Abstract
Fructus Psoraleae (FP), widely used in traditional medicine, is increasingly reported to cause serious hepatotoxicity in recent years. However, the main toxic constituents responsible for hepatotoxicity and the underlying mechanisms are poorly understood. In the present study, psoralen, a main and quality-control constituent of FP, was intragastrically administered to Sprague-Dawley rats at a dose of 60 mg/kg for 1, 3 and 7 days. Blood and selected tissue samples were collected and analyzed for biochemistry and histopathology to evaluate hepatotoxicity. The results showed that psoralen could induce hepatotoxicity by enhanced liver-to-body weight ratio and alterations of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total cholesterol after administration for 3 days. In addition, histopathological examinations also indicated the hepatotoxicity induced by psoralen. Furthermore, the mRNA and protein levels of hepatic bile acid transporters were significantly changed, in which MRP4, ABCG5 and ABCG8 were repressed, while the protein level of NTCP tended to increase in the rat liver. Taken together, psoralen caused liver injury possibly through affecting bile acid transporters, leading to the disorder of bile acid transport and accumulation in hepatocytes.
Introduction
Fructus Psoraleae (FP), the dried fruit of Psoralea corylifolia L., is a commonly used traditional Chinese medicine (TCM) with numerous therapeutic applications in osteoporosis, osteomalacia, and skin diseases such as vitiligo, psoriasis and cutaneous T-cell lymphoma. 1 The application of FP in TCM prescriptions is very extensive, and there are more than 30 kinds of commonly used proprietary Chinese medicines containing FP. In recent years, hepatotoxicity of FP has attracted widespread attention. Since the first clinical report about the acute hepatitis associated with large consumption of FP in 2005, there are more and more clinical and experimental studies as to its hepatotoxicity. 2 –4 For example, a 53-year-old woman was diagnosed with acute cholestatic hepatitis associated with FP after treated with Qubaibabuqi tablets for approximately 7 months. 5 Meantime, Zhuangguguanjie Wan and Baishi pills, whose main components include FP, have been reported to induce adverse reactions characterized by acute cholestatic liver damage by the State Food and Drug Administration of China (National center for ADR Information Bulletin, China, 2008). 1 So far, it is definitely certain that FP is associated with acute cholestatic hepatic injury. However, the exact toxic compound contributing to the hepatotoxicity of FP and the related mechanism remain unclear.
The main constituents of FP include, among others, fatty oil, resin, bakuchiol, psoralen and isopsoralen. Psoralen and isopsoralen are the quality-control markers in FP and the main components in blood after oral administration of FP. Psoralen is also found naturally in the common fig, celery, parsley, West Indian satinwood and in most citrus fruits. Modern pharmacological studies showed that psoralen exert anti-inflammatory, antipyretic, anticancer, antibacterial, and estrogenic activities. 6,7 In clinic, psoralen is primarily used to treat various skin diseases such as psoriasis, vitiligo, and chronic graft versus host disease. 8 However, there have been several reports of liver injury caused by psoralen which might reveal the hepatoxic compound of FP in part at least. 9,10 Up to now, the evidence for psoralen hepatotoxicity especially in vivo are not sufficient and the mechanisms still remain ambiguous.
Bile acids (BAs) are produced by hepatocytes from cholesterol that play an important role in the intestinal absorption and transport of lipids, nutrients, and vitamins. The hepatic uptake and efflux processes of bile acids are maintained by distinct transporters located at liver cells. On one hand, canalicular transporters are in charge of clearance and secretion of bile acids across the canalicular membrane of hepatocytes into bile, including bile salt export pump (BSEP), multidrug resistance-associated protein 2 (MRP2) and so on. 11 Meanwhile, ATP-binding cassette subfamily G member 5/8 (ABCG5/8) have been identified to play a role in the hepatobiliary excretion of cholesterol. 12 On the other hand, basolateral transporters play a key role in the uptake of bile acids from the sinusoidal blood plasma, such as sodium taurocholate co-transporting polypeptide (NTCP). 13,14 However, multidrug resistance-associated protein 4 (MRP4) and organic solute transporter α (OSTα), also located at the basolateral membrane, are responsible for pumping out bile acids from liver to blood. When the balance of uptake and efflux of bile acids is impaired, which may be due to a disturbance of the canalicular and basolateral transporters, cholestatic liver damage has occurred.
