Abstract
Introduction:
Environmental arsenic contamination is a major toxicological problem worldwide due to its carcinogenic and nephrotoxic potential.
Aim:
The purpose of this observational study was to determine the suspected association between urinary arsenic (uAs) and urinary leucine (or leucyl) aminopeptidase 3 (uLAP3) to evaluate uLAP3 as a candidate biomarker of exposure to airborne arsenic.
Materials and Methods:
A total of 918 adults occupationally and/or environmentally exposed to airborne arsenic were enrolled in the study. Baseline information (age; sex; history of smoking; alcohol, fish and seafood consumption) was gathered. Total uAs concentrations [μg/L] of 918 subjects, as well as the sum of arsenic species (ΣiAs) in 259 subjects, were obtained. Urinary LAP3 was measured by an immune-enzymatic assay using an ELISA kit. Urinary creatinine concentration was assessed with the IB/lAB/1289 research protocol (version II, 2015-09-17). The values of uAs and uLAP3 were recalculated per unit of creatinine. The association between uAs and uLAP3 was assessed using a logistic regression model adjusted for confounders.
Results:
The study identified a positive correlation between the logarithm of uAs and the logarithm of uLAP3 in the study population (r = 0.1737, p < 0.0000) and between urinary creatinine and uLAP3 concentration not adjusted for creatinine level (r = 0.1871, p < 0.001). In the logistic regression model, there was also an association between increased (≥15 µg/L) uAs and decreased (below the 25th quartile) uLAP3 [OR uLAP3 = 1.22 (95% CI 1.03 to 1.44, p < 0.02)].
Conclusions:
These data suggest that urinary LAP3 may be a potential biomarker of arsenic exposure, which warrants further study.
Introduction
Environmental arsenic contamination is a relevant toxicological, geochemical and ecological issue worldwide. Millions of people are exposed to arsenic through drinking water, mainly in Taiwan 1 and Bangladesh 2 ; however, exposure to airborne arsenic has been recognized as a local problem in areas with copper mines and copper smelters. 3
Arsenic has been associated with the development of cancer, 4,5 kidney damage, 6 cardiovascular diseases 7,8 and diabetes mellitus. 9 Ingesting drinking water containing a high concentration of arsenic at is a risk factor for lung cancer; however, it is unclear whether there is such an association in the case of low levels of exposure to arsenic. 10 Consuming drinking water polluted with arsenic has also been found to be a risk factor for basal cell and squamous cell skin cancers, as well as renal, ureteric and bladder cancers. 4,5,11 Less is known about the carcinogenic effects of airborne arsenic. According to World Health Organization (WHO) arsenic guidelines (2019), tobacco smoking is associated with exposure to natural inorganic arsenic. The source of the inorganic arsenic in tobacco is soil and lead arsenate-containing insecticides used in tobacco cultivation.
Kidneys are particularly sensitive to the toxic effects of arsenic that enters the body via both the alimentary and inhalatory routes. Arsenic nephrotoxicity generally depends on the blood arsenic concentration. 12 In subjects chronically exposed to arsenic, toxic and carcinogenic effects may be exerted through oxidative stress and inflammation. 13,14 Oxidative stress is associated with an increase in specific markers, such as urinary isoprostanes, whereas inflammation is associated with an upregulation of tumor necrosis factor-α and interleukin-6.
In humans, leucine aminopeptidases are early markers of nephropathy 15 and cancer. 16 –18 These enzymes are associated with tumor cell proliferation, invasion and angiogenesis. LAP3 expression is significantly upregulated in hepatocellular carcinoma cells and closely correlates with decreased differentiation, metastasis to lymphatic nodes and a poor prognosis. 17 Overexpression of LAP3 contributes to the development of human esophageal squamous cell carcinoma. 19 LAP3 promotes glioma progression by regulating the proliferation, migration and invasion of glioma cells. 20 In breast cancer, LAP3 overexpression plays a crucial role in tumor metastasis. 21
Leucine (leucyl) aminopeptidases (LAPs) preferentially catalyze the hydrolysis of leucine residues at the N-terminus of peptides and proteins. These enzymes are active in the presence of manganese, magnesium and zinc ions. LAPs have been identified in bacteria, parasites, plants and humans. In several bacterial species, LAP acts as a DNA-binding protein and repressor or activator of operon regulation of virulence-associated genes. 16 LAP from Plasmodium falciparum is being studied as a promising target for novel antimalarials. 22 Impaired hydrolysis of RNA and proteins in rice seedlings due to the inhibitory effects of arsenic on RNase and LAP activity has been observed. 23
The established role of arsenic and the suspected involvement of LAPs in carcinogenesis and nephrotoxicity was the rationale for conducting our investigation. The purpose of this observational study was to determine the suspected association between urinary arsenic and urinary LAP3 and to evaluate LAP3 as a useful marker of exposure to airborne arsenic in a population residing within a copper smelter impact zone. In this zone, the airborne arsenic concentration exceeded the accepted limit, whereas the concentration of arsenic in drinking water was low.
