Abstract
Introduction:
Increasing evidence indicated that miR-2682-5p acted as a tumor suppressor in various cancers. The current study aimed to investigate the biological function of exosomal miR-2682-5p in non-small cell lung cancer (NSCLC).
Methods:
The expression of miR-2682-5p in NSCLC tissues and adjacent non-tumor tissues, NSCLC cell lines and human embryonic lung fibroblast, as well as serum and serum exosomes of NSCLC patients and healthy donors was detected by RT-qPCR. The effects of miR-2682-5p on the viability, migration, and apoptosis of NSCLC cells were detected by CCK-8, Transwell, and flow cytometry assays. Dual-luciferase reporter gene and RNA immunoprecipitation assays were used to evalutate the relationship between miR-2682-5p and HDAC1.
Results:
Low expressed miR-2682-5p was found in tumor tissues, cell lines, serum, and serum exosomes of NSCLC patients. MiR-2682-5p overexpression suppressed NSCLC cell viability and migration and promoted apoptosis, while miR-2682-5p knockdown showed the opposite results. Furthermore, exosomes from healthy donor serum inhibited NSCLC cell viability and migration and promoted apoptosis. Dual-luciferase reporter gene and RNA immunoprecipitation assays verified that HDAC1 was a target of miR-2682-5p. HDAC1 overexpression abolished the effects of miR-2682-5p mimic on NSCLC cell viability, migration, and apoptosis. Chromatin immunoprecipitation assay indicated that HDAC1 bound to the promoter region of ADH1A. Upregulation of ADH1A counteracted the effects of HDAC1 overexpression on NSCLC cell viability, migration, and apoptosis.
Conclusion:
Taken together, exosomal miR-2682-5p inhibited NSCLC cell viability and migration and promoted apoptosis by the HDAC1/ADH1A axis, and this result might provide a novel therapeutic target for NSCLC.
Introduction
Lung cancer is a malignant tumor with the most rapidly increasing morbidity and mortality. 1 Non-small cell lung cancer (NSCLC) accounts for about 80% of all lung cancers, and about 75% of patients are found at the middle and advanced stages.2,3 Although many therapy strategies show responses during early treatment of NSCLC, the 5-year survival rate for NSCLC patients is not satisfactory. 4 Therefore, it is essential to investigate effective therapeutic targets of NSCLC.
MicroRNAs (miRNAs) are defined as a subtype of non-coding RNAs with 19–25 nucleotide length. Aberrant expression of miRNAs involve in various biological processes such as cell proliferation, apoptosis, metastasis and epithelial-mesenchymal transition (EMT). 5 For instance, miR-25, as an oncogene, promoted cell proliferation and metastasis of NSCLC. 6 Xiao et al. found that miR-7-5p acted as a tumor suppressor in NSCLC progression, which could restrain tumor metastasis by upregulating NOVA2 expression. 7 Previous literatures uncovered that the expression of miR-2682-5p was downregulated in various tumor tissues, such as pancreatic cancer, oral squamous cell carcinoma and bladder cancer, and miR-2682-5p could suppress the proliferation, migration and invasion of tumor cells.8–10 However, the expression and biological function of miR-2682-5p in NSCLC have not been clearly defined and need to be further explored.
Exosomes are packaged vesicles with a diameter of 30–150 nm secreted by a variety of cells. They contain genetic material (miRNAs, lncRNAs, cricRNAs, tRNAs, and mRNAs), proteins, lipids or cytokines, and mediate intercellular communication. 11 Exosome-derived miRNAs have been demonstrated to have different effects in various cancers, including NSCLC. For example, exosome-derived miR-126 inhibited cell growth and induced loss of malignancy of NSCLC cells. 12 Xu et al. reported that exosomal miR-let-7e from serum inhibited cell proliferation, migration and invasion in vitro as well as delayed tumor growth in vivo by downregulating the expression of SUV39H2. 13 A current study indicated that the role of exogenous miRNAs in tumors was similar to that of endogenous miRNAs transported by exosomal. 14 Moreover, due to the double-layer structure of exosomes, exosomal miRNAs are remarkably stable compared to miRNAs that are not contained in exosomes. Therefore, delivery of miRNAs to NSCLC cells may be a potential strategy for NSCLC treatment.
In this study, we found that miR-2682-5p expression level was remarkably downregulated in tumor tissues, cell lines, serum, and serum exosomes of NSCLC patients. Furthermore, we revealed that exosome-delivered miR-2682-5p could inhibit the malignant biological behaviors of NSCLC cells by the HDAC1/ADH1A axis.
