Abstract
Exosomes are small, cell-derived vesicles of 30–100 nm that participate in cell-to-cell communication. They are released by many cells, such as dendritic cells (DC), lymphocytes, platelets, epithelial cells, endothelial cells (EC), and are found in most body fluids, including blood, saliva, urine, and breast milk. The exosomes released from cells within the cardiovascular system may contain either inhibitors of calcification in normal physiological conditions or promoters in the pathological environment [atherosclerosis (AS), and Chronic Kidney Disease (CKD)]. The exosomes of the vascular smooth muscle cells (VSMCs) are novel players in vascular repair processes and calcification. Several studies have shown that the cytoplasmic contents of exosomes are rich in a variety of proteins, nucleic acids, and lipids. Currently, exosomal micro RNAs and proteins are increasingly being recognized as biomarkers for the diagnosis of several diseases, including those of kidney and liver, as well as different types of cancer. In this review, we summarize recent advances in the role of exosomes in vascular calcification and their potential applications as diagnostic markers as well as a brief overview of the role of stem cell-derived exosomes in cardiovascular diseases.
Keywords
Introduction
Extracellular vesicles (EVs) are secreted in the extracellular milieu on fusion of the late endosomes or multivesicular bodies (MVB) with the plasma membrane and are synthesized as intraluminal vesicles (ILVs) in multivesicular bodies (MVB).1,2 Studies suggest, exosomes are important mediators of intercellular communication besides, acting as means of cellular waste disposal. 3 The comparatively smaller size and unified shape allow exosomes to successfully escape clearance by the mononuclear phagocyte system, thereby prolonging their circulation time, as well as implying their contribution toward cell–cell communication. 4 In mid 1960s, Bonucci, reported the involvement of extracellular vesicles, also called matrix vesicles (MVs) in the calcification process. 5 Matrix vesicles are produced by cells like osteoblasts, odontoblast and chondrocytes, exhibiting features similar to the cells from where they originate.6,7 With regard to lipid composition, MVs harbor low levels of neutral phospholipids such as, lysophospholipids and phosphatidylcholine and comparatively high levels of acidic phosphatides, phosphatidylserine (PS) and sphingomyelin. 8 Under oxidative stress, Ca2+ containing vesicles bind with acidic phospholipid, PS, in complex with either phosphate ions (Pi) or annexin, with the release of MVs outside the cytosol.9,10 The interaction of MVs with glycosaminoglycans, a negatively charged polysaccharide marks the beginning of mineralization. 11
Depending on the parent cell that secrete exosomal vesicles, the exosomes produced from different cell types exhibit pleiotropic biological activities, either protective or detrimental, which is determined mainly by their origin and present status. 12
The clinical consequences of arterial and renal vascular calcification (VC) include cardiovascular morbidities such as arterial stiffening, impaired vasodilation, myocardial infarction (MI), stenosis of arteries, congestive heart failure, valvular heart disease ischemia in both coronary and peripheral arteries, atherosclerosis and death.13,14
Generally, cellular communication via exosomes affects each phase of atherosclerosis. Studies have shown that the genesis of AS is initiated by endothelial dysfunction, which is fundamentally due to local disturbances in blood flow along the endothelium. 15 Several microRNAs(miRNAs) present in the cytoplasm of exosomes, contribute significantly to different stages of vascular calcification. Recent studies have demonstrated CKD patients develop extensive and progressive VC due to the loss of anti-calcific mechanisms, differentiation of VSMCs, chronic inflammation as well as the release of exosomes and other extracellular vesicles. 16
In this review, we have tried to summarize the process of generation of the extracellular vesicles, namely exosomes and their role in calcification of the vascular system and kidneys and give new insights into the role of exosome derived microRNAs (miRNA) in the pathogenesis associated with calcification. We have also addressed another area which is attracting much attention, namely the role of stem cell derived exosomes in the atherosclerotic process.
