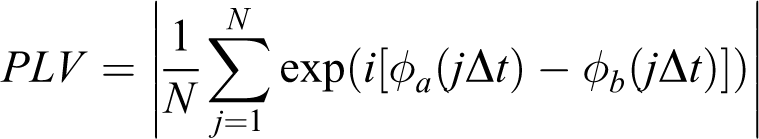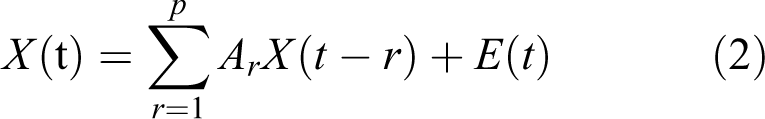Abstract
Although melamine exposure induces cognitive deficits and dysfunctional neurotransmission in hippocampal Cornus Ammonis (CA) 1 region of rats, it is unclear whether the neural function, such as neural oscillations between hippocampal CA3–CA1 pathway and postsynaptic receptors involves in these effects. The levels of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR) subunit glutamate receptor (GluR) 1 and GluR2/3 in CA1 region of melamine-treated rats, which were intragastric treated with 300 mg/kg/day for 4 weeks, were detected. Following systemic or intra-hippocampal CA1 injection with GluR2/3 agonist, spatial learning of melamine-treated rats was assessed in Morris water maze (MWM) task. Local field potentials were recorded in CA3–CA1 pathway before and during behavioral test. General Partial Directed Coherence approach was applied to determine directionality of neural information flow between CA3 and CA1 regions. Results showed that melamine exposure reduced GluR2/3 but not GluR1 level and systemic or intra-hippocampal CA1 injection with GluR2/3 agonist effectively mitigated the learning deficits. Phase synchronization between CA3 and CA1 regions were significantly diminished in delta, theta and alpha oscillations. Coupling directional index and strength of CA3 driving CA1 were marked reduced as well. Intra-hippocampal CA1 infusion with GluR2/3 agonist significantly enhanced the phase locked value and reversed the melamine-induced reduction in the neural information flow (NIF) from CA3 to CA1 region. These findings support that melamine exposure decrease the expression of GluR2/3 subunit involved in weakening directionality index of NIF, and thereby induced spatial learning deficits.
Keywords
Introduction
Melamine was now a well-known food adulterant and food contaminant. 1 The toxicity studies on melamine were mostly relevant to nephrotoxicity 2,3 till lately studies revealed that melamine was able to impair hippocampal function and cause spatial cognition impairment associating with oxidative stress, 4 –6 inhibiting neural excitability, 7,8 and disturbing the neurotransmission by reducing the levels of postsynaptic glutamate receptors. 9 –11
Cortical neurons collectively have synchronous or oscillatory patterns of activity the frequencies and temporal dynamics of which are associated with distinct behavioral states. However, these oscillations with properties changing under cognitive disorders such as schizophrenia, 12 Alzheimer’s disease, 13 and attention-deficit hyperactivity disorder. 14 Ample evidence supports that the hippocampal oscillations link CA3 to CA1 for information storage 15 and their synchronization are related to cognitive processes, 16 indicated by delta oscillations contribute to the consolidation of memory traces, theta oscillations reflect the encoding of new information, whereas alpha oscillations display search and retrieval processes in semantic long-term memory. 17 Furthermore, hippocampal oscillations are regulated by neuromodulatory substances. 18,19 Rhythms in the delta, theta and gamma frequency bands are fairly common, and vary with behavioral conditions. 19 Interestingly, neuromodulators with their functional receptors can shift the states of cells, giving rise to oscillatory activity or switching from one oscillatory regime to another. 20 Experiments in invertebrates show that neurons can perform such switching, with highly specific behavioral consequences. 21,22
Neural oscillations are implicated in cognitive and memory performance, but in a complex and partly nonlinear way. Therefore, the measurement of neural information flow (NIF) direction in various physiological frequency bands, rather than full frequency range, could be a possible way to discover the underlying association between cognitive function and neural oscillation. However, traditional computational methods, such as coherence, cross-correlation coefficient and cross-correlogram cannot provide any inference of causality, i.e. the direction of NIF, which is constrained by synaptic neurotransmission, revealing how transmit information passes through nerve pathways. Recently, partial directed coherence (PDC), which is based on the AR model, was proposed along with its improvement called general partial directed coherence (gPDC). 