Materials and methods
Patients and tissue samples
A total of 20 paired NSCLC tumor tissues and adjacent non-tumor tissues were collected from patients who underwent surgery at Peking University Shenzhen Hospital from September 2016 to November 2018. All tissue samples were stored at −80°C. Blood samples from 20 healthy donors and 20 NSCLC patients were obtained and centrifuged at 3000 rpm for 15 min, then serum samples were harvested and stored at −20°C. The use of clinical tissues was approved by the ethics committee of Peking University Shenzhen Hospital. Each patient and donor provided written informed consent.
Cell culture
NSCLC cell lines (A549, NCI-H23, NCI-H1299, and HCC827) and human embryonic lung fibroblast (MRC-5) were purchased from the Chinese Academy of Sciences Cell Bank (Shanghai, China). A549 and MRC-5 cells were cultured in high-glucose Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum (FBS; Thermo Fisher Scientific, Inc). NCI-H23, NCI-H1299, and HCC827 cells were cultured in RPMI 1640 medium (HyClone, Utah, USA). All cells were cultured in a humidified atmosphere of 5% CO2 at 37°C.
Exosome isolation and identification
Exosomes were isolated from NSCLC patient and healthy donor serum samples with ExoQuick TC (EXOQ20A-1, System Biosciences, USA). Serum samples were first filtered through a 0.45-μm polyvinylidene fluoride filter (Millipore, Billerica, MA, USA). Afterward, 126 μL of ExoQuick solution was added in 500 μL of serum samples and incubated for 30 min at 4°C. Then, the mixture was centrifuged at 3000 rpm for 30 min at room temperature. The exosomes were collected and washed with sterilized PBS. Subsequently, the exosomes were resuspended in PBS and filtered through 0.22-μm filters (Millipore, Billerica, MA, USA). The contents of exosomal proteins were measured by the BCA Protein Assay Kit (Beyotime, Nanjing, China). The levels of CD63, TSG101, CD9, and CD81 (the biomarkers of exosomes) were determined by using Western blotting analysis. The isolated exosomes were also identified by transmission electron microscopy (Tecnai 12; Philips).
Quantitative real-time polymerase chain reaction
Total RNA was extracted from cells and tissues with a GENzolTM TriRNA Pure Kit (Invitrogen, CA, USA). Total RNA in serum was purified by using miRNeasy Serum/Plasma Kit according to the manufacturer’s protocols (Qiagen). Exosomal RNA was isolated from serum by using miRNeasy Micro Kit (Qiagen). Reverse transcription was performed by using a PrimeScript RT reagent kit (Takara Biotechnology, Dalian, China). RT-qPCR was performed by using SYBRTM Green qPCR Master Mix (Thermo Fisher Scientific) on a 7900HT system (Applied Biosystems). The RT-PCR cycling conditions were as follows: 95°C for 10 s followed by 40 cycles at 95°C for 5 s, 60°C for 30 s, and 72°C for 15 s. The RT-qPCR reaction system was as follows: 2 μL of cDNA was added to 10 μL of the 2× SYBR green PCR master mix with 0.4 μL of Taq polymerase enzyme (RiboBio, Guangzhou, China), 0.8 μL of each primer, and 6 μL of ddH2O to a total volume of 20 μL. Relative gene expression was normalized to U6 or GAPDH. The results were calculated by using the 2−ΔΔCT method and then converted to fold changes.
Cell transfection
A549 and NCI-H1299 cells were seeded into 6-well plates and transfected by using Lipofectamine 2000 reagent (Invitrogen, CA, USA) in serum-free medium. MiR-2682-5p mimics, inhibitors and their negative controls, HDAC1 and ADH1A overexpression plasmids and their small interfering RNAs (siRNAs) were used to transfect into cells. Then, the culture medium was replaced to serum-containing medium after transfection for 6 h.
Cell viability assay
Cell viability was evaluated by CCK-8 assay. A549 and NCI-H1299 cells were seeded in 96-well plates. After incubation for 72 h, the cells were incubated with the CCK-8 solution (Amyjet Scientific, Wuhan, China) for 4 h. The absorbance was measured at 450 nm by using a microplate reader (Molecular devices, Shanghai, China).
Cell migration assay
A Transwell chamber (Corning, New York, USA) migration method was used to determine the migratory capacity of A549 and NCI-H1299 cells. The transfected cells (105/mL) or the cells incubated with exosomes were transferred into the upper chamber in serum-free medium. In addition, the medium containing serum was added into the lower compartment. After incubation for 24 h, cells migrating to the bottom of the membrane were fixed with 4% paraformaldehyde and stained by using a Three-Step Stain Set (Thermo Fisher Scientific). The number of migration cells was counted under an inverted microscope. Each experiment was performed three times.