Material and methods
In 2017, a group of 918 volunteers environmentally exposed to airborne arsenic was enrolled in the study. The inclusion criteria were aged over 18 years and living in a zone in which the arsenic air concentration exceeded the upper limit recommended by European Environment Agency (EEA), i.e. 6 ng/m3. All participants were from a region where the average annual arsenic air concentration was 30.2 ng/m3 in 2017 and 10.04 ng/m3 in 2018, according to the Provincial Inspectorate for Environmental Protection, whereas the arsenic concentration in the drinking water was lower than 10 µg/L. The exclusion criterion was fish, seafood or alcohol consumption in the 4 days preceding blood sample collection to minimize the effects of dietary intake of arsenic on the measured parameters.
Baseline information (age, sex, history of smoking, alcohol consumption, recent fish and seafood consumption) was gathered via a questionnaire. Participants were divided into subgroups based on age, namely, below 40, between 40 and 60 and above 60 years old, as well as smoking habits: current smokers, former smokers and nonsmokers (the reference group). Further subgroup division was based on alcohol consumption, which was defined as regular (at least once a week), occasional (less than once a month) and abstinence (the reference group). Among the 918 people, 15 did not provide information about smoking, 22 about alcohol consumption, and 25 about fish and seafood consumption. Considering the whole sample, the missing data represented less than 2.7% and did not significantly affect the final results.
The study population was divided into two exposure groups: a large group of subjects who were environmentally exposed to arsenic and a small sample of subjects who were exposed to arsenic both environmentally and occupationally. The second group included 79 men who were copper miners or copper smelters, aged 38.3 ± 8.0 years and occupationally exposed to arsenic for at least 10 years.
Given the increasing evidence that urinary arsenic may differ between sexes, 24 potential differences between men and females environmentally exposed to arsenic were also explored.
This study was conducted in accordance with the Declaration of Helsinki (1964). Participants provided written, informed consent to participate in the study. The project received a positive opinion from the local ethical committee (No: KB-125/2015). The methodology followed the STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) checklist.
Urinary arsenic
The total urinary arsenic (uAs) concentrations [μg/L] in the 918 participants, as well as the sum of arsenic species (ΣiAs), including inorganic arsenic (iAs), monomethylarsonic acid (MMA) and dimethylarsinic acid (DMA) [μg/L] of 259 subjects, were measured at the Central Institute of Occupational Medicine (Lodz, Poland) using HPLC-ICP-MS. 25
Urinary LAP3 and creatinine concentrations
Biological samples were collected between September 29, 2017, and December 8, 2017. Urine samples taken in the morning after overnight rest and samples for the determination of arsenic in urine, were simultaneously collected in 10 milliliter tubes with a descriptive code and placed in a freezer at −80°C. Biochemical measurements were performed between January and May 2018. Urinary LAP3 was measured by an immune-enzymatic assay using an ELISA kit (Wuhan EIAab Science Co., Ltd., Wuhan, China) and a Multimode reader SYNERGY/lX (BioTekSynergy/lX, Vermont, USA). Positive and negative controls were tested to ensure interlaboratory quality control for uLAP3 determinations. For each set of test samples, a standard curve was constructed according to the manufacturer’s instructions, the target enzyme concentration in each sample was determined in duplicate, and the obtained values were correlated with the curve.
Urinary creatinine (ICD: M37) was measured according to the IB/LAB/1289 research protocol (version II, 2015-09-17), with a reference range 28–217 mg/dL, in an accredited laboratory (PCA No. AM003).
Urinary arsenic and LAP3 values, expressed in μg/L and μg/mL, respectively, were recalculated per unit of creatinine.
Statistical analysis
Summary statistics were calculated for each variable; means ± SDs or medians and quartiles were calculated for continuous variables, and n (%) was calculated for categorical variables for the whole study population and for each subgroup. A natural logarithmic transformation [ln(x)] was applied to the variables with skewed distributions: uAs and sum iAs+MMA+DMA (ΣuAs). This transformation allowed for the stabilization of variances for parametric model assumptions and reduced the influence of extreme values. Multiple regression analysis was used to assess the effect of urinary arsenic on uLAP3. Our final logistic regression models were built by the backwards stepwise method and adjusted for sex, age, smoking status, alcohol consumption, and occupational exposure to arsenic. In the first logistic regression step, qualitative and quantitative predictions were defined. The coding of qualitative variables with sigma limitations (quasi-experimental) was performed. Potential predictors were evaluated by logistic regression, a one-factor analysis. A p-value of 0.05 was used as the cutoff to indicate statistical significance. The TIBCO Statistica 13.3 program was used.
Results
The characteristics of the study population are presented in Table 1. The most numerous subgroup in the study was females over the age of 40. The number of participating males was approximately half that of participating females. Seventy-nine men were not only environmentally but also occupationally exposed to arsenic.
Characteristics of the studied population.

