Abstract
Epididymis is a complex tubular structure of male reproductive system where spermatozoa undergo maturation and gain the fertilizing ability. Epididymal pseudostratified columnar epithelium with different cell types play imperative role by their secretory properties and enrich the luminal microenvironment necessary for achieving spermatozoal motility. During epididymal transit several secretory proteins like P26h, SPAG11, HSPD1 and many others are deposited on spermatozoal surface. At the same time spermatozoal proteins are also modified in this intraluminal milieu, which include cyritestin, fertilin, CE9 and others. Natural and anthropogenic activities disclose various environmental pollutants which affect different physiological systems of animals and human being. Likewise, reproductive system is also being affected. Fluoride causes structural alterations of caput and cauda segments of epididymis. Redox homeostasis and functional integrity are also altered due to diminished activities of SOD1, GR, Crisp2, Lrp2 and other important proteins. On the contrary arsenic affects mostly on cauda segment. Redox imbalance and functional amendment in epididymis have been observed with arsenic revelation as evidenced by altered genomic appearance of SOD, GST, catalase, Ddx3Y, VEGF and VEGFR2. This review is dealt with structure-function interplay in normal epididymal spermatozoal maturation along with subsequent complications developed under fluoride and arsenic toxicities.
Introduction
Spermatogenesis is exclusively dependent on close liaison in-between Sertoli and Leydig cells of testis. 1 However, the spermatozoa formed by the testis are immature and infertile.2,3 The spermatozoa need to be transported via epididymis to mature entirely and thus attain the competence to fertilize.4,5 The epididymis is sustained by an array of homeostatic control systems operated by hormonal and non-hormonal features, which coalesce to ensure spermatozoal maturation and fortification. It is a unique and fascinating accessory sex organ of the male reproductive system made up of super coiled and convoluted tubule restraining a nutritious luminal fluid required by the spermatozoa for the attainment of fertility.6,7 Structural orientation has sub-divided epididymis into three different regions like caput, corpus and cauda. 6 They are composed of various types of cells which in turn by their secretory activities help to create and maintain the luminal fluid composition for spermatozoal nourishment. 7 The cauda portion of the epididymis stores the matured spermatozoa and also plays an active role in spermatozoal nutrition and transportation. Thus, epididymal maturation of spermatozoa is considered crucial for the establishment of male fertility.
Quite a lot of natural and anthropogenic bustles bring about environmental pollution through exposure of several organic, inorganic, metallic and non-metallic compounds present in nature which lead to blemish of both terrestrial and aquatic ecosystems. Among the inorganic contaminants, as per the present day scenario, the increased presence of fluoride and arsenic in the environment is one of the key concerns of developing and developed countries. Both the geogenic and anthropogenic sources are responsible for simultaneous increase of fluoride and arsenic. Several industries such as pharmaceutical, microelectronics, mining, beverages, pesticide, glass and food are the major anthropogenic sources liable for environmental contagion with fluoride and arsenic. 8 It has been reported that above 50 percent population through the entire globe utilize ground water for household and drinking purposes 9 and most of people from various parts of the world are suffering from the toxic effects of fluoride and inorganic arsenic through the consumption of groundwater.10,11 Worldwide coexistence of fluoride and arsenic has been reported in Asia (Japan, Pakistan, Korea, China and India), Africa (Tanzania, Ethiopia, Nigeria and Ghana) and also in Latin America (Mexico, Argentina, Peru, Bolivia, Colombia and Chile). 12 Extensive survey revealed that fluoride and arsenic are the two major groundwater contaminants causing a set of health symptoms known commonly as fluorosis and arsenicosis respectively. 8 Reproductive toxicities are also associated with the health hazards also caused by these two pollutants. Waste water discharge in an indecent manner enhances the possibility of combined existence of fluoride and arsenic in the ground water. 8 The present literature summarizes the toxic effects of these two contaminants on the epididymal spermatozoal maturation which is considered as a very important feature in connection with fertility issue. Fluoride is the 13th most abundant element on earth crust and considered as potent environmental toxicant but unlike other toxicants it acts as di-sword weapon because at low concentration it has mitogenic effects but increased exposure might result in serious toxic effects on animals and human beings. 13 Arsenic being 20th most abundant element on earth crust is also considered as human carcinogen, exposure of which causes unpleasant human health consequences, which include hepatic, renal, cardiovascular disorders and also cancer in lung, liver, kidney, skin and urinary bladder. 14
Various reports have suggested that individually these elements are reprotoxic and cause reproductive disorders in both male and female systems. Fluoride has been found to impede with the reproductive system of animals. In male animals the consequences include diminished activity of the steroidogenic enzymes, reduced serum testosterone levels, decreased sperm count and motility, testicular damages, structural defects in spermatozoa along with oxidative stress.15–21 In female Sprague-Dawley rats, administration of sodium fluoride in drinking water notably reduces in the number of viable fetuses and increases in the resorption rate were observed. 22 Fluoride induced free radical toxicity was also reported in mice ovary.23,24 Some experimental studies on animals have testified that arsenic exposure is linked with spermatozoal toxicities,25,26 diminished testicular steroidogenic activity, reduction of testicular weight and also weight of accessory reproductive organs. 27 Arsenic intoxication is also associated with the inhibition of ovarian steroidogenesis and as well as secretion of gonadotrophins, 28 elevated adrenocortical steroidogenesis and level of plasma corticosterone. 29 Epidemiological study demonstrated that chronic arsenic exposure through drinking water causes adverse pregnancy conditions like premature delivery, neonatal death and spontaneous abortion. 30 Literature reports even suggested that exposure to high fluoride concentrations in drinking water is also linked with decreased human birth rates. 31 But the knowledge of the joint action of these two elements is lacking under male reproduction involving epididymis and the results derived from previous studies were inconclusive on epididymis regarding spermatozoal maturation under such reprotoxic environment. This review emphasizes normal structural and functional integrity of epididymis and altered spermatozoal maturation under toxic effects of fluoride and arsenic.
