Abstract
Some traditional Chinese decoctions, such as Zhuyu Annao, exert favorable therapeutic effects on acute cerebral hemorrhage, hemorrhagic stroke, and other neurological diseases, but the underlying mechanism remains unclear. This study aimed to determine whether Zhuyu Annao decoction (ZYAND) protects the injured brain by promoting angiogenesis following intracerebral hemorrhage (ICH) and elucidate its specific mechanism. The effect of ZYAND on the nervous system of mice after ICH was explored through behavioral experiments, such as the Morris water maze and Rotarod tests, and its effects on oxidative stress were explored by detecting several oxidative stress markers, including malondialdehyde, nitric oxide, glutathione peroxidase, and superoxide dismutase. Real-time quantitative RT-PCR and WB were used to detect the effects of ZYAND on the levels of prolyl hydroxylase domain 3 (PHD3), hypoxia-inducible factor-1α (HIF-1α), and vascular endothelial growth factor (VEGF) in the brain tissues of mice. The effect of ZYAND on the NF-κB signaling pathway was detected using a luciferase reporter gene. A human umbilical cord vascular endothelial cell angiogenesis experiment was performed to determine whether ZYAND promotes angiogenesis. The Morris water maze test and other behavioral experiments verified that ZYAND improved the neurobehavior of mice after ICH. ZYAND activated the PHD3/HIF-1α signaling pathway, inhibiting the oxidative damage caused by ICH. In angiogenesis experiments, it was found that ZYAND promoted VEGF-induced angiogenesis by upregulating the expression of HIF-1α, and NF-κB signaling regulated the expression of HIF-1α by inhibiting PHD3. ZYAND exerts a reparative effect on brain tissue damaged after ICH through the NF-κB/ PHD3/HIF-1α/VEGF signaling axis.
Introduction
Intracerebral hemorrhage (ICH) is a neurological disorder with a high mortality rate in the early stage and in most cases a poor prognosis, resulting in stroke, amyloid angiopathy, arteriovenous malformations, brain tumors, and traumatic brain injury.1–5 The high morbidity and mortality rates of ICH are associated with both primary and secondary injuries. 6 Primary brain injury is predominantly caused by mechanical insult, and blood metabolites after ICH appear to be an important source of secondary brain injury. 7 Efficacy has been achieved for primary injury, but the mechanisms of secondary injury are not well-defined. Increasing evidence suggests that oxidative stress following ICH is a key factor that exacerbates ICH-induced secondary brain injury. 8 Oxidative stress damages protein molecules, disrupts cell signaling, and causes neuronal death.9,10 The dysfunction caused after ICH is associated with the release of blood metabolites, such as heme, and an increase in redox-active iron, which are important factors in secondary brain injury.11,12 At present, there are no effective surgical or pharmacological treatments available to improve the prognosis of patients with ICH; thus, it is of great significance to explore effective treatment options.
Brain angiogenesis after ICH is involved in the repair of damaged brain tissue, during which the expression of many relevant cytokines, chemokines, and proteases is upregulated, including hypoxia-inducible factor-1α (HIF-1α), vascular endothelial growth factor (VEGF), and angiopoietin 1.13,14 HIF-1α is a nuclear transcription factor for various pro-angiogenic factors and plays a central regulatory role. The HIF-prolyl hydroxylase domain (PHD) enzyme is a type of oxygen sensor that plays a major role in the regulation of HIF-1α. 15 Iron chelators inhibit HIF-PHDs under normoxic conditions, inhibiting the oxygen-dependent hydroxylation of HIF and protecting against ICH-induced brain damage. 16 Iron chelators also stabilize HIF-1a and activate a range of related genes, and appropriate concentrations of iron chelators protect neurons from oxidative death. 17 HIF-PHD protein-mediated hydroxylation is a key post-translational modification that regulates the activity and stability of HIF-1α. 18 Under hypoxic conditions, oxygen-dependent PHD is inhibited and HIF-1α is degraded. Therefore, drugs targeting PHD may allow the control of PHD levels by inhibiting HIF-1α degradation to promote angiogenesis following ICH.
PHD inhibitors can pharmacologically activate the innate endogenous adaptive response to low oxygen levels; thus, they have become attractive therapeutic targets for the treatment of local ischemia. Zhuyu Annao decoction (ZYAND) is a classic traditional Chinese regimen for the treatment of ICH. ZYAND can reduce brain edema, improve the function of the nervous system, reduce inflammation, and exert a protective effect on the brains of ICH mice.
19
The main ingredients of ZYAND are leech, ground beetle, Chinese trumpet creeper, peach kernel, lumbricus,
The present study first investigated the effects of ZYAND on the nervous systems of mice after ICH in terms of animal behavior to determine whether it had a protective effect on injured brain tissue. This was followed by a series of cellular molecular experiments to elucidate the molecular mechanism by which ZYAND protects injured tissues after ICH. Finally, we investigated the effect of ZYAND on angiogenesis after ICH using the human umbilical cord vascular endothelial cell (HUVEC) angiogenesis test.
Materials and methods
Establishment of the mouse model of ICH
Male C57BL/6 mice (8–10 weeks of age; Southern Model Animal Research Institute) were anesthetized with 2–5% isoflurane and fixed in prone position. During this procedure, a thermal blanket was placed under the mice to maintain their temperature at 37°C. A burr hole was drilled into the skull with a cranial drill, and 0.0375 U of collagenase in 0.5 µL of normal saline was injected into the right basal ganglia at a flow rate of 0.1 ul/min using a microinfusion pump and Hamilton syringe. The stereotactic coordinates of the injection were as follows: 0.2 mm anterior, 3.5 mm ventral, and 2.5 mm lateral to the anterior reg. In the control group, equal amounts of normal saline were injected. Appropriate postoperative care was provided until the animal had fully recovered. After injection, the needle was left in place for an additional 10 min, and then withdrawn at a rate of 1 mm/min. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This study was approved by the Ethics Committee of The First Affiliated Hospital of Guangxi University of Chinese Medicine on 25 April 2019 (Approval No. 2019-033-02).