*, ** statistically significant differences between groups of men and women; * p < 0.05, ** p < 0.01.
The distribution of urinary creatinine across the study population is presented in Table 2 (group sizes are the same as those in Table 1).
The distribution of urinary creatinine in studied groups.

p—the level of statistical significance of the difference between women and men.
The distribution of urinary LAP3 (uLAP3) and total urinary arsenic (uAs) values was lognormal. The median uLAP3 (Q25; Q75) values among 310 males and 608 females environmentally exposed to airborne arsenic were similar, while the median uAs in males were higher than those in females. However, after adjustment for urinary creatinine level, the median uLAP3 in males was lower than that in females (p < 0.001), whereas the median uAs concentration was similar in both groups (Table 3).
Urinary leucine aminopeptidase 3 (LAP3) and arsenic (uAs) before and after adjustment for creatinine level.

**, *** statistically significant differences between groups of women and men: **p < 0.01, *** p < 0.001.
These differences may be because the mean urine creatinine concentration was approximately 25 mg/dL higher in males than in females (Table 2).
The multiple regression analysis showed a positive correlation between ln(uAs) [μg/g creatinine] and ln(uLAP3) [μg/µg creatinine] in the entire study group (Figure 1).

Relationship between logarithm of total urinary arsenic and logarithm of urinary leucine aminopeptidase 3 in the entire studied population (r = 0.1737, F = 28.52, b = 0.1654, t = 5.34, p < 0.0000).
Additionally, a positive linear correlation between ln(uAs) and urinary creatinine was found (Figure 2).

