Abstract
Background:
Benzonatate is a commonly prescribed medication that can be lethal in acute overdose of a small number of capsules.
Objective:
This was a systematic review to describe the course of severe poisoning and deaths from benzonatate supplemented with the National Poison Data System (NPDS) fatalities module.
Methods:
The NPDS was queried from 2000 to 2018 for benzonatate fatalities. Pubmed, Cochrane, Embase, and Google Scholar were searched for combinations of benzonatate and “poisoning,” “overdose,” and “toxicity.” References of relevant articles were searched for additional publications. Articles were included if they described the clinical course of at least one patient suffering from benzonatate poisoning and available in English. Dual independent review and extraction were performed.
Results:
Seventeen cases from NPDS and 19 published reports met the inclusion criteria resulting in 36 cases, mostly (28/36) self-harm ingestions. Most patients were young [17 (11–29), median (IQR)] and female (22). Onset of toxicity was rapid at <5 min (9). Most common symptoms included cardiac arrest (29), seizures (24), and dysrhythmias (24). Treatments included intubation (26), cardiopulmonary resuscitation (28), vasopressors (20) and others. Return of spontaneous circulation was achieved in 23/28 patients, but most had significant neurologic deficits or other end organ damage and 5 survived with a good neurologic outcome.
Conclusion and relevance:
Overdose ingestions of benzonatate can cause significant toxicity with a rapid onset. Interventions performed were generally supportive in nature. Duration of directly toxic effects is short, but dramatic with neurologic devastation and resuscitated patients often still have a poor outcome.
Introduction
Benzonatate is a commonly prescribed cough suppressant with over 3 million prescriptions per year in the US in 2016. Benzonatate shares structural similarities with other local anesthetics such as benzocaine and tetracaine, and alleviates cough through numbing the peripheral stretch receptors in the respiratory system. 1 Calls to poison centers about benzonatate exposures have risen drastically (doubling since the 2012 FDA warning) with a concomitant increase in the number of severe poisonings from approximately 120 in 2012 to 240 in 2019. 2 Sporadic cases of severe benzonatate poisoning are reported in the literature.3–5
This was a descriptive study using the National Poison Data System (NPDS) fatalities module in combination with a systematic review to describe the course of severe poisoning and deaths from benzonatate.
Methods
Two independent authors performed searches of Pubmed, Cochrane, Embase, and Google scholar for combinations of benzonatate and “poisoning,” “overdose,” and “toxicity” from database inception until November 2019 (benzonatate AND (poisoning OR overdose OR toxicity)). Additionally, references of relevant articles were searched for additional papers. The only exclusion on the type of article was English language. Articles were included if they described the clinical course of at least one patient poisoned from benzonatate. Studies and articles that provided only coded data were excluded from analysis unless they described individual severe cases. Cases identified in both NPDS and published literature were only presented once. Severe cases were defined by a case that had life-threatening symptoms or symptoms that resulted in significant residual disability or disfigurement. Abstracts were included as long as the clinical course was described. Due to the descriptive nature, no meta-analysis was planned. The review was not pre-registered. We used Covidence systematic review software (Version v2553 b219db6b, Veritas Health Innovation, Melbourne, Australia) to manage the systematic review and followed the PRISMA guidelines for reporting systematic reviews (see supplemental material) 6
Both authors independently reviewed titles, abstracts, and full-text of each article. After a decision of which articles met inclusion, both authors extracted data from each article. Discrepancies were resolved through discussion.
Poison Centers in the United States submit cases to the NPDS. In cases that result in fatalities, the poison center submits a fatality abstract providing further information surrounding the case. The fatality review team assigns a Relative Contribution to Fatality (RCF) on a rank of one to six: 1—undoubtedly responsible, 2—probably responsible, 3—contributory, 4—probably not responsible, 5—clearly not responsible, 6—unknown. Benzonatate fatality abstracts were requested from the NPDS from 2000 to 2018 (pre-2000 data is archived and more difficult to obtain). Cases were included if the RCF was 1—undoubtedly responsible or 2—probably responsible for the death.