Cell apoptosis analysis
Flow cytometry was used to evaluate cell apoptosis ratio. A549 and NCI-H1299 cells were suspended with binding buffer and stained by using Annexin V-fluorescein isothiocyanate and Propidium Iodide (PI) (BD Biosciences, CA, USA) for 20 min. Then, flow cytometry analysis was carried out within 1 h. These data were analyzed by using BD CellQuest Pro software 5.1 (BD Biosciences, CA, USA).
Western blot analysis
The A549 and NCI-H1299 cells were ruptured by using RIPA buffer (Beyotime, Shanghai, China) containing 1% protease inhibitor (Roche, CA, USA). Equal amounts of proteins (20 μg/lane) were separated by 12% SDS-PAGE at 70 V for 30 min, followed by 120 V for 90 min, then transferred to 0.22-μm polyvinylidene fluoride (PVDF) membranes (Millipore) at 300 mA for 2 h and blocked with 5% non-fat milk. The membranes were incubated with primary antibodies against N-cadherin (1:800, ab76011, Abcam), E-cadherin (1:1000, ab40772, Abcam), CD63 (1:1000, ab134045, Abcam), CD81 (1:1000, ab79559, Abcam), HDAC1 (1:800, ab109411, Abcam), ADH1A (1:1000, ab108203, Abcam), TSG101 (1:1000, ab125011, Abcam), CD9 (1:1000, ab92726, Abcam), and GAPDH (1:2000, ab8245, Abcam) at 4°C overnight and then incubated with horseradish peroxidase (HRP) conjugated goat anti-rabbit IgG (1:1500, ab6721, Abcam) for 2 h at ambient temperature. Finally, the bands were detected and quantified with the ImageQuanTM LAS 4000 (GE Healthcare Life Sciences, Pittsburgh, PA, USA) and the ChemiDocTM XRS + (Bio-Rad, Hercules, CA, USA).
RNA immunoprecipitation (RIP) assays
Magna RIP Kit (Millipore) was used for RNA immunoprecipitation assay. A549 cells were ruptured by using RIP lysis buffer, and 100 μL of lysates was incubated with magnetic beads conjugated with the argonaute2 (Ago2) antibody (Anti-Ago2). Immunoglobulin G (IgG) was regarded as a negative control. Proteinase K digestion and extraction of RNA were performed with TRIzol reagent. The immunoprecipitated RNA was analyzed by RT-qPCR assay.
Chromatin immunoprecipitation (ChIP) assay
ChIP assay was performed by using the EZ-ChIP kit (Millipore) according to manufacturer’s protocol. A549 and NCI-H1299 cells were supplemented with 1% formaldehyde and incubated for 10 min to produce DNA-protein crosslinks. Then, crosslinked chromatin DNA was sonicated to yield chromatin fragments of 200–300 base pairs, and incubated with anti-HDAC1 (Millipore) or IgG at 4°C. The precipitated chromatin DNA was amplified by RT-qPCR assay. The sequence information of ChIP primers were shown as follows: forward primer: GGG GCT CAG TCA ATG CTG AT; Reverse primer: AAA GAT GAC AGC CAG GAG CC.
Dual-Luciferase reporter gene assay
The wild-type and mutant-type HDAC1 3′UTR (HDAC1-WT/MUT) reporter vectors were generated by General Biosystems (Anhui, China). A549 and NCI-H1299 cells were transfected with miR-2682-5p mimic and the above reporter vectors by using Lipofectamine 2000 (Invitrogen, CA, USA). After transfection for 48 h, the luciferase activities were measured by using Dual-Luciferase Reporter Assay System (Promega).
Statistical analysis
SPSS 22.0 (La Jolla, CA, USA) was used to analyze the data. Statistical significances were analyzed by using Student’s t-test for two group, and ANOVA for multiple groups. Differences were considered statistically significant when p < 0.05.
Results
The expression of miR-2682-5p was downregulated in NSCLC tissues and cell lines
We examined the expression of miR-2682-5p in 20 pairs of NSCLC tissues and adjacent non-tumor tissues by RT-qPCR. The results revealed that the expression of miR-2682-5p was downregulated in NSCLC tissues compared to the adjacent non-tumor tissues (Figure 1(a)). We then detected the expression of miR-2682-5p in NSCLC cell lines (A549, NCI-H23, NCI-H1299, and HCC827) and human embryonic lung fibroblast cell line (MRC-5), and found that miR-2682-5p expression was lower in NSCLC cell lines than that in MRC-5 cells (Figure 1(b)). Expression of miR-2682-5p was downregulated in the NSCLC tumor tissues and cell lines. (a) MiR-2682-5p expression level in tumor tissues and adjacent non-tumor tissues was detected by RT-qPCR assay (n = 20). (b) MiR-2682-5p expression level in NSCLC cell lines (A549, NCI-H23, NCI-H1299, and HCC827) and normal embryonic lung fibroblast cells (MRC-5) was detected by RT-qPCR assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.