Biology of epididymis
Spermatozoa after development within testis go through one long, narrow and convoluted tubule which is known as epididymis. Spermatozoa undergo maturation after interacting with secretory proteins in the epididymal lumen and obtain progressive motility to fertilize ovum. According to histomorphological and ultrastructural features epididymis has been differentiated into three regions, which include head (caput), body (corpus) and tail (cauda). In some species such as mouse, the most proximal segment of epididymis is known as initial segment which is in addition to the earlier nomenclature. 32
Cellular distribution
Epididymis is lined by pseudostratified epithelium having different cell types, namely apical cell, basal cell, clear cell, halo cell, narrow cell and principal cell. The principal cells constitute about 80% of epithelium and responsible for maximum secretion within lumen. 32 The thickness of epididymal epithelium gradually decreases along the tubule 33 and due to water reabsorption; sperm concentration subsequently increases down the epididymal segment. All these factors are responsible for gradually increasing luminal diameter along the epididymis. 34 The other notable features along with functions of different cells of epididymal epithelium have been well characterized and are presented here (Figure 1).
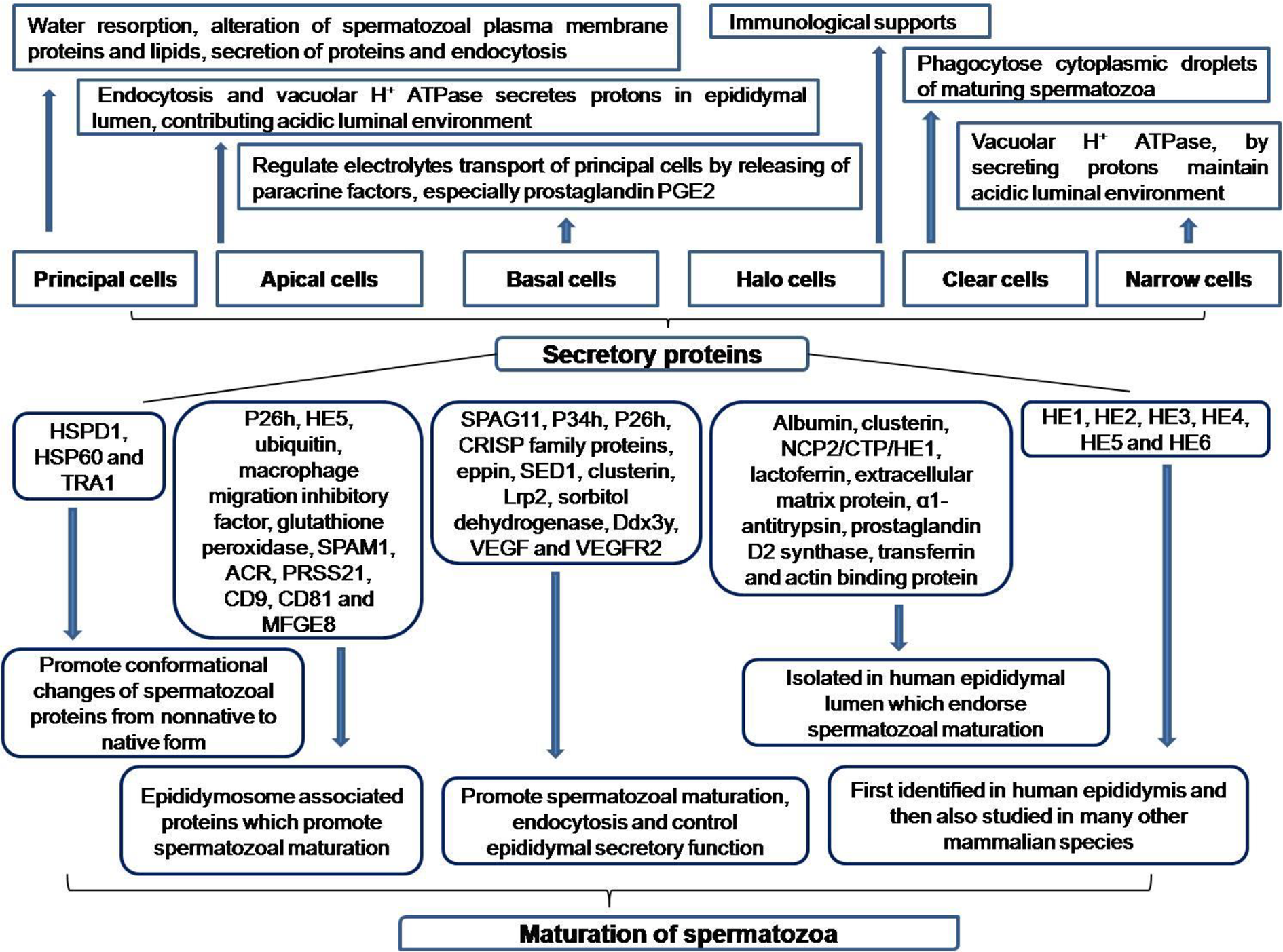
Functions of Epididymal cells and their secretory proteins.
Principal cell
The major constituents of epididymal epithelium are principal cells having secretory function in nature. Nuclei of principal cells of first portion of caput are spindle shaped but become gradually infolded in distal region of caput, corpus and especially in cauda. The intense nuclear lobation is an important characteristic feature of cauda region. Often a nuclear lobe is connected to another lobe by attenuated fold in nuclear envelope and in some instances, nuclear envelope displays continuation with endoplasmic reticulum, and thereby nucleus is invested with endoplasmic reticulum profile. 35 Numerous stereocilia, extended several micrometer, cover apical surface of the principal cells. Well developed rough endoplasmic reticulum and different sized mitochondria with distinctive lamellar cristae are other characteristic features of these cells. 35 Claudin and occludin are transmembrane proteins, responsible for formation of tight junctions, are also present between principal cells and thus form blood-epididymis barrier, which offers immunoprotective region in epididymal lumen necessary for maturation of spermatozoa. 36 Connexin, an integral protein forms gap junctions at apical and lateral surfaces of neighboring principal cells and permit the transportation of molecules having <1 kDa molecular weight. 32
Apical cell
Apical cells, found consistently in epididymal epithelium but not in huge number, extend to the lumen from basal lamina. It is narrow throughout its length but become wider near the lumen and luminal surface contains few short microvilli. Apically located nucleus appears as round or oval shaped having shallow infolding on nuclear envelope. One salient feature of apical cells is the presence of huge number of mitochondria, located in apical region. 35
