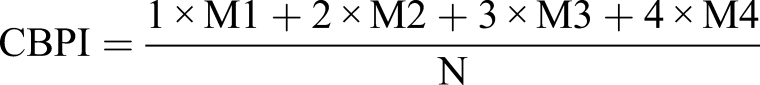Abstract
Tetrachlorvinphos is an organophosphate that is classified as a carcinogen in humans by several authorities. Due to very limited data regarding the genotoxic potential, we aimed to comprehensively investigate in vitro genotoxic potential of tetrachlorvinphos. We performed our study by applying the cytokinesis-block micronucleus cytome and sister chromatid exchange (SCE) assays to human peripheral blood lymphocytes. We evaluated micronucleus (MN) and SCE frequencies and cytokinesis-block proliferation index in both exposed and non-exposed lymphocytes. We also calculated the chromosomal instability level in response to exposure by combining the results of MN and SCE. We found that MN frequency did not increase with exposure to tetrachlorvinphos (0–50 µg/ml). In contrast, we observed that SCE frequencies significantly increased with exposure to ≥5 µg/ml tetrachlorvinphos. Furthermore, exposure to tetrachlorvinphos at concentrations of 50 µg/ml induced a significant increase in chromosomal instability level (p < 0.05). Cytokinesis-block proliferation index level did not significantly decrease in response to tetrachlorvinphos exposure. Our findings reveal that tetrachlorvinphos resulted in different DNA damages that were measured by two assays. Furthermore, our findings suggested that exposure to tetrachlorvinphos increased chromosomal instability that is a hallmark of many malignancies. We conclude that although tetrachlorvinphos does not significantly increase the MN level, the significant increase of both SCE and CIN frequencies indicates the genotoxic potential of tetrachlorvinphos in human peripheral lymphocytes. Additionally, tetrachlorvinphos is not cytotoxic in the range of tested concentrations.
Introduction
Pesticides are widely used worldwide for many purposes including fighting against epidemics, increasing food production, and protecting cultivated areas and forests. Substantial evidence has indicated that pesticides contaminate air, water, and food; can accumulate in the tissue; and have the potential hazard to non-target organisms and the environment.1,2 Due to the widespread use of pesticides both domestically and industrially, the effects of pesticides at the molecular level, such as the genotoxic effect, are a major concern for public health. 3 Many previous studies showed a relationship between exposure to pesticides and elevated cancer risk and other non-malignant diseases.4–7 Therefore, the application of many pesticides due to their carcinogenic and/or genotoxic properties is prohibited or restricted. 1
Tetrachlorvinphos is an organophosphate (OP) group insecticide and is classified by the International Agency for Research on Cancer as a potential carcinogen for humans (group 2B). 8 According to the United States Environmental Protection Agency (US EPA), tetrachlorvinphos has the potential to be slightly toxic in acute toxicity tests. It was reported that tetrachlorvinphos has the potential to induce adrenal cortical adenomas and thyroid C-cell adenomas hepatocellular carcinoma, hepatocellular adenomas, and granulomatous lesions of the liver in mouse and rats. There was no observation reported in terms of developmental toxicity in rats. Contradictory results have been reported about the mutagenicity potential of the tetrachlorvinphos. (EPA 1995). It is used to combat fleas and tick collars and powders in domestic animals. In addition, it is used as a larvicide in cattle, hog, goat, and horse feed and cattle feedlots and poultry houses. It is applied to various crops including cotton, cereals, fruits, and vegetables for agricultural purposes.8,9 General pathways of exposure to tetrachlorvinphos are occupational exposure in farmers and dietary exposure due to the application of tetrachlorvinphos in farmland. 10 It has also been reported that children and adults in the general population are contaminated by their pets’ flea collars containing tetrachlorvinphos. 11 Another exposure pathway is inhalation due to spraying in open areas with tetrachlorvinphos for controlling public health pests. 12 The use of tetrachlorvinphos was banned in the European Union countries. However, it continues to be used in many countries such as Canada, South Africa, United States, and Australia.8,13
Although it has been announced as a carcinogen, very few studies have been performed to investigate the toxic potential of tetrachlorvinphos. Furthermore, the effects of tetrachlorvinphos on human cells are extremely limited.8,14 Genotoxicity studies are useful tools for estimating the potential risk of chemicals. 1 Furthermore, the genotoxic potential is the leading risk factor for effects such as degenerative diseases, reproductive toxicology, and carcinogenesis. 15 Sister chromatid exchange (SCE) is one of the powerful tools for determining DNA damage resulting from chemical (genotoxic, mutagenic, and carcinogenic substances), physical (radiation), and biological (viruses and bacterial products, aging) agents and diseases (cancer, falconi’s anemia, and bloom syndrome). 16 In genetic toxicology, another excellent tool is the cytokinesis-block micronucleus (CBMN) assay that can measure nuclear anomalies such as micronucleus, nucleoplasmic bridge, and nuclear bud, as well as apoptosis, necrosis, and cytostatic effect. 17
To the best of our knowledge, there are no published data regarding the genotoxic and cytotoxic potential of tetrachlorvinphos on human peripheral lymphocytes. Therefore, the current study aimed to investigate in vitro genomic damage induced by tetrachlorvinphos using the CBMN and SCE assays on human peripheral lymphocytes.