Morris water maze test
The learning ability and spatial memory were measured through a water maze test using a circular pool (inner diameter: 95 cm, depth: 35 cm). The pool was divided into four quadrants and water was poured to a depth of 22 cm, with its temperature maintained at 24 ± 1°C. Nontoxic white powder was added to the pool, covering the surface of the water, resulting in the view of the water beneath the powder being blocked. Each mouse was tested four times per day for 5 days. When the mice swam to the platform, the time spent on the platform was recorded and the test was terminated. If the mice did not find the platform within 60 s, they were guided to the platform by the test administrator, where they remained for 15 s. On the sixth day, the platform was removed. Each mouse was released into a quadrant of the pool and allowed to swim freely for 60 s. All tests were recorded using an overhead CCD camera, and a video tracking system was used to measure the trajectory of the mice and the time taken to reach the platform.
Rotarod test
We performed automated Rotarod (Ugo Basile, Comerio, Italy) to measure motor function. Mice were trained four times a day, with the rod rotating at a constant speed of 40 rpm, for 4 days prior to surgery. The average latency of falling from the rod was recorded, and the maximum cutoff time was set as 180 s.
Pole test
The pole test was performed according to previously described procedures. Each mouse was placed on a ball (diameter: 2.5 cm) glued to the top of a wooden pole (length: 50 cm, diameter: 1 cm). The time it took the mice to climb down the pole was measured four times, with the maximum cutoff time set to 100 s.
Traction test
The severity of limb damage was assessed using a traction test. A stainless steel rod (50 cm long, 2 mm diameter) was connected to two vertical supports, and a front paw of each mouse was placed on the middle of the rod. The mice were scored as follows: three points, hanging from the rod with two hind paws; two points, hanging from the rod with one hind paw; one point, hanging from the rod with two front paws; 0 points: falling off the rod.
Oxidative stress measurements
The peripheral basal ganglia around the hematoma were collected from each group and homogenized on ice, followed by centrifugation at 4000 g at 4°C for 5 min. The concentrations of superoxide dismutase (SOD) [Superoxide Dismutase (SOD) assay kit, A001-3-2], glutathione peroxidase (GPx) [Glutathione Peroxidase (GSH-PX) assay kit, A005-1-2], nitric oxide (NO) [Nitric Oxide (NO) assay kit, A012-1-2], and malondialdehyde (MDA) [Malondialdehyde (MDA) assay kit, A003-1-2] in the supernatant were determined using commercially available kits (Nanjing Jiancheng Bioengineering Institute, Nanjing). NADPH oxidase activity was measured using a NADPH assay kit [NADPH oxidase assay kit, A127-1-1]. To determine the production of total ROS, brain slices were placed with dichlorofluorescin diacetate (10 mmol/L) at 37°C for 30 min in darkness. After washing with PBS three times, images were taken with a fluorescence microscope at 488 nm excitation and 525 nm emission, and fluorescence intensity was quantified using ImageJ (1.52V) software.
Real-time quantitative PCR
Total RNA was isolated from brain tissues using a miRNeasy Mini Kit (Qiagen). Equal amounts of mRNA were reverse transcribed using a cDNA synthesis kit (Life Technologies), and experiments were performed using TaqMan premix. All gene expression levels were standardized to β-actin gene expression levels as determined by VIC-labeled probes. All experiments were performed using a 7500 real-time PCR system (Applied Biosystems).
Western blotting (WB)
The brain tissue surrounding the hematoma was isolated for WB analysis. The basal ganglia surrounding the hematoma were washed and grated with PBS, and the homogenate was lysed with RIPA lysis buffer containing PMSF. The protein concentration of each sample was determined by BCA quantification. Samples were electrophoresed on 8–10% SDS-PAGE gels then transferred to nitrocellulose membranes (Millipore, MA, USA). The membranes were closed in 5% skim milk and incubated with one antibody. The membranes were incubated overnight with the following primary antibodies: anti-VEGF (1:1000, Cell Signaling, USA), anti-HIF-1α (1:1000, Abcam, UK), anti-PHD1 (1:1000, Abcam, UK), anti-PHD2 (1:1000, Abcam, UK), anti-PHD3 (1:500, Abcam, UK), and anti-GAPDH (1:1000, Abcam, UK). Afterward, the WB results were observed using a HRP-embedded secondary antibody (Beyotime Biotechnology Institute) with an enhanced chemiluminescence kit. The intensity of each lane was analyzed using ImageJ software for relative levels of proteins.
Immunofluorescence
Mice were anesthetized via the injection of 1% pentobarbital sodium peritoneal, and the aorta was perfused with 4% paraformaldehyde to euthanize the mice. After fixation and dehydration, brain specimens were embedded in paraffin for immunofluorescence experiments, and 25-mm-thick coronal brain sections were stained. Immunofluorescence methods were used to determine CD34+-positive cells, which are predominantly found in neovascular endothelial cells. The antibody used for immunofluorescence was anti-CD34 (1:100, Cell Signaling, USA). Nuclei were labeled with DAPI (1:1000, Life Technologies, R37606). We incubated the brain sections overnight at 4°C with the primary antibody, then observed them using a Zeiss LSM 710 confocal microscope system (Zeiss, Germany). Cells with fluorescent markers were considered CD34+-positive and represented a type of neovascularization. The number of CD34+-positive cells was counted for each slice using five different fields of view.
Cell culture
HEK293T cells were cultured in DMEM liquid medium containing 10% inactivated fetal bovine serum (FBS), placed in a 37°C, 5% CO2 incubator, and passaged every 2 days with Trypsin-EDTA (0.25%). After 30 s, the cells became rounded, and a single-cell suspension was prepared by complete dissociation. Approximately one-fifth of the cells were retained and cultured. Primary HUVECs were purchased from ATCC and cultured in 1640 medium with +10% FBS.
Reporter gene assays
Luciferase activity assay: the transfected cells were washed with PBS buffer, then 100 µL of cell lysate was added to each well. The samples were placed on ice or in a −20°C freezer for 30 min to allow complete cell lysis. Then, 5 µL of luciferase assay buffer was added to each well of a 96-well plate, followed by the addition of 45 µL of cell lysate and 100 µL of luciferase assay buffer, and the luciferase activity was immediately measured using a lucifluorescence chemiluminescence instrument.
HUVEC angiogenesis assay
First, 50 µL of substrate glue was placed in each well of a 96-well plate, followed by incubation at 37°C for 30 min. HUVECs were inoculated at a density of 5 × 104 cells onto the stromal layer and treated with ZYAND. After 12 h, the tubular structure of the endothelial cells was stained with Calcein-AM (Molecular Probes, Eugene, OR, USA) and photographed using ImageXpress Micro (Molecular Devices, CA, USA), followed by the observation of angiogenesis.