In the present study, we investigated the toxicity effect of psoralen on SD rats treated for 1, 3 and 7 days. It was observed that administration with psoralen induced liver injury determined by relative liver weight, blood biochemistry and histopathology of liver. The gene and protein expression of several bile acid transporters were also assessed for the purpose to explain the underlying mechanism for hepatotoxicity.
Materials and methods
Reagents
Psoralen was purchased from Tianjin Yueyahu Co., Ltd. (Tianjin, China) and the content is more than 98%. Gum tragacanth powder was purchased from Beijing Solarbio Science & Technology Co., Ltd. (Beijing, China).
Animals and treatment
Male and female Sprague-Dawley rats, weighting 170–200 g, were purchased from Beijing Huafukang Bioscience Co. Inc. (Beijing, China). All the animals were kept in the Specific Pathogen Free (SPF) animal room with free access to laboratory food and water and were maintained under a 12 h light/dark cycle. After 5 days of acclimatization, the rats were randomly divided to four groups, 12 animals in each group with 6 females and 6 males. The experimental procedure was shown in Figure 1. The psoralen groups were treated with psoralen at 60 mg/kg (p.o.) for 1, 3 or 7 consecutive days, while the control group was treated with 5% gum tragacanth solution. The volume of administration is 10 ml/kg. After the drug treatment, the animals were anesthetized with tribromoethanol. Then the blood was taken from the abdominal aorta and the organs were collected. All animal studies were carried out according to the Guidelines for the Care and Use of Laboratory Animals and approved by Animal Ethics Committee of Tianjin University of Traditional Chinese Medicine.

Experimental design for animal treatment. The rats were treated with vehicle (5% gum tragacanth solution) or psoralen (60 mg/kg) by gavage once a day (n = 12) for 1, 3 or 7 days. Serum and liver samples were collected for further analysis.
Blood biochemistry
Blood samples were collected in anticoagulant-free tubes. Serum was separated by centrifugation for 10 min at 4000 g. Serum biochemical analysis was performed with Automatic Analyzer (7020, Hitachi). Hepatic injury was assessed by the levels of alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase (ALP), total bile acid (TBA), total bilirubin (TBIL), triglycerides (TG), and total cholesterol (TC) in serum.
Histopathology
The liver samples were fixed in 4% buffered paraformaldehyde, embedded in paraffin, cut into 4-μm-thick sections and stained with hematoxylin–eosin (H&E). A light microscope was applied to assess the histopathological changes. An random scope was given to each sample viewed at magnifications of ×200.
Real-time PCR
The liver samples were lysed with TRIzol reagent (Invitrogen, USA) according to the manuscript with some modifications. First, cut up the liver samples with scissors. Then the samples were disrupted by Vibra-cell processors (Sonics, USA). One microgram of RNA was reverse transcribed to cDNA using the TransScript All-in-One First-Strand cDNA Synthesis SuperMix (Transgen Biotech, Beijing, China). The primers used for real-time PCR were described in Table 1 (Sequences of primers for real-time PCR). Gene expression was detected with the TransStart Tip Green qPCR SuperMix (Transgen Biotech, Beijing, China), and was determined by normalizing to GAPDH using the CFX96 Real-Time System (Bio-Rad, USA).
Sequences of primers for real-time PCR.