Basal cell
The base of epididymal epithelium contains small cells, which reside on basal lamina throughout epididymal tubule and cytoplasm of these cells do not extend into the lumen. Round or ovoid nuclei are having with or without invaginations, few nucleolar and small heterochromatin elements. One key feature of these cells is the interdigitations of its plasma membrane with bordering principal cells. 35 The cells also contain coated pits on its apical plasma membrane and which are responsible for receptor mediated endocytosis of molecules derived from principal cells and blood. Accumulation of secretory materials in the Golgi saccules is the other characteristic feature of basal cells. 35
Halo cell
The cells contain round or intended nuclei, which are highly infolded occasionally having abundant heterochromatin. Cytoplasm possesses little amount of endoplasmic reticulum and few mitochondria. The key feature of this cell type is presence of cytoplasmic pseudopodia protruding between principal cells. 35 In adult animals, these cells are having helper T lymphocytes, cytotoxic T lymphocytes and as well as monocytes but not the B lymphocytes. Through the advancement with age, region specific enhancement of the number of each immune cell is observed along with occasional appearance of B lymphocytes and eosinophils. 37
Clear cell
The cells are prevalently present in cauda segment and are considered as primary immune cells in epididymis. 32 These cells are described by appearance of endosomes, lysosomes, coated pits, vesicles and multivesicular bodies in apical region whereas basal region contains nucleus and lipid droplets. Endocytic activity of these cells is much greater than adjacent principal cells, particularly in cauda region as executed by endosomes and lysosomes. Cytoplasmic droplets as released from spermatozoa during transmission through lumen are taken up by these cells. 33
Narrow cell
These cells are narrower than that of neighboring principal cells and are attenuated. The cells send fine processes of cytoplasm to reach the basement membrane. The key characteristic feature of these cells are the presence of numerous apically positioned cup-shaped vesicles which are involved in endocytosis and secretion of H+ ions into lumen. 38
Spermatozoal maturation in epididymal lumen
During epididymal transition from proximal to distal part, spermatozoa undergo several biochemical, morphological and physiological alterations, finally attain the progressive motion and also fertilizing ability. 32 Spermatozoal remodeling includes changes in dimension and the internal appearance of acrosome, alterations in the dimension of nucleus, migration of cytoplasmic droplets along sperm tail and structural changes in the intracellular organelles. 39 Phospholipid and cholesterol contents of spermatozoal plasma membrane are also changed. 40 Spermatozoa obtain the capacity for regulating cell volume by uptake of several luminal components, which function as osmolyte reserves, so that after exposure in female reproductive tract, spermatozoa do not undergo osmotic shock. 41