Statistical analysis
Data are reported as the mean ± SD of three replicates of multiple individual experiments; **P < 0.01 represents a significant difference. Statistical analyses were performed using GraphPad Prism 6, unless otherwise noted. When two groups were compared, a two-tailed t-test was used.
Results
ZYAND protects against ICH-induced brain injury
Neurobehavior is significantly altered when brain injury occurs. The results of the Morris water maze test showed that the administration of ZYAND had little effect on healthy mice. Following ICH surgery, the time taken to reach the platform was shorter in the group treated with ZYAND compared to the controls (Figure 1(a)). Mice remained on the rotating rod for a shorter period of time after ICH, but this trend was reversed after the administration of ZYAND (Figure 1(b)). Compared with the controls, mice receiving ICH surgery had significantly lower scores, but this was significantly improved with ZYAND (Figure 1(c)). In addition, for pole tests, the activity time of the mice increased significantly following ICH but decreased significantly after treatment with ZYAND (Figure 1(d)). The results of these behavioral experiments indicate that neurological function is significantly impaired in mice after ICH. However, ZYAND could ameliorate the neurological deficits caused by ICH.
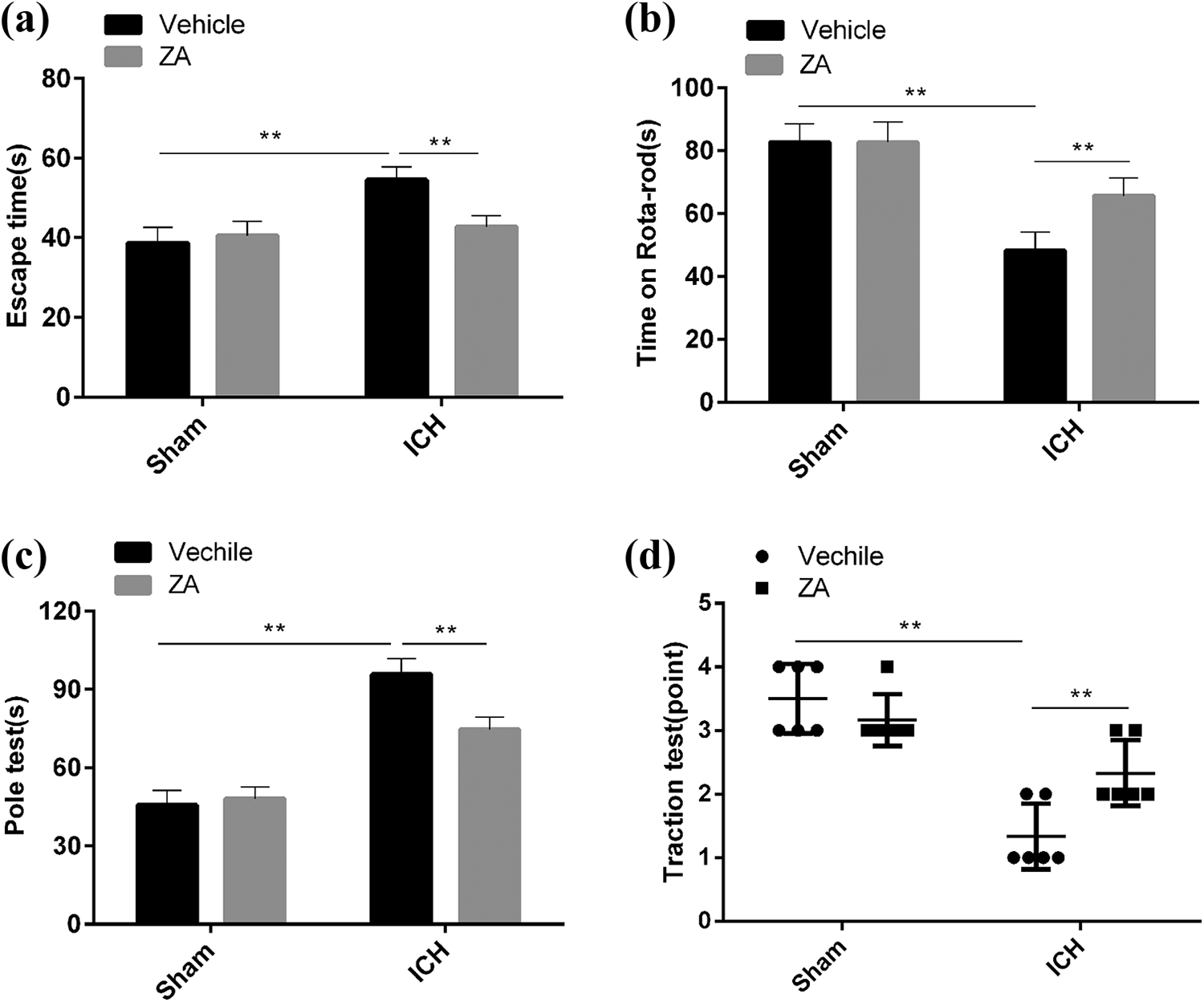
Zhuyu Annao decoction protects against ICH-induced neurological deficits. (a) Sham-operated or ICH mice were injected intraperitoneally with Zhuyu Annao decoction (10 mg/kg) or the corresponding control solvent for 48 h. The mice were tested by Morris water maze test for learning ability and spatial memory. The learning ability and spatial memory ability of the mice were tested by Morris water maze test. (b) and (c) Motor function of mice was assessed by the rotating bar test (b) and pole test (c). (d) Limb motor function of mice was measured and scored by the traction test. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
ICH-induced oxidative stress is inhibited by ZYAND
Given that oxidative stress plays a key role in the pathogenesis of ICH, we next investigated the effects of ZYAND on oxidative stress. The levels of markers of oxidative stress, such as MDA, NO, GPx, and SOD, did not change significantly in healthy mice after administering ZYAND. Three days after ICH surgery, MDA and NO levels were elevated in ICH mice compared to controls, while SOD levels were significantly reduced. However, after ICH mice were treated with ZYAND, MDA and NO levels were suppressed, and GPx and SOD levels were elevated again (Figure 2(a)–(d)). In addition, the intracellular ROS level significantly increased in ICH mice but was significantly suppressed in ICH mice treated with ZYAND (Figure 2(e)). These results indicate that ICH-induced brain damage was ameliorated by ZYAND through the suppression of oxidative stress-induced oxidative damage.
