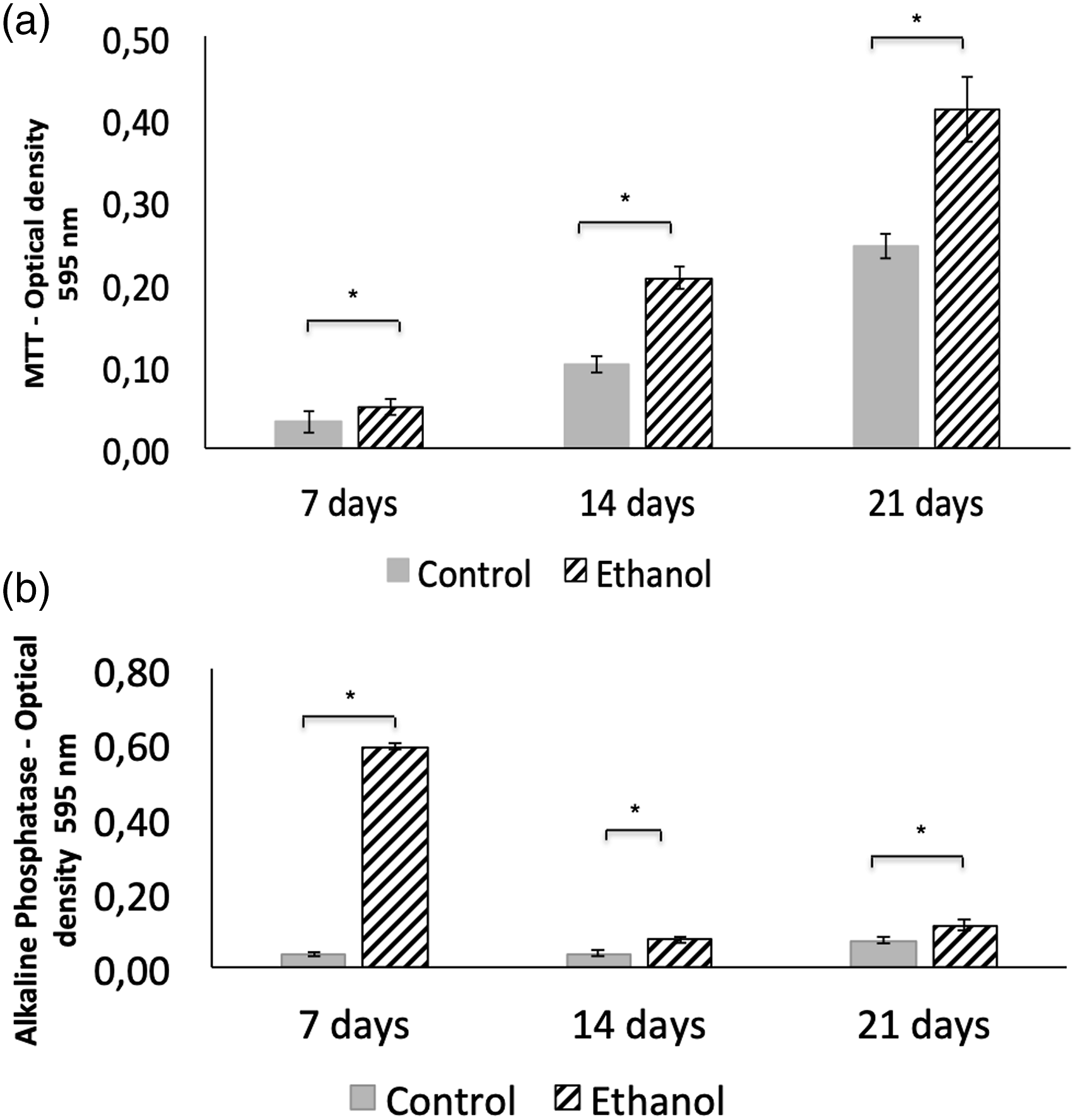
Percentages of areas covered by cells and PAS+ areas
The cultures of the ethanol group in monolayer showed a significantly higher percentage of area covered by cells compared to the culture of the control group at all experimental timepoints (Figure 2). In the 3D culture, there was no significant difference between groups in the percentage of PAS+ areas (Figure 3). (a) Photomicroscopy of the percentage area covered by cells in monolayer (2D) chondrocyte cultures from neonates, born to untreated rats (control group) and to rats that consumed ethanol during gestation (ethanol group), at 7, 14, and 21 days of culture in chondrogenic medium. (b) Average and standard deviation of the percentage area covered by cells of the control and ethanol groups. * p < .05. (a) Photomicroscopy of the percentage of PAS+ area of 3D chondrocyte cultures from neonates, born to untreated rats (control group) and to rats that consumed ethanol during pregnancy (ethanol group), at 21 days of culture in chondrogenic medium. (b) Average and standard deviation of the percentage of PAS+ area in the control and ethanol groups. *p > .05.