23,24 This algorithm which is proposed based on weak coupling dynamical oscillators is appropriate for analyzing the interacting neural subsystems because of the weakly synchronous character in neural signals. This approach has been proved to make a good performance not only in model systems, 24 but also in real organism, such as to electroencephalogram (EEG) data from Parkinson’s disease patients 25 and anesthesia and kainic acid rats. 26
Here, we hypothesized that subacute melamine exposure affected local field potential (LFP) which would characterize the alternations of the NIF in CA3–CA1 pathway. The performance of spatial learning was examined by the Morris water maze (MWM) task. Local field potentials (LFPs) were recorded in the CA3–CA1 pathway before and during the behavioral test. The algorithm of general partial directed coherence (gPDC) was employed to determine the coupling directionality of NIF between CA3 and CA1 over narrow frequency bands. Given the critical roles of AMPA receptors on the LFPs 27,28 and suppressive effects of melamine on the expression of glutamate receptors rather than presynaptic glutamate release, 11,29 the levels of CA1 AMPA receptor subunits, GluR1 and GluR2/3, were assessed and the reversible effects by the agonist were further evaluated to certify the possible mechanisms.
Experimental procedure
Experimental animals
Totally, 69 male Wistar rats, 3-week-old, were obtained from the Laboratory Animal Center, Academy of Military Medical Science of People’s Liberation Army, and reared in clear plastic cages in a colony room (12-h light-dark cycle, 21 ± 2°C; 45 ± 5% humidity) with ad libitum access to food and water. Experiments were conducted during the light period (between 14:00 and 17:00) and experimenters were blind to the treatment of the animals. All the experiments were conducted at about postnatal day 53 (range from 50 to 56). All procedures were performed in accordance with the Care and Use of Animals Committee of Guizhou University of Traditional Chinese Medicine (SCXK-2013-0020) and in accordance with the practices outlined in the NIH Guide for the Care and Use of Laboratory Animals.
According to our previous studies demonstrating significant effects of melamine on spatial learning and memory, 30 –32 rats in the melamine group were administered with melamine solution (30 mg/mL, dissolved in 1% carboxymethylcellulose (CMC)) at a dose of 300 mg/kg/day, while the rats in control group were received the same dose of 1% CMC. Gavage was performed and melamine or CMC was given once a day for 4 weeks.
For the systemic studies with the GluR2/3 agonist, rats were treated with 1.0 mg/kg, intraperitoneal injection of LY379268 30 min before the initiation of the behavioral test as previous reports. 33,34
Totally, 63 rats were randomly selected to each group. Fifteen rats were assigned to detect hippocampal GluR1 and GluR2/3 levels (control: n = 7; melamine: n = 8). Forty-eight rats were used for the MWM test (control: n = 7; melamine: n = 8; LY379268(CA1): n = 6; melamine + LY379268(SYS): n = 8; melamine + LY379268(CA1): n = 8; melamine + LY379268(CA3): n = 6; melamine + AMPA(CA3): n = 5). Data form the neuronal recording was collected from the above behavioral groups.
Spatial learning in MWM task
Hippocampal dependent spatial learning was tested using the MWM task, which was conducted as described previously. 11,35 A 150-cm-diameter circular pool was filled with water opacified with nontoxic black ink and kept at 25 ± 1°C. The tank divided into four equal quadrants and named clockwise I, II, III, and IV. A clear 10-cm-diameter platform was positioned in the center of quadrant III with its surface 2 cm below the water surface. The pool was surrounded by light blue curtains with several clearly visible, distinctive cues. Swimming behavior was monitored by a computerized video tracking system (Ethovision 2.0, Noldus, Wagenigen, Netherlands), through which data were collected for off-line analyzing.