The expression of miR-2682-5p was downregulated in serum and serum exosomes of NSCLC patients
By evaluating the expression level of miR-2682-5p in serum samples, we found that miR-2682-5p in the serum of NSCLC patients had decreased expression compared with that of the healthy donors (Figure 2(a)). To test the expression of miR-2682-5p in exosomes, we purified the exosomes from the serum of NSCLC patients and healthy donors. The isolated exosomes were visualized as round particles (50–150 nm) by transmission electron microscopy (TEM) (Figure 2(b)). The isolated exosomes were also identified by the expression of exosome markers CD63, TSG101, CD9, and CD81. Western blotting results showed that all exosome markers could be detected in the isolated exosomes but not the centrifuged supernatant, which indicated that serum-derived exosomes were successfully obtained in our experiments (Figure 2(c)). We then assessed the expression of miR-2682-5p in exosomes isolated from the serum samples and centrifuged supernatant. The results showed that the expression level of exosomal miR-2682-5p was also significantly downregulated in NSCLC patients compared to healthy control (Figure 2(d)). However, no change in miR-2682-5p level was found in centrifuged supernatant (Figure 2(e)). Expression of miR-2682-5p was downregulated in serum and serum exosomal of NSCLC patients. (a) MiR-2682-5p expression level in serum of NSCLC patients and healthy donors was detected by RT-qPCR assay (n = 20). (b) Transmission electron microscopy image of exosomes derived from the serum of patients and controls (×16500). Scale bars represent 1.0 μm. (c) Western blotting was used to detect the presence of CD63, TSG101, CD9, and CD81 in exosomes and centrifuged supernatant. (d)–(e) MiR-2682-5p expression level in serum exosomes of NSCLC patients and healthy control and centrifuged supernatant was detected by RT-qPCR assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.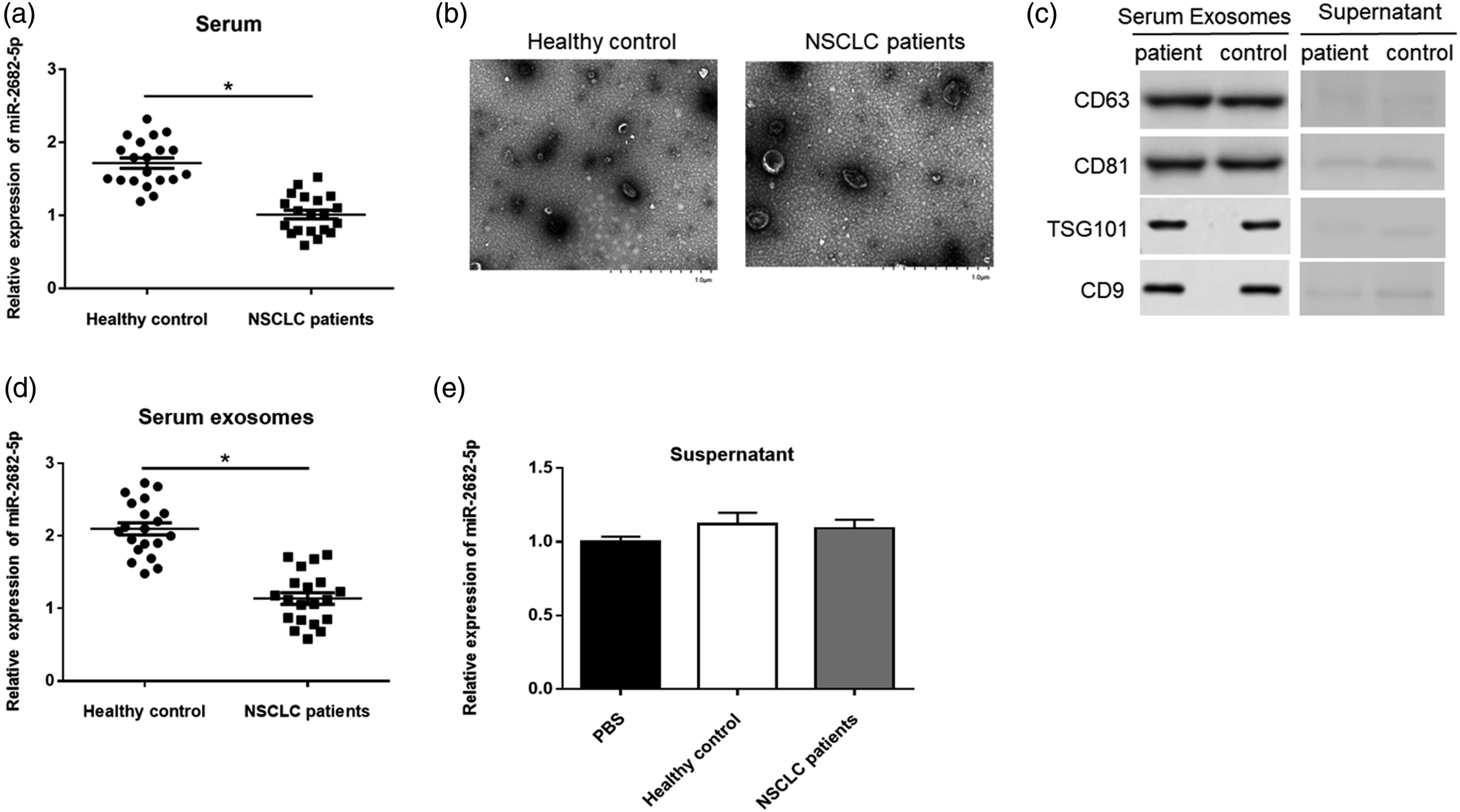
miR-2682-5p repressed NSCLC cell viability and migration and promoted apoptosis To explore the biological role of miR-2682-5p, we transfected miR-2682-5p mimic or miR-2682-5p inhibitor in A549 and NCI-H1299 cells. RT-qPCR results indicated that the expression of miR-2682-5p was remarkably lower in the miR-2682-5p inhibitor group and significantly higher in the miR-2682-5p mimic group than that in NC inhibitor and NC mimic group (Figure 3(a)). Overexpression of miR-2682-5p significantly suppressed A549 and NCI-H1299 cell proliferation, and miR-2682-5p knockdown led to the opposite effects (Figure 3(b)). Additionally, Transwell assay showed that the number of migrated cells was distinctly decreased in the miR-2682-5p mimic group, and significantly increased in the miR-2682-5p inhibitor group (Figure 3(c)). Flow cytometry results revealed that miR-2682-5p overexpression enhanced cell apoptosis, and miR-2682-5p silencing inhibited cell apoptosis (Figure 3(d)). The expression of Vimentin was significantly decreased in the miR-2682-5p mimic group and increased in the miR-2682-5p inhibitor group. On the contrary, the expression of E-cadherin was markedly increased in the miR-2682-5p mimic group and repressed in the miR-2682-5p inhibitor group in A549 and NCI-H1299 cells (Figure 3(e)). MiR-2682-5p inhibited viability and migration and promoted apoptosis of NSCLC cells. A549 and NCI-H1299 cells were transfected with miR-2682-5p mimic (50 nM) or miR-2682-5p inhibitor (50 nM). (a) RT-qPCR analysis of miR-2682-5p overexpression and knockdown efficiencies in A549 and NCI-H1299 cells. (b)–(d) The proliferation ability, migration ability, and apoptosis ability of A549 and NCI-H1299 cells were detected by CCK-8, Transwell, and flow cytometry assays, respectively. (e) The expression of E-cadherin and Vimentin in A549 and NCI-H1299 cells was detected by Western blot assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.
Exosomal miR-2682-5p inhibited NSCLC cell viability and migration and promoted apoptosis
A549 and NCI-H1299 cells were incubated with exosomes at different concentrations (0, 50, 100, and 150 μmol/L). The results of RT-qPCR showed that the expression of miR-2682-5p was increased in A549 and NCI-H1299 cells after incubation with exosomes (Figure 4(a)). As the amount of miR-2682-5p-EXO increased in the A549 and NCI-H1299 cells, cell proliferation and migration were reduced, while cell apoptosis ratio was elevated (Figure 4(b)–(d)). Furthermore, Western blot assay revealed that the increasing concentration of miR-2682-5p-EXO inhibited the expression of Vimentin and promoted the expression of E-cadherin (Figure 4(e)) MiR-2682-5p-EXO inhibited viability and migration and promoted apoptosis in A549 and NCI-H1299 cells. A549 and NCI-H1299 cells were incubated with 0, 50, 100 and 150 μg/mL miR-2682-5p-EXO. (a) MiR-2682-5p expression was detected by RT-qPCR assay. (b)–(d) The cell viability, migration ability and apoptosis ratio of cells were detected by CCK-8, Transwell and flow cytometry assays, respectively. (e) The expression of E-cadherin and Vimentin was detected by Western blot assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05.