NTCP: Sodium-taurocholate co-transporting polypeptide; BSEP: Bile salt export pump; MRP2: Multidrug resistance-associated protein 2; MRP4: Multidrug resistance-associated protein 4; OSTα: Organic solute transporter α; ABCG5: ATP-binding cassette subfamily G member 5; ABCG8: ATP-binding cassette subfamily G member 8; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Western blotting
The liver samples were lysed in lysis buffer for 30 min in ice-bath after cut up by scissors and disrupted by Vibra-cell processors (Sonics, USA). Then the sample was centrifuged for 30 min at 12000 revolutions/min and the supernatant was collected. The protein concentration of the sample was assessed by Pierce BCA Protein Assay Kit (Invitrogen, USA). Equal amounts of protein were separated by SDS-PAGE gel and followed by subsequently transferred to PVDF membrane (Millipore, USA). The membrane was blocked with 3% BSA (Sigma–Aldrich, USA) and then incubated with primary antibodies, including NTCP (1:1000 dilutions; Invitrogen, USA), BSEP (1:500 dilutions; Santa Cruz Biotechnology, USA), MRP2 (1:500 dilutions; Boster Biological Technology, China), MRP4 (1:1000 dilutions; Cell Signaling Technology, USA), OSTα (1:500 dilutions; Boster Biological Technology, China), ABCG5 (1:500 dilutions; Invitrogen, USA), ABCG8 (1:500 dilutions; Boster Biological Technology, China) and β-actin (1:1000 dilutions; Cell Signaling Technology, USA). Horseradish peroxidase-conjugated goat anti-rabbit IgG antibody was applied as the secondary antibody (1:5000 dilutions; KPL, USA). Protein bands were detected by the Enhanced Chemiluminescence Plus Detection System (GE, USA). The Gel-Pro Analyzer software (Media Cybernetics) were applied to quantify the specific protein brands.
Statistical analysis
The data were presented as means ± SEM in each group. The data were statistically analyzed by one-way ANOVA followed by Holm-Sidak test using GraphPad Prism. The difference was determined to be significant if the P value was < 0.05.
Results
Psoralen-induced liver injury
As depicted in Figure 2, the body weight of each group showed no difference before treatment. After treatment with psoralen for 1, 3 or 7 days, the body weight still showed no difference compared with the control group. However, the liver weight in the group treated for 3 days increased markedly. The liver/body weight ratio was also much higher in the group treated with psoralen. These results indicated that psoralen oral administration might induce hepatotoxicity. Hence, liver injury in rats was evaluated by measuring the concentrations of ALT and AST in serum, the biomarkers of liver health. As shown in Figure 3, serum ALT and AST levels were increased in psoralen-treated rats, which were indicative of hepatocellular injury. The levels of serum ALP, TBA, TBIL and TG showed no difference after psoralen treatment. Notably, the levels of serum TC increased profoundly in psoralen-treated rats compared with the control. Furthermore, treatment with psoralen for 3 days significantly enhanced the serum TC level compared with 1-day treatment. According to the above data, treatment with psoralen for 3 days seemed to induce the hepatic injury.

(a) Body weight, (b) liver weight and (c) liver/body weight ratio in control and psoralen (1, 3 or 7 days)-treated rats. *P<0.05, **P<0.01 and ***P<0.001 vs. control (means ± SEM, n = 12).
