Abstract
Autism Spectrum Disorder (ASD) is a complex neurodevelopmental disease characterized by cognitive and sensorimotor impairment. Numerous research findings have consistently shown that alteration of Smo-Shh (smoothened-sonic hedgehog) signaling during the developmental process plays a significant role in ASD and triggers neuronal changes by promoting neuroinflammation and apoptotic markers. Purmorphamine (PUR), a small purine-derived agonist of the Smo-Shh pathway, shows resistance to hippocampal neuronal cell oxidation and decreases neuronal cell death. The goal of this study was to investigate the neuroprotective potential of PUR in brain intoxication induced by intracerebroventricular-propionic acid (ICV-PPA) in rats, with a focus on its effect on Smo-Shh regulation in the brain of rats. In addition, we analyze the impact of PUR on myelin basic protein (MBP) and apoptotic markers such as Caspase-3, Bax (pro-apoptotic), and Bcl-2 (anti-apoptotic) in rat brain homogenates. Chronic ICV-PPA infusion was administered consecutively for 11 days to induce autism in rats. In order to investigate behavioral alterations, rats were tested for spatial learning in the Morris Water Maze (MWM), locomotive alterations using actophotometer, and beam crossing task, while Forced Swimming Test (FST) for depressive behavior. PUR treatment with 5 mg/kg and 10 mg/kg (i.p.) was administered from day 12 to 44. Besides cellular, molecular and neuroinflammatory analyses, neurotransmitter levels and oxidative markers have also been studied in brain homogenates. The results of this study have shown that PUR increases the level of Smo-Shh and restores the neurochemical levels, and potentially prevents morphological changes, including demyelination.
Introduction
Neurological disorders are a heterogeneous group of diseases of the nervous system having distinct etiologies that reflect illness in the brain’s specific regions and nervous tissues that regulate essential physiological functions, including learning and memory, posture and synchronization of nerve/muscle movements. Autism spectrum disorder (ASD) is also one such neurodevelopmental disorder that involves neurological abnormalities in brain regions such as the neocortex, hippocampus, amygdala, and basal ganglia.1,2 ASD includes several symptoms that differ in severity and variability between individuals. 3 ASD influences the number of social and communication deficits, coupled with repeated and irregular motor-sensory behaviors that affect the person’s daily routine. 4
Single chromosomal or genetic aberrations are among the causative factors that affect neurodevelopment in ASD. 5 Recent studies were based on multiple genes and copy number variations (CNVs). ASD-associated CNV, usually a duplication or deletion of neuropsychiatric phenotypes at a specific chromosomal locus, 16p11.2, strongly indicates an additive association between inherited polygenic and de novo risk. Another study examined the phenotype associated with CHD8 (Chromodomain helicase DNA binding protein 8) gene mutations in several exons sequencing studies associated with ASDs. 6 The process of neurodevelopment during pre and early postnatal development can be altered by environmental agents, such as chemical toxins, including thalidomide, valproic acid, ethanol, and propionic acid (PPA).4,7
PPA is a short-chain fatty acid and a metabolic end-product of enteric bacteria species such as Clostridia, Desulfovibrio&Bacteriodetes contained in the gut. 8 The leading cause of autism nowadays is the massive intake of processed foods with high rates of PPA as a preservative by pregnant women, which increases the risk of autism in newborns. 9 PPA can easily pass through the placental barrier and reach the fetus. Once PPA penetrates and accumulates in the undeveloped fetus brain, it severely affects the neural stem cells and other glial cells such as astrocytes and oligodendrocytes by downregulating the smoothened-sonic hedgehog (Smo-Shh) signaling pathway. Reduction in oligodendrocytes can result in demyelination, which is indicated by a decreased auditory brainstem response velocity.10,11
PPA and other short-chain fatty acids such as butyrate and acetate affect different physiological processes such as neurotransmitter synthesis and release, 12 increase in oxidative stress, 13 and release of neuroinflammatory markers such as TNF-α and IL-1β. 14 PPA also severely influences the apoptotic markers such as caspase, Bax, and Bcl-2, further affecting Smo-Shh signaling.15,16
Shh is a fascinating signaling molecule, become active on the patched binding receptor (Patch1). This binding relieves Ptch-mediated inhibition on the Smo receptor. Further, it promotes the transcription factor “Glioma-associated oncogene” (Gli) translocating to the nucleus and regulating the target gene’s transcription and expression.17,18 Shh plays an essential role in the forebrain’s growth, hindbrain, spinal cord and proliferation, differentiation, and axonal targeting. 19 Shh pathway also activates during adult brain injuries, neurodegenerative disorders, and neurogenesis. 20 Shh signaling is responsible for embryonic brain development by regulating cell differentiation and proliferation. Smo-Shh’s downregulation leads to the activation of different neuropathic markers in the brain and affects neurogenesis. This further affects cell proliferation and cell survival among neuronal cells and alters intercell communications. 21 Also, various brain mediators increase their expression, trigger neuroinflammation, oxidative stress, and excitotoxicity. All modification contributes to the underdevelopment of the fetus brain, and after birth, triggers multiple cognitive and other behavioral abnormalities.22,23
Purmorphamine (PUR), a purine derivative, and a small molecular modulator of Smo-Shh signaling induce Smo agonism and promote the multipotent mesenchymal differentiation progenitor cells. PUR directly interacts with the Smo receptor and responsible for its agonism. 24 PUR protects neurons present in the hippocampus against oxidative stress and shows neuroprotective and regenerative properties. 25 PUR has generally been discovered to preserve the cortical neurons and restore neurological disabilities in rats following ischemic stroke. 26 The upregulation of Shh signaling by PUR may treat and delay the progression of neuro abnormalities linked with autism and serve as a preventive treatment for ASD. 27 In several studies, PUR has also been shown to facilitate barrier formation and plays a role in stimulating the central nervous system’s endogenous anti-inflammatory response.
A recent study by Chechneva et al., 26 PUR reveals a defensive mechanism against deficits in long-term memory and learning and beneficial effects on synaptic impairments and reduction in neuroinflammation and ROS level. The relevance of PUR as a small Smo-Shh molecular agonist position it as a treatment target of several neurological disorders like autism, 5 multiple sclerosis (MS), 28 amyotrophic lateral sclerosis (ALS), 29 Alzheimer’s disease (AD), 30 Parkinson’s disease (PD), 21 Huntington’s disease (HD) 31 and many more. Apart from neurological PUR, it is actively involved in osteogenesis to promote osteoblast markers in C3H10T1/2 cells.32–34
The activation of SHH with its agonist, PUR, may act as a potential preventive therapy for symptomatic relief in autism. Therefore, throughout this study, we explore PUR’s neuroprotective role on behavioral, neurochemical, histopathological, and morphological parameters in ICV-PPA-induced experimental rat model of autism. We confirmed the importance of the Smo-Shh signaling mechanism for autism pathogenesis and found that PUR has significant neuroprotective effects in ASD.