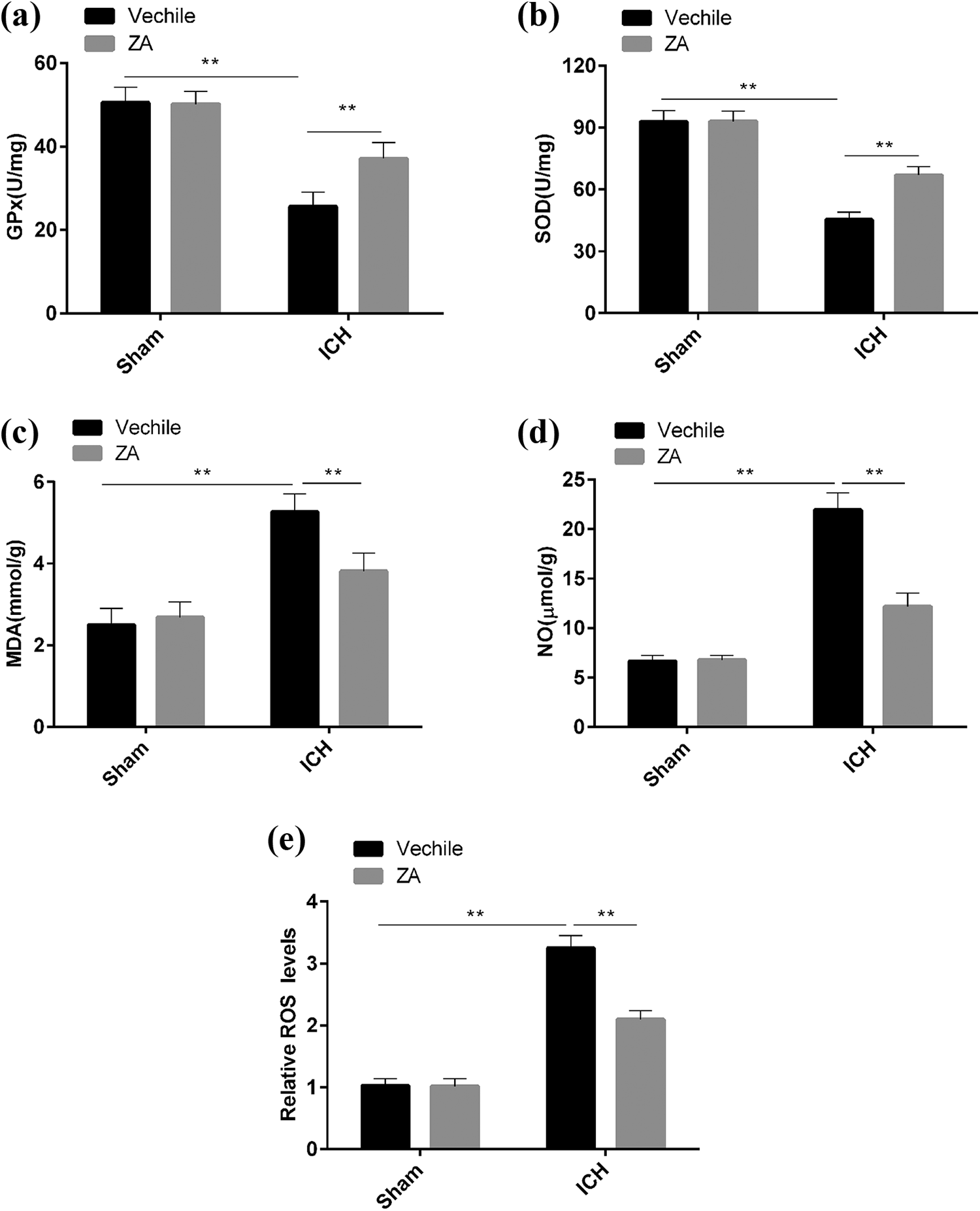
Zhuyu Annao decoction (ZA) inhibits ICH-induced oxidative stress. (a)–(d) Concentrations of oxidative stress indicators glutathione peroxidase (GPx) (a), superoxide dismutase (SOD) (b), malondialdehyde (MDA) (c) and nitric oxide (NO) (d) in brain tissues were determined by commercial kits as described in the Methods section. (e) Intracellular ROS production in brain slices was assessed by H2DCF-DA staining, and the relative ROS level of each group was determined by an enzymograph. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
ZYAND activates the PHD3/HIF-1α signaling pathway in brain tissue
The real-time quantitative RT-PCR (qRT-PCR) assay showed that the expression level of PHD3 mRNA was significantly elevated in the brain tissue of the ICH group compared to the controls, while the expression level of HIF-1α mRNA was significantly decreased, but the mRNA trends of PHD and HIF-1α were reversed in ICH mice administered by ZYAND, which had no effect on the expression of PHD1 and PHD2
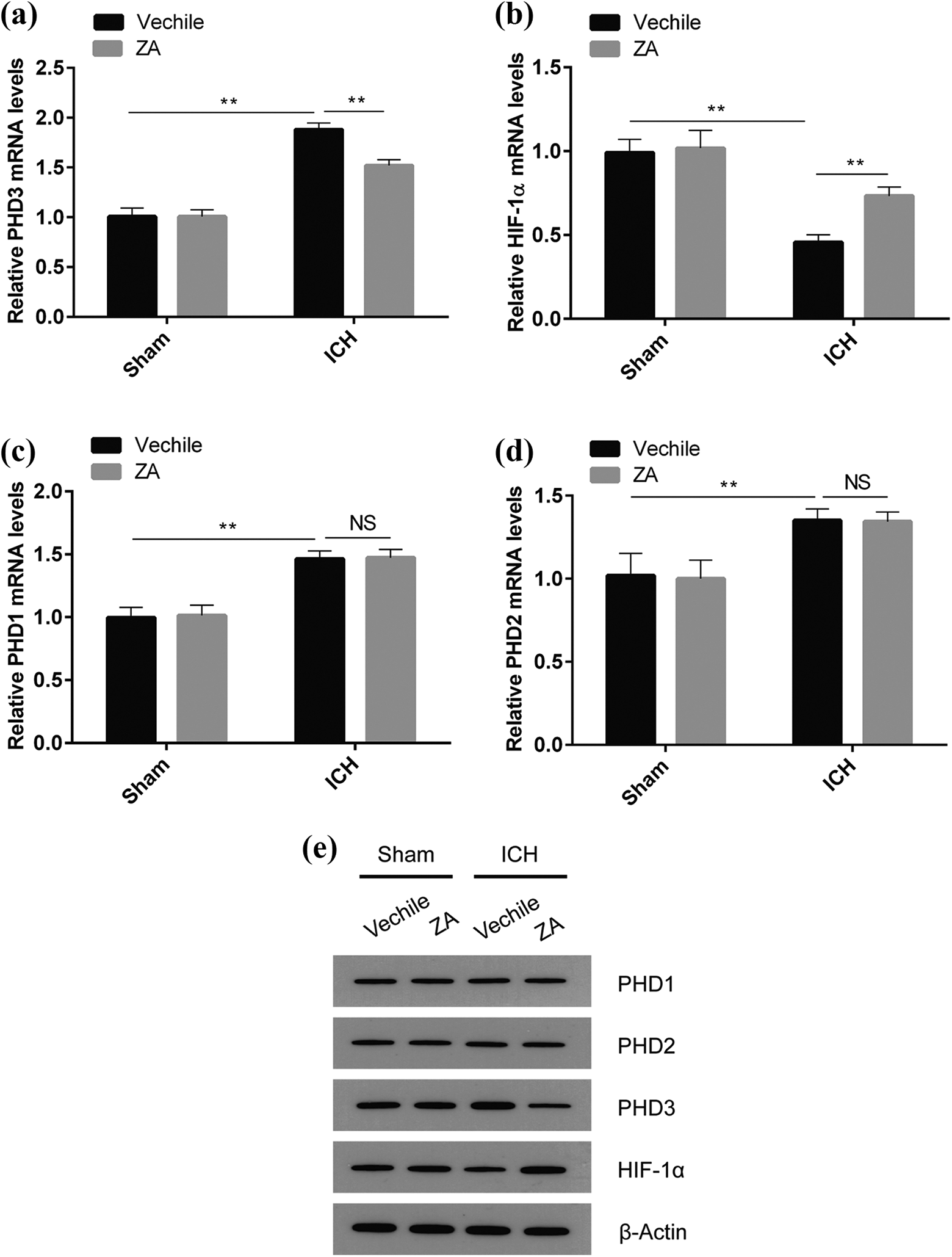
Zhuyu Annao decoction (ZA) activates the PHD3/HIF-1α signaling pathway. (a)–(d) The effects of Zhuyu Annao decoction on PHD1, PHD2, PHD3 and HIF-1α mRNA expression in ICH-induced mouse brain tissues were detected by real-time quantitative PCR. (e) Western blot detects the effects of Zhuyu Annao decoction on PHD1, PHD2, PHD3 and HIF-1α protein expression in ICH-induced brain tissues. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
ZYAND promotes VEGF-induced angiogenesis