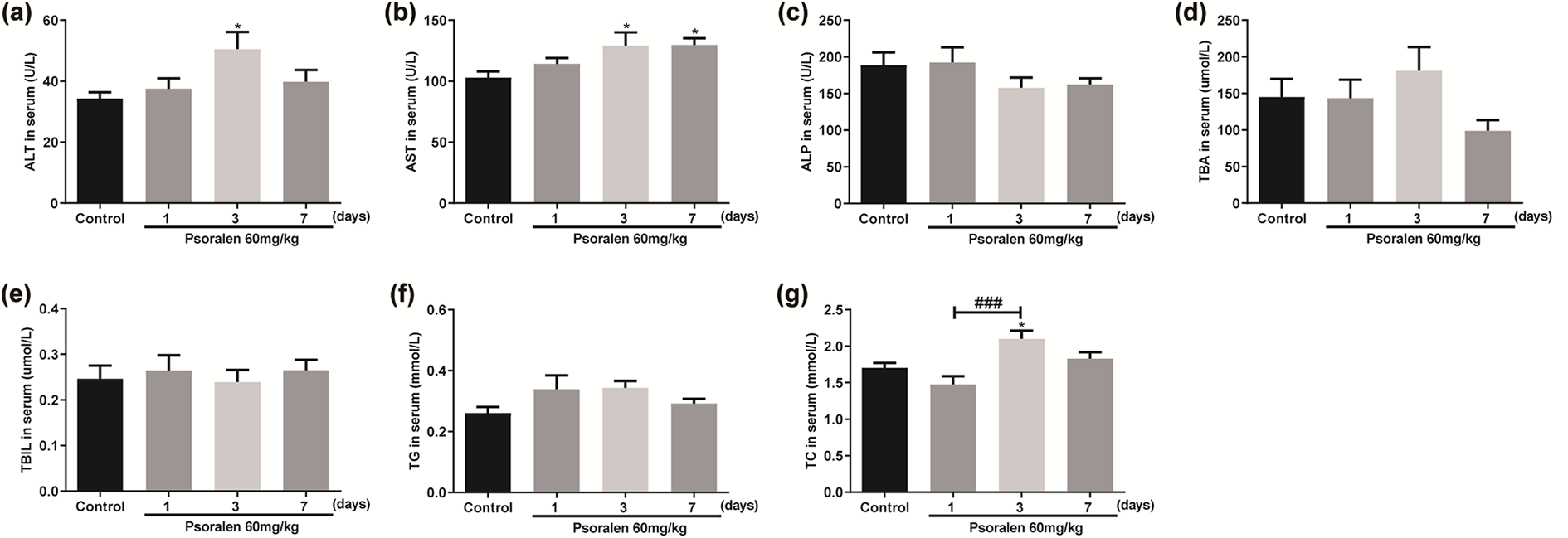
(a) ALT, (b) AST, (c) ALP, (d) TBA, (e) TBIL, (f) TG and (g) TC in control and psoralen (1, 3 or 7 days)-treated rats. *P<0.05 vs. control, ###P<0.001 vs. psoralen-treated for 1-day group (means ± SEM, n = 12). ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; TBA: total bile acid; TBIL: total bilirubin; TG: triglyceride; TC: total cholesterol.
The effect of psoralen on liver pathology
To determine the hepatic injury induced by psoralen, we conducted histological examination. According to microscopic examination, control rats had no appreciable pathological changes as shown in Figure 4. Rats treated with psoralen for 1 day showed almost no difference with the control. After 3 days of psoralen administration, minor necrosis of liver cells and ballooning degeneration were observed. The hepatocyte necrosis and degeneration were further exacerbated in rats after psoralen was administered for 7 days. The results of both histopathology and blood biochemistry suggested administration of psoralen for 3 days might cause hepatic injury.

Histopathological examination of liver in the rats of (a) control group, (b) psoralen for 1 day group, (c) psoralen for 3 days group and (d) psoralen for 7 days group by H&E. Images were captured using an microscope with 200 magnification. H&E: hematoxylin and eosin.
The effect of psoralen on hepatic bile acid transporters
Since excessive of bile acids have been considered as a common reason of liver injury and bile acid transporters contribute mostly to the regulation of bile acids, we furthermore measured the gene expression of bile acid transporters. In our study, the mRNA levels of NTCP remarkedly decreased after treated with psoralen for 1 or 3 days. Compared with the 1-day group, the mRNA levels of NTCP of 3-day and 7-day group were significantly increased. As shown in Figure 5(b), the mRNA levels of BSEP were significantly decreased after treated with psoralen for 7 days compared with the control. However, the mRNA level of BSEP in 3-day group was markedly improved compared with 1-day and 7-day group. Interestingly, the ratio of BSEP/NTCP was increased after treated for 1 day, while decreased significantly after treated for 3 or 7 days. The mRNA levels of transporters taking charge of transporting bile acids from liver to bile duct, including MRP2, ABCG5 and ABCG8, were much lower compared with control group. The other transporters responsible for pumping out bile acids from liver to serum, such as MRP4 and OSTα, were also decreased remarkably (Figure 5). The above results indicated that the treatment of psoralen altered the balance between roll in and roll out of bile acids.