Exosomes
Classification
The term exosome was coined by Dr. Rose Johnstone in a quest to study the maturation of erythrocyte from immature reticulocytes. 17 Exosomes are membrane vesicles bound by a lipid bilayer and contain transmembrane proteins and a hydrophilic core containing proteins, mRNAs, and miRNA. These small vesicles are 50–100 nm in diameter, present inside large multivesicular endosomes. They contain transferrin receptors, a marker that mediates endocytosis and the recycling of cell-surface proteins that are engulfed by the plasma membrane. Exosomes originate as vesicles from late endosomes, and are characterized by the presence of transferrin receptors in the extracellular domain. 18 Every mammalian cell releases these bilayer membrane vesicles for intercellular communication. 19 The protein composition of exosomal vesicles resemble MVB since they arise from endosomes and include various fusion proteins (e.g., GTPases, annexins and flotillins), tetraspanins (e.g., CD9, CD63 and CD81) as well as heat shock proteins (e.g., HSP70). 20 In contrast to other microparticles, exosomes differ in their size and their formation within the multi-vesicular endosomes. 21 Eukaryotic extracellular vesicles are mainly classified into four different types; exosomes and microvesicles are shedding microparticles, apoptotic vesicles are apoptotic bodies or apoptotic blebs.22,23 Ectosomes, are membrane vesicles distinguished from exosomes based on their physicochemical characteristics, including size, density, appearance under microscopy, sedimentation rate, lipid composition, protein markers, and subcellular origin. 22 The ExoCarta database is a useful resource for the composition of exosomes. 24
Biogenesis
Multi-vesicular bodies which form a part of the endocytic pathway fuse with the plasma membrane to exude vesicles in the extracellular fluids.25,26 The large MVBs are characterized by the presence of ILV in their lumen. The ILVs release vesicles, referred to as “exosomes” upon fusion with the plasma membrane.4,27 During this process, certain proteins are incorporated into the invaginating membrane, and the cytosolic components are engulfed and enclosed within the ILVs. 28 Alternatively, they are degraded in the lysosome.26,29–31 MVBs fuse with the plasma membrane and subsequently release their cargo in the extracellular milieu through exocytosis (Figure 1).

Schematic representation of exosome biogenesis, and release of intraluminal vesicles. Several molecules of the ESCRT machinery participate in the genesis and transport of ILVs. Formation of primary endocytic vesicles triggers the formation of early endosomes (EE) which undergoes two pathways, either return the cargo to the plasma membrane as the “recycling endosomes” or transform into “late endosomes” or MVBs. The destiny of MVBs is either to fuse with the lysosome or with the plasma membrane, eventually releasing their cargo outside the cells. Several RAB proteins, RAB 27A and RAB 27B and SNARE (soluble NSF attachment proteins SNAPs) protein complexes aids in the transfer of MVBs to plasma membrane and the release of exosomes.
The formation of these membrane vesicles has an endocytic origin, accompanying secretion processes of the endosomal system. Endocytic vesicles, early endosomes, late endosomes, and lysosomes are the several components of this endosomal system. Endocytic vesicles arise through clathrin or non-clathrin-mediated endocytosis at the plasma membrane, which is then transported to early endosomes. Late endosomes develop from early endosomes by acidification. The early endosomes display a tubular appearance and are located at the outer margin of the cell, whereas late endosomes are spherical in shape, located close to the nucleus. The critical step in the formation of MVBs from late endosomes is reversed budding. During this process, the limiting membrane of late endosomes buds into their lumen, which results in a continuous enrichment of intraluminal vesicles. 25
Endosomal sorting complex required for transport in MVB biogenesis
During MVB generation, the entire Endosomal sorting complex required for transport
Different elements of the ESCRT complex:
ESCRT-0 complex plays vital role in MVB biogenesis as it binds and clusters ubiquitinated protein on the surface of the cell, and aids in binding lipids to endosomal membrane, subsequently recruiting the tagged proteins to endosomes. These proteins are taken into endosomes, forming multi vesicular bodies (MVB) which are then degraded in the lysosomes. 32 Ubiquitinated proteins are clustered by ESCRT-I and act as a bridge between ESCRT-0 and ESCRT-II complexes. 33 ESCRT-I also recruits ESCRT-III, thereby forming the constriction zone for the separation of cells. 34 However, ESCRT-II complex functions primarily during the biogenesis of multi-vesicular bodies (MVBs) and delivery of ubiquitin tagged proteins to the endosomes. Proteins tagged with ubiquitin passes from ESCRT-0 to ESCRT-I and then to ESCRT-II. Subsequently, ESCRT-II associates with ESCRT-III, and pinches the cargo containing vesicle. 35
MicroRNAs
The emerging role of miRNA as key regulators of biological function has gained momentum in the past few years and with the identification of presence of miRNAs in exosomes, the concept of cell communication has widened. Exosomal miRNAs are conserved, small non-coding, single stranded RNAs ranging in size between 21–23 nucleotides and have been found to play a critical role in the regulation of physiological and pathological processes. 36 These miRNAs exert their role by binding to the 3’ UTR region of the mRNA and post-transcriptionally silence gene expression.
The miRNAs have been shown to be stable in exosomes as they are protected from degradation and can travel through body fluids and be taken up by various cells, thereby leading to changes in their gene expression (Figure 2). The interesting aspect of miRNAs is that they can be used as non-invasive biomarkers of the disease state, as the exosomes in which they are present carry signatures of the cells from which they were shed. Profiling of exosomal miRNA has gained significance for not only diagnosing, but also as a marker for disease prognosis.

Brief insights into the role of exosome derived microRNAs (miRNA) in the pathogenesis associated with calcification. DC-dendritic cells; VC-vascular calcification; HT-hypertension; AS-atherosclerosis; MI-myocardial infarction.