The results of qRT-PCR showed that, compared with the sham-operated group, the expression of VEGF in the ICH group decreased, but the VEGF expression level significantly increased after treatment with ZYAND (Figure 4(a)). WB also showed similar results, that is, the expression of VEGF protein in the brain tissues of ICH mice could be significantly increased by ZYAND (Figure 4(b)). Immunofluorescence results showed that CD34+ cell levels were significantly increased in the brain tissues of ICH mice after treatment with Zhuyu Annao decoction (Figure 4(c) and (d)). These results indicate that ICH regulates VEGF-induced angiogenesis by promoting the expression of HIF-1α.
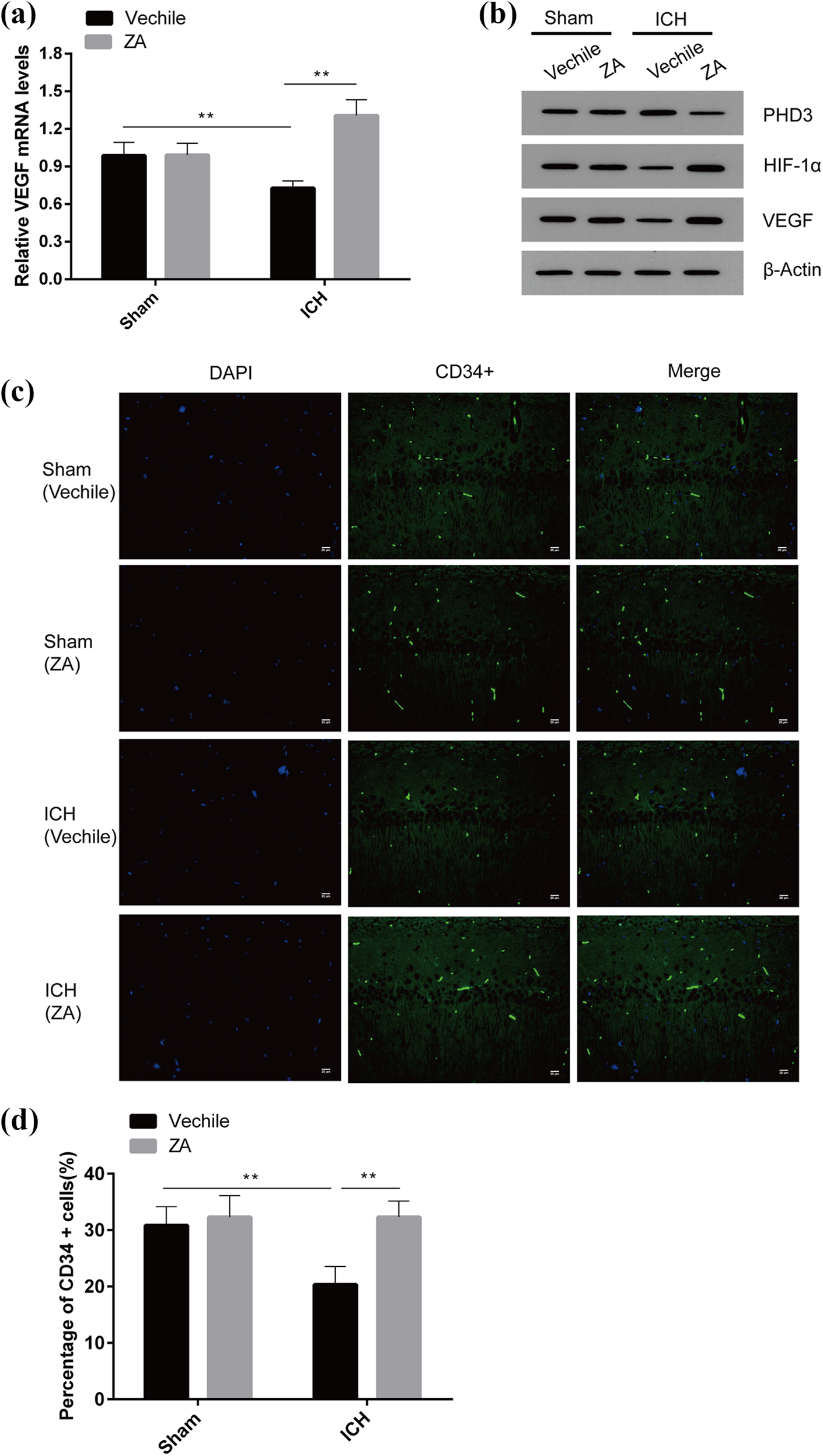
Zhuyu Annao decoction (ZA) promotes angiogenesis after ICH. (a) The effect of Zhuyu Annao decoction on the expression of the angiogenic factor VEGF by real-time quantitative PCR. (b) Western blot was performed to detect the effect of Zhuyu Annao decoction on the expression of VEGF in ICH-induced mouse brain tissues. (c) Immunofluorescence detection of the number of CD34+ cells in mouse brain tissue and assessment of angiogenesis in mice. (d) Percentage of CD34+ cells in mice treated with Zhuyu Annao decoction or control solvent. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
ZYAND inhibits PHD3 expression by activating the NF-κB signaling pathway
It was found that cytokine-mediated PHD3 expression is NF-κB-dependent, suggesting that the inhibition of PHD3 expression by ZYAND may be related to the activation of the NF-κB signaling pathway. To investigate whether ZYAND is associated with the activation of the NF-κB signaling pathway, we constructed the NF-κB reporter gene and transfected it into 293T cells. The results of the luciferase assay showed that the fluorescence intensity in the mice treated with ZYAND was significantly higher than in those without
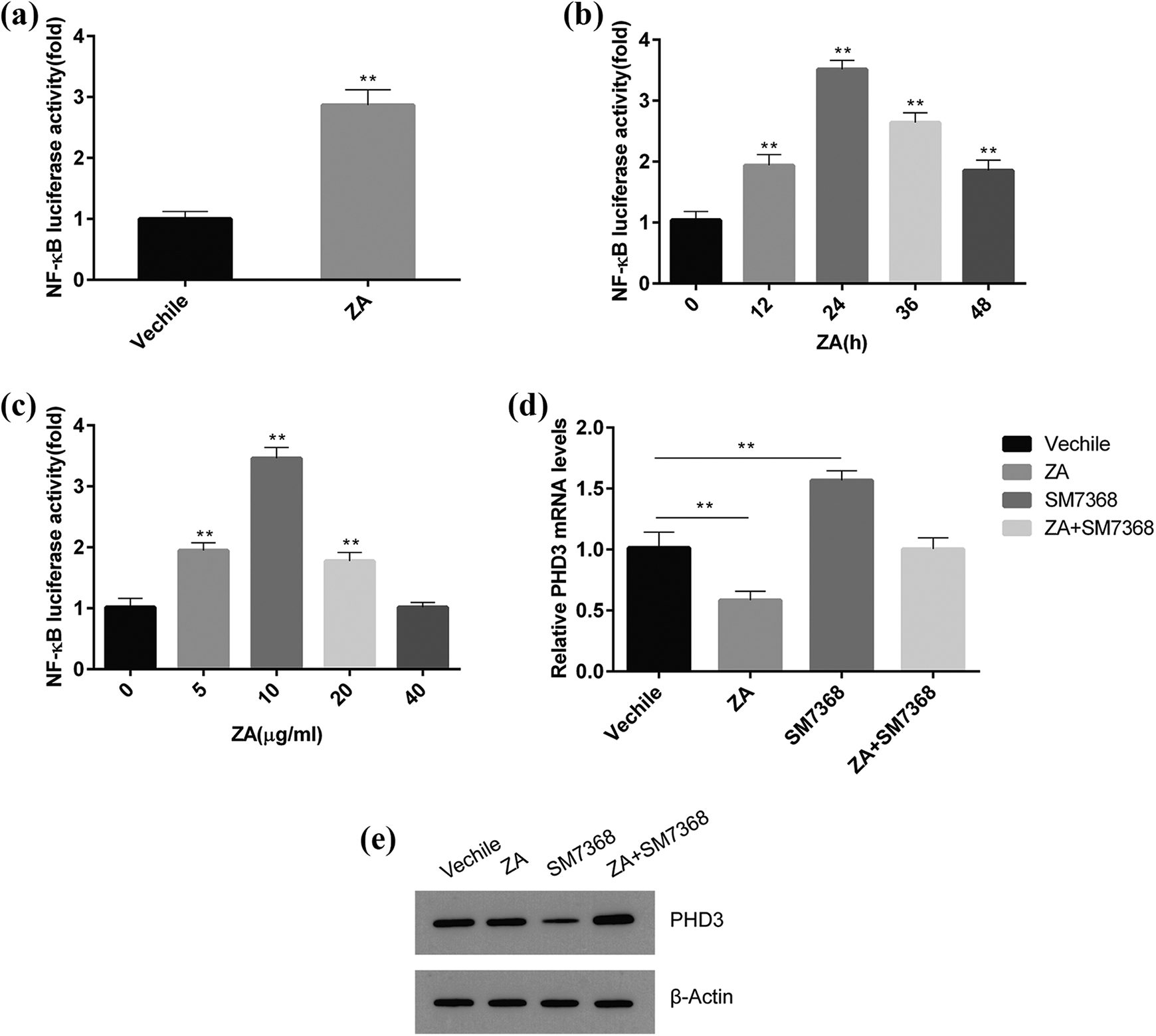