The mRNA levels of (a) NTCP, (b) BSEP (c) BSEP/NTCP, (d) MRP2, (e) MRP4, (f) OSTα, (g) ABCG5 and (h) ABCG8 in the livers of control and psoralen (1, 3 or 7 days)-treated rats. *P<0.05, **P<0.01 and ***P<0.001 vs. control, #P<0.05 and ###P<0.001 vs. psoralen-treated for 1-day group, @@P<0.01 vs. psoralen-treated for 3 days group (means ± SEM, n = 5).
To validate the protein levels of NTCP, BSEP, MRP2, MRP4, OSTα, ABCG5 and ABCG8 of rats administered with psoralen for 1, 3 or 7 days, Western Blotting was performed (Figure 6). The protein content of NTCP showed an upward trend after treated with psoralen. Instead, the other bile acid transporters, including MRP4, ABCG5 and ABCG8, were decreased significantly compared with control group. Furthermore, the protein level of ABCG8 in 7-day group was significantly reduced compared with the 1-day and 3-day group. The protein levels of MRP2 and OSTα decreased slightly without statistically difference. However, there was little change in the protein level of BSEP after administration of psoralen. The above results confirmed that psoralen-induced bile acids excretion decreased, and uptake increased. Therefore, the content of bile acids was increased in rat liver, which may cause cholestatic liver damage.
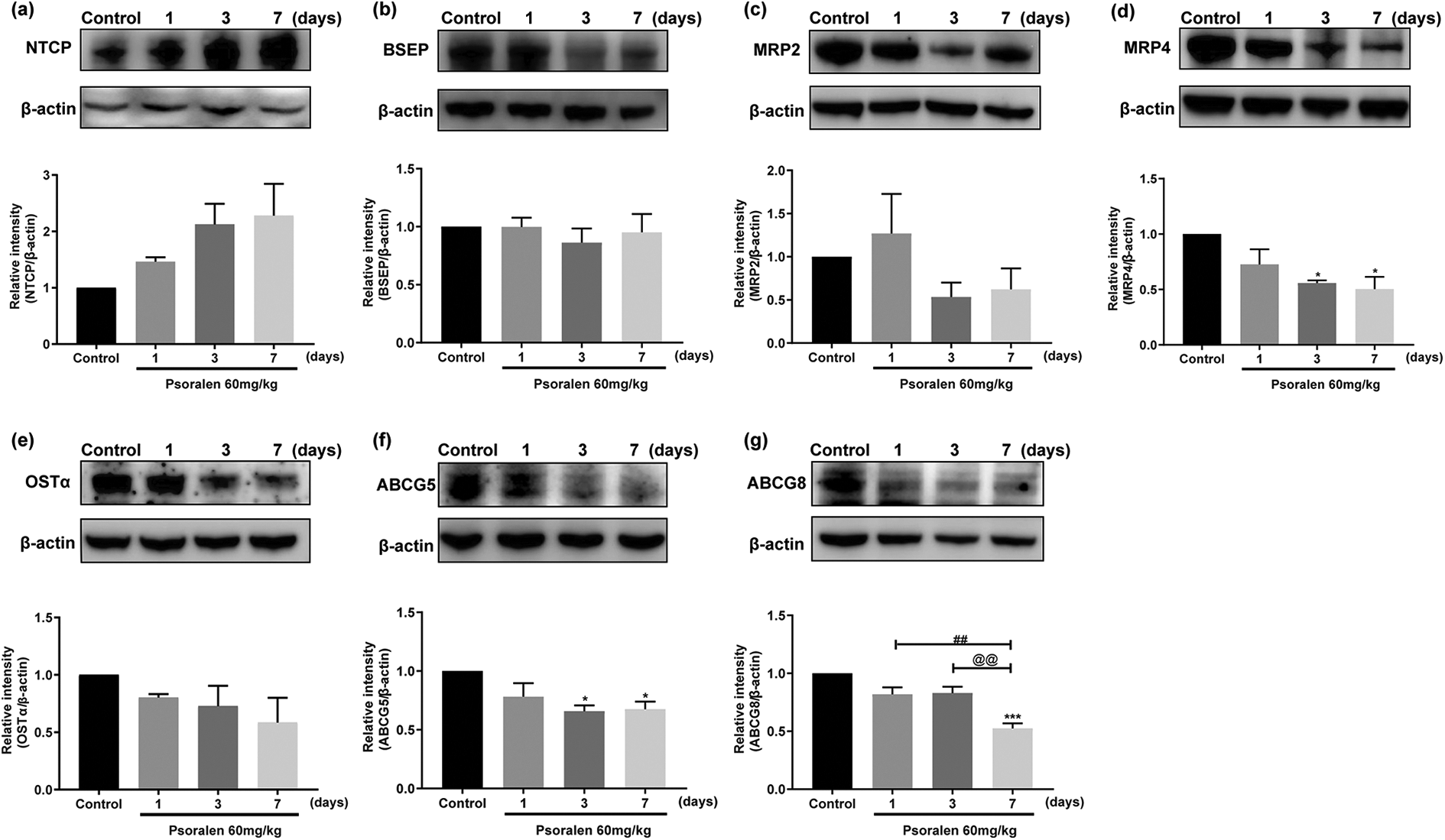
The protein levels of (a) NTCP, (b) BSEP, (c) MRP2, (d) MRP4, (e) OSTα, (f) ABCG5 and (g) ABCG8 in the livers of control and psoralen (1, 3 or 7 days)-treated rats. *P<0.05 and ***P<0.001 vs. control, ##P<0.01 vs. psoralen-treated for 1-day group, @@P<0.01 vs. psoralen-treated for 3 days group (means ± SEM, n = 3).