Correlation between logarithm of urinary total arsenic and urinary creatinine (r = 0.3108, F = 97.9, b = 0.3108, t = 9.89, p < 0.0000).
A total urine arsenic concentration exceeding the upper limit accepted by WHO, i.e. ≥15 μg/L, was observed in 259 people, including 90 males and 169 females. In these males and females, the median (25Q; 75Q) total uAs concentrations were 28.15 (20.5; 66.0) μg/L and 30.4 (20.0; 57.0) μg/L, respectively. The sums of arsenic species (arsenite—iAsIII, arsenate—iAsV, MMAV and DMAV) were measured in these groups. A positive correlation between the logarithm of the sum of arsenic species ln(ΣAs) and ln(uLAP3) was observed (Figure 3).
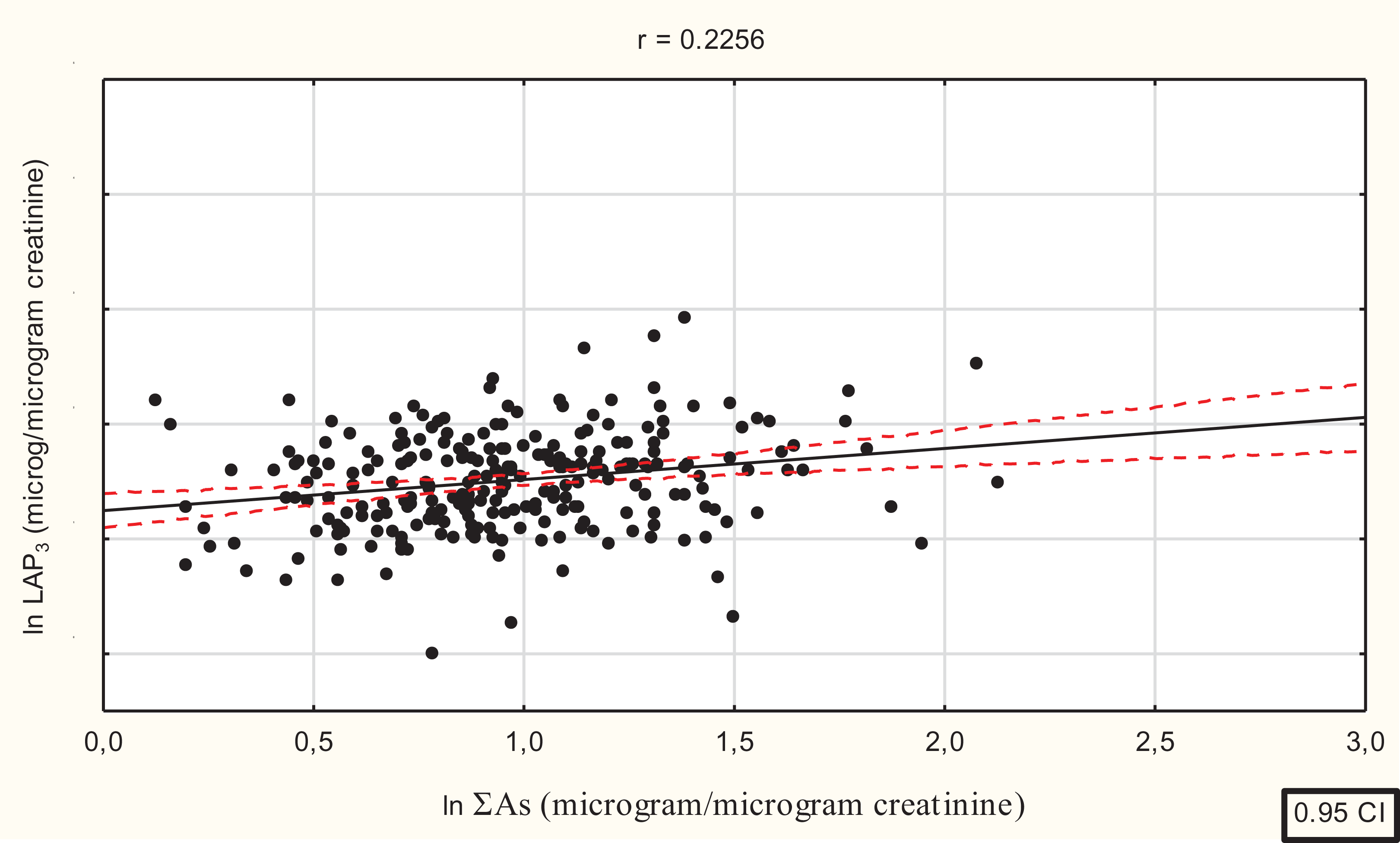
Relationship between logarithm of the sum of urinary arsenic species (lgΣuAs) and logarithm of uLAP3 in people with total arsenic urine concentration exceeding the upper norm accepted by WHO (r = 0.2256, F = 13.57, df = 1.25, b = 0.2256, p < 0.001).
In the entire study population, uLAP3 [μg/µg of creatinine] was positively correlated with age (Spearman’s rho = 0.2671; r = 0.14805, p < 0.001). There was a gradual increase in uLAP3 with age in both males and females as well as smokers and nonsmokers (Table 4). No relationship between age and uLAP3 expression in ng/mL was found (r = −0.0148, p > 0.05). This discrepancy was probably caused by the reduction in urinary creatinine concentration with age (Table 2).
Urinary LAP3 (uLAP3) in smokers vs non-smokers exposed to airborne arsenic.
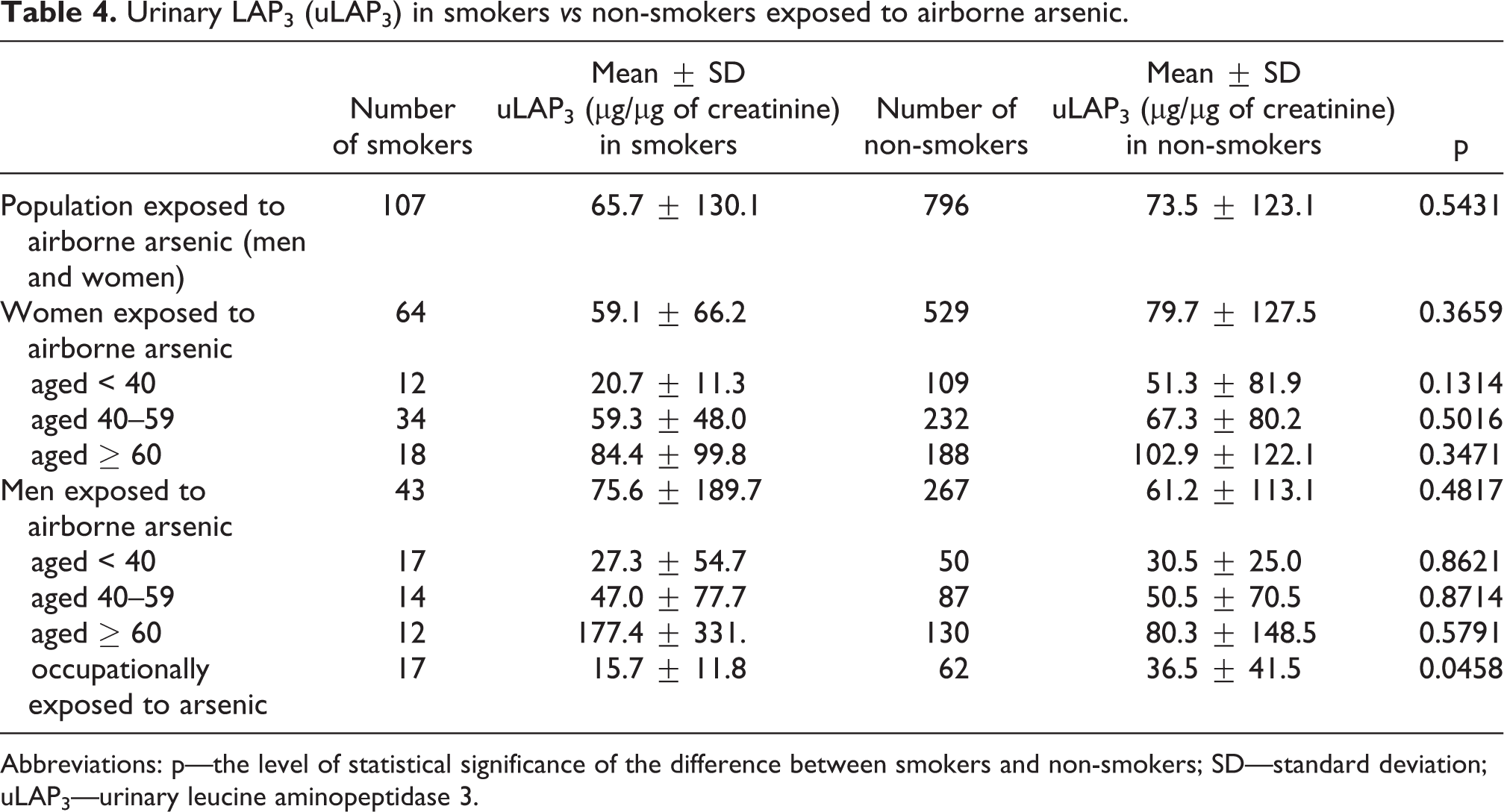
Abbreviations: p—the level of statistical significance of the difference between smokers and non-smokers; SD—standard deviation; uLAP3—urinary leucine aminopeptidase 3.