Data were dually extracted from each case included age, sex, dose, onset of symptoms, specific symptoms during acute toxicity, therapies, duration of effect, and sequelae of the poisoning. Missing data were not imputed. Any discrepancies in data extracted were resolved through discussion.
Results
The literature searches yielded 169 articles of which 107 were removed as duplicates. One additional article was found from searching of the citations. After screening for eligibility, 18 abstracts or full-text manuscripts were included and yielded 19 cases (Figure 1). From the NPDS, 52 cases included benzonatate as a substance listed. Of these 52 cases, 17 were included for a total of 36 cases between the two data sources. No cases were duplicated in the published literature and NPDS deaths. The majority patients were young [17 (11–29), median (IQR)], female (22), and mostly intentional self-harm exposures (28) (Table 1). Onset of symptoms was <5 min in 9/16 patients where a time of onset was known. Most commonly reported symptoms included cardiac arrest (29), seizures (24), and dysrhythmias (24). Specific dysrhythmias included asystole, pulseless electrical activity, ventricular tachycardia, and ventricular fibrillation. Case descriptions are available in Table 2.
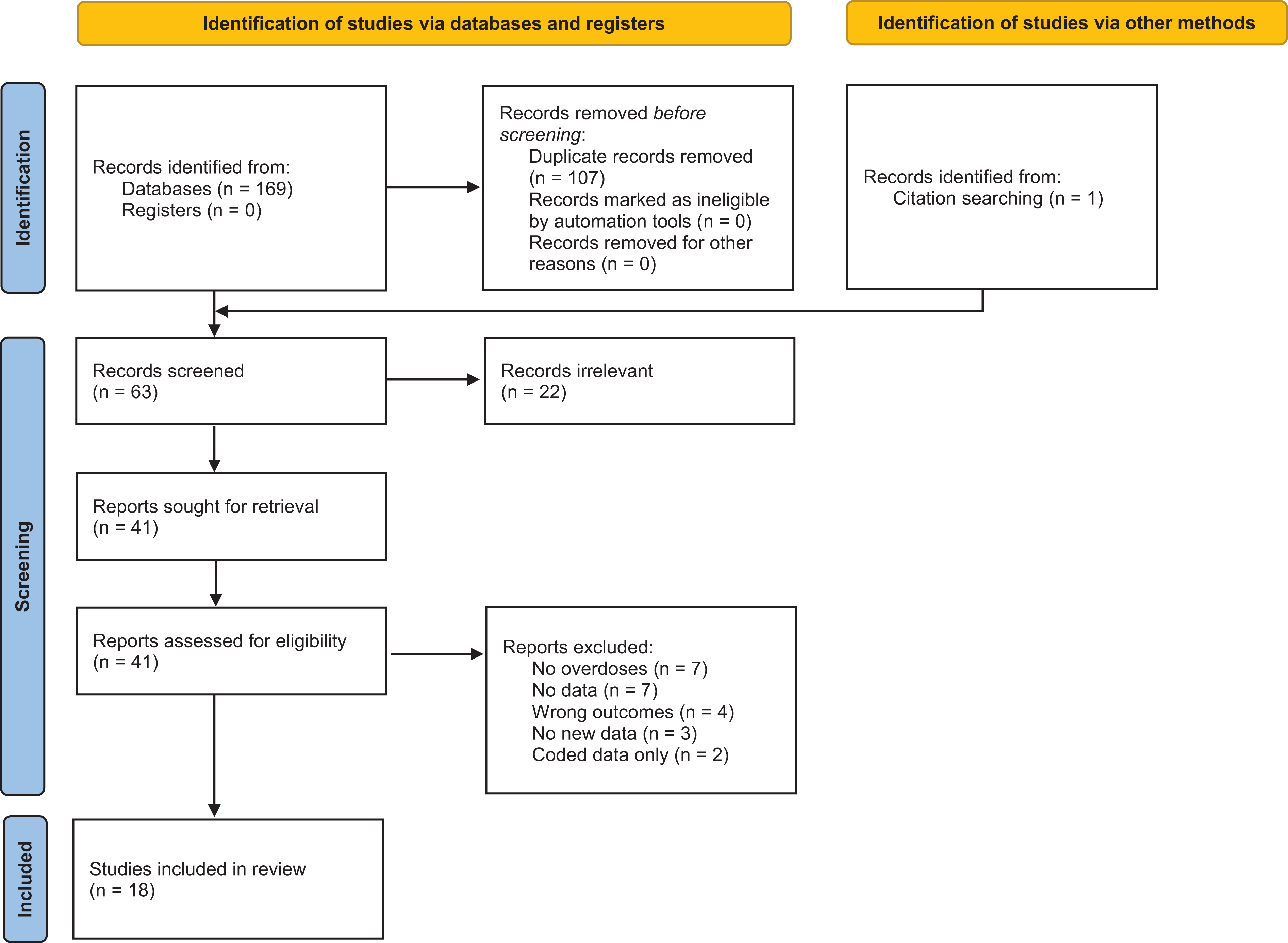
Included studies.