Material and methods
Experimental animals
Adult Wistar rats (250–300 g, total animal 36, total group six, each group contain six animals: 18 male and 18 female) obtained from the Central Animal House, ISF College of Pharmacy, Moga, Punjab. The animals with a wire mesh top and soft bedding were housed in polyacrylic cages. All animals were kept under standard husbandry conditions of 12 h reverse light-cycle with food and water (ad libitum), maintained at 23 ± 2°C. The experimental protocol was approved by the Institutional Animal Ethics Committee (IAEC) with registration no. 816/PO/ReBiBt/S/04/CPCSEA as protocol no. ISFCP/IAEC/CPCSEA/Meeting No.25/2019/Protocol No 427 as per the guidelines of the Government of India. Animals were acclimatized to laboratory conditions before the experiment.
Drugs and chemicals
PPA was purchased from Sigma–Aldrich (USA). PUR obtained from Cayman Chemicals Co Pro lab Marketing, New Delhi India. All other chemicals used in the study are of analytical grade. Drug and chemicals solutions were prepared fresh before use. PUR dissolved in DMSO (soluble in water) injected intra-peritoneally (i.p.).
Experimental protocol schedule
The total experimental protocol schedule was 44 days. 7 Animals were randomly assigned to six groups. Group 1 Vehicle Control; Group 2 Sham control; Group 3 PUR perse (10 mg/kg, i.p.); Group 4 PPA (10 µl/0.26 M, i.c.v.); Group 5 PPA (10 µl/0.26 M, i.c.v.) + PUR (5 mg/kg, i.p.); Group 6 PPA (10 µl/0.26 M, i.c.v.) + PUR (10 mg/kg, i.p.). Chronic administration of the ICV-PPA is given to induce autism. ICV-PPA administration was given from day 1st to 11th, whereas the protocol drug PUR started on day 12th and continue until day 44th. Behavioral parameters such as MWM, locomotor activity, beam crossing task, and FST were carried out on specific days. On the 44th day of the protocol schedule, all the same, animals on which behavioral parameters were performed were sacrificed by decapitation and their brains isolated for biochemical, inflammatory, and neurochemical estimations. The experimental protocol is summarized in Figure 1.

Experimental protocol schedule.
Experimental model of ASD
The PPA-induced experimental model of autism in the rat was conducted according to the method established by Sharma et al. 7 Rat acclimatized to the laboratory conditions and then anesthetized through intraperitoneal ketamine injection (75 mg/kg, i.p.). The anesthetized rat’s body was placed on a warm padded surface with the head placed in the stereotaxic frame (Stoelting Co., Wood Dale, IL, USA). The head was positioned in a symmetrical position with the aid of the incisor bar and ear bars. Before starting the surgical procedure, the head position was adjusted so that the bregma and lambda coordinates aligned at the same level. After the rat head has been shaved, the scalp is washed with 70% ethanol. A small incision is made of a scalpel (mid-sagittal), skin retracted, and skull exposed to bregma and lambda to facilitate the ICV injection co-ordinates’ determination. Cotton buds are used to prevent bleeding. The cotton swabs are immersed in normal saline in the eyes of rats to prevent dehydration.
Under the aseptic conditions, rats were surgically implanted with the 2.5 cm long cannula at the following coordinates concerning Bregma: anterior/posterior (AP) −1.4 mm; medial/lateral (ML) 1.8 mm; dorsal/ventral (DV) −3.0 mm. The Burr hole was then repaired using dental cement, and the surgical incision was then sutured using an absorbable surgical suture and sterile surgical needle. A removable plastic ear pin was used to seal the cannula. After surgery, all rats were immediately injected intraperitoneally with gentamycin (35 mg/kg) to prevent sepsis, and lignocaine gel was applied to the sutured region to reduce pain. Neosporin powder was sprinkled to avoid bacterial skin infections and followed up to three days.
From day 1st to 11th, PPA (10 µL of a 0.26 M solution) was injected through a 0.4 mm outer diameter hypodermic needle attached to a 10-µl Hamilton microliter syringe in the left lateral cerebral ventricle for 10 min (1 μl/min) duration. The Hamilton® microneedle did not displace for 5 min after injection to promote drugs’ diffusivity in CSF. 35 In post-operative treatment, rats were housed individually in polyacrylic cages containing warm cloth and husk, and special care was performed till spontaneous movement was restored. After anesthesia, around 2–3 h 36 ; Room temperature was held at 25 ± 3°C. Milk and glucose water was held in the cages for 2–3 days to prevent any physical trauma following surgery. Clinical signs were routinely tracked, including general body condition and dehydration. After 7 days, rats were given a proper diet and water intake and recovered spontaneous mobility showing signs of recovery.
Parameters assessed
Measurement of body weight
Bodyweight was observed on the experimental protocol schedule day 1st, 13th, 23rd, 33rd, and 43rd. 37
Behavioral parameters
Beam crossing task
Each animal’s motor coordination ability was tested on day 1st, 13th, 23rd, and 43rd of the protocol schedule. The number of slips in each trial was recorded, and additionally, the motor performance of rats was scored on a scale ranging from 0 to 4. A score of 0 was assigned to an animal that could readily transverse the beam. Scores 1, 2, and 3 were given to animals that demonstrated mild, moderate, and severe impairment, respectively. Score 4 was assigned to the animals completely unable to walk on the beam. 38
Spontaneous Locomotor activity
Animals were tested for locomotor activity on day 1st, 13th, 23rd, 43rd, using an actophotometer (INCO Group of Companies Dubai, United Arab Emirates). Each animal was observed in a square, the closed arena for 5 min, equipped with photocells sensitive to infrared light. A digital actophotometer value started as counts per 5 min. 14
Forced swim test (FST)
Testing for FST was performed on day 1st, 13th, 23rd, and 43rd to evaluate rats’ depressive behavior because depression is also the predictor of suicidal thoughts or attempts among children with autism. In the training period, the first exposure of rats in the tank is for 15 minutes, and the second is performed 24 h after the first, with an exposure period of 5 min. The rat’s testing period consists of a single 6-min exposure, with the first 2 min serving as a habituation period and the last 4 min consisting of the test itself, which yields the duration of immobility. 39
Morris water maze task (MWM)
On day 40th, 41st, 42nd, 43rd, and 44th of the protocol schedule, spatial learning and memory of animals tested in Morris water maze (INCO Group of Companies Dubai, United Arab Emirates). Water (25 ± 1°C) was filled in a circular water tank to a depth of 40 cm, and the hidden platform was submerged down 2 cm in water. 40 The acquisition phase includes training sessions of 4 trials per session (once from each starting point) for 5 days of day 40th, 41st, 42nd, 43rd, and 44th, and escape latency (ELT) to reach the hidden platform was noted, having ceiling time of 120 s. If the rat failed to locate the hidden platform within the maximum time of 120 s, it was gently placed on the platform and allowed to remain there for 20 s. Twenty-four hours after the acquisition phase, a probe test (day 44) was conducted by removing the platform. Rats were allowed to swim freely in the pool for 120 s, and TSTQ (Time spent in target quadrant) was recorded. TSTQ indicates the degree of memory consolidation, which had taken place after learning. 41
Estimation of biochemical parameters
Brain homogenate preparation
On the 45th day of the protocol schedule, animals were sacrificed by decapitation; brains were removed and washed with ice-cold isotonic saline solution. Brain samples were then homogenized with 10 times (w/v) ice-cold 0.1 M phosphate buffer (7.4). The homogenate was centrifuged at 10,000×g for 15 min, the supernatant was separated, and aliquots were used for biochemical estimation. 42
Measurement of cellular and biochemical marker
Measurement of SMO-SHH levels
Rat ELISA kit from Elabsciences, China, was used to measure SMO-SHH levels in rat’s brain homogenate supernatant, and the values are expressed in ng/ml.