The effect of cigarette smoking on uLAP3 concentration was generally not statistically significant. Lower uLAP3 concentrations were found in smokers than in nonsmokers, except for males over 60 years old (Table 4). In the entire study population, there were no significant differences in uLAP3 between nonsmokers (N) and former smokers (F). Both nonsmokers and former smokers had higher uLAP3 concentrations than current smokers (S) (Figure 4). The urinary creatinine concentration was only slightly higher in smokers than in nonsmokers (105.1 ± 60.9 mg/dL vs 98.4 ± 63.2 mg/dL, respectively, ns).

Urinary LAP3 concentration in non-smokers (N), past-smokers (P) and current smokers (S). Statistical differences in Kruskal-Wallis test, * p < 0.05.
In males, another LAP3-modifying factor was occupational exposure to arsenic. Urinary LAP3 was lower (p < 0.001) in the 79 males occupationally exposed to arsenic than in the 231 nonexposed males (32.0 ± 38.1 and 74.5 ± 142.9 μg/µg creatinine, respectively).
The urinary arsenic concentration in the group of any smokers was higher than that in the group of nonsmokers, but the difference was not significant (p > 0.05). There were also no significant difference in arsenic concentration between smokers and nonsmokers in the group of workers occupationally exposed to As (Table 5).
Urinary arsenic (uAs) in smokers vs non-smokers exposed to airborne arsenic.

Abbreviations: p—the level of statistical significance of the difference between smokers and non-smokers; SD—standard deviation; uAs—urinary arsenic.