Secretory proteins and their roles
Numerous luminal secretory proteins and a number of spermatozoal proteins, which are modified during luminal transport and also the signaling event in caudal segment are documented here (Figure 1). Apocrine secretions through exosome and epididymosome are the main mode of transfer of luminal proteins to sperm plasma membrane.
42
Apical region of principal cells release blebs, known as epididymosome and it has been reported to occur in rat, bovine, mice, horse and human.
43
Caput being the most metabolic active region of epididymis, secrets about 70–80% of total protein released in epididymal lumen and during transit through this region about 99% of fluid is resorbed.
44
Most secretory proteins undergo trafficking through Golgi apparatus then undergo packaging and ultimately released from secretory granules to be delivered to sperm surface.
32
Proteins, secreted in luminal microenvironment are responsible for conformational change of the sperm proteins from non-native, misfolded and denatured form to native form, which include tumor rejection antigen 1 (TRA1), a member of HSP 90 family and heat-shock protein 1 (HSPD1 and HSP60).
32
A number of proteins, associated with epididymosome are transported to spermatozoa during their luminal transit and these are P26h (zona pellucida binding protein), HE5, ubiquitin, macrophage migration inhibitory factor and glutathione peroxidase.
45
Beside these proteins, SPAM1 protein, secreted from epididymal epithelial cells is incorporated in spermatozoal head through epididymosome and this protein helps to break the cumulus layer of egg.
46
Apart from SPAM1, Other two proteins serine protease PRSS21 and acrosin (ACR) play pivotal role in fertilization process by promoting spermatozoal motility and acrosome reaction.
46
Other transmembrane proteins, CD9 and CD81, which also represent epididymosome and promote signal transduction. MFGE8 is also present in epididymosome and is responsible for maturation of spermatozoa.
46
Quite a few secretory proteins interact with spermatozoa and provoke sperm maturations, which include SPAG11, P34h, P26h, CRISP family proteins, Eppin, SED1 and Clusterin.47–53 Other important proteins, secreted through the epididymis are Low-density lipoprotein receptor-related protein 2 (Lrp2), involved in endocytosis and sorbitol dehydrogenase, which controls epididymal secretory function.
54
Some proteins play important role in sperm maturation, which include Ddx3y, vascular endothelial growth factor (VEGF) and VEGF receptor 2 (VEGFR2).55,56 Several other secretory proteins, identified in human epididymal lumen are albumin, α1-antitrypsin, prostaglandin D2 synthase, clusterin, lactoferrin, extracellular matrix protein, transferrin NCP2/CTP/HE1 and actin binding protein.
57
Many novel genes had been first identified in human epididymis and then studied in many other mammalian species, which include HE1, encodes cholesterol binding protein; HE2, a β-defensin; HE3, having unidentified function; HE4, a proteinase inhibitor family; HE5 and HE6.