There are various risk factors of renal and arterial calcification which include the traditional ones like age, hypertension (HTN), diabetes, lipid imbalance and genetics along with the non-traditional risk factors such as inflammation, oxidative stress, and altered mineral metabolism. 37 Although several studies have shown an association between calcification and hypertension, but whether hypertension or arterial calcification is the precipitating factor is a matter of debate. However, certain groups have suggested that pre-existing coronary arterial calcification (CAC) is positively associated with the development of HTN 38 according to the updated 2017 ACC/AHA guidelines. 39
In a recent report microRNA profiles were significantly altered in exosomes from VSMCs undergoing calcification. 40 Expression profiling has also revealed the significance of various miRNAs in controlling pulmonary vascular paracrine signaling in both arterial and pulmonary hypertension (PAH), which is a critical risk factor for calcification. Research on the mechanisms and therapeutic potential of microRNAs in hypertension has revealed the significant contribution of over 50 miRNA. 41 These miRNAs have been implicated in the pathways promoting HT by modulating proliferation, apoptosis, differentiation and vascular ECM remodeling 42 ; TGF-β pathway targeted by MiR-125-3p, miR-148-3p, and miR-193 43 miR-204 modulating the RhoA-ROCK pathway 44 ; miR-27b (Notch pathway); miR-214 (PTEN) 45 ; miR-130/301(Stat3, PPARγ) 46 ; miR-145 (KLF4).
Reports show that differential expression of specific miRNAs lead to the pathogenesis of hypertension. The up-regulation of circulating miR-130/301 could be a useful biomarker of pulmonary HT. The modulation of these miRNAs could represent a novel strategy for the management of hypertension which could prove to be beneficial for preventing or attenuating calcification.
Coronary artery calcification
Vascular calcification is a well-established hallmark and surrogate marker of AS, where narrowing of arteries occurs due to deposition of plaques in the arterial walls. The subendothelial retention of apolipoprotein B (apo B) in the focal areas of arteries marks the beginning of this disease. 47 The process by which coronary arteries of the heart calcify involves osteogenic alterations of the valve interstitial cells and smooth muscle cells (SMCs). The beginning of calcification does not require participation by specific cells, however, the progression of lesion is driven by chondrocytes and linked with the expression of inflammatory factors, such as cytokines. Vascular cells can undergo chondrogenic or osteogenic differentiation, causing mineralization and formation of bones. Calcification and inflammation both play essential roles in the progression of AS. 48 Inflammation marks the beginning of plaque formation and calcification of the vessel wall occurs in the subsequent stage, characterized by repeated cycles of inflammatory damage and repair.49,50
Types and mechanism of calcification in atherosclerosis
Depending upon the location of calcification, VC can be classified into two distinct forms, within the intima (intimal calcification), and medial calcification in the vascular medial layer. 48 Medial calcification is characterized by the loss of elasticity of peripheral arteries of lower extremities and occurs frequently in people affected with peripheral vascular disease. 51 Alternatively, intimal calcification forms at the site of atherosclerotic lesion and calcifying vascular cells are derived from local smooth muscle cells, as well as circulating hematopoietic stem cells. It resembles endochondral bone formation, and cytokines, and chondrocyte-like cells are associated with the advancement of the lesion. 52 There are two stages of vascular calcification; microcalcification, the initial stage of calcification and macrocalcification, the subsequent stage of macroscopic calcium formation. 53 Microcalcification represents the early stages of intimal calcium formation and may directly contribute to the rupture of fibrous plaques. 54 Calcification reduces inflammation, but if the inflammation continues and more microcalcification occurs, this results in macrocalcification, thereby stabilizing the plaque. There is progressive calcification, and the stability of plaque increases as plaque inflammation becomes pacified, thereby lowering the risk of plaque rupture. 48 Coronary artery calcification scoring is used as a predictive risk indicator in CHD patients. 38
Atherosclerotic plaques are built by oxidized lipoproteins, and cytokines, produced at the site of plaques by tissue macrophages and foam cells. The retention of apolipoprotein B (apo B) and lipoprotein (LPS) in the subintima initiates the formation of calcified atheroma, resulting in diffuse intimal thickening (DIT), composed of proteoglycans which bind to lipoprotein cholesterol and oxidized lipoprotein cholesterol. Eventually, chemoattractant peptides are produced, and monocytes are recruited to the site of the lesion. Macrophages are produced from monocytes and subsequently, these macrophages differentiate into foam cells, loaded with low density lipoproteins, forming “atherosclerotic plaques.” Macrophages, on the other hand, phagocytose the remnants of apoptotic cells that produce anti-inflammatory cytokines, such as interleukin (IL)-10 and transforming growth factor (TGF)-beta, which inhibit atherosclerosis development. However, the advancement of the lesion is marked by phagocytosis of apoptotic detritus, and instead of eliminating the apoptosomes, there is retention of residual cellular components within the lesion. The membrane integrity is lost, and toxic contents are released in the environment, causing further inflammation. There are two categories of macrophages that play an essential role in inflammation associated with atherosclerosis. The role of M1 macrophages is to produce inflammatory cytokines and continue inflammation, whereas M2 macrophages attenuate inflammation. Therefore, the calcifying cells show osteoblast-like or chondrocyte transdifferentiation to attenuate inflammation. Apoptotic bodies are released by the nucleation of matrix vesicles (MV) and hydroxyapatite on the extracellular vesicles in the process of oncosis from macrophages and smooth muscle cells, leading to microcalcification.