Discussion
The occurrence of hepatotoxicity cases linked to FP has raised serious concerns regarding FP safety. There are several patients experienced acute liver injury when only FP was used to treat vitiligo. 15 Psoralen, a furocoumarin, is one of the main active constituents of FP. A previous study has found that psoralen showed hepatotoxicity in mice which may be associated with FP-induced cholestatic liver injury. 16 The main cause of liver damage may lie in that psoralen impairs the bile acids flow in the liver, resulting in the accumulation of toxins in hepatocytes. 17 Hence, we are going to identify the effect of psoralen on hepatic bile acid transporters in rats.
In this study, we explored the hepatotoxicity effect of psoralen on rats treated for 1, 3, or 7 days. After administration, the liver coefficients were increased remarkably. In addition, there were clear increased trends in the levels of ALT, AST and TC in the serum of rats after 3 days administration of psoralen. Histopathological examination showed that hepatocyte necrosis and ballooning degeneration occurred after psoralen treated for 3 or 7 days. Our results showed that psoralen might cause liver injury in rats after administration for 3 days. However, several lines of previous researches indicated that psoralen caused hepatotoxicity after about 28 days or even longer time exposure at a mild dose in rats. 17 ,18 Our data suggested that psoralen might cause hepatotoxicity after administration for just 3 days at 60 mg/kg.