58
Conformational changes of spermatozoa proteins in epididymal lumen
Some spermatozoal proteins, which are synthesized during spermatogenesis, undergo modification during epididymal transit in terms of proteolytic processing or deglycosylation. These proteins include ADAM family members cyritestin and fertilin, CE9, α mannosidase, endoplasmin, β-galactosidase, Grp78/Hsp70, phosphatidylethanolamine binding protein, basigin, α-enolase, lactate dehydrogenase, cytokeratin, testis lipid-binding protein, and β-subunit of F1-ATPase (Figure 2).59–64
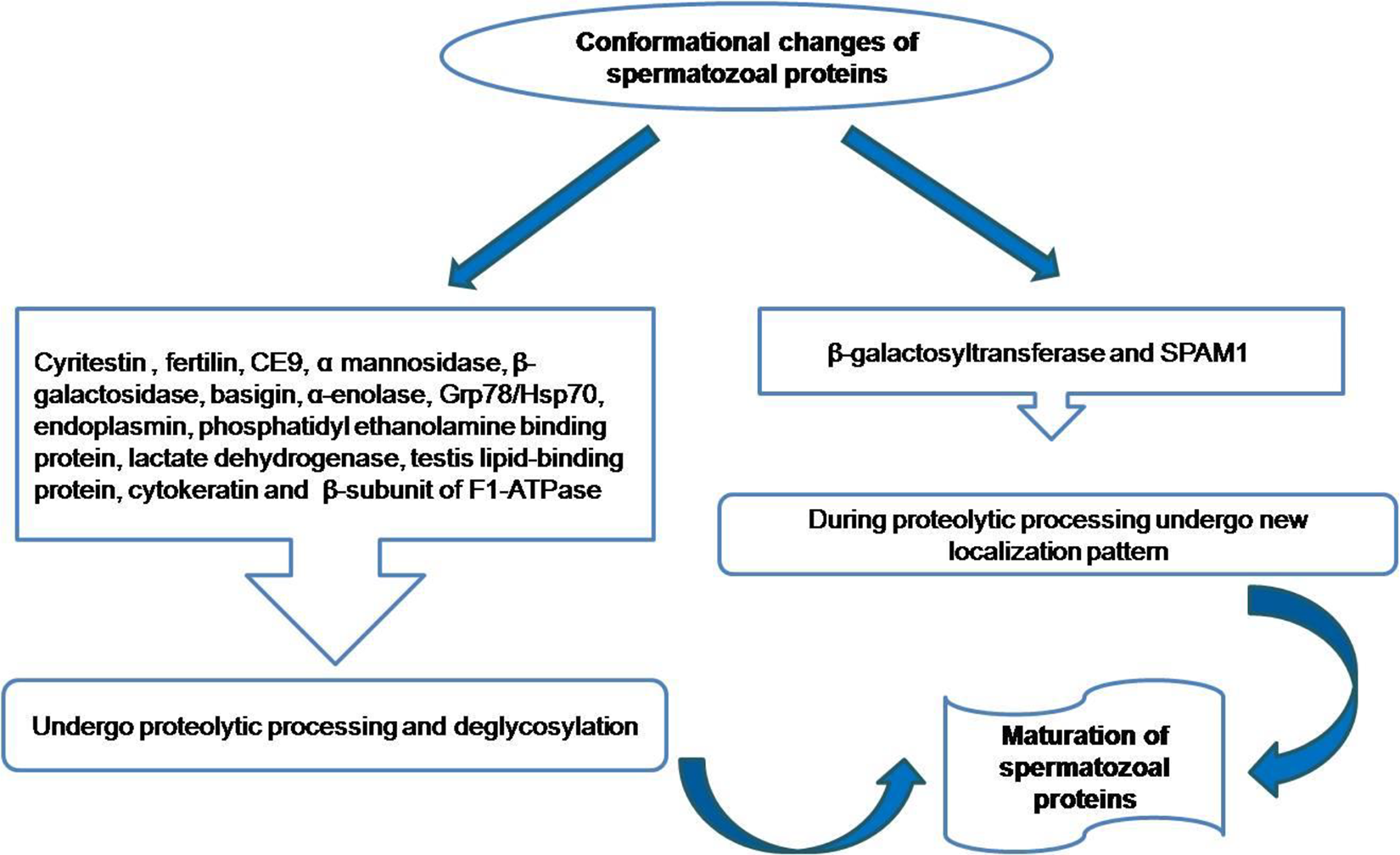
Conformational changes of epididymal spermatozoal proteins.
During proteolytic processing in epididymal lumen, some spermatozoal proteins undergo new localization pattern, which include β-galactosyltransferase and SPAM1(Figure 2). 65
Final maturation in cauda and molecular cascade
Study on spermatozoal motility clearly indicated that asymmetrical beating of flagella as observed in caput was seen to be changed to symmetrical forward motion in cauda (Figure 3).
66
Scientific data regarding the achievement of spermatozoal motility is still unclear but the available information pointed out that intracellular cyclic adenosine monophosphate (cAMP) generation by activating the enzyme adenylate cyclase and subsequent phosphorylation of protein kinase A and many other proteins are the key factors.
67
It was also observed that the concentration of cAMP in spermatozoa increases gradually from corpus to cauda
57
that leads to the increase of metabolic competence.