NF-kB signaling positively regulates angiogenesis after ICH. (a) The effect of Zhuyu Annao decoction on NF-kB reporter gene activity. (b) Effect of luciferase assay plus Zhuyu Annao decoction at different times on the activity of NF-kB reporter gene. (c) The effect of different concentrations of Zhuyu Annao decoction on the activity of NF-kB reporter gene by luciferase assay. (d) Real-time quantitative PCR was used to detect the effect of Zhuyu Annao decoction and SM7368 on PHD3 expression. (e) Western blot of the effect of Zhuyu Annao decoction and SM7368 on PHD3 expression. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
NF-κB signaling pathway regulates angiogenesis in ICH mice
The results of qRT-PCR showed that after microinjection of the NF-κB signaling inhibitor SM7368 into the brains of ICH mice, the expression levels of VEGF mRNA and its upstream HIF-1α were significantly reduced (Figure 6(a) and (b)). Similar results were obtained at the protein level, that is, SM7368 inhibited VEGF expression in the brain tissues of the ICH mice (Figure 6(c)). Immunofluorescence results showed that the number of CD34+-positive cells in the brain tissues of ICH mice significantly increased after treatment with ZYAND, but the number of CD34+ cells significantly decreased after microinjection of SM7368 (P < 0.01) (Figure 6(d) and (e)). When cultured on matrix gel, HUVECs can spontaneously form three-dimensional capillary-like tubular structures. Therefore, we simulated in vitro angiogenesis experiments to investigate the effects of ZYAND and NF-κB signaling on angiogenesis. As shown in Figure 6(f), ZYAND could significantly promoted the formation of HUVEC microvessels, but angiogenesis was significantly hindered by SM7368. These results suggest that ZYAND promotes the formation of microvessels in the brain tissue of ICH mice by activating NF-κB signaling.