miR-2682-5p could bind with HDAC1
We then found that the expression of HDAC1 was upregulated in NSCLC tissues and cell lines (Figure 5(a) and (b)). To investigate the relationship between miR-2682-5p and HDAC1 in NSCLC, we conducted a bioinformatics analysis and uncovered that miR-2682-5p directly targeted the 3′UTRs of HDAC1 mRNA (Figure 5(c)). Dual-luciferase reporter gene assay results showed that miR-2682-5p overexpression significantly decreased the luciferase activity of HDAC1-WT in A549 and NCI-H1299 cells, whereas had no effect on the luciferase activity of HDAC1-MUT (Figure 5(d)). In addition, RIP assay showed that anti-Ago2 precipitated a large number of miR-2682-5p and HDAC1 mRNA (Figure 5(e)). Furthermore, A549 and NCI-H1299 cells were transfected with miR-2682-5p mimic or miR-2682-5p inhibitor to detect the effect of miR-2682-5p on HDAC1 expression. As expected, overexpression of miR-2682-5p downregulated HDAC1 protein level, and miR-2682-5p knockdown upregulated HDAC1 protein level in A549 and NCI-H1299 cells (Figure 5(f)). MiR-2682-5p negatively regulated HDAC1. (a) The expression of HDAC1 in NSCLC tumor tissues and adjacent non-tumor tissues was detected by RT-qPCR assay. (b) The expression of HDAC1 in NSCLC cell lines (A549, NCI-H23, NCI-H1299 and HCC827) and normal embryonic lung fibroblast cells (MRC-5) was detected by RT-qPCR assay. (c) The binding sites of HDAC1 on miR-2682-5p was predicted by miRmap. (d) The luciferase activity was detected by dual-luciferase reporter gene assay. (e) RIP assay was performed to determine the enrichment of miR-2682-5p and HDAC1 in Anti-Ago2 or IgG. (f) The expression of HDAC1 was detected by Western blot assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.
miR-2682-5p inhibited NSCLC cell viability and migration and promoted apoptosis via HDAC1 downregulation
To investigate the effects of HDAC1 in NSCLC, we transfected miR-2682-5p mimic alone or together with HDAC1 overexpression plasmid (pcDNA-HDAC1) into A549 and NCI-H1299 cells. The expression level of HDAC1 was significantly reduced by miR-2682-5p overexpression, which was reversed by transfection of pcDNA-HDAC1 (Figure 6(a)). CCK-8, Transwell, and flow cytometry assay results revealed that overexpression of HDAC1 could effectively reverse the inhibitory effect of miR-2682-5p on cell proliferation and migration and the promoting effect on apoptosis in A549 and NCI-H1299 cells (Figure 6(b)–(d)). Likewise, the decrease of Vimentin protein level and the increase of E-cadherin protein level indicated that overexpression of HDAC1 inverted the effects of miR-2682-5p mimic on Vimentin and E-cadherin expression in A549 and NCI-H1299 cells (Figure 6(e)). HDAC1 reversed the effects of miR-2682-5p on the NSCLC cell viability, migration, and apoptosis. A549 and NCI-H1299 cells were transfected with miR-2682-5p mimic or co-transfected with miR-2682-5p mimic (50 nM) and HDAC1 overexpression plasmid (50 nM). (a) The expression of HDAC1 was detected by RT-qPCR assay. (b)–(d) The cell viability, migration ability, and apoptosis ratio of A549 and NCI-H1299 cells were detected by CCK-8, Transwell, and flow cytometry assays, respectively. (e) The expression of E-cadherin and Vimentin in A549 and NCI-H1299 cells was detected by Western blot assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.
HDAC1 interacted with ADH1A
HDAC1 has been reported to be a common regulator of ADH1A expression. RT-qPCR results showed that ADH1A expression level was downregulated in NSCLC tissues and cell lines compared with adjacent non-tumor tissues and MRC-5, respectively (Figure 7(a) and (b)). Overexpression of HDAC1 declined the expression of ADH1A, and knockdown of HDAC1 increased the expression of ADH1A in A549 and NCI-H1299 cells (Figure 7(c)). Afterward, we performed chromatin immunoprecipitation (ChIP) assay, and the results confirmed that HDAC1 was recruited to the ADH1A promoter regulatory region (Figure 7(d)). HDAC1 regulated ADH1A expression in A549 and NCI-H1299 cells. (a) The expression level of ADH1A in NSCLC tumor tissues and adjacent normal tissues was detected by RT-qPCR assay. (b) The expression level of ADH1A in NSCLC cell lines (A549, NCI-H23, NCI-H1299, and HCC827) and normal embryonic lung fibroblast cells (MRC-5) was detected by RT-qPCR. (c) The effect of HDAC1 silencing or overexpression on the protein level of ADH1A was measured by Western blot assay. (d) ChIP assay was performed to investigate HDAC1 binding in the ADH1A promoter in A549 and NCI-H1299 cells. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.