43
Measurement of myelin basic protein (MBP)
Rat MBP level measured using ELISA kit from Elabsciences, China in rat’s brain homogenate supernatant, and the values are expressed in μg/mg total protein.
44
Measurement of Caspase 3
Caspase-3 levels were measured by ELISA kit (Elabsciences, China) in the brain homogenate. The procedure was performed according to the instructions of the manufacturer. The assay employs the enzyme immunoassay competitive method. The level is expressed in ng/gm protein.45,46 Measurement of Bax and Bcl-2 level
Bcl-2 protein and Bax protein levels were measured by ELISA kit (Elabsciences, China) in the brain homogenate. The procedure was performed according to the instructions of the manufacturer. The level is expressed in ng/mg protein. 47
Neurotransmitter’s evaluation
Serotonin levels
The serotonin level in rat brain homogenate analyzed by HPLC-ECD Waters, USA, fitted with an electrochemical detector. The mobile phase consisted of buffer (sodium citrate), and acetonitrile (ACN) in the ratio of 87:13, v/v (pH 4.5). Itis maintained for smooth separation at a flow rate of 1 ml/min, and findings are reported as ng/mg protein.
48
Glutamate levels
Following derivatization with o-phthaldehyde / β-mercaptoethanol (OPA / β-ME), glutamate was quantified. A quantitative analysis was performed using an electrochemical detector (ECD), high-performance liquid chromatography by HPLC-ECD Waters, USA in the tissue sample, and findings are reported as ng/mg protein.
49
Dopamine (DOPA) levels
Using an electrochemical detector (ECD), high-performance liquid chromatography (HPLC) ECD Waters, USA was used to measure dopamine levels in the rat brain. The mobile phase was buffered with sodium citrate (pH 4.5)–acetonitrile (87:13, v/v). The Sodium citrate buffer comprises 10 mM of citric acid, 25 mM of NaH2HPO4, 25 mM of EDTA (ethylene diamine tetraacetic acid) 2 mM of 1-heptane sulfonic acid. The experiment’s electrochemical conditions were +0.75 V, and sensitivity ranged from 5 to 50 nA. The separation was performed at a flow rate of 0.8 ml / min. The samples (20 μl) were manually injected. Samples of rat brains were homogenized in a homogenizing solution containing 0.2 M perchloric acid and were centrifuged for 5 min at 12,000 g. Results are expressed as ng/mg protein.
48
Acetylcholine (Ach) levels
The level of acetylcholine in rat brain homogenate was measured using a diagnostic kit from Krishgen Diagnostics India. Both samples and reagents had been prepared according to the description of the kit. The optical density of the reaction mixture in the microtiter plate was determined at 540 nm, and the results are expressed as ng/mg protein. 50
Evaluation of neuroinflammatory cytokines
TNF-α level
The level of TNF-α in rat brain homogenate was measured using a diagnostic kit from Krishgen Diagnostics India. All samples and reagents were prepared according to the description of the kit. The optical density of the reaction mixture in the microtiter plate was determined at 450 nm, and the results are expressed as pg/mg protein.
51
IL-1β levels
The level of IL-1β in rat brain homogenate was measured using a diagnostic kit from Krishgen Diagnostics India. Both samples and reagents had been prepared according to the description of the kit. The optical density of the reaction mixture in the microtiter plate was determined at 450 nm, and the results are expressed as pg/mg protein. 52
Evaluation of oxidative stress markers
Acetylcholinesterase (AChE) levels
The quantitative measurement of acetylcholinesterase activity in the rat brain was performed by UV spectrophotometer (Pharmaspec, Shimadzu, USA). The assay mixture contained 0.05 ml supernatant, 3 ml 0.01 M sodium phosphate buffer of pH 8, 0.10 ml iodide acetylthiocholine, and 0.10 ml DTNB (Ellman reagent). Spectrophotometrically, the absorbance was immediately measured at 412 nm. The enzymatic activity of the supernatant is expressed as protein μM/mg.
53
Lactate dehydrogenase (LDH) levels
The level of lactate dehydrogenase in rat brain homogenate was measured using a diagnostic kit from Coral diagnostics, India, and is expressed as unit/mg protein.
54
Superoxide dismutase (SOD) levels
The SOD activity was measured at pH 10.4 by spectrophotometric auto-oxidation of epinephrine. The supernatant (0.2 ml) was mixed with a 0.8 ml 50 mM glycine buffer, pH 10.4, and the reaction was initiated by adding 0.02 ml of epinephrine. Spectrophotometrically, the absorbance was measured at 480 nm after 5 min. SOD activity is expressed in the form of μM/mg protein.
55
Reduced glutathione (GSH) levels
To estimate the level of reduced brain glutathione, 1 ml supernatant was precipitated with 1 ml of 4% sulfosalicylic acid and cold digested at 4°C for one h. For 15 minutes, the samples were centrifuged at 1200 g. Then 2.7 ml of phosphate buffer (0.1 M, pH 8) and 0.2 ml of 5.5′-dithiobis-(2-nitrobenzoic acid) (DTNB) to 1 ml of the supernatant were added. The yellow color was developed and was immediately measured with a spectrophotometer at 412 nm. The concentration of glutathione is expressed as μM/mg protein in the supernatant.
54
Nitrite levels
The accumulation of nitrite in the supernatant, an indicator of nitric oxide production (NO), is determined through a colorimetric assay using Greiss reagent (0.1% N-(1-naphthyl) ethylenediamine dihydrochloride, 1% sulfanilamide, and 2.5% phosphoric acid). Equal volumes of supernatant and Greiss reagent were combined; the mixture incubated in the dark at room temperature for 10 min, and the absorbance was spectrophotometrically measured at 540 nm. The supernatant’s nitrite concentration was measured using the standard sodium nitrite curve and expressed as μM/mg protein.