Exosomes in calcification of coronary arteries
Calcification of arteries is a common complication in patients with atherosclerosis and CKD. 55 The exosomes are derived from several atherosclerosis associated cells like; vascular endothelial cells, vascular smooth muscle cells, macrophages or monocytes, dendritic cells, and platelets.
Exosomes released from vascular endothelial cells
The vascular endothelium consists of a monolayer of endothelial cells (EC) and lines the inner walls of arteries, veins, and capillaries, and therefore it is in direct contact with the components and cells of the blood. The endothelium regulates the degree of vascular relaxation and constriction, and the extravasation of solutes, and macromolecules as it is a barrier between blood and tissues. 56 Endothelial dysfunction is linked to several diseases including AS and is marked by vasodilation, suppression of smooth muscle cell growth, and inhibition of inflammatory responses. 57 Among them, many of these effects are largely mediated by nitric oxide, the most potent endogenous vasodilator. Nitric oxide (NO) opposes the effects of endothelium-derived vasoconstrictors and inhibits oxidation of low-density lipoprotein (LDL). 15 Foam cells are formed by the deposition of lipids and monocytes in the subendothelial space due to altered morphology of endothelial cells and subsequently causing debilitation of vascular endothelial barrier function. The reduction in the synthesis of nitric oxide and rise in the secretion of endothelin lessens endothelium-dependent relaxation response, marking the onset of atherosclerosis. 58 Therefore, endothelium performs athero-protective roles by maintaining a balance between vasodilation and vasoconstriction as well as leukocyte adhesion and proliferation of smooth muscle cells (SMC).58–60 When this balance is disturbed, endothelial dysfunction occurs, resulting in the deposition of plaques in the arterial wall (Figure 3).

Endothelial dysfunction and Atherosclerosis. Cardiovascular and several emerging risk factors give rise to endothelial dysfunction (ED). Endothelial cell activation, caused by proinflammatory cytokines, turbulent flow and advanced glycation end products (AGEs) via transcription factor, NF-κB thereby induces several cell adhesion molecules, vascular cell adhesion molecule 1 (VCAM-1), Intercellular Adhesion Molecule 1 (ICAM-1), E-selectin, subsequently resulting in endothelial dysfunction. Loss of nitric oxide (NO), the most potent vasodilator results in increased activation of endothelial cells. Therefore, the decrease in production of nitric oxide (NO) leads to endothelial dysfunction, resulting in coronary artery calcification by increased vasoconstriction, smooth muscle cell proliferation, platelet aggregation, leukocyte adhesion and LDL oxidation.
Several microparticles derived from endothelial cells undergoing apoptosis are known to play an important role in the progression of cardiovascular disease.61,62 The vascular endothelial cells and vascular smooth muscle cells as well as cells of the immune system communicate with each other by exosomes synthesized by the endothelium and subsequently, play a distinguishing role in the detection of endothelial cell function and AS. 61 Upon tissue injury, a CXC chemokine CXCL12 is produced, and exerts its affect by binding to its receptor CXCR4. CXCL12 retards programmed cell death by mobilizing endothelial progenitor cells. Similarly, during the progression of atherosclerosis, the vascular endothelial cells produce apoptotic bodies, enriched with miR-126, which also promotes the production of CXCL12 and removes macrophages from the site of plaque formation, thereby reducing inflammation and exerting atheroprotective effects. 63
Exosomes released by vascular smooth muscle cells
VSMCs are the cellular components of the normal blood vessel wall that contribute to the structural integrity of the blood vessels and regulate the diameter of the vessels in response to vasoactive stimuli. 64 They are the primary cells that constitute the vascular wall, and therefore any structural or functional changes in them form the basis of various cardiovascular diseases. The secretion of exosomes from VSMCs is driven by Platelet-derived growth factor (PDGF) and TNFα. 65 The VSMCs undergo chondrogenic transformation in response to pathological signals such as, mineral imbalance or expression of inflammatory cytokines, resulting in the expression of bone-related proteins and release of matrix vesicles (MVs) (Figure 4). However, the mechanism of the origin and release of these particles are poorly understood.66,67 Electron microscopy (EM) studies have shown that matrix vesicles (MVs) form the nidus for mineralization.55,68 MVs are VSMC-derived exosomes enriched with exosomal tetraspanins CD9 and CD63 and CD81 and originate at the site of medial calcification from both living and apoptotic VSMCs, as well as from macrophages, endothelial cells, and atherosclerotic plaques.65,69