68
Presence of calcium and bicarbonate in the luminal fluids directly control spermatozoal cAMP concentration and subsequent activation of phosphorylation of other proteins and also spermatozoal motility.
69
Another In-vitro study explains that the combined presence of calcium and bicarbonate help in attainment of flagellar motion of mature spermatozoa.
70
Mass spectrometry analysis has revealed alterations of protein profile between immature spermatozoa from caput and mature spermatozoa from cauda. Presence of phosphorylated proteins like heat-shock protein 70, β-tubulin, actin, glucose-regulating protein, lactic acid dehydrogenase, mitochondrial protein aconitase and also β subunit of F1 ATPase as observed only in caudal spermatozoa which may be considered as the marker of matured sperm.
71
Hence the presence of regulatory molecules in cauda and phosphorylation of sperm proteins symbolize a key maturational step of spermatozoa. Another vital protein, predominantly secreted from cauda region is beta-defensin 126, absorbed in spermatozoa.
72

Molecular cascades involved in maturation caudal spermatozoa.
Effects of fluoride toxicity
Structural amendments
Fluoride is responsible for a lot of changes in testicular structure and also has a large impact on histomorphometry of entire epididymis. The associated changes in both caput and cauda are documented (Figure 4).

Effects of fluoride and arsenic toxicity on structure—function of epididymis.
Caput: The first portion of epididymis from untreated animals structurally characterized by many tubules covered with pseudostratified columnar epithelial cells having numerous stereocilia in the luminal surface and intertubular interstitium present between tubules. Secretory granules are abundantly present in epithelial cells and tubular lumen is filled with bundles of sperm. An experimental study by intoxication with sodium fluoride to mice at 10 and 20 mg/kg body weight/day for 30 days revealed increased tubular diameter, nuclear pyknosis of secretory epithelial cells and absent of tubular spermatozoa. 73 When rabbits were exposed with NaF at 10 mg/kg body weight/day for the duration of 23 months it results drastic changes such as presence of fewer stereocilia or complete absent of stereocilia in many areas on epithelial cell line, presence of less abundant secretory granules in tubule and even in epithelial cells, increase of luminal diameter and decrease of height of epithelium 74 but the same experimental study for the duration of 20 months results no significant structural alterations.
Cauda: This region of untreated animals are morphologically characterized by presence of circular and regular tubules having larger diameter than caput, presence of pseudostratified columnar epithelium in tubular lining, presence of intertubular interstitium, presence of blood vessels in extratubular space, dense layer of stereocilia protruding from apical surface of principal cells toward luminal side and tubular lumen occupied by healthy spermatozoa having well defined head and tail. According to Chinoy et al., mice from treated group (10 and 20 mg/kg /day for 30 days) showed a massive changes of caudal structure in terms of decreased height of epithelial cells, larger tubules having denudation of cells within lumen and tubules are devoid of spermatozoa. 73 Extensive morphological alterations also found in sodium fluoride treated (10 mg/kg body weight/day for 23 months) rabbits and these alterations has been exemplified as bald appearance of pseudostratified epithelium due to less dense or absence of stereocilia, fewer spermatozoa in the tubular lumen, reduced number of secretory granules, decreased epithelial height and increased tubular diameter but animals having same treatment dose for 20 months devoid of such alterations 74 . Fluoride is also responsible for alterations the caudal structure in a dose dependent manner (100, 200, 300 ppm of NaF/day for 40 days) in rats. Structural evaluation by histology and Scanning Electron Microscopy (SEM) revealed that treatment with 100 ppm of NaF results disruption of basement membrane, vacuole formation in germinal epithelial cells, reduced sperm number and less stereocilia in lumen. Structural analysis of cauda in second treated group (200 ppm) showed epididymal hyperplasia, less spermatozoa and presence of cellular debris, disintegrated connective tissues and presence of very less stereocilia. Ultrastructural analysis by SEM divulges same outcome. Severe alterations have been noted in highest treated group (300 ppm) such as reduced lumen diameter with very few spermatozoa and huge exfoliated germ cells, inflammatory infiltration having lymphoid aggregates, distortion of basement membrane, vacuolation in germinal epithelium, focal infolding in epithelium with pseudo-glandular appearance and hyperplasia was also noted in caudal region. SEM analysis revealed bald appearance due to absence of stereocilia and presence of fragments in epithelial surface. 75
Functional alterations
A recent experimental study has explored altered epididymal protein expression, which in turn impedes epididymal functions (Figure 4). Lrp2 is expressed through the entire length of epididymis, which participates endocytosis and as well as lysosomal degradation of several ligands and this protein is known to be expressed anomalously upon fluoride exposure. 54 Sorbitol dehydrogenase, encoded by a key gene Sord is involved to maintain epididymal secretory functions and fluoride has been known to alter the expression of Sord gene and disrupts its function. 54 An important protein, secrets preferentially from cauda epididymis is beta-defensin 126, which is absorbed through post acrosomal region and tail of spermatozoa 72 and regulates spermatozoa activity by increasing the swimming ability in cervical mucus and also ensure protection from immune system within female reproductive tract. 76 Fluoride also amends the expression of beta-defensin 126. 