However, the multivariate analysis of variance showed a significant interaction (p < 0.05) between sex, smoking and uAs impacting uLAP3 [β = 0.1311; 95% confidence interval (CI), 1.71–26.81]. Smoking males with normal urine arsenic concentrations had higher uLAP3 levels than nonsmoking males, whereas smoking males with elevated (≥15 µg/L) uAs had lower uLAP3 levels than nonsmokers (Figure 5).
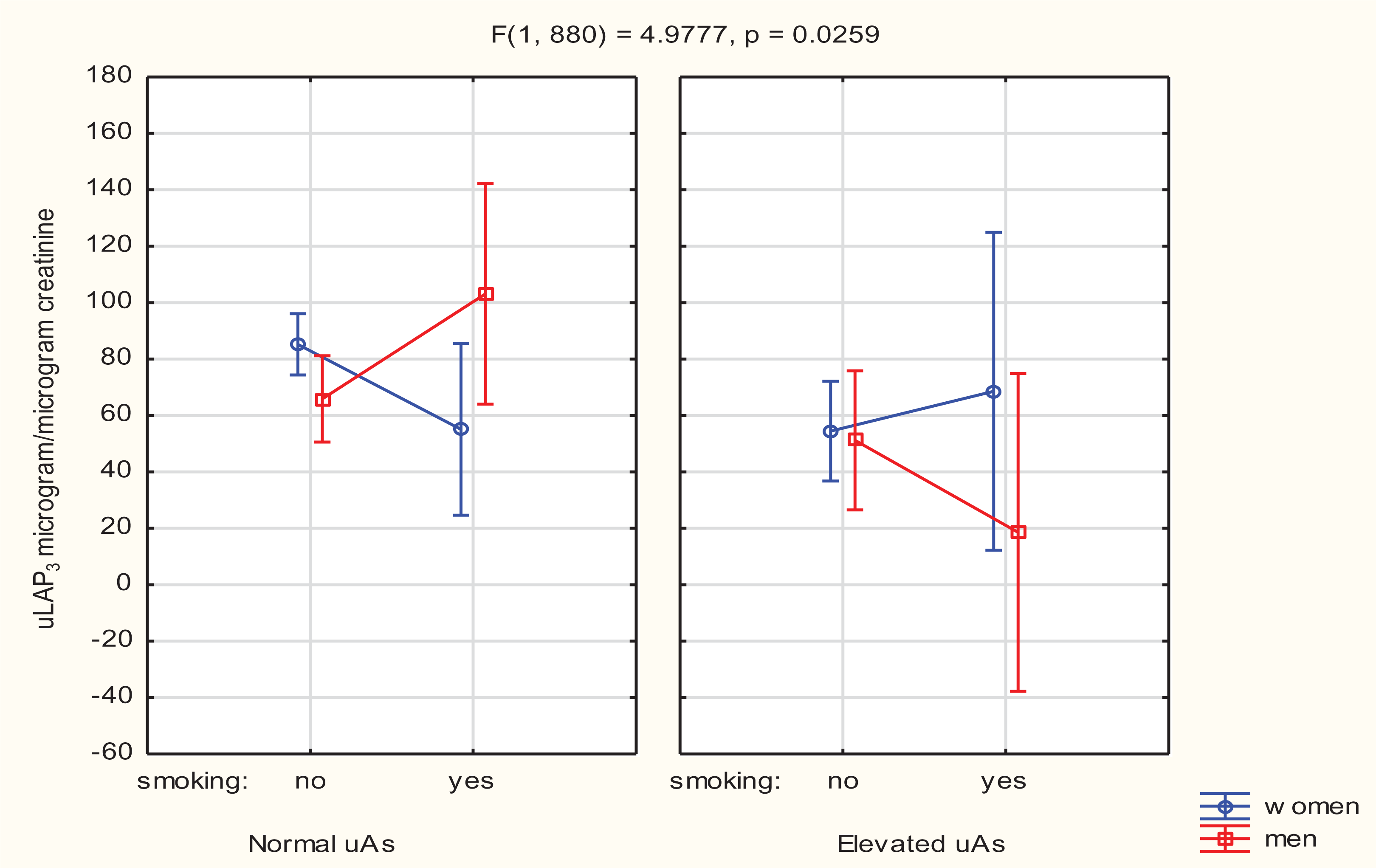
Interaction between sex, smoking and total urinary arsenic in the study population.
Alcohol consumption had no significant effect on uLAP3 or uAs concentrations. The urinary LAP3 concentrations were 64.9 ± 122.3 μg/µg creatinine in occasional drinkers (n = 99), 73.9 ± 114.1 μg/µg creatinine in regular drinkers (n = 182) and 72.9 ± 124.2 μg/µg creatinine in abstainers (n = 635). The mean uAs concentrations were 25.2 ± 55.2 μg/g creatinine in occasional drinkers, 24.4 ± 54.2 μg/g creatinine in regular drinkers and 24.9 ± 56.3 μg/g creatinine in abstainers.
In all the studied subjects (n = 918), the uLAP3 concentration expressed in ng/mL increased linearly with urinary creatinine concentration (r = 0.1871, p < 0.001), Figure 6.
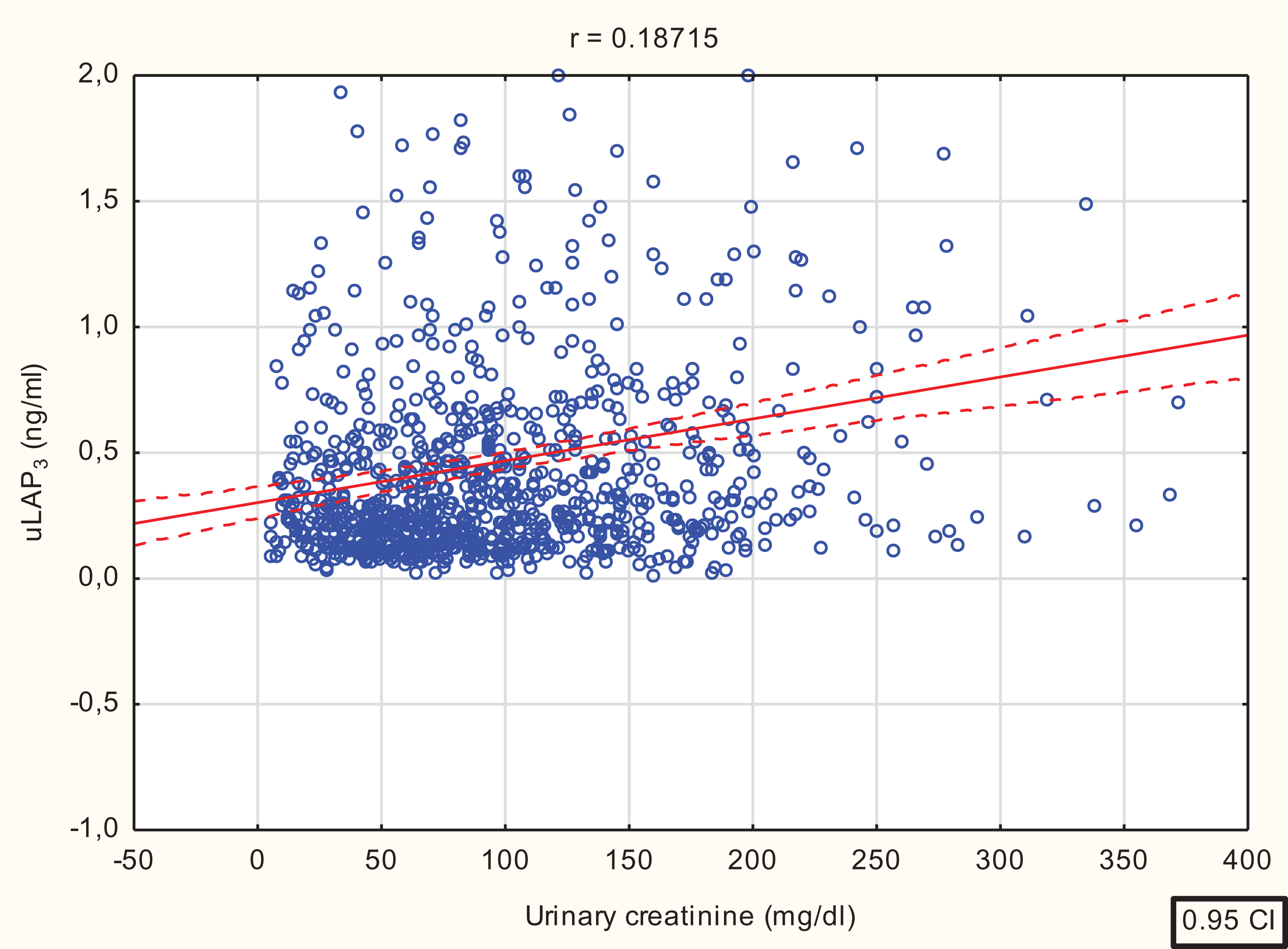
Correlation between urinary creatinine concentration and uLAP3 (r = 0.1871, F = 35.06, b = 0.1871, t = 5.92, p < 0.0000).
Multiple logistic regression model
A multiple logistic regression model was constructed to determine whether there were significant associations of normal and abnormally high (≥15 μg/L) total urinary arsenic (a qualitative predictor) with an increased concentration of uLAP3 as the dependent variable. For further statistical analyses, the value of the 75th quartile (Q75) for uLAP3 (0.54 ng/mL = 72 μg/µg creatinine) was adopted as the cutoff point. Values higher than the Q75 were considered elevated in the study population. The influence of confounders such as sex, age, smoking, alcohol consumption, occupational exposure to arsenic and urinary creatinine was taken into account.