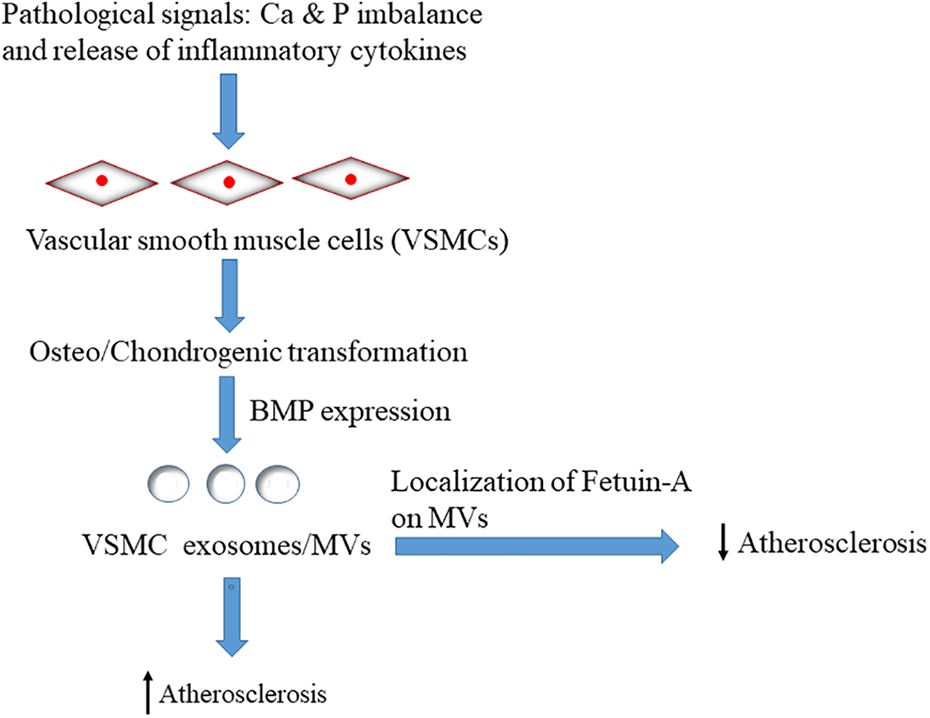
Factors involved in the release of exosomes from VSMCs. The pathological signals like; Ca(calcium) & P(phosphorous) imbalance along with the presence of inflammatory cytokines stimulates the release of exosomes or MVs from the vascular smooth muscle cells by the expression of bone morphogenetic protein (BMP), resulting in the advancement of atherosclerosis. Alternatively, the localization of fetuin-A, a calcification inhibitor on these exosomes reduces the development of atherosclerotic plaques.
A hallmark of arterial calcification is the phenotypic transdifferentiation of VSMCs to osteoblast-like cells. Generally, healthy VSMCs prevent VC by expressing calcification inhibitors. Fetuin-A, a potent calcification inhibitor is synthesized in the liver and secreted in the blood. VSMCs are involved in recycling of Fetuin-A, and loading it into VSMC-derived exosomes, thus preventing mineralization. However, when there is prolonged exposure of VSMCs to procalcific factors such as high Ca/P, the circulating Fetuin-A levels are reduced in patients with calcification. Elevated levels of extracellular calcium induce the expression of sphingomyelin phosphodiesterase 3 (SMPD3), and calcifying exosomes are secreted from VSMCs. The calcium stress induces dramatic changes in the composition of VSMC exosomes by the accumulation of phosphatidylserine (PS), Annexin A6, and matrix mettaloproteinase-2, subsequently converting exosomes into a center for calcification. On the other hand, chemical inhibition of SMPD3 prevents VSMC calcification.65,70 Consequently, when there is disturbance in the calcification activator and inhibitor, the exosomes of vascular smooth muscle cells speed up the process of VC. 65
Exosomes released by monocyte/macrophage
Monocytes and macrophages comprise the mononuclear phagocytic system (MPS), which play a key role in immune response. 71 At the site of pathogenic infection and inflammation, monocytes and macrophages are recruited, producing inflammatory cytokines and chemokines. The macrophages present at the site of atherosclerotic lesions engulf lipid droplets and form foam cells. Subsequently, the atherosclerotic plaque grows by the increasing number of foam cells at the site of lesion. 72 Cytokines and chemokines are produced by the macrophages causing further inflammation. The plaque grows and forms necrotic core consisting of dead macrophages. 73
Exosomes derived from monocytes are known to initiate apoptotic pathway in the endothelial cells thereby giving rise to several endothelial disorders. 74 The exosomes contain miRNAs in circulating blood cells, and monocytes/macrophages are released into circulation in response to various inflammatory stimuli. Vascular endothelial cells and monocytes/macrophages communicate with each other through miR-150 under pathological conditions, including chronic inflammation as seen in AS and hyperglycemia. Granulocyte-macrophage colony-stimulating factor (GM-CSF)) and Phorbol 12-myristate-13-acetate (PMA) stimulate the production of exosomes from monocytes. The healthy macrophages also shed exosome like microvesicles for the maintenance of homeostasis and assembly of immune cells. 75
Exosomes released from dendritic cells
Dendritic cells (DCs) are antigen presenting cells of the mammalian immune system that process antigen and present it to the T lymphocytes. They act like biosensors of the cellular microenvironment by detecting the presence of signals that determine T-cell tolerance and immunity. 76 Dendritic cells are found in the arterial walls as well as within atherosclerotic lesions. Atherosclerosis is marked by sub-endothelial retention of apolipoprotein B in the focal areas of the arteries triggering a chronic, non-resolving inflammatory disorder mediated by activation of the innate and adaptive arms of the immune system. 77 The retained lipoproteins undergo oxidative modification activating the endothelial cell and eventually, recruiting monocytes into the subendothelial region, in the arterial intima. These monocytes differentiate into macrophages and DCs and ingest the retained lipoproteins to become cholesterol ester-laden foam cells which mount a maladaptive and non-resolving immune response, promoting the progression of atherosclerotic process.78,79