56
Malondialdehyde (MDA) levels
The level of the malondialdehyde determined the lipid peroxidation in rat brain homogenate. The amount of MDA is measured by spectrophotometer after its reaction with thiobarbituric acid at 532 nm. MDA concentration is expressed as the nM/mg protein. 4
Assessment of gross pathology and morphology
Animals were sacrificed on day 45 of the protocol schedule by decapitation; brains were removed for gross pathological analysis. After observation of the whole rat brain, coronal sections were taken.57,58 Sectioned 2-mm thick pieces of the brain (coronally from the anterior pole to the posterior cerebral cortex poles) were mounted on glass slides. A digital camera (Nikon, Japan) was used for observing all areas of the brain covering the entire striatum. It took less than 5 minutes to capture digital photographs of all parts of the brain. The digital images obtained were analyzed to prepare the figures and converted for image analysis into TIFF files. The demyelination region (mm) in each brain segment was measured on day 45th after completing the procedure through MOTICAM-BA310 image plus 2.0 analysis software. The volume of the demyelination scale (mm) was calculated for each coronal brain segment by converting the demyelination region (mm). The demyelination size (mm3) in each brain section was determined from the dark greyish area near the striatum by image analysis on the 45th day. The injury’s size was calculated in each coronal 2-mm-thick brain section by calculating the demyelination area (l × b × h).
Statistical analysis
Data were analyzed using two-way ANOVA followed by Post hoc test Bonferroni and one-way ANOVA repeated measures followed by Post hoc test Tukey’s multi comparison test. P < 0.0001 was considered statistically significant. Data was found to be normalized, and the sample size was calculated by checking the normality distribution by the Kolmogorov Smirnov test. All statistical results were performed out by GraphPad Prism version 5.03 for Windows (GraphPad Software, San Diego, CA, USA). Statistical results were expressed as the mean±standard error of mean (SEM).
Results
Effect of PUR on body weight in PPA-treated autistic rats
Bodyweight was measured on day 1st, 13th, 23rd, 33rd, and 43rd of the experimental protocol schedule. On day one, no significant difference was observed among all the treatment groups. The rats’ body weight treated with ICV-PPA was gradually decreased during administration on day 23rd, 33rd, and 43rd. A significant decrease in the weight was recorded at the end of the protocol schedule. Chronic treatment with PUR 5 mg/kg and 10 mg/kg on days 33 and 43 significantly and dose-dependently restored body weight loss [two-way ANOVA F(20,120) = 1319, p < 0.0001] as compared to the ICV-PPA treated group (Figure 2).
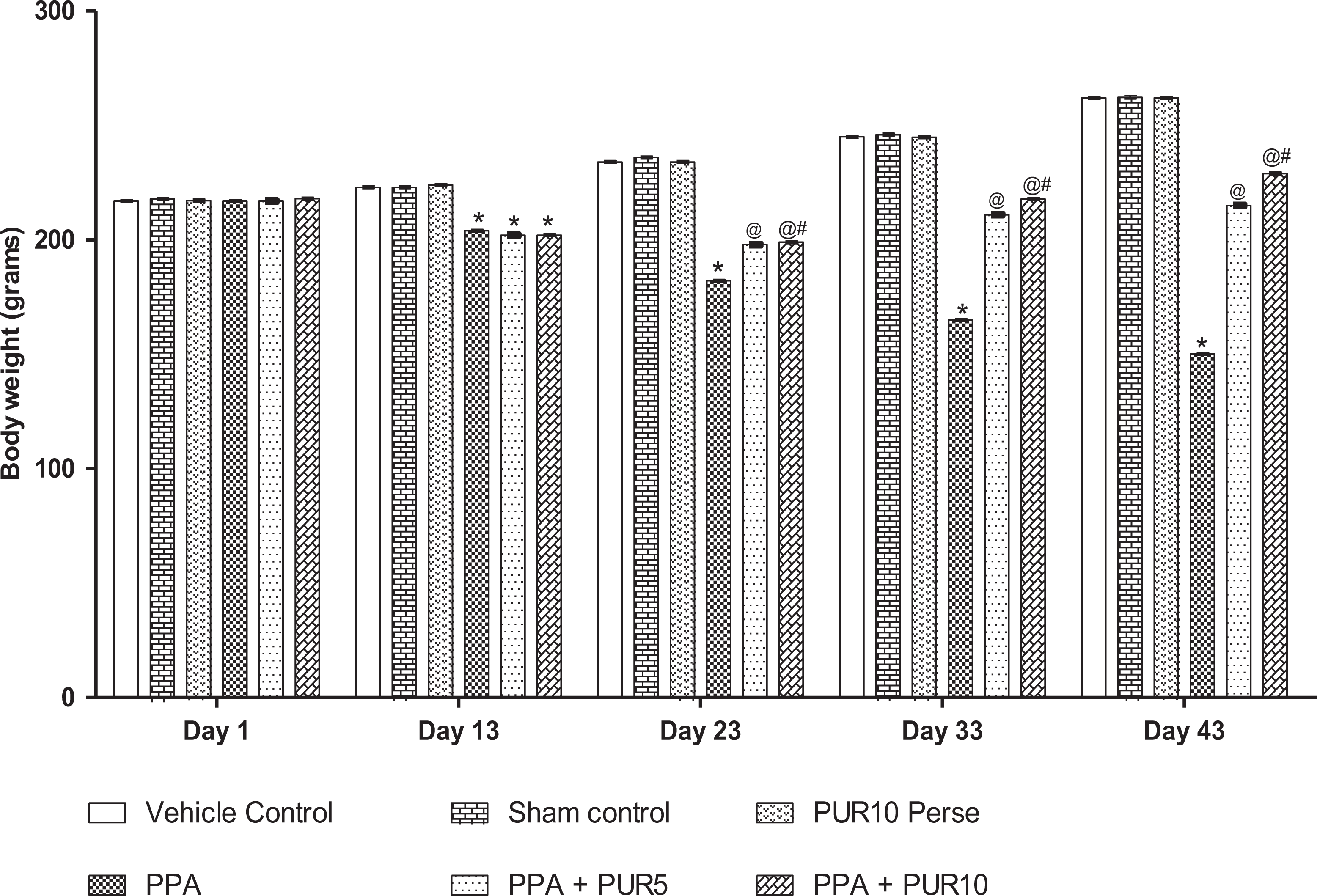
Effect of PUR on body weight in PPA-treated autistic rats. Values expressed as mean ± SEM,
Behavioral parameters
Effect of PUR on beam crossing task in PPA-treated autistic rats
The beam crossing task was performed for monitoring neuromuscular coordination on day 1st, 13th, 23rd, and 43rd of the experimental protocol schedule. On day one, no significant difference was observed among all treatment groups. Neuromuscular coordination was reduced as an increased number of slips and walking impairment alleviated as assessed by beam crossing tasks on day 13 in PPA-treated rats than in normal and PUR-treated rats. On days 23 and 43, the number of slips was remarkably reduced, and the balance and walking were improved in 5 mg/kg, and 10 mg/kg PUR-treated rats than in PPA-treated rats [two way ANOVA F(15,90) = 26.45, p < 0.0001]. Furthermore, 10 mg/kg was more significant than 5 mg/kg, and improved balance beam-walking performance (Figure 3).