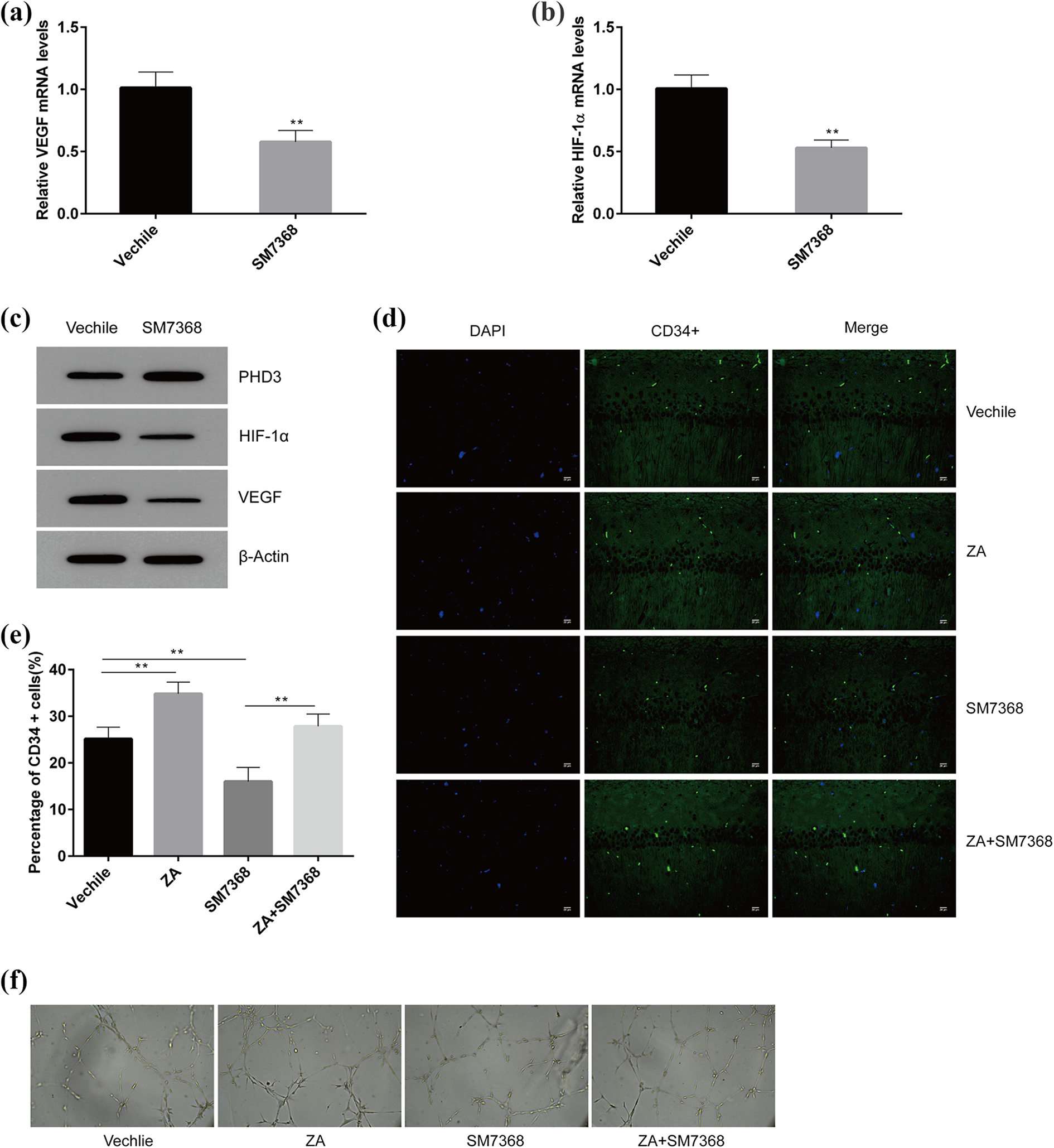
(a) and (b) Real-time quantitative PCR detection of the effect of NF-kB inhibitorSM7368on VEGF and HIF-1α expression. (c) Western blot detection of the effect of SM7368 on PHD3, VEGF and HIF-1α expression. (d) Immunofluorescence detection of effects of Zhuyu Annao decoction and SM7368 on angiogenesis in mouse brain tissue (as assessed by CD34+ cell number). (e) The angiogenic capacity of HUVEC cells was detected by angiogenesis assay. All data are expressed as standard deviation ± SD. **P < 0.01 represents significant difference, n = 6 for per group.