HDAC1 promoted NSCLC cell viability and migration and reduced apoptosis by suppressing ADH1A expression
To evaluate the role of ADH1A in NSCLC, we transfected pcDNA-HDAC1 alone or together with ADH1A overexpression plasmid (pcDNA-ADH1A) in the A549 and NCI-H1299 cells. HDAC1 overexpression decreased the protein level of ADH1A, while overexpression of ADH1A restored ADH1A protein level in A549 and NCI-H1299 cells (Figure 8(a)). The results of CCK-8 and Transwell assays revealed that the viability and migration ability of A549 and NCI-H1299 cells co-transfected with pcDNA-HDAC1 and pcDNA-ADH1A were decreased compared with cells transfected with pcDNA-HDAC1 alone (Figure 8(b) and (c)). Moreover, upregulation of ADH1A also reversed the inhibition effect of HDAC1 overexpression on cell apoptosis in A549 and NCI-H1299 cells (Figure 8(d)). The expression of Vimentin was increased and the expression of E-cadherin was decreased in A549 and NCI-H1299 cells in response to HDAC1 overexpression, which could be abolished by ADH1A overexpression (Figure 8(e)). HDAC1 promoted NSCLC progression via the regulation of ADH1A. A549 and NCI-H1299 cells were transfected with HDAC1 overexpression plasmid (50 nM) alone or together with ADH1A overexpression plasmid (50 nM). (a) The expression of ADH1A was detected by RT-qPCR assay. (b)–(d) The cell viability, migration ability and apoptosis ratio of A549 and NCI-H1299 cells were detected by CCK-8, Transwell, and flow cytometry assays, respectively. (e) The expression of E-cadherin and Vimentin in A549 and NCI-H1299 cells was detected by Western blot assay. Data were presented as mean ± standard deviation with three replicates. *p < 0.05. NSCLC: Non-small cell lung cancer.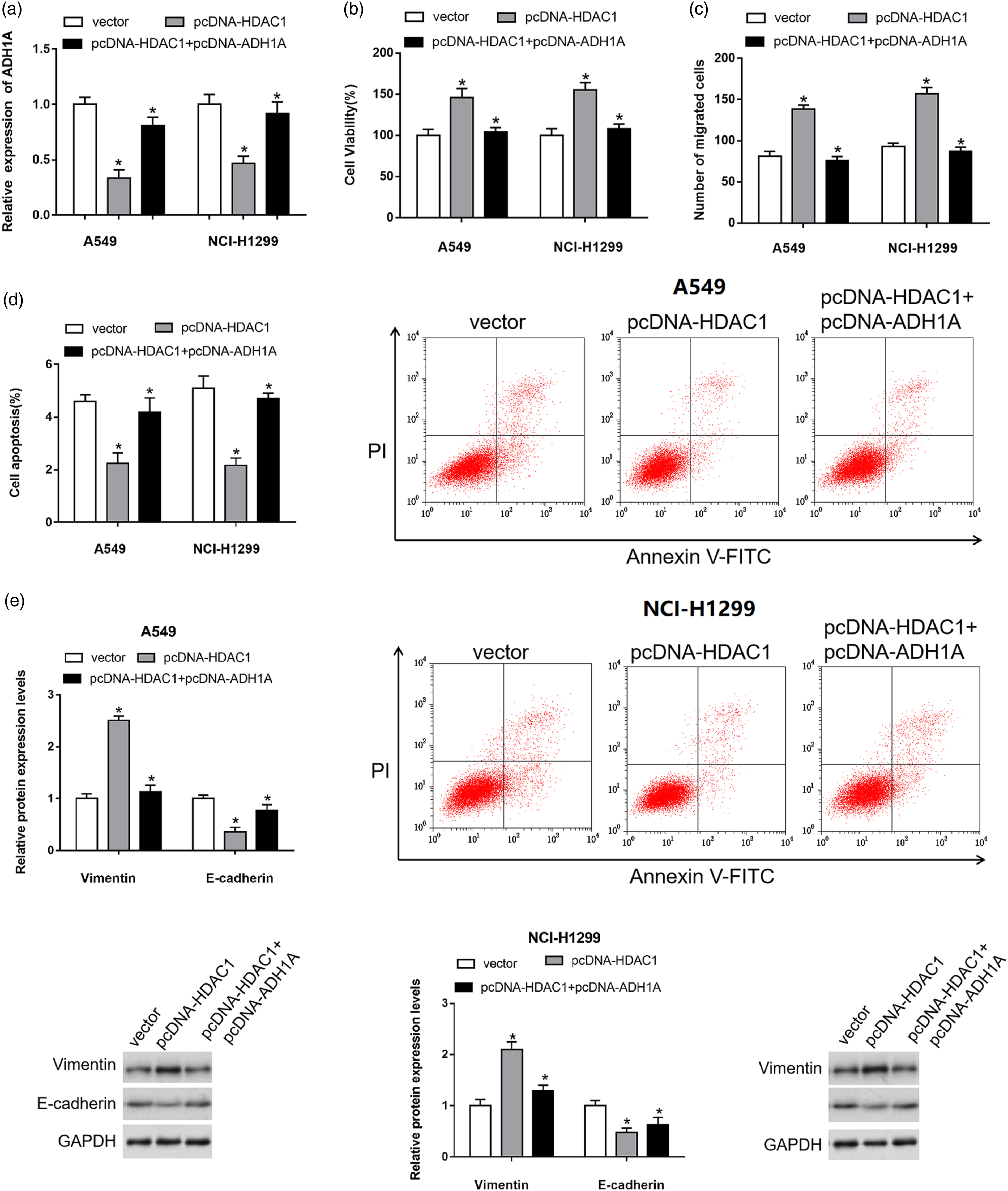
Discussion
In our study, we found that miR-2682-5p expression was noticeably downregulated in NSCLC tissues, overexpression of miR-2682-5p inhibited NSCLC cell viability and migration and promoted apoptosis, while miR-2682-5p knockdown showed the opposite effects. Increasing studies suggested that miRNAs could act as an oncogene or a tumor inhibitor in NSCLC. It was reported that miR-2682-5p was a downstream gene of LINC01006, and could abolish the facilitating effect of LINC01006 on pancreatic cancer cell proliferation and metastasis. 8 Likewise, miR-2682-5p downregulation was found in oral squamous cell carcinoma, ectopic expression of miR-2682-5p suppressed cell proliferation, and migration by upregulating HOXB8. 9 Li et al. uncovered that miR-2682-5p was low expressed in bladder cancer cells and tissues, overexpression of miR-2682-5p could promote cell proliferation, migration, and EMT by strengthening the expression of ELK1. 10