Effect of PUR on beam crossing task in PPA-treated autistic rats. Values expressed as mean ± SEM,
Effect of PUR on locomotion activity using in PPA-treated autistic rats
As per the protocol schedule, the locomotor activity was observed using an actophotometer on day 1st, 13th, 23rd, and 43rd. On day 1, there was no significant difference found between all the treatment groups. On day 13th, rats treated with ICV-PPA showed a considerable decrease in ambulatory movements than the vehicle, sham, and PUR 10 mg/kg per se group. On day 23 and 43, PUR 5 and 10 mg/kg treated rats showed significantly elevated locomotion counts than PPA-treated. Long-term administration of PUR 5 mg/kg and 10 mg/kg showed an increase in the locomotor activity [two way ANOVA F(15,90) = 4659, p < 0.0001] when compared with ICV-PPA treated group. Among the selected doses, PUR 10 mg/kg showed a significant dose-dependent increase in rats’ locomotor activity compared to 5 mg/kg (Figure 4).

Effect of PUR on locomotion activity in PPA-treated autistic rats. Values expressed as mean ± SEM,
Effect of PUR on immobility phase in PPA-treated autistic rats
As per the protocol schedule, immobility time was observed using a forced swim test on day 1st, 13th, 23rd, and 43rd. On day 1 of the observation, there was no significant difference found between all the treatment groups. The chronic administration of ICV-PPA in rats showed a significant increase in immobility time, recorded at the end of the protocol schedule. On days 13, a substantial increase in immobility was founded in PPA-treated rats than in normal rats. The decrease in immobility was seen during chronic treatment with PUR 5 mg/kg and 10 mg/kg on day 23rd and 43rd than in PPA-treated rats [two way ANOVA F(15,90) = 740.9, p < 0.0001]. Chronic treatment with 10 mg/kg PUR significantly and dose-dependently found to be more effective in regaining mobility exhibiting a loss in depressive behavior (Figure 5).
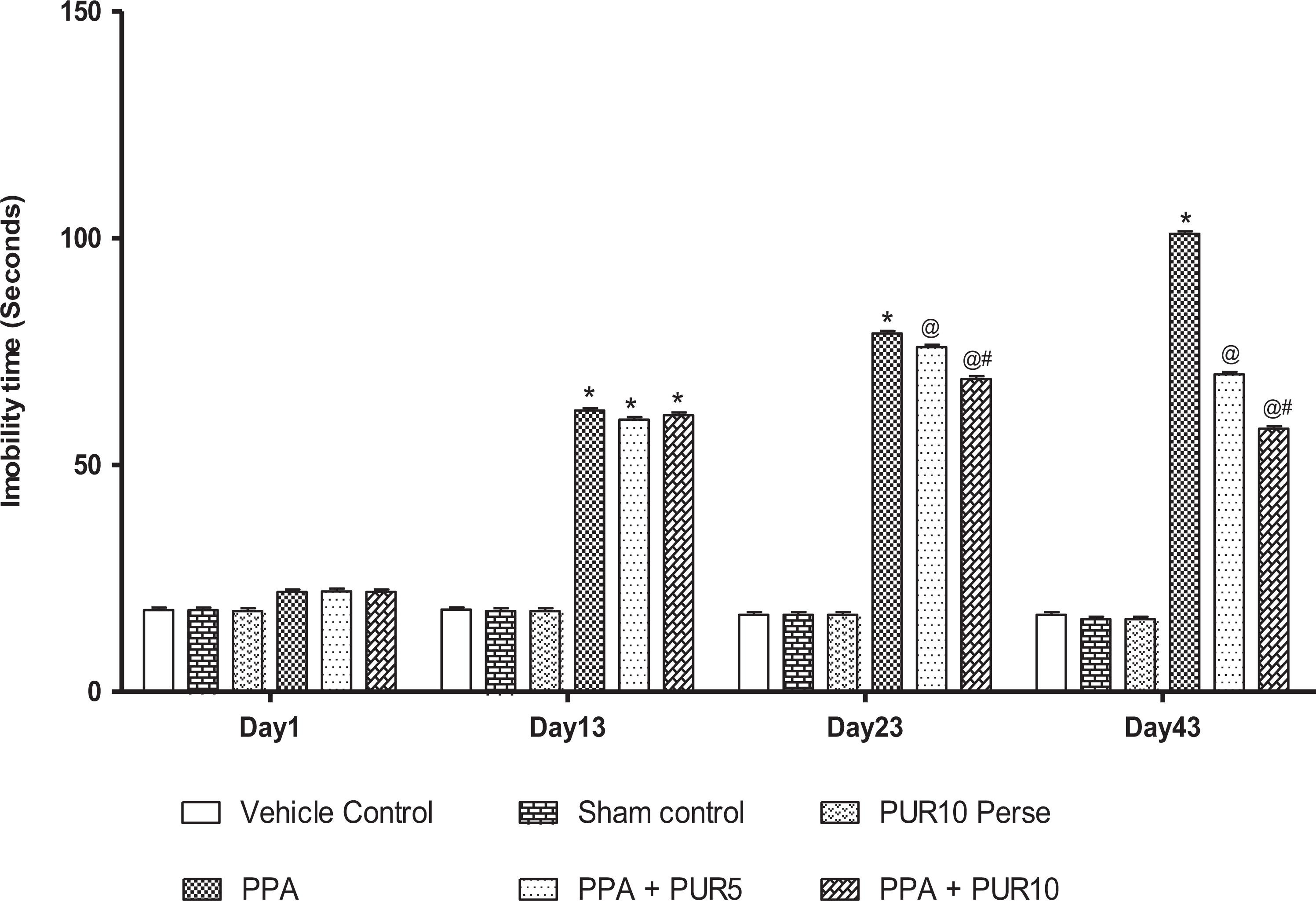
Effect of PUR on immobility phase in PPA-treated autistic rats. Values expressed as mean ± SEM,
Effect of PUR on spatial learning and memory in PPA-treated autistic rats
As per the protocol schedule, ELT was observed on days 40, 41, 42, and 43, and on day 44th TSTQ was noted. Based on the observations, long-term administered ICV-PPA treated rats showed a gradual increase in ELT and decreased TSTQ during administration. On days 40 to 43, a significant reduction in escape latency was observed in PUR10 mg/kg-treated autistic rats than in PUR 5 mg/kg-treated autistic rats [two way ANOVA F(15,90) = 60.49, p < 0.0001] (Figure 6). TSTQ was performed on day 44. PPA-treated rats showed a major reduction in TSTQ when compared to normal and PUR-treated rats. Administration of 5 mg/kg PUR led to a significant increase in TSTQ [One way ANOVA F(5,25) = 14.80, p < 0.0001]when compared with the PPA-treated group. Administration of 10 mg/kg PUR caused effective restoration in memory function when compared 5 mg/kg PUR in PPA-intoxicated rats (Figure 7).