Expression of aggrecan, SOX-9, collagen type II, collagen X, Runx-2, and VEGF.
There was no statistical difference between the expression of aggrecan, Runx-2, collagen X, or VEGF gene transcripts from the ethanol group when compared to those of the control group. However, there was greater expression of the type II collagen gene transcript in the ethanol group, along with less SOX-9 expression (Figure 4). Relative quantification of gene expression (mean ± standard deviation) in chondrocyte cultures from neonates, born to untreated rats (control group) and to rats that consumed ethanol during pregnancy (ethanol group), at 21 days of culture in chondrogenic medium. * p < .05.
Discussion
The present study demonstrates that ethanol consumption during pregnancy alters the phenotype and activity of offspring chondrocytes, characterized by higher AP activity, cellularity, and collagen type II expression, and lower SOX-9 expression, which may be mechanisms by which endochondral bone formation is compromised by maternal ethanol consumption.
The effects of ethanol are considered dose-dependent 23–25; that is, they can vary according to the volume consumed daily, with consumption classified as discrete, moderate, or intense. 26 In the present study, rats in the ethanol group received a daily dose that would correspond to approximately 15–30 g of ethanol per day in humans, classified as moderate-to-intense consumption. 26 The oral via was selected to mimic human consumption, and administration was performed via gavage to control the amount of alcohol consumed by each animal. The dose and the period of initiation of ethanol treatment in our study were determined based on the results of a previous study. 19 This study demonstrated that a dose of 4 g/kg of ethanol and treatment initiation on the ninth day of pregnancy were effective in promoting the toxic effects of ethanol on the offspring, leading to the emergence of fetal alcohol syndrome. 19 It is believed that the date chosen for the beginning of ethanol supply to mothers is compatible with the beginning of the organogenesis period in rats, which is around the 8th day of gestation. 27
Ethanol consumption did not change litter size or weight gain during pregnancy. These results are in contrast to another study that demonstrated a reduction in weight gain in rats consuming ethanol from the first to the last day of gestation, with half the alcoholic concentration used in the present experiment, as well as a reduction in litter size. 28 In the present study, although there was no significant difference in the size of the pups, neonates in the ethanol group were significantly lighter than those of the control group. It is believed that the reduced weight of neonates in the ethanol group may be associated with a lower bone mass at birth. This inference is supported by an earlier study using a similar methodology, in which fetuses at 20 days of gestation underwent PEE that produced reduction of the femoral length and dramatic reduction of the bone matrix due to significant accumulation of chondrocytes in the hypertrophic zone of growth cartilage. 19
The effects of maternal ethanol consumption on chondrocyte histomorphometry of offspring growth cartilages have been studied.1,11,19 15-day-old rats, offspring of mothers who received a liquid diet with 36% alcoholic content from 30 days before pregnancy until the end of lactation, showed a significant reduction in the size of the growth plate due to a significant reduction in hypertrophic chondrocytes. 29 Another study demonstrated that 21-day-old rats subjected to PEE showed an intense reduction in the resting zone and abnormal accumulation of chondrocytes in the hypertrophic zone. 11 The heterogeneity of results is probably due to methodological differences regarding the concentrations of the alcoholic solution administered to the mother, as well as the period of administration and the age of the studied offspring. The results of these studies were based on histological analysis of growth cartilage, unlike the present study, in which the chondrocytes of each group were extracted and cultured to be evaluated for phenotype and synthesis activity in an attempt to clarify some of the mechanisms by which these changes occur in vivo.