The exosomes released by dendritic cells are involved in inflammation associated with atherosclerosis. Endothelial cells are invaded by exosomes from aorta aggravating inflammation via the exosome NF-κB pathway. 80 Dendritic cell derived exosomes play a crucial role in T-cell activation as they express several immune regulatory molecules, such as MHC class I and II, CD80 and CD86 and may show either immunostimulatory or immunosuppressive effects depending on their stage of maturation. According to Alexander et al., exosomes released from dendritic cells are enriched with miRNAs, which have inflammatory effects. Exosomal miR-155 enhances inflammatory responses whereas, miR-146a reduces inflammation associated gene expression. 81
Exosomes released from platelets
Platelet activation and endothelial damage play essential roles in AS. 82 Beyond the traditional role of platelets in promoting homeostasis and thrombosis, they are increasingly recognized as critical players in inflammation and AS. The interaction between platelets and leukocyte regulates the initiation, development, and progression of AS. Activated platelets stimulate the release of platelet-derived pro-inflammatory cytokines, promoting endothelial cell activation, subsequently enhancing monocyte adhesion and tissue infiltration. 83 Platelet adhesion and activation occurs at the site of endothelial dysfunction promoted by the pro-inflammatory microenvironment present there. This stimulates the release of platelet derived chemokines and the recruitment of neutrophils. Low density lipoproteins (LDL) are oxidized, inducing the production of reactive oxygen species (ROS) by neutrophils. 84 Hence, platelets contribute to LDL oxidation and endothelial dysfunction. In the course of infection, platelets communicate with leukocytes and control excessive inflammation. During AS, platelets hasten the formation of foam cells by interacting with monocytes, neutrophils, and dendritic cells, disrupting the plaques leading to their rupture. 85
Platelets secrete vesicular bodies which have been found to promote atherosclerosis by facilitating cell-cell interaction and adhesive interaction between vascular walls and blood. 86 The matrix vesicles stimulate the production of vascular smooth muscle cells as well as endothelial cells, facilitating atherosclerosis progression. 87 The exosomes secreted by platelets are rich in several miRNAs linked with platelet stimulation. The miR-223, miR-339 and miR-21 inhibits platelet-derived growth factor receptor-beta (PDGFRβ) expression in VSMCs and promotes platelet aggregation and coagulation processes, accelerating atherosclerosis. 88 Pro-inflammatory molecules; caspase-3 and RANTES (regulated on activation, normal T cell expressed and secreted protein) are transferred by these platelet MVs promoting monocyte adhesion and VC. 89 (Figure 5)

Pleiotropic effects of exosomes on vascular calcification. Exosomes can add to the progression of calcification in the vascular system by promoting endothelial dysfunction (ED) and apoptosis of endothelial cells (ECs). Under pathological conditions, EC derived exosomes release miRNAs which hastens endothelial dysfunction by the elevation of p38 (mitogen activated protein kinases), reducing the synthesis of endothelial nitric oxide synthase (eNOS) and nitric oxide (NO). Several matrix vesicles (MVs) in circulation stimulate the release of pro-inflammatory cytokines (IL-6 &IL-8) and express cell adhesion molecules (ICAM-1, VCAM-1 and E-selectin), enhancing monocyte adhesion and transmigration and aggravating inflammation and plaque development in the arteries. Similarly, the exosomes released by platelets promote VC either by their miRNAs (miR-223, miR-339 and miR-21) inhibiting platelet-derived growth factor receptor-beta (PDGFRβ) and promoting platelet aggregation and coagulation or by release of pro-inflammatory molecules; caspase-3 and RANTES (regulated on activation, normal T cell expressed and secreted protein) and increases monocyte adhesion and inflammation. The monocyte derived exosomes increase VC by stimulating the release of ICAM-1, CCL2 (chemokine C-C motif ligand 2) and IL-6 resulting in the apoptosis of ECs.