Effect of PUR on ELT in PPA-treated autistic rats. Values expressed as mean ± SEM,

Effect of PUR on TSTQ in PPA-treated autistic rats. Values expressed as mean ± SEM,
Biochemical estimations
Effect of PUR on SMO-SHH and MBP level in PPA-treated autistic rats
As compared to the vehicle control, sham control, and PUR perse group, chronic administration of neurotoxin PPA in rats showed a significant decrease in the expression of SMO-SHH in rat brain homogenate. Chronic PUR treatment with 5 mg/kg and 10 mg/kg for 44 days significantly increased in the Smo-Shh level [one way ANOVA F(5,25) = 0.755, p < 0.0001]. As compare to PUR 5 mg/kg, influential restoration in the Smo-Shh level was seen in PUR 10 mg/kg.
MBP protein level in brain homogenates was investigated using an ELISA kit. The findings show that the MBP level significantly decreased in the ICV-PPA treated rats compared with the vehicle, sham control, and PUR perse group. Chronic treatment with PUR 5 mg/kg and 10 mg/ kg presented a significant improvement in the level of MBP [one way ANOVA F(5,25) = 1.034, p < 0.0001] compared to ICV-PPA rats. A significantly high dose of PUR shows a more promising restoration of MBP level compared to a low dose (Table 1).
Effect of PUR on cellular and molecular markers in PPA-treated autistic rats.
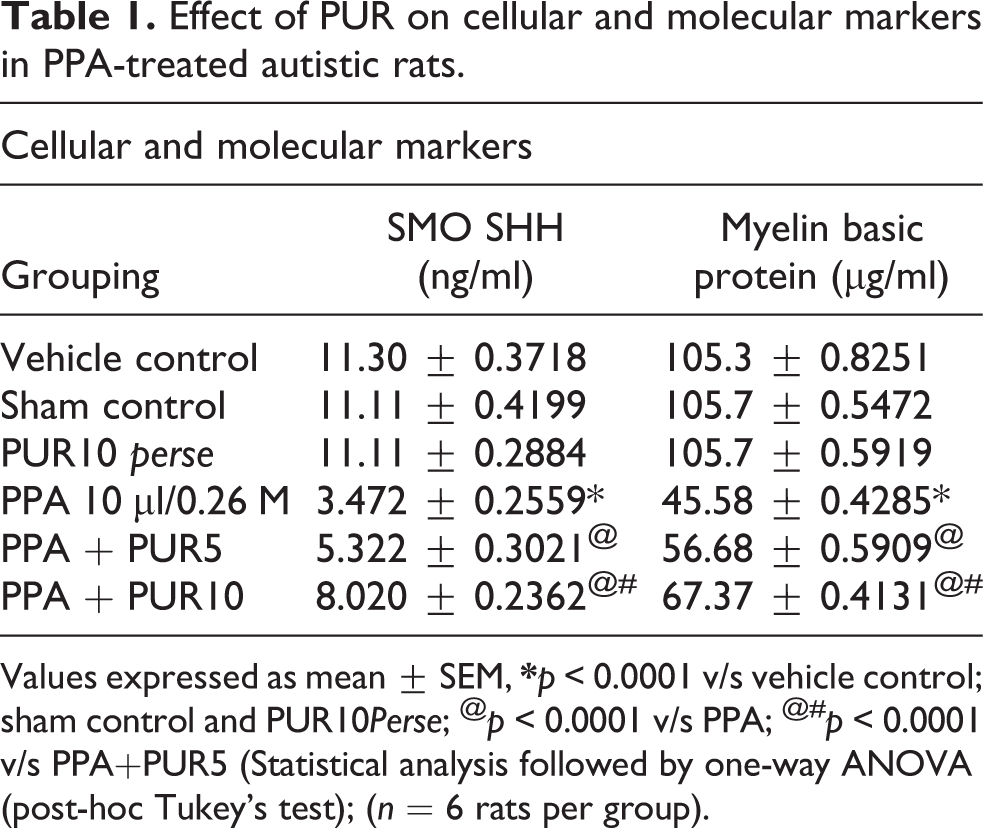
Values expressed as mean ± SEM,
Effect of PUR on apoptotic markers in PPA-treated autistic rats
The level of apoptotic markers after the chronic administration of toxin PPA in rats showed a significant increase in the level of Bax as compared to the vehicle, sham, and PUR 10 per se group. A significant decrease in Bcl-2 level was observed in PPA treated rats compared to vehicle, sham, and PUR 10 per se group. Pre-treatment with PUR 5 mg/kg and 10 mg/kg decreased the level of Bax, [one way ANOVA F(5,25) = 1.180, p < 0.0001]. The level of Bcl-2 [one way ANOVA F(5,25) = 0.2779, p < 0.0001] was elevated significantly compared to the PPA group. Interestingly, the caspase-3 activity was also showed a similar pattern to the Bax. PPA treated rats show an increase in caspase-3-level. Whereas, after chronic treatment with PUR 5 mg/kg and 10 mg/kg resulted in a significant decrease in the PPA-induced increase in caspase-3-level [one way ANOVA F(5,25) = 1.307, p < 0.0001]. Persistent treatment with a high dose of PUR shows a more substantial improvement in the level of apoptotic markers compared to low dose (Table 2).
Effect of PUR on apoptotic markers level in PPA-treated autistic rats.

Values expressed as mean ± SEM,
Effect of PUR in neurotransmitter level in PPA-treated autistic rats
Neurotransmitter levels after chronic administration of neurotoxin PPA in rats showed a significant decrease in dopamine, serotonin, acetylcholine levels, and glutamate concentration in the brain homogenate. Constant PUR 5 mg/kg and 10 mg/kg treatment for 44 days showed significantly and dose-dependently, increase the level of dopamine[one way ANOVA F(5,25) = 0.5773, p < 0.0001], serotonin[one way ANOVA F(5,25) = 1.273, p < 0.0001], acetylcholine[one way ANOVA F(5,25) = 0.9310, p = 0.4778], whereas significantly decrease in the glutamate levels[one way ANOVA F(5,25) = 1.218, p < 0.0001]. Compared to the low dose of PUR, 10 mg/kg PUR treated rat show more promising and significant restoration of the neurotransmitter level (Table 3).
Effect of PUR in neurotransmitter level in PPA-treated autistic rats.