Chondrocytes from the ethanol group showed a higher conversion of MTT into formazan crystals at all timepoints evaluated, and the same was true of AP activity and percentage of area covered by cells. Both the MTT assay and AP activity measured using the BCIP/NBT method are colorimetric tests influenced by the number of cells in the culture. Despite the initial number of cells in each culture being the same between groups, it was shown that the culture of chondrocytes in the ethanol group showed greater cell coverage than the control group at all timepoints, suggesting greater proliferative activity or reduced apoptosis of the chondrocytes, thus justifying the highest MTT and AP activity values.
Despite the greater cellularity of the culture observed in the ethanol group, there was no significant difference between groups in the percentage of PAS+ areas that reflect the proteoglycan content of the matrix. It is known that proliferating chondrocytes are responsible for the synthesis of the cartilaginous matrix rich in proteoglycan and type II collagen.5,30 As the chondrocytes undergo hypertrophy, the synthesis of proteoglycans and type II collagen decreases, resulting in the synthesis of type X collagen. 5 Expression of similar levels of the aggrecan transcript between the groups may explain the similar proteoglycan content shown by the PAS+ areas since aggrecan is the main proteoglycan present in the cartilage. Despite this, the relative expression of type II collagen was significantly higher in the ethanol group. The cartilaginous matrix is composed mainly of collagen fibrils associated with proteoglycan macromolecules that bind to sulfated glycosaminoglycan molecules. The synthesis of type II collagen is more pronounced in the early stages of endochondral bone formation, and this collagen is responsible for providing cartilage with resistance. 31 As these chondrocytes mature and hypertrophy, the synthesis of type II collagen is reduced and replaced by collagen type X.5,32
In vitro studies demonstrate that mesenchymal cells, derived from facial primordia in chicken embryos at 21 days, show greater chondrogenic differentiation when cultured with ethanol by increasing the expression of aggrecan and type II collagen. 21 In vitro, the addition of ethanol in chondrocyte cultures of neonatal rats drove alteration of the phenotype and synthesis activity due to the reduction of AP activity, reduction of PAS+ matrix, and decrease of aggrecan expression; these effects were dose-dependent. 33 One possible explanation for the difference between the results is that methodological differences existed, since the ethanol added directly to the cell culture excludes likely alterations resulting from the metabolic processing of the substance within the organism. No other study has evaluated the effects of maternal ethanol consumption on the phenotype and in vitro synthesis activity of joint chondrocytes from offspring at the time of the present study.
Although Runx-2 and VEGF are important in the process of endochondral bone formation,34–36 prenatal exposure to ethanol did not alter the expression of their transcripts. However, the ethanol group showed less expression of SOX-9 when compared to the control group. SOX-9 is a fundamental gene transcript in the initial stages related to chondrocyte differentiation and maturation, initiating the process of hypertrophy.5,37 However, as well-differentiated chondrocytes undergo hypertrophy, SOX-9 expression decreases.38–40 Reduction of SOX-9 expression is essential for hypertrophy of chondrocytes, as well as the subsequent expression of VEGF. 3 By correlating the results of the lowest expression of the SOX-9 transcript with the highest expression of type II collagen, we can assume that prenatal exposure to ethanol leads to chondrocyte differentiation and proliferation without, however, reaching the peak of hypertrophic chondrocyte maturity. This hypothesis is corroborated by another study that demonstrated a reduction in the size of the growth plate due to significant reduction in hypertrophic chondrocytes in rats born to mothers who received a liquid diet with 36% alcoholic content. 29
Future studies on the effect of prenatal exposure to ethanol on the proliferative activity of the chondrocytes are pertinent and would help understand the increase in cellularity observed in the ethanol group. In addition, evaluating the expression of the same chondrogenic factors used in the present study on the growth cartilage of neonates would make it possible to compare the in vitro and in vivo results.