In the total study population exposed to airborne arsenic, a significant association between urinary creatinine and increased uLAP3 was observed [ORuLAP3 = 1.020 (95% CI 1.016 to 1.024; p < 0.001)]. Creatinine was also associated with increased uAs [ORcreatinine = 1.006 (95% CI 1.004 to 1.009; p < 0.001)].
In the logistic regression model, an association between normal (lower than 15 μg/L) urinary arsenic and elevated urinary leucine aminopeptidase 3 was shown [ORuLAP3 = 1.33 (95% CI 1.11 to 1.59), p = 0.0017].
The odds ratio for the association between elevated uAs (≥15 μg/L) and increased uLAP3 (ORuLAP3) was 0.73 (95% CI 0.60 to 0.88, p < 0.001). The proportions of people with elevated uLAP3 were 47/259 (18.1%) in the group with elevated uAs and 186/659 (28.2%) in the group with normal uAs.
In the next step, a logistic regression model using uLAP3 values below the 25th quartile (Q25) as the dependent variable was constructed. Urinary LAP3 values lower than the Q25 (0.16 ng/ml = 18,9 μg/µg creatinine) were observed in 242 people (133 females and 109 males), with a mean concentration of 0.11 ± 4.52 ng/mL. There was an association between increased uAs (≥15 μg/L) and decreased (below Q25) uLAP3 [ORuLAP3 = 1.22 (95% CI 1.03 to 1.44, p < 0.02)].
Discussion
In this study, an association between kidney function (assessed on the basis of urinary creatinine concentration) and urinary arsenic was shown in the population environmentally exposed to airborne arsenic. Moreover, in people with normal urinary arsenic levels (< 15 μg/L), a positive relationship between uAs and uLAP3 was found.