Clinical effects and treatments of benzonatate poisonings.

CPR, cardiopulmonary resuscitation; IQR, interquartile range; n, number; ROSC, return of spontaneous circulation; SD, standard deviation.
N’s for individual parameters if different from the total N defined above are stated within the respective column.
Individual included cases.

ABI, anoxic brain injury; AC, activated charcoal; AKI, acute kidney injury; APAP, Acetaminophen; ASA, aspirin; BZD, benzodiazepine; CAR, cardiac arrest; CPR, cardiopulmonary resuscitation; DIC, disseminated intravascular coagulopathy; GL, gastric lavage; HDI, high dose insulin euglycemic therapy; HoTN, hypotension; IBU, ibuprofen; INT, intubation; ILE, intravenous lipid emulsion; IVF, intravenous fluids; LEV, levetiracetam; mo, month; NaB, Sodium bicarbonate; NAC, n-acetylcysteine; NAL, naloxone; PEA, pulseless electrical activity; PHB, phenobarbital; PHY, phenytoin; PROP, propofol; QRSw, QRS interval widening; ROSC, return of spontaneous circulation; SE, status epilepticus; SZ, seizure; TTM, targeted temperature management; UNK, unknown; VASO, vasopressor; VEC, vecuronium Vfib, ventricular fibrillation; Vtach, ventricular tachycardia; VPA, valproic acid; yr, year.
* Unintentional exposure.
Treatments included intubation (26), cardiopulmonary resuscitation (28), vasopressors (20) and others. Six patients received intravenous lipid emulsion (ILE) therapy, one of which survived. Return of spontaneous circulation was achieved in 23/28 (83%) patients, but most had significant neurologic deficits or other end organ damage and only 5 survived with a good neurologic outcome. Three young children (18 months, 2 years, 4 years) died possibly as the result of aspiration of the capsule, possibly from numbing of the oropharyngeal zone. Patients who survived had resolution of cardiovascular symptoms within 2 hours and most were extubated within the day. Some had prolonged hospitalization due to aspiration.
Discussion
This study highlights the clinical effects of severe benzonatate ingestions. Published cases were identified as early as 1986 and were infrequently published up through 2019. Data from poison centers was only available through 2018 and deaths occurred almost every year. All cases were primarily large intentional ingestions in adolescents or adults, but 7/36 were under 5 years of age; a little over half were female (22/36). Published cases had a greater than 60% survival. In general, onset of effects is rapid and within minutes after ingestion, although some cases may have been delayed up to 2 h.4,7,8 However, when evaluating the pharmacokinetics of benzonatate, this timeframe does not make sense.