During the training phase, each rat was trained for 12 consecutive trials (30 s intertrial interval) to find the platform. Rats were released into the water individually facing the pool wall from one of four starting points. The order of starting points was used pseudorandom (III, I, IV, II; IV, III, I, II; III, II, IV, I) but the same for all animals. Rats that failed to find the submerged platform within 60 s were guided to the platform and remained on it for 20 s before being returned to a holding cage. With the ample training of this protocol, rats could effectively and efficiently acquire the spatial information.
Surgery and microinjection
Rats were anesthetized with isoflurane and placed in a stereotaxic frame (SN-3, Narishige, Japan) for surgery. 36 –38 Stainless steel guide cannulae (22-gauge; Plastics One, Inc.) were bilaterally implanted to the dorsal CA1 (AP: −3.3 mm, ML: ±2.2 mm, DV: 2.4–2.8 mm) or CA3 (AP: −4.2 mm, ML: 3.5 mm, DV: 2.3–2.6 mm) region of the hippocampus (HPC). Obdurators (30-Ga, Plastics One Inc.) were inserted into guide cannula to prevent obstruction. Rats were allowed to recover for 7 to 10 days.
Infusions were performed by inserting custom needles (30-gauge, Small Parts Inc.) connected through PE-50 tube into an infusion pump (Harvard Apparatus), extended 1.0 mm pass the end of the cannulae. The GluR2/3 agonist LY379268 (2 nmol in 1 µl artificial CSF), AMPA (0.5 nmol in 1 µl artificial CSF) or artificial CSF (ACSF) was infused into the HPC (0.5 μL/min/side for 2 min) 30 min before testing began. The needles were left for 3–5 min to allow the diffusion of the drug. Dose and route of administration were chosen based on our and other previous studies. 11,33,34,39 On each drug treatment day, the treatments were reversed or counterbalanced designs. One week before the treatment, infusion procedure was habituated on 4 separate days. The infusion sites were identified with the aid of The Rat Brain in Stereotaxic Coordinates (1997, third edition). Only data from animals with correct implants were analyzed (Figure 1d and Figure 1e).

The reduction of non-directional interaction index PLV between CA3 and CA1 in low frequency bands. Western blot analysis of GluR1 (a) and GluR2/3 (b) in the CA1 region. (c) Escape latency was significantly prolonged following subacute melamine exposure (Melamine), while systemic injection of GluR2/3 agonist LY379268 (Melamine + LY379268(SYS)) reversed spatial learning deficits induced by melamine treatment. (d) The mean escape latency was comparable between intra-hippocampal CA1 infusion (Melamine + LY379268(CA1)) and systemic injection (Melamine + LY379268(SYS)) of LY379268. (e) Intra-hippocampal CA3 infusion of AMPA (Melamine + AMPA(CA3)) or LY379268 (Melamine + LY379268(CA3)) could not mitigate melamine-induced learning impairment. Note: Insets indicated the infusion sites. *P < 0.05. The number of rats in each group was used is indicated in each column or figure.
Western blot analysis
After neuronal recording, entire hippocampi were separated bilaterally and CA1 region was micro-dissected under a dissection microscope as previously described. 40 –42 Samples were randomly selected from one of hemispheres and prepared for this test. The rests were stored for melamine concentration assay. Samples were homogenized in ice-cold lysis buffer (pH 7.4) containing a cocktail of protein phosphatase and proteinase inhibitors (Sigma, MA, USA). The samples were centrifuged at 12,000×g and 4°C for 10 min and the supernatant were collected. Protein concentrations were detected by bicinchoninic acid assay (Bio-Rad Lab). Equal amount of proteins were resolved by 10–15% SDS-PAGE and then transferred onto PVDF membranes (Pall, Florida, USA) for immunoblotting. The membranes were blocked with 5% non-fat skimmed milk for 1 hour and incubated with the primary rabbit anti-GluR1 (1:1000; Chemicon, CA, USA), rabbit anti-GluR2/3 (1:1000; Chemicon, CA, USA). Mouse anti-β-actin (1:20,000; Sigma, MA) was used as an internal control. After three washes with Tris buffered saline Tween (TBST) buffer (10 min in each), the membranes were incubated with horseradish-peroxidase (HRP)-conjugated secondary goat anti-rabbit or anti-mouse IgG (1:1000; Southern Biotechnology Associates, AL) incubated for 1 hour. After three washes with TBST buffer, immunoreactivity was detected by ECL Detection Kit (CWBIO, China) as our previous reports. 9,43
LFPs recording
Microelectrodes were arranged in two 4 by 4 matrix using 25-μm-diameter platinum/iridium wire, coated with polyimide (California Fine Wire Company) in a 16-gauge silica tube (World Precision Instruments). It was then attached via gold pins to an EIB-36-PTB board (Neuralynx Inc.), which was assembled to microdrive (Harlan 8-drive; Neuralynx). The electrode tips were gold-plated to maintain the impedance to 200–600 kΩ measured at 1 kHz. Electrodes were checked daily for spontaneous cellular activity via a pre-amplification headstage (NB labs, Denison, TX; sampling frequency of 26–32 kHz).