According to Li et al., miR-223 modulates the MAPK and NF-κB pathways by downregulating ICAM-1 expression in TNF-α-stimulated human umbilical vein endothelial cells (HUVECs). 82 The role of different exosomal RNAs in vascular damage and calcification is summarized in Table 1.
Role of various exosomal RNAs in atherosclerosis processes.

Calcification of arteries in chronic kidney disease
Chronic kidney disease affects 8–16% people globally, and cardiovascular diseases accounts for a large number of deaths in patients with CKD. 108 Though, vascular calcification is a pathological hallmark of aging but is accelerated in several diseases like diabetes, CKD, and AS and, therefore associated with higher risk of cardiovascular mortality in patients with CKD. 109 Furthermore, the majority of patients with chronic kidney disease have excessive vascular calcification. 110 Dysregulation of calcium (Ca) and phosphate (P) metabolism is common in CKD patients as the elevated levels of calcium and phosphorous promote osteogenic and chondrogenic differentiation of VSMCs, thus providing a major stimulus for calcification in these patients, thereby leading to vascular calcification. 111 Calciprotein particles (CPP), a complex comprised of Fetuin-A and minerals, are normally present in the serum in low amounts but in CKD patients its level is high. The calcification inhibitors; GRP (Gla rich protein) and Fetuin-A are downregulated in CKD patients and interact with the serum CPP resulting in increased calcification of VSMCs 112 (Figure 6).

Vascular smooth muscle cell derived exosomes in vascular calcification. The levels of calcium (Ca), Phosphorous (P) and its product calcium phosphate (CaP) is high in CKD patients and they have increased incidence of arterial calcification. Calciprotein particles (CPP) on interacting with Extracellular vesicles (EVs) released from VSMCs along with low levels of Gla rich protein (GRP) and Fetuin-A promote osteogenic/chondrogenic differentiation resulting in inflammation and calcification of VSMCs. On the other hand, healthy VSMCs inhibit calcification by forming complex with calcification inhibitors; Fetuin-A and Matrix Gla protein (MGP).
Defects in endogenous anti-calcification factors such as matrix Gla protein, Fetuin-A, and Klotho may play an essential role in complication associated with CKD.113,114
Role of Klotho in CKD
Klotho was originally identified as an anti-aging gene. However, its role in the calcification of kidneys was found much later. Klotho, a transmembrane protein functions as a co-receptor of fibroblast growth factor (FGF), and is expressed widely, but its level is highest in kidneys. 115 Chang et al., observed that the early stage of CKD is marked by graded reduction in urinary Klotho, and it ultimately progresses to loss of renal function. Studies have also shown that transgenic mice that overexpressed Klotho had improved renal function and reduced calcification. Klotho directly inhibits principal renal phosphate transporter, NaPi-2a leading to hypophosphatemia and phosphaturia. Thus, Klotho ameliorates VC as it enhances phosphaturia, preserves glomerular filtration, and directly inhibits phosphate uptake by VSMCs. 116
Matrix Gla protein
Matrix Gla protein (MGP), a member of vitamin-K2 dependent, Gla-containing proteins is a 10-kD protein. The protein acts as an inhibitor of vascular mineralization and plays a vital role in bone organization, as it is secreted by chondrocytes and VSMCs in the arterial media.117,118 It acts as a calcification inhibitor in vivo, probably by directly inhibiting calcium precipitation and crystallization in the vessel wall and inhibits bone morphogenetic protein-2 (BMP-2), which regulates osteoblast differentiation, and thus bone formation and also induces VC. 10 In earlier studies, it was shown that, MGP only exerts its anticalcifying activity after post-translational-glutamyl carboxylation of five glutamate residues, a crucial activation step that depends on the availability of vitamin K. 119 Moreover, MGP must undergo further phosphorylation on three serine residues in order to become fully active. 120 Subsequently, some studies have demonstrated an inverse association between total uncarboxylated MGP (ucMGP) and VC, 121 although data for its association with early stages of CKD are still lacking.
Fetuin-A and exosomal calciprotein particles: Inhibitors of calcification
Fetuin-A, secreted by the liver parenchyma cells, is an abundant, circulating protein present in the serum. 122 It is known to inhibit extra-skeletal calcification in states of serum supersaturation and also serves as a buffer of serum calcium and phosphate. 123 Several studies have shown that low levels of Fetuin-A are associated with cardiovascular mortality. 124 The development of calcific lesions is marked by loss of anti-calcific mechanisms, VSMC differentiation, chronic inflammation, increased ECM remodeling, as well as the release of EVs.65,112 CKD is associated with disturbances in mineral metabolism such as elevated calcium, phosphorous as well as its product, CaP, which are often associated with VC and mortality in CKD. 111 In physiological conditions also, the serum is nearly supersaturated with respect to Ca and P and therefore inhibitory mechanisms must exist to prevent extra-skeletal mineralization.