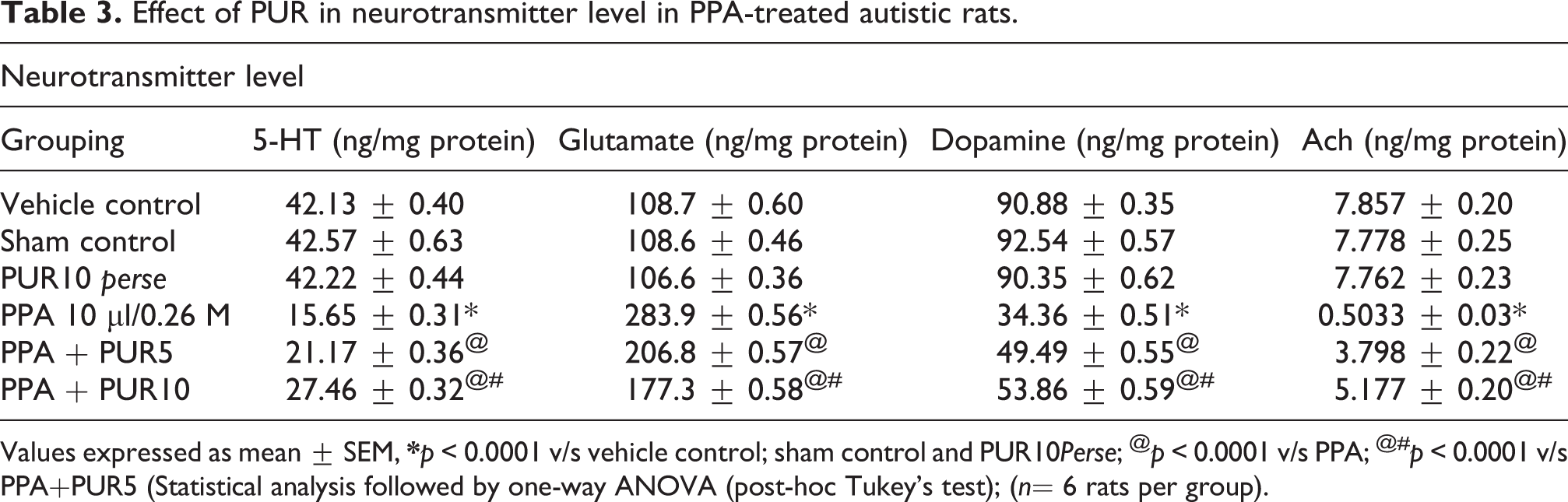
Values expressed as mean ± SEM,
Effect of PUR on neuroinflammatory markers in PPA-treated autistic rats
There were no significant differences in TNF-α and IL-1β levels between normal and PUR-perse rats. The chronic administration of PPA in rats showed a significant increase in pro-inflammatory cytokines. Chronic treatment with PUR 5 mg/kg and 10 mg/kg consequentially decrease TNF-α [one way ANOVA: F(5,25) = 0.7395, p < 0.0001] and IL-1β levels [one way ANOVA: F(5,25) = 0.3295, p < 0.0001] than ICV-PPA treated rats. A high dose of 10 mg/kg of PUR significantly reduces TNF-α and IL-1β levels than low doses of 5 mg/kg of PUR (Table 4).
Effect of PUR on neuroinflammatory markers in PPA-treated autistic rats.

Values expressed as mean ± SEM,
Effect of PUR on oxidative stress markers in PPA-treated autistic rats
Neurochemical levels of AchE, MDA, LDH, and nitrite were significantly increased. In contrast, reduced GSH and SOD levels were significantly decreased in ICV-PPA treated rats compared to sham control and PUR 10 per se. PUR 5 mg/kg and 10 mg/kg significantly decrease in levels of AchE [one way ANOVA F(5,25) = 2.142, p < 0.0001], MDA [one way ANOVA F(5,25) = 1.643, p < 0.0001], LDH [one way ANOVA F(5,25) = 1.058, p < 0.0001] and nitrite [one way ANOVA F(5,25) = 0.6669, p < 0.0001] and remarkably restore the anti-oxidant levels of reduced GSH [one way ANOVA F(5,25) = 1.923, p < 0.0001] and SOD [one way ANOVA F(5,25) = 0.3503, p < 0.0001] as compared with PPA control group. PUR treatment at a high dose of 10 mg/kg was significantly more effective than low doses of 5 mg/kg of PUR treated rats (Table 5).
Effect of PUR on oxidative stress markers in PPA-treated autistic rats.

Values expressed as mean ± SEM,
Effect of PUR on gross pathological evaluation in PPA-treated autistic rats
Assessment of the whole brain in PPA-treated autistic rats
The ICV-PPA treated rat brain showed damaged clotted outer layer with breach meninges versus vehicle control, sham control, and PUR perse. In the vehicle control, sham control, and PUR 10 mg/kg per se group’s rat’s brain showed optimally sized, undamaged proper shape with clearly observable meninges. PUR 5 mg/kg and 10 mg/kg remarkably restored the morphological alterations in ICV-PPA-induced rats (Figure 8).

Effect of PUR on gross pathological changes (whole rat brain) in PPA-treated autistic rats. (a) Vehicle control. (b) Sham control. (c) PUR10 perse. (d) PPA. (e) PPA + PUR5 (f) PPA + PUR10. (Scale bar = 2 mm). Note: Yellow circles are pointing to the site of the brain injury.
Assessment of the brain sections in PPA-treated autistic rats
Morphologically optimally sized, unharmed proper shape with clearly observable basal ganglia, cortex, and hippocampus tissue was observed in Vehicle control, Sham control, and PUR 10 mg/kg perse treated rat brain. The ICV-PPA treated rat brain shows hippocampus and cortical atrophy, as well as atrophy in subcortical structures such as the caudate nucleus, putamen, and atrophy in the medial thalamus, atrophy of the internal medullary lamina when the comparison was made with vehicle control, sham control and PUR perse, treated rats. PUR5 mg/kg and 10 mg/kg remarkably reduce the pathological changes (Figure 9).

Effect of PUR on gross pathological changes (brain sections) in PPA-treated autistic rats. (a) Vehicle control (i) Cerebral cortex (ii) Hippocampus (iii) basal Ganglia. (b) Sham control. (c) PUR10 perse. (d) PPA. (e) PPA + PUR5 (f) PPA + PUR10. (Scale bar = 5 mm). Note: Yellow circles are pointing to the injured site.
Assessment of the demyelination volume in PPA-treated autistic rats
The normal and vehicle control groups showed no significant effect on the demyelination region’s size compared to PUR 10 mg/kg perse. However, chronic administration of PPA neurotoxin substantially increased the area of demyelination compared to normal, vehicle, and PUR 10 mg/kg perse. Long-term treatment with PUR 10 mg/kg perse treatment showed no change compared to normal rats. Treatment with PUR5 mg/kg and 10 mg/kg significantly reduced the demyelination region relative to those groups where only PPA was used [one-way ANOVA: F(5,25) = 0.4017, p < 0.0001] (Figure 10).

Effect of PUR on demyelination volume in PPA-treated autistic rats. Values expressed as mean ± SEM,
Discussion
Smo-Shh signaling is a key element that plays a significant role in developing the embryo’s central nervous system—disruptions in Smo-Shh lead to significant post-natal neurodevelopmental disorders. 43 Infusions of ICV-PPA in rodents have been shown to induce behavioral changes similar to those seen in ASD and investigated as a possible adult ASD rodent model. 59 This model is validated by work investigating the link between ASD and phenotypic behavioral abnormalities and is clinically similar to other models of autism. 60 In addition, it can also have a significant influence on the signaling system, such as Smo-Shhh, and treatment with its agonist PUR appears to have a beneficial effect on disorders caused by high PPA rates. 61 In this study, chronic PUR treatment following ICV-PPA has been shown to protect against acute brain injury and long-term memory and learning disability in rat brains.