Discussion
In this study, we demonstrated that ZYAND reduces neurological deficits and promotes the formation of microvessels in damaged brain tissues in a mouse model of ICH. Possible mechanisms include an increase in HIF-1α expression after treatment, which is achieved via the activation of the NF-κB signaling pathway. We identified a new signaling axis, namely, ZYAND/NF-κB/NF-κB/PHD3/HIF-1α, which contribute to regulate angiogenesis and tissue repair in damaged brain tissues after ICH (Figure 7). In addition, we demonstrated that ZYAND inhibits the oxidative damage caused by ICH, thus protecting the damaged brain tissues.
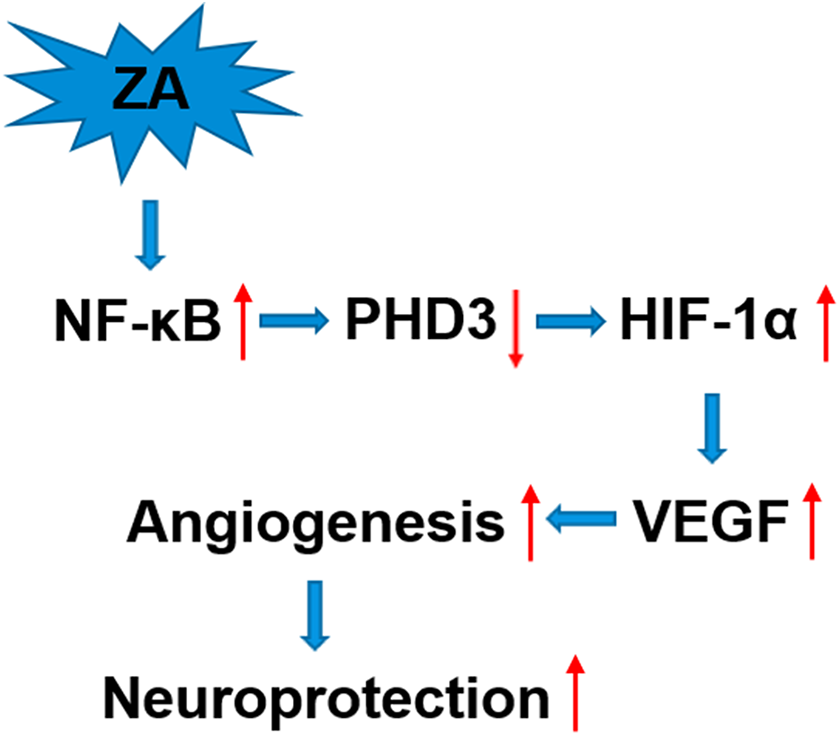
The effect of angiogenesis assay on PHD3 and its target gene HIF-1α. By inhibiting PHD3 protein expression through the activation of NF-kB signaling pathway, Zhuyu Annao decoction can promote the expression of HIF-1α gene and increase angiogenesis, and thus protect the brain tissues of mice after ICH.
In this study, the neurobehavioral experiments demonstrated the neurological deficits caused by ICH and the therapeutic effects of ZYAND. The results of the Morris water maze test, rotating bar test, traction test, and polar test indicated that the administration of ZYAND primarily improves motor function after ICH, providing suggestions for addressing relevant clinical issues in human ICH.
The activation of NF-κB signaling is a promising neuroprotective mechanism,21–23 and the present study showed that ZYAND activates the NF-κB signaling pathway. PHD enzymes play a crucial role in neuroprotection.23–25 The injection of PHD1 antisense oligonucleotides into the lateral ventricle reduces infarct size and neurological deficits. 26 Neuron-specific inhibition of PHD subtype PHD2 is sufficient to attenuate acute neuronal loss in stroke mice. 27 The loss of PHD proteins (PHD1, PHD3) stabilizes HIF-1α and promotes neovascularization in ischemic mice. 28 Our results showed that PHD3 is an important downstream molecule of NF-κB signaling that activates the NF-κB signaling pathway to protect brain tissues after ICH.
The mature and stable neovascular microvascular system after brain injury is essential for restoring the entire vascular network, facilitating the exchange of oxygen and metabolites and removing necrotic debris.29–34 VEGF plays a crucial role in promoting the proliferation, differentiation, and angiogenesis of vascular endothelial cells.35–37 HIF-1α is a central regulator of nuclear transcription factors and angiogenesis as well as an upstream regulator of VEGF. ICH-induced angiogenesis usually occurs along the edges of a hematoma and gradually extends to its center.38–40 When PHD3 is activated by hypoxia caused by ICH, the hydroxylation and degradation of HIF-1α are enhanced. Our in vivo and in vitro experiments demonstrated that ZYAND upregulates the protein expression level of HIF-1α by targeting the inhibition of PHD3 expression, thereby promoting VEGF expression and angiogenesis. Another study of ours also found neurological impairment in the TLR4 knockout group; after knockout of TLR4, the infiltration of monocytes and neutrophils in mice was reduced, peripheral inflammation was reduced, and functional recovery was also improved. 41 TLR4 itself does not mediate phagocytosis; it merely acts as a recognition receptor. Thus, knockout of TLR4 promotes the absorption of edema. The possible mechanism for alleviating inflammatory damage is that after the occurrence of ICH, TLR4 is co-expressed by microglia and peripheral mononuclear macrophages in the central nervous system, which mediates the injury of ICH.
Conclusion
In conclusion, to the best of our knowledge, this is the first study to demonstrate that ZYAND protects the nervous system by promoting angiogenesis after ICH, and we preliminarily investigated the specific mechanisms underlying its regulation. ZYAND activated the intracellular NF-κB signaling pathway, suppressed the expression of PHD3, increased the protein level of HIF-1α, and promoted the expression of the downstream target gene, VEGF, which, in turn, promoted angiogenesis. These findings will assist in improving our understanding of brain repair following hemorrhagic injury and in developing new therapeutic strategies and agents accordingly. However, the neuroprotective effects of the stasis-easing formula require further investigation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of National Natural Science Foundation of China (81760847, 82060844), the Key Project of Guangxi Natural Science Foundation (2018GXNSFDA050018), Guangxi Medical and Health Appropriate Technology Development and Application Project (S2019020), Open Topics of Guangxi University of Chinese Medicine (2019XK018, 2019XK021), the Project of Guangxi Key Laboratory of Chinese Medicine Foundation Research (No. 19-245-14-05), the High-level Talent Team Cultivation Project of Qihuang Project of Guangxi University of Chinese Medicine (2018003) and Academic Team of the First Affiliated Hospital of Guangxi University of Chinese Medicine (No. 2018 (146)).