Recent studies have demonstrated the role of EVs in the process of calcification of soft tissues (ectopic calcification). CPP membrane-less particles, are fetuin-mineral complexes found in circulation, predominantly composed of Ca and P minerals. Fetuin-A is known to act as a potent inhibitor of ectopic calcification through the binding of small clusters of calcium and phosphate thereby lowering the supersaturation of these minerals and hence preventing their growth, aggregation, and precipitation. 125 CPP is formed when fetuin-A is present along with other proteins and can be considered as a mineral chaperone with a role in stabilization, transport, and recycling of water-insoluble minerals, to inhibit crystal formation or maturation. CPPs present in the circulation of healthy individuals differ from the CPPs present in CKD patients in terms of mineralization inhibitor levels and mineral crystal maturation.16,125 GRP is a constitutive component of both CPP and circulating EVs. It has been demonstrated both in vivo and in vitro that an increase in mineral maturation is associated with deficiency of both GRP and Fetuin-A. Incidentally, CPPs and EVs showing decreased levels of GRP and Fetuin-A were shown to promote calcification of VSMCs. 16
Stem cell derived exosomes
Considering the morbidity and mortality associated with calcification leading to AS and CKD, there is an urgent need for not only the early diagnosis but also for the management of these conditions. Stem cells are pluripotent in nature and therefore, capable of forming various specialized cell types within the body. Stem cell therapy offers hope for patients suffering from AS and CKD. However, barriers in stem cell therapy lies in the limited supply of stem cells that can be used for transplantation. As with the other cell types present in the body, many vesicular bodies including exosomes are released by stem cells.
Stem cell derived exosomes offer an exciting and novel strategy for the management of these disease conditions (Figure 7). These exosomes with their interior cargo of miRNA, proteins and lipids can be taken up by cells and facilitate paracrine signaling and thereby modulate key pathways involved in calcification. 126 Mesenchymal stem cell (MSC) derived exosomes have been shown to downregulate inflammation in the cardiovascular system through the action of miRNAs which target macrophage infiltration and polarization and inhibit autophagy. 127

Stem cell derived exosomes in the management of disease outcome.
Stem cell derived exosomes promote angiogenesis and are less liable for immune rejection, and subsequently may serve as favorable treatment option for CVD. 128 The secretion of stem cell derived exosomes having the ability to protect against AS can be enhanced by treatment with drugs such as statins 129 and hence offers an attractive treatment design.
However, conflicting reports exist regarding the role of stem cell derived exosomes. Studies show that cardiac progenitor cell-derived exosomes (CPC-exo) play a crucial role in the progression of atherosclerosis as they induce oxidative stress, apoptotic cell death and inflammatory changes at the site of lesion. 130 Exosomes are involved in the formation of fibrous caps in the atherosclerotic plaques, a key feature of microcalcification.131,132 The fact that exosomes are abundantly present in almost all body fluids, provides the ease of its non-invasive collection and subsequently, serve as biomarkers for a number of diseases including different types of tumors and cancers of the prostate gland and ovaries, besides CVD. 128
Clinical findings of phase 2/3 trials of stem cell derived exosomes in CKD have shown promise 133 and an improvement in eGFR, creatinine and inflammation was noted. However, the field of stem cell-derived exosome for effective diagnosis and treatment is still in its infancy and hence further investigations are warranted.
Conclusion
In this review the role of exosomes in pathological conditions such as, arterial calcification and chronic kidney disease has been explored. Recent studies highlight the key role of exosomes in cardiovascular diseases and the findings suggest that the contents and quantities of exosomes are variable under different cardiovascular conditions. The contribution of exosomal miRNAs in the calcification process can be exploited for designing miRNA drugs targeting specific genes for effective management of cardiovascular diseases. Liquid biopsies which contain the exosomes can be used for the non-invasive sampling of biological fluids for the diagnosis as well as the prognosis of the extent of calcification. The changes in the composition of exosomes can be used strategically to monitor the response to various treatments and miRNA signatures can be developed for monitoring the presence, as well as, extent of calcification. The role of exosomes derived from different atherosclerotic cells as well as stem cells in the progression of cardiovascular diseases, through communication between several cells associated with vascular calcification has also been highlighted. However, the establishment of exosomes as biomarkers in CVDs is still years away as it requires efficient and standardized detection methods and robust clinical trials.
Footnotes
Acknowledgment
The authors are thankful to Amity Institute of Biotechnology, Amity University, Noida for providing technical support and Indian Council of Medical Association (ICMR), New Delhi for the Junior Research fellow (JRF) grant. We are very grateful to Dr Manoj Garg, AIMMSCR, for helping in the preparation of the figures by the Biorender application.
Author contributions
All authors have contributed to the intellectual content of this paper and have met the following requirements: drafting or revising the article and significant contributions to the conception and design of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