Materials and methods
Blood samples
We took blood samples (4 mL) from two healthy volunteer donors into sterile heparin tubes. Voluntary consent forms were obtained from the donors. Both donors were aged between 40 and 45 who were non-smoker, non-alcoholic, and not exposed to radiation for occupational and diagnostic purposes. In addition, they did not always expose to any chemicals and take any drug during and before blood sampling. Finally, both donors were healthy and had no diseases. The ethical permission was granted from Clinical Research Ethics Committee, Çanakkale Onsekiz Mart University, Turkey (Decision number: 2019–10).
Chemicals
Chemicals used in the study were purchased from the following suppliers: tetrachlorvinphos (99.5% of purity, Lot number 4886300); RPMI 1640, fetal calf serum, mitomycin C (MMC), cytochalasin B: Sigma (USA); phytohaemaglutinin (PHA): Biological Industries (Israel); and methanol, acetic acid, dimethyl sulfoxide, and ethanol: Merck (Germany).
In vitro CBMN assay
The CBMN and SCE assays were conducted according to Fenech’s and Moorhead’s methods, respectively17,18 (Figure 1). Briefly, 4 mL of medium (RPMI 1640), 1 mL of fetal calf serum, and 0.2 mL of phytohemagglutinin (PHA) were mixed to prepare the cell culture medium. 0.5 mL of whole blood was added to the culture medium for each concentration. We performed a preliminary study to determine the cytotoxic potential of different concentrations. We found that the highest concentration of tetrachlorvinphos (50 µg/ml) induced less than 60% cytotoxicity, as suggested by Krisch–Volders.
19
Cytotoxicity was calculated according to Lorge.
20
Four different concentrations (1, 5, 25, and 50 µg/ml) of tetrachlorvinphos (dissolved in dimethyl sulfoxide) were added to each culture 24 h after initiation of the cultures. The cultures were incubated at 37°C for 72 h. For the CBMN assay, 6 µg/ml of cytochalasin B was added to block dividing cells at the binucleated stage at the 44th hour of the cultures. At the end of the 72 h, the cells were harvested. In this step, the cells were treated with 0.075M KCl as a hypotonic solution, and the cells were then treated three times with methanol-acetic acid (7:1). Microscopic evaluation was performed at 1000× magnification with light microscope according to criteria suggested by Fenech et al.
21
For the determination of MN frequency, 1000 binucleated cells were evaluated per slide. Therefore, in total, 4000 binucleated cells were evaluated for each concentration (2 donors × 2 replications). According to Eastmond and Tucker,
22
500 cells per slide (mononucleated, binucleated, trinucleated, and tertanucleated cells) were counted to calculate cytotoxicity. Thus, in total, 2000 cells were evaluated for each concentration (2 donors × 2 repetitions) to determine cytostasis (CBPI values) with the following formula A: Binucleated cell with micronuclei generated in CBMN assay. B: SCE excnahge generated by SCE assays. MN: Micronuclei, SCE: Sister Chromatid Excahnge.
In this formula, M1, M2, M3, and M4 represent 1, 2, 3, and 4 nucleated cells, whereas N represents the total number of counted cells. % cytotoxicity was calculated as described by Lorge et al. for each concentration. 20
In vitro SCE assay
The SCE assay was conducted in a similar cell culture condition performed for the CBMN assay. For the SCE assay, at the 24th hour of the cultures, 10 µg/ml of BrdU was added to each concentration and the negative and positive controls. After adding BrdU, all the culture tubes were kept in the dark until at the end of 72 h. At hour 70, colcemid (0.3 µg/ml) was added to stop metaphases. At the end of the 72 h, the cells were harvested. In this step, the cells were treated with 0.075M KCl as a hypotonic solution (heated at 37°C) for 10 min at 37°C. Finally, the cells were rinsed with methanol-acetic acid (3:1) for three times. The slides were stained with fluorescence plus Giemsa as described by Perry and Wolff. 23 Twenty-five well-spread metaphases (in total 100 metaphases, 2 donors × 2 duplicates) containing 46 chromosomes were evaluated for each concentration and the negative and positive controls at 1000× magnification with the light microscope.
Statistical analysis
All the results are presented as the mean (±SE) from two parallel experiments. After in vitro exposure, the results obtained for each concentration of tetrachlorvinphos were compared with the negative control using one-way analysis of variance and then Bonferroni’s multiple comparison tests for MN, SCE, CBPI, and CIN results. Correlation analysis was performed to show the association between the concentration and parameters. The data analyses were performed in the Prism software (GraphPad Software Inc) and Excel (Microsoft).