Accumulated evidence proofed that exosomes participated cellular communication and loaded exogenous cargo,15,16 as well as exerted an important effect in a variety of cancer progression. 17 For example, exosome-transmitted UFC1 promoted NSCLC cell proliferation, migration and invasion. 18 A study revealed that exosomal miR-146a-5p was downregulated in serum of NSCLC patients and closely correlated with shorter survival. 19 Zhang et al. found that exosome-delivered of miR-214 remarkably enhanced gefitinb resistance in NSCLC. 20 Our study showed that miR-2682-5p expression was lower in serum and serum exosome of NSCLC patients than healthy donors. Exosomes packed with miR-2682-5p could be bound and internalized by recipient cell, leading to changes in the behavior of the recipient cell. Exosomal miR-2682-5p remarkably suppressed NSCLC cell viability and migration, and promoted apoptosis. Exosomes specifically packed with miR-2682-5p may be a useful target for NSCLC therapy.
Histone deacetylase 1 (HDAC1) has been reported as an epigenetic factor which belongs to histone deacetylase family. 21 It was reported that HDAC1 was involved in cell proliferation, differentiation, and apoptosis in cancer development by regulating gene expression. 22 HDAC1 overexpression could inhibit GAS5-AS1 expression, and subsequently promote tumor metastasis via upregulation of several key EMT markers. 23 MiR-34a could induce senescence in cells lacking functional p53 because of its ability to upregulate p21 via downregulating HDAC1 in NSCLC. 24 Herein, our study found that miR-2682-5p could bind with HDAC1. Overexpression of miR-2682-5p restrained NSCLC cell viability and migration, and induced apoptosis by suppressing HDAC1 expression. Our results might provide more information for the regulatory network of HDAC1 in cancers.
Recently, HDAC1 has been reported to participate in cancer progression by regulating the expression of downstream gene, such as interleukin-8, 25 hypoxia-inducible factors, 26 and p38 MAPK. 27 Zahid et.al. reported that HDAC1 was a transcriptional regulator of ADH1A and inhibited ADH1A expression. 28 Upregulation of ADH1A was significantly associated with good survival rate in hepatocellular carcinoma patients. 29 In this study, we uncovered that the expression of ADH1A was downregulated in NSCLC tissues and cell lines, and HDAC1 could bind to ADH1A promoter region and reduce its expression, thus promoting NSCLC cell viability and migration and declining apoptosis.
In conclusion, our findings proofed that exosomal miR-2682-5p noticeably suppressed NSCLC cell viability and migration and induced apoptosis. MiR-2682-5p exerted an anticancer role in NSCLC through regulating the HDAC1/ADH1A axis. These results showed that miR-2682-5p might function as a tumor suppressor and be a potential target for the treatment in NSCLC. Although this study provided a mechanistic understanding of the role of miR-2682-5p in NSCLC, it could not be ruled out that miR-2682-5p might play its role in NSCLC through other mechanisms.
Footnotes
Acknowledgements
The authors showed their gratitude to all participants in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