Time to maximal concentration (Tmax) occurs at 30 min with a rapid decline to nearly undetectable serum concentrations by the 2-h mark. 19 The primary analytical metabolite, 4-(butylamino)benzoic acid (BABA) peaks at the same time as benzonatate with a similar rapid decline. 19 Although both may have detectable concentrations at 2 h after ingestion, these low serum concentrations are not expected to cause significant toxicity. Additionally, BABA does not have appreciable sodium channel inhibition. 20 Another argument for the delayed onset of toxicity could be potential pharmacobezoar formation delaying the dissolution and absorption phases. Anecdotally, capsules are known to stick together in a container when exposed to moisture, so this may be possible, but pharmacobezoar formation with liquid filled capsules seems unlikely. The more likely case, as is with most intentional ingestions, is that the reported timeframe of ingestion is not accurate.
Early symptoms reported in many of the patients were either seizures or dysrhythmias. This is consistent with other sodium channel blocking local anesthetics like lidocaine, bupivacaine, etc. which makes sense pharmacologically as benzonatate is structurally related to tetracaine and procaine.21,22 Twenty-four patients experienced seizures and six these developed status epilepticus. Resolution of acute drug effect was fairly rapid, correlating with the 1 h half-life of benzonatate. Return of spontaneous circulation was reported in the majority of cases. Despite rapid resolution of direct drug effect, many had anoxic brain injuries and/or cerebral edema that ultimately resulted in mortality. Two patients that survived noted to have long-term neurologic dysfunction.8,17
Confirmatory concentrations were infrequently reported in these cases. Previous methods of determining benzonatate in plasma had several practical issues that limited detection and identification of benzonatate as the cause of death.19,22 Benzonatate and its primary degradation metabolite, 4-(butylamino)benzoic acid (BABA) are not routinely measured in hospital laboratories and are probably found by specific investigation by medical examiners rather than mass screening. 22 Systemic concentrations reported included 2.5 mcg/mL in plasma and 0.680 mcg/mL to 35 mcg/mL in blood. 7 In one of the few pharmacokinetic studies, peak concentrations of benzonatate were under 1 mcg/mL. 19 Difficulties with determining benzonatate concentrations means that death from benzonatate is likely significantly underreported.
In general, patients received only supportive measures. Six patients received ILE. Intravenous lipid emulsion is recommended for life-threatening local anesthetic systemic toxicity but, it is unclear that it would benefit these patients. 23 Benzonatate is a unique anesthetic as it does not have a logD (0.32) that is favorable for ILE therapy, and as displayed in Table 1, only 1 out of 6 survived. It was unclear whether ILE benefited this patient with the numerous interventions ongoing in their care. Regardless it does not appear that ILE has a benefit in benzonatate ingestions.
This was a retrospective study and carries all the expected limitations with this type of research. As the majority of cases do not reach a full manuscript and many only in abstract form, there is significant publication bias; we were limited in what information was available for analysis from the published literature along with selection bias by limiting it to only the English language. Additionally, from the NPDS portion, poison control centers are receiving information about cases secondhand for the purposes of patient care and not research. Minor details of cases and information are often not reported in poison center data systems resulting in reporting biases. The addition of the fatality abstracts however adds more description to the cases we evaluated from NPDS. Although we included all fatalities, we did not include any “major” outcome cases as the limited information from just the coded data may not be as useful and studies utilizing coded data have already been performed.2,24 This exclusion of major or moderate cases biases the outcomes toward fatality. There is a risk of duplicating cases from the published literature and the NPDS fatality module. However, since we only included cases with descriptions of the clinical course, there were none which appeared to be the same case. There was one case published as both poster and manuscript that was resolved by reaching out to authors.
Conclusion
Overdose ingestions of benzonatate can cause significant toxicity with a rapid onset. Interventions performed were generally supportive in nature. Duration of directly toxic effects is short, but dramatic with neurologic devastation and resuscitated patients often still have a poor outcome.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_09603271211030560 - A description of the clinical course of severe benzonatate poisonings reported in the literature and to NPDS: A systematic review supplemented with NPDS cases
Supplemental Material, sj-pdf-1-het-10.1177_09603271211030560 for A description of the clinical course of severe benzonatate poisonings reported in the literature and to NPDS: A systematic review supplemented with NPDS cases by FS Minhaj and JB Leonard in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