Bile acids, derived from cholesterol in the liver, constitute the main components of enterohepatic circulation of bile, which play an important role in liver function, liver physiology and metabolic regulation. Bile acids could not only facilitate the biliary excretion of cholesterol, prevent the formation of gallstones, but also promote the absorption of lipids and nutrients in the intestine. In cholestatic liver diseases, a large amount of bile acids accumulate in the hepatocyte, leading to liver injury. 19 Hepatic bile acid transporters are responsible for transporting bile acids and drugs. Therefore, any effect on these transporter systems can result in accumulation of potentially harmful bile acids or elevation of hepatic uptake of exogenous chemicals, and then may arouse liver cell damage. In addition, reduced or even absent expression of hepatic bile acid transporters, is a critical cause for various cholestatic liver diseases. 20
At the basolateral membrane of hepatocyte, Na+-dependent bile salt uptake pathway is regarded as the major bile acid transport system due to the uptake of 80% of the total taurocholate. 21 As one of them, NTCP recruits one molecule of conjugated bile acid along with two Na+ down its gradient into the hepatocytes. 22 While, at the canalicular membrane of hepatocyte, BSEP is a critical transporter in charge of bile acids secretion and maintaining low intracellular levels of poisonous bile acids. 23 Earlier molecular and genetic researches demonstrated that genetic mutations in BSEP gene were closely related to progressive familial intrahepatic cholestasis (PFIC). Due to the exclusive role of BSEP in the bile salt excretion from the hepatocyte, the amount of primary bile salts in the bile of patients with serious PFIC-2 decreased to about 1% of normal. 24 MRP2, members of the MRPs subfamily, is responsible for pumping divalent BAs into the bile canaliculi. 25 In mutant rats lacking Mrp2, the bile flow exhibits a reduction by about 50%. 26,27 The previous studies have certified that both BSEP and MRP2 are major contributors to bile acid-independent and bile acid-dependent bile flow, respectively. 13,28 –30 Besides BSEP and MRP2, MRP4 and OSTα are also involved in the efflux of bile acids. MRP4, belonging to the efflux pumps of basolateral hepatocyte membrane, have been identified to play a significant compensatory role in cholestasis and devote to the balance between uptake and efflux of substances during the vectorial transport from sinusoidal blood into bile. 31 According to the study showing in mice lacking Ostα distinctly decreased intestinal bile acid absorption, serum bile acid concentration and bile acid pool size, it seems that OSTα is the principal basolateral bile acid transporter in the intestine and many other epithelial cells. 32,33 Apart from those, the secretion of biliary free cholesterol mediated by ABCG5/8 is also an important way to clear hepatic cholesterol. For example, the biliary cholesterol concentration markedly decreased in mice lacking ABCG5 and ABCG8 while reversed in mice transgenic expression of ABCG5 and ABCG8. 34 In summary, impaired bile acids secretions disrupt bile flow and lead to cholestasis or cholesterol gallstone disease.
Our study found that psoralen decreased the mRNA levels of NTCP, BSEP, MRP2, MRP4, OSTα, ABCG5 and ABCG8. And, most transporters’ mRNA levels changed significantly after the first day of administration. However, as the most essential transporters of BAs, the ratio of BSEP and NTCP mRNA levels was significantly increased after treated for 1 day, while tended to decrease after treated for 3 or 7 days. Further, the protein level of NTCP tended to increase by psoralen after administration for 1, 3 or 7 days, while the expression of BSEP didn’t show significant difference. The above results suggested that psoralen may enhance the uptake of BAs in hepatocyte from the sinusoidal blood by maintaining high protein level of NTCP. Due to the reduced protein levels of MRP4, ABCG5 and ABCG8, psoralen may reduce the efflux of BAs in hepatocyte. Hence, our data suggested that psoralen could cause the disorder of bile acids balance by regulating hepatic bile acid transporters, which may be the underlying mechanism of psoralen-induced liver injury. Since the levels of hepatic bile acid transporters are tightly controlled by nuclear receptor superfamily, such as the farnesoid X receptor (FXR) and the pregnane X receptor (PXR), our future work will focus on the exact target by which psoralen affects the hepatic bile acid transporters.
In summary, the present study elucidated that psoralen induced liver injury in rats after 3 days administration at 60 mg/kg. The possible mechanism of liver injury was associated with hepatic bile acid transporters (Figure 7), by which psoralen broke the balance between roll in and roll out of bile acids. Our research would provide experimental basis to evaluate the safety of clinic application of psoralen and Fructus Psoraleae.

Effect of psoralen on related bile acid transporters in rat liver.
Footnotes
Author contributions
Juyang Huang and Kun Zhou proposed the study concepts and designed the study; Juyang Huang, Yanan Bi, Hong Shi and Qin Wang performed the experiments and data analysis; Mengying Chen, Yanan Bi and Hong Shi helped to prepare the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National Natural Science Foundation of China (No. 81673826), National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (2014ZX09304307-001-005) and the Science & Technology Development Fund of Tianjin Education Commission for Higher Education (2017KJ136).