Urinary arsenic mainly indicates recent exposure to inorganic arsenic. Most clinical studies on inorganic arsenic and renal outcomes have focused on proteinuria, showing that exposure to both high and low arsenic levels is associated with increased proteinuria. 6 The nephrotoxic effect of arsenic had also been demonstrated in experimental studies. 26,27 Due to high metabolic activity, the microsomal fraction of the brush border in proximal tubule cells is the part of the nephron that is most susceptible to toxic damage. 28,29 Leucine aminopeptidases are kidney brush border enzymes. Urinary levels of these enzymes indicate damage to the microsomal or membrane fraction. 30 In many studies, urinary leucine aminopeptidase was identified as a sensitive indicator of early renal damage. 15,31,32
In our study, an increase in uLAP3 was correlated with an increase in total arsenic as well as the sum of arsenic species. It remains unclear whether the relationship between uAs and uLAP3 results from the direct effect of arsenic on proximal tubule cells and/or from the indirect effect on other parts of the nephron, such as glomeruli. The latter is supported by the observation of a significant association between creatinine and increased uLAP3 as well as between creatinine and increased uAs in the multiple logistic regression model. Since impaired glomerular filtration is the most likely cause of creatinine increase, the existence of these associations could indicate complex nephron dysfunction.
Based on our research, we concluded that the determination of urinary LAP3 together with urinary creatinine may be useful in the assessment of kidney function in populations exposed to airborne arsenic. One of the key findings was that in all the studied subjects, the uLAP3 concentration expressed in ng/mL increased linearly with the urinary creatinine concentration. Additionally, an association between creatinine and increased uLAP3 was shown in the logistic regression model. This indicates a significant influence of creatinine level on uLAP3. This relationship is clearly visible in the analysis of uLAP3 by subject age; decreases in creatinine levels due to age were accompanied by increases in the uLAP3 values in both males and females. However, this was observed for only uLAP3 values expressed in nanograms per microgram of creatinine; the uLAP3 values expressed in nanograms per milliliter of urine were not influenced by age. As urinary creatinine and uLAP3 increased together with urinary arsenic, nephron dysfunction was most likely caused by arsenic exposure.
Similar to other authors, 33 we observed sex differences in urinary excretion of LAP3. After adjustment for creatinine level, uLAP3 was higher in females than in males. This may be due to lower urinary creatinine levels in females than in males, resulting in an increased uLAP3/creatinine quotient.
The relationship between smoking and uLAP3 seems to be ambiguous and depends on sex. Among normo-arsenic-exposed females, uLAP3 levels were lower in smokers than in nonsmokers, whereas among normo-arsenic-exposed older males, uLAP3 levels were higher in smokers than in nonsmokers. The interaction between smoking, urinary arsenic and sex may impact the uLAP3 concentration (Figure 5), explaining these differences. This observation is in alignment with the results from another study, which demonstrated that an interaction between sex and smoking influenced the risk of bladder cancer, and differences in arsenic exposure effects in males and females were noted. 34 In a different study, a positive interaction between smoking and urinary arsenic, affecting the risk of lung cancer, was shown. 35
In recent years, there has been increasing interest in the role of leucine aminopeptidase 3 in the neoplastic process. LAP3 is involved in pro-oxidative, inflammatory and proliferative processes. 14,15 LAP3 overexpression is a known predictor of various human malignancies, e.g. acute promyelocytic leukemia, 36 ovarian cancer, 18 breast cancer, 21 prostate cancer 37 and hepatocellular carcinoma. 17 Novel LAP3 inhibitors are being investigated as potential agents in cancer therapy.
It should be noted that uLAP3 below the Q25, observed in 242 (26%) people, was associated with elevated uAs (≥15 μg/L) in our study. This association between elevated uAs and decreased uLAP3 may warrant further research.
Why exposure to arsenic, a known carcinogen, 35 is associated with low urinary levels of LAP3, as LAP3 normally increases in cancer cells and tissues, remains unclear. Inhibition of LAP3 by arsenic could explain the antitumor effect of arsenic in some proliferative diseases. Arsenic has long been used in the treatment of leukemia. Currently, research is focused on both the carcinogenic and anticancer effects of inorganic arsenic and its methylated metabolites. 36,38
Our study has several limitations. First, detailed data on participants’ lifestyles and health statuses were missing. Second, while urinary arsenic species are the recommended biological indicators of exposure to inorganic arsenic rather than total urinary arsenic species, they were measured only in 26% of the total study population. Moreover, although urinary arsenic concentration is a result of short-term exposure and therefore probably sensitive to considerable fluctuation, it was measured only once in the studied subjects, and its variability over time was not assessed.
Despite these weaknesses, we believe that urinary leucine aminopeptidase 3 could be a potential biomarker of arsenic exposure. Although this study did not provide evidence of arsenic nephrotoxicity, the positive relationship between urinary excretion of arsenic and uLAP3 in normo-arsenic-exposed subjects may indicate proximal tubular dysfunction. The observed association between elevated uAs and decreased uLAP3 warrants further research.
Conclusions
The results of our study provide evidence that urinary LAP3 is associated with the excretion of arsenic in urine. Establishing the role of LAP3 in urine as a biomarker of arsenic exposure should be the subject of future research.
Footnotes
Acknowledgements
Results of total urinary arsenic concentrations as well as sum of arsenic species were provided courtesy of The Copper Health Centre.
Data availability statement
The detailed data can be made available at the Editor’s request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wroclaw Medical University (Grant Number KO/68/U/17).