Conclusion
Thus, we conclude that prenatal exposure to ethanol alters the phenotype and activity of offspring chondrocytes, characterized by higher AP activity, cellularity, and collagen type II expression and lower SOX-9 expression (Figure 5), which may be mechanisms by which endochondral bone formation is compromised by maternal ethanol consumption. Graphical abstract showing the effect of prenatal exposure to ethanol on the phenotype and synthesis activity of newborns’ chondrocytes.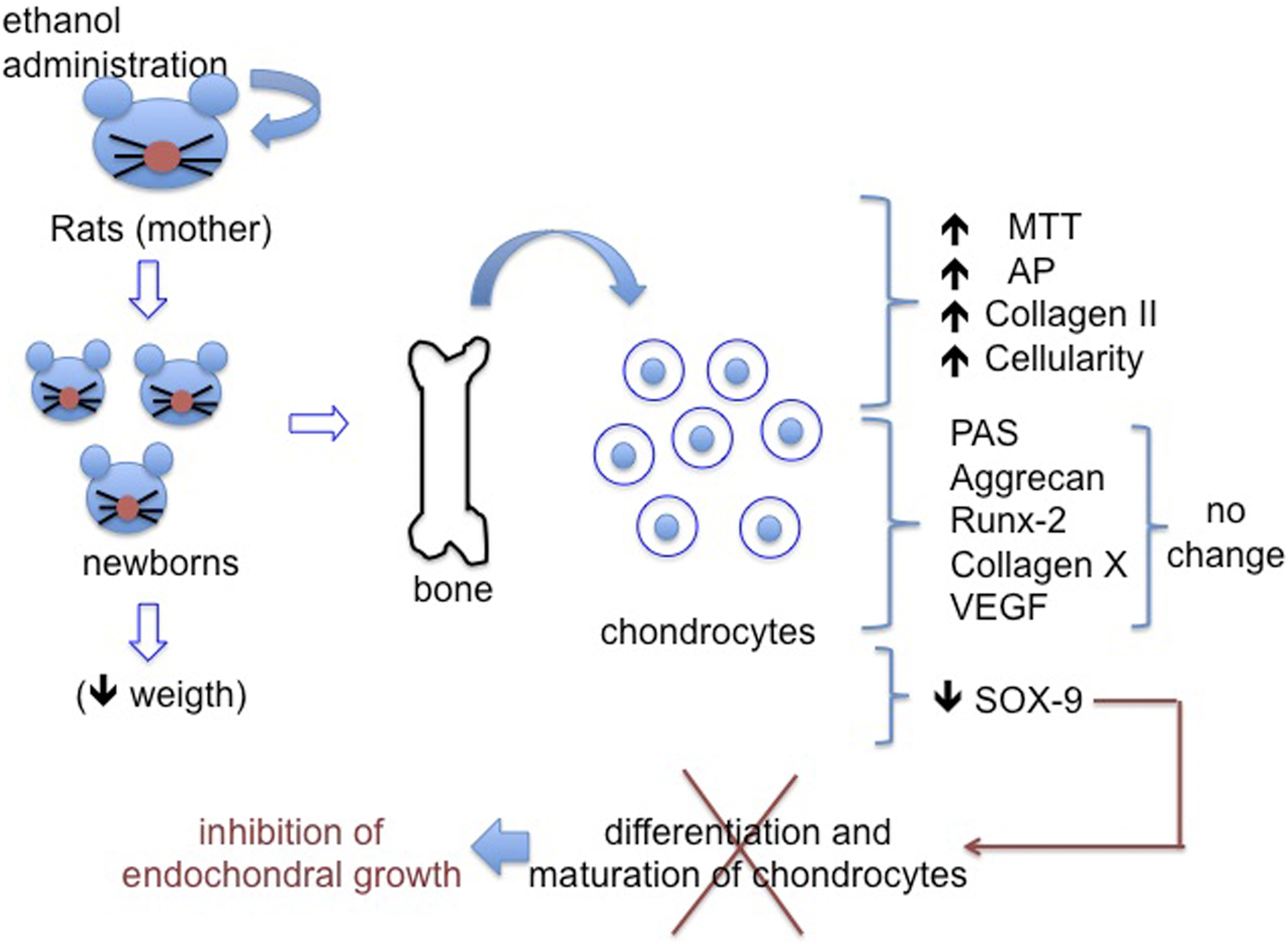
Footnotes
Declaration of conflicting interests
The authors declare that they have no conflicts of interest with respect to their authorship or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Fundação de Amparo à Pesquisa de Minas Gerais (Fapemig), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