54 Crisp1 and Crisp2 are family members of cysteine rich secretory protein (Crisp). Crisp1 glycoprotein secreted from epididymal epithelium inhibits sperm capacitation and also suppresses sperm-egg fusion 77 ; Crisp2 is also involved in male fertility. 78 Altered expression of these epididymal proteins has been reported upon sodium fluoride intoxication and that leads to male infertility. 54 Recent report also revealed that fluoride downregulates the expression of SPAM1, CD9 and CD81 proteins in entire cauda epididymis, which cause obstruction of the sperm maturation and as well as inhibition of sperm egg binding. 46 This study has revealed that spermatozoa deficient with ACR protein show decreased progressive motility toward the cumulus cells than wild type spermatozoa and PRSS21 disrupted spermatozoa exhibit fragile neck in addition to slower motility rate. The study further states that fluoride exposure down-regulates both mRNA and protein expression of ACR and PRSS21 protein in cauda epididymis 46 and this might be one possible mechanism by which fluoride affects spermatozoal progressive motility and fertilizing ability. CRISP2, a CRISP family protein which promotes spermatozoal motility and sperm egg fusion has known to be declined after fluoride exposure. 79
Redox balance
It is significantly approved that redox balance is crucial for standard physiological function of a living system and this balance is sustained following combat between internal antioxidant mechanisms (both enzymatic and nonenzymatic) with reactive oxygen species (ROS) generated in the living system. However, in living system, interactions with any kind of xenobiotic compound triggers potentially damaging events, which can directly or indirectly confront redox homeostasis. Fluorine, being a highly ionized element can interrupt oxygen metabolism and promotes increased generation of O2- free radicals, which in turn promotes the formation of other free radicals like hydroxyl, hydrogen peroxide and peroxynitrite. 80 In case of mammalian tissues super oxide dismutase (SOD) is a central antioxidant enzymatic component having three different isozymes, CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3). 80 Among these SOD1 is crucial scavenging enzyme catalyzes O2- into H2O2 and another vital enzyme to remove free radicals is catalase (CAT). 81 An experimental study by intoxication with sodium fluoride through drinking water to mice at different doses (25, 50 and 100 mg NaF/L) for 10 weeks results significant decreased mRNA expression of SOD1 and CAT in epididymis at all three treated groups. 54 From this it can be concluded that fluoride alters normal homeostasis between oxidative stress and antioxidant enzymes in epididymal tissues (Figure 4).
Glutathione subsists in thiol-reduced (GSH) and disulfide-oxidized (GSSG) forms and both are found in about all mammalian tissues and defend against oxidative stress. 82 When glutathione is converted from reduced (GSH) to oxidized (GSSG) form, GSH peroxidase (GPx) reduces lipid peroxide and hydrogen peroxide and during conversion from GSSG to GSH, GSSG reductase (GR) is involved at the expense of NADPH. 82 Organic peroxides (ROOH) are reduced by either GPx or by another enzyme glutathione S-transferase (GST). 82 An experimental study by exposure of NaF at 25 and 100 mg/L for 60 days to mice results decreased mRNA and protein expression of GPx, GST and GR in epididymis in a dose dependent manner and this study has explored that fluoride affects epididymal antioxidant status by changing glutathione related enzymes. 54
Effects of arsenic toxicity
Structural amendments
Several studies have stated that arsenic toxicity is responsible for the decrease in the weight of testes along with epididymis at the organ level.83–85 Structural detoriation of epididymis has also been reported upon arsenic threat (Figure 4). Histological observations in arsenic affected epididymis of goat (obtained from arsenic prone zone) revealed altered histoarchitecture in terms of irregular and dense connective tissue, elastic fibers and also collagen fibers as compared to control animal group. Lesser number of spermatozoa in the lumen has also been noted in treated animals. 86 Photomicrographs of cauda epididymis from sodium arsenate (25 ppm for 70days) exposed rat highlighted architectural disarray of pseudo-stratified columnar epithelium with fewer or devoid of spermatozoa in the lumen. 84 A recent study on arsenic exposed epididymis of pre-pubertal rats (52 and 82 days age) reported cribiform appearance in the epithelium, lack of spermatozoa in the lumen, presence of vacuoles and cell debris indicating disruption of luminal microenvironment leading to impairment of spermatozoal maturation. 87
Functional alterations
It is well established that arsenic after entering the normal homeostatic system, increases ROS generation and oxidative stress. 88 Literature survey revealed that apoptosis is triggered by high levels of ROS in the testis and accessory reproductive organs 89 ; eventually cause low sperm count, motility and finally triggering altered spermatozoal quality.27,83 It has been reported that arsenic accumulates in male reproductive tissues, including prostate, seminal vesicles, testis and also in epididymis.83,90 Epididymal arsenic accumulation leads to low spermatozoal viability 90 as a consequence of spermatozoal DNA damage coupled with spermicidal activity.91,92 According to the report of Li et al., 55 the sub-chronic exposure of arsenic significantly down-regulates the expression of Ddx3y gene and protein in the epididymis of mice, suggesting that this may affect maturation of sperms in the epididymis. Although their results showed that arsenic exposure had detrimental effects on spermatogenesis and sperm development through down-regulating the expression of Ddx3y gene but its effect on fertility of the exposed animals are yet to be confirmed. Further investigation is required to justify adverse effect of sub-chronic exposure of arsenic on fertility of the male animals through down-regulating the expression of this gene and also to know the precise molecular mechanisms on suppression of this gene.