PPA was injected stereotaxically in the ICV region of the Wistar rat. PPA selectively and specifically damages neuronal cells, leads to neural cell acidification, and changes cell metabolism. 60 During the 44-day protocol schedule, PUR’s neuroprotective effect was examined by conducting behavioral parameters and molecular markers, inflammatory cytokines, neurotransmitters, and oxidative stress markers in rat brain homogenates.
Current findings show that ICV injected PPA in rats induces significant changes in body weight, locomotive behavior, neuromuscular coordination, and immobility time. However, chronic PUR treatment revealed a substantial change in PPA mediated modification’s dose-dependent behavior, with the higher dose of PUR showing more significant effects relative to the lower dose. Overall, high doses of PUR in rats display significant body weight changes, locomotion, decreased number of slips, improved mobility time, and reduced immobility more effectively than in low-dose groups during injection days.
In our behavioral findings, there was a marked increase in ELT and a decrease in TSTQ in rats with chronic PPA treatment in MWM, which according to previous studies, indicates spatial memory impairment. 62 When treated with a high dose of PUR, there was a substantial decrease in ELT and an increase in TSTQ, indicating a significant restoration of memory loss compared to a low dose.
During our research to determine the molecular mechanism, we investigated PUR’s effect on the Smo-Shh level in rats’ brains. Previous studies have shown that the downregulation of Smo-Shh signaling in the hippocampus has led to neuronal apoptosis in the hippocampus. 63 In support of this Shh signal agonist, it helps prevent cognitive deficits in rats. In contrast, the Shh agonist’s anti-apoptotic effect was mediated by an increase in the Bcl-2 level. Our findings showed an improvement in Smo-Shh in rat brain homogenate compared to PUR 5 mg/kg, and 10 mg/kg for ICV-PPA treated animals. Significantly high doses of PUR suggest more promising low-dose outcomes, increasing the Smo-Shh pathway. In support of cellular and molecular processes, the ELISA level of MBP is studied, suggesting a gradual decrease in MBP levels of ICV-PPA rats. For chronic PUR therapy, the amount of MBP has been shown to positively impact in a dose-dependent manner. The level of MBP indicates a significant increase in the high dose of PUR. In addition, caspase-3 & Bax (pro-apoptotic) and Bcl-2 (anti-apoptotic) levels significantly increase and decrease in ICV-PPA-treated rats. Chronic treatment with a high and low PUR dose showed a substantial decrease in caspase-3 & Bax, whereas Bcl-2 seems to have increased significantly compared to ICV-PPA rats.
In the current research, long-term PUR administration has shown significant restoration of neurotransmitter levels, neuroinflammation, and oxidative damage in autistic rats, indicating its neuroprotective efficacy toward ICV-PPA mediated pathogenesis. Chronic infusion of ICV-PPA to rats has an important effect on the number of neurotransmitters in rats’ brain homogenates. Dopamine, serotonin, and acetylcholine levels have decreased in ICV-PPA-treated rats, whereas glutamate levels have risen significantly, indicating neuronal excitotoxicity. Chronic therapy with PUR 5 mg/kg and 10 mg/kg restores the amount of neurotransmitters in rats’ brains.
Oxidative damage and neuroinflammation play an essential role in neurodegeneration and neurodevelopmental changes, and TNF-alpha and IL-1β are the main contributors to these processes. There are reports also shows a large increase in the number of these cytokines in various neurological disorders. 64 Chronic administration of PUR 5 mg/kg and 10 mg/kg showed a substantial decrease in both TNF-alpha and IL-1β levels compared to ICV-PPA rats and demonstrated defense against neuroinflammation. However, the level of oxidative stress markers such as MDA, Nitrite, LDH, and AchE appears to be increasing. On the other side, the amount of antioxidant markers such as GSH and SOD has decreased. The level of antioxidant markers has been restored during treatment with PUR for 44 consecutive days, and a substantial decrease in oxidative stress markers has been observed. Our research looked at the morphological structure, the whole brain section, and the volume of demyelination. Results showed that chronic doses of PUR 5 mg/kg and 10 mg/kg, demyelination could be stopped and remyelination facilitated. Our study found that the hippocampal area was relatively vulnerable to PPA therapy. Histological and morphological evidence suggests differences in the size and shape of the brain of PPA-treated animals. Chronic high-dose PUR medication showed significant improvements in morphological and histological changes in autistic rats rather than low dosage.
However, current results are only associations in which the activation of Smo-Shh primarily examines the neuroprotective activity of PUR in PPA-induced neurobehavioral, molecular and morphological alterations in Autism-treated rats mediated signaling pathways. A further mechanism of action, such as overexpression or deletion of the Smo-Shh signaling pathway, as well as additional molecular evidence, such as immunoblotting and immunohistopathology, must be validated.
Conclusion
In conclusion, we prove that PUR can control the dysregulation of neuronal cells and reduces apoptosis in ICV-PPA treated rats through the up-regulation of the Smo-Shh pathway. This indicates that Smo-Shh can induce neuronal cell growth by inhibiting the apoptotic pathway and increasing myelin-based protein levels in rats’ brains. PUR also improves antioxidant and anti-inflammatory activity by reducing the production of oxidative stress markers and decreasing the levels of inflammatory cytokines such as TNF-alpha and IL-1β. In addition, PUR modulates the anti-apoptotic effect by reducing caspase-3 and Bax while increasing the level of Bcl-2 (anti-apoptotic marker) by specifically regulating and restoring Smo-Shh signaling in damaged neuronal cells. Besides, histological and morphological changes in the whole brain and brain sections indicate that PUR’s neuroprotective ability against ICV-PPA induces brain alterations. Demyelination due to PPA’s chronic administration has also been shown to enhance the induction of protocol drugs in rats’ brain tissue. Such interesting findings suggest that PUR could be a potential approach to neurodevelopmental disorders such as autism. Also, Smo-Shh can be compared as a futuristic pharmacological intervention with other standard drug therapies. Apart from the fact that our study has some limitations, we have not performed any western blot immunohistopathology studies that should be considered for the future.
Footnotes
Acknowledgments
The authors express their gratitude to Chairman, Mr. Parveen Garg, and Director, Dr. G.D. Gupta, ISF College of Pharmacy, Moga (Punjab), India, for their excellent vision and support.
Author contributions
S.R.: thesis research work, performed experimental animal studies. R.G.: compilation of statistical research data, revision of research manuscript. A.S.: clinical and pre-clinical survey, revision of research manuscript. S.M.: original research hypothesis, guide, and compilation of all manuscript data. All authors approved the final version of this study.
Data availability statement
All data generated or analyzed during this study are included in this article. There are no separate or additional files.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All applicable institutional guidelines for the care and use of animals were followed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by institutional grants by the Institutional Animal Ethics Committee (IAEC) with registration no. 816/PO/ReBiBt/S/04/CPCSEA as protocol no. ISFCP/IAEC/CPCSEA/Meeting No.25/2019/Protocol No 427 as per the guidelines of the Government of India and approved by RAB Committee, ISFCP, Moga, Punjab, India.