Results
Level of MN and CBPI in response to tetrachlorvinphos exposure.

MN: the total number of micronuclei; MMC: mitomycin C; SE: standard error; CBPI: cytokinesis-block proliferation index.
Level of SCE in response to tetrachlorvinphos exposure.

MMC: mitomycin C; SE: standard error; SCE/cell: total number of sister chromatid exchange (SCE) in a cell. *p < 0.05, ***p < 0.001.
Chromosomal instability in response to tetrachlorvinphos exposure.

Chromosomal instability (CIN) indicated the sum of MN and SCE frequencies for each concentration, SE: standard error, *p < 0.05.
Discussion
Although tetrachlorvinphos is used for many different purposes, to the best of the author’s knowledge, there is only one study performed on human cultured lymphocyte presenting findings for chromosomal aberration. In our study, we showed that tetrachlorvinphos did not induce a statistically significant increase in MN formation at any concentration. In a previous study. It was reported that Gardona®, the commercial formulation of tetrachlorvinphos, resulted in an increased CA frequency. 24 In another study, the genotoxic effects of Gardona® were evaluated using SCE and CA on mouse spleen cells, and it was reported that Gardona® had a mutagenic effect at concentrations of 0.25, 0.50, 1.0, and 2.0 μg/mL. 25 There are several biomonitoring studies related to the evaluation of the genotoxic potential of other OP insecticides. In these studies, OP pesticides were found out to be genotoxic in domestic use. 26 Additionally, chronic occupational exposure to OP pesticides induced DNA damage and oxidative stress.27–30
In our study, two parameters were measured to evaluate potential chromosomal instability induced by tetrachlorvinphos. MN can contain acentric chromatid/chromosome, whole chromatid/chromosome, or broken anaphase bridges. 31 Acentric fragments in MN originate from DNA breaks. Additionally, MN containing whole chromatid or chromosome is formed as a result of centromeric DNA hypomethylation, defected cell cycle control system, damaged kinetochore, or mitotic spindle failure. 32 The results obtained in our study show that tetrachlorvinphos increased the MN frequency on human peripheral lymphocytes in all concentrations (1.0, 5.0, 25, and 50 μg/mL) although it was not statistically significant. This result indicates that tetrachlorvinphos may cause significant increases in human peripheral lymphocytes with longer exposure times. It suggests that chronic exposure to tetrachlorvinphos might induce chromosomal instability via the mechanism and/or mechanisms mentioned above. We also determined CBPI values which represent the cytostatic effects of tetrachlorvinphos. Our results showed that tetrachlorvinphos did not statistically significantly decreased CBPI values at tested concentrations. We concluded that tetrachlorvinphos did not affect cell cycle kinetic on human peripheral lymphocytes in the range of the evaluated concentration in our study. However, since we found a negative association between concentration and CBPI values, we can infer that higher concentration may induce a significant decrease in CBPI.
SCE is a simple, rapid, and sensitive method for the genotoxic evaluation of physical, chemical, or biological factors. 16 In our study, the spontaneous SCE formations in the negative control and the induced SCE formations in response to tetrachlorvinphos exposure were compared. The result shows that SCE frequencies caused by 5, 25, and 50 µg/ml concentration of tetrachlorvinphos were found to be statistically significantly higher than those obtained for the negative control. These results suggest that tetrachlorvinphos might affect DNA repair and replication enzymes. SCE occurs via two sister chromatids break and then physically exchange at homologous loci during the S phase of the cell cycle. Repair and replication enzymes might play a role in SCE formation. Conditions that increase single-strand breaks (SSB), such as XRCC1 deficiency and inhibition of poly (ADP-ribose) polymerase 1 (PARP-1), often induce SCE formations.33,34 In addition, it was reported that DNA intercalators targeted DNA-topoisomerase II complex, and inhibition of topoisomerase II enzymes was associated with SCE formation. 35
Chromosomal instability was defined as a number of whole chromosomes or chromosomal segments in a cell. 36 In our study, we calculated chromosomal instability as a sum of the frequencies of MN and SCE. We observed a significant increase of chromosomal instability in a dose-dependent manner. Furthermore, the highest concentration of tetrachlorvinphos induced a significant increase of chromosomal instability that was a typical feature of many tumors.36,37 We used two assays, CBMN and SCE, and various different events can be evaluated through the consideration of both methods. Thus, chromosomal instability level in response to exposure has the potential to represent the potential of tetrachlorvinphos. We concluded that tetrachlorvinphos had the potential to induce chromosomal instability or alter the chromosomal integrity, which may shed light on its unknown side.
Conclusion
We conclude that although tetrachlorvinphos does not significantly increase the MN level, the significant increase of both SCE and CIN frequencies indicates the genotoxic potential of tetrachlorvinphos in human peripheral lymphocytes. Additionally, tetrachlorvinphos is not cytotoxic in the range of tested concentrations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Coordination Unit of Çanakkale Onsekiz Mart University, Turkey (Project number: 2020-3303).