VEGF being involved in reproductive system endorses angiogenesis and also plays a vital role to resistance apoptosis. VEGF and VEGFR2 are involved in sperm maturation and expressed in testis, epididymis, prostate and seminal vesicle. It has been reported that the expression of mRNA and protein level has been declined in epididymis of rat upon arsenic threat, which alter epididymal microenvironment and sperm maturation. 56 This study suggested that it might be one of the mechanisms of male infertility caused by arsenic poisoning (Figure 4).
Redox balance
Expression of genes, which instruct antioxidant enzymes are used to assess the extent of toxicity of any environmental pollutant. It has been reported that sodium arsenite causes declined mRNA expression of epididymal SOD1, SOD2, CAT and GST, which undoubtedly impede epididymal redox status and leads to reproductive disorders (Figure 4). 93 A recent finding has further elucidated that arsenic enhances the NO2/NO3 levels in the epididymis and thereby promotes nitrosative stress. 94 Another study on arsenic exposed epididymis of post-natal rats (52 days age) has reported a significant up-regulation of SOD1, CAT and GSTK1 genes in response to combat with the arsenic-induced oxidative stress. 87 Increase of MDA level and decrease of enzymatic CAT activity have also been observed also suggesting altered redox state. 87 Recently Souza ACF 93 has reported that arsenic exposure on post-natal rats of 14 and 28 days caused increased activity of CAT to neutralize arsenic induced reactive species but exhaustion occurred finally reducing the activity below normal level. Souza ACF 93 has also stated that arsenic induced reduction of epididymal GST activity in post-natal rats of 28 days validating the malfunction of antioxidant enzyme that act against arsenic mediated oxidative stress.
It is well known that micronutrients are essential for production of antioxidant enzymes. Manganese is required for the activity of SOD in mitochondria, whereas zinc and copper are essential for the same enzyme activity in cytosol. Similarly iron is essential for catalase activity and selenium is required for GPx activity. Imbalance of these trace elements has also been reported in rat epididymis upon arsenic exposure, which might be probable reason underlying altered antioxidant defense. 94
Conclusion
Descendent of testicular spermatozoa through the epididymis is prerequisite to gain progressive motility and fertilizing ability. The functional maturation of spermatozoa is accomplished by incessant interaction with epididymal intraluminal milieu. Secretory activities of epithelial cells endow with several proteins which are deposited on spermatozoa and a number of spermatozoal proteins are also modified during luminal transportation. Over exposure of fluoride and arsenic affects structural integrity as well as intraluminal microenvironment of epididymis resulting distorted epithelial lining along with decreased luminal spermatozoa. Altered redox homeostasis is another disadvantageous outcome as evidence by anomalous genomic and proteomic expression of antioxidant molecules. Expression of crucial biomarkers, responsible for spermatozoal maturation gets altered under such reprotoxic state. Cumulative outcome of these experimental findings clearly pointed out that excessive exposure of fluoride and arsenic decline the structure-function entity of epididymis which gradually impair male fertility.
Footnotes
Abbreviations
Author contributions
PP and SB equally contributed in literature search and writing of the manuscript. PKM is responsible for revision of the article. All authors read and approved the final manuscript.
Acknowledgment
The author’s would like to acknowledge the FRPDF grant of Presidency University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