Rats were anesthetized with isoflurane and prepared for surgery using previously reported procedures. 42,44,45 Two electrode arrays were chronically implanted: one was located at the CA1 region (AP: −3.5 mm, ML: 2.5 mm, DV: 2.0 mm) and the other one was located at the CA3 (AP: −4.2 mm, ML: 3.5 mm, DV: 2.5 mm) of the hippocampus in the ipsilateral hemisphere. The hemisphere was implanted randomly but counterbalanced across rats.
The recording was performance with a Digital Cheetah system (Cheetah software, Neuralynx Inc.). LFPs were sampled at 32 kHz and filtered at 0.1–9,000 Hz from each electrode. In the experimental room, neural signals were transferred through a slip-ring commutator (Neuralynx) to the data acquisition system. The animals’ behavior was monitored by a digital ceiling camera (Neuralynx Inc.) and sand the CCD camera’s signal was fed to a frame grabber (sampling rate, 1 MHz) with the experimental time superimposed for off-line analysis.
The recording was conducted 30 min before behavioral test in their home-cage and during the whole behavioral tests. After the completion of all recording sessions, selective positions of individual electrodes were marked by electrolytic lesions (10 μA current for 10 s). Brains were sectioned and recording sites were identified using standard protocols with reference to The Rat Brain in Stereotaxic Coordinates (1997, third edition). Only data from rats with probes contained within both targeted regions were included in the analysis (Figure 2a).

The reduction of the interaction index PLV and directional coupling index d and c2 in CA3–CA1 pathway. The mean value of PLV before
Phase locked value (PLV)
PLV is defined to analyze the strength of phase synchronization. Extracting the phase of two signals,
with N stands for the length of time series,
General partial directed coherence(gPDC) algorithm
PDC, whose definition is based on the notion of linear Granger causality, is proposed to describe the causal relationship between multivariate time series. Its core meaning is based on the decomposition of multivariate partial coherences computed from multivariate autoregressive models. two-variate process PDC algorithm was introduced as following.
Considering a two dimensional process
Granger causality within a two-variate process defined by
with
Taking the Fourier Transformation of the VAR coefficients:
yields a frequency-domain representation of the VAR model.
Defining the matrix:
And then PDC from variable xj to xi is defined as
It has been shown that large differences in the variances of the modeled time series can lead to distortions in the resulting PDC values.
46,47
To avoid this, a variation of the original PDC which is called generalized PDC (gPDC)
47
is presented. In gPDC, the coefficients
The denominator in (5) is a normalization that bounds the gPDC coefficients to values from 0 to 1. The choice of scaling means that
Quantitation of melamine in the hippocampus
After weighing, the rest samples from the blotting test were rinsed in 0.1 M phosphate buffer (pH = 7.4) and homogenized with ice-cold saline to be 10% (w/v) homogenates. The mixtures were homogenized using a glass homogenizer for 5 min, centrifuged at 3000 rpm at 4°C for 15 min, and the supernatant was collected. The melamine content was determined by using a melamine ELISA kit according to the instructions provided by the manufacturer (Huaan Magnech Bio-Tech Co., Ltd. Beijing, PR China., cat. number sc0067/sc0068). The detection range of this kit is 0.1–100 μg/mL.
Data and statistical analysis
All the data were expressed as Mean ± SEM. All analyses were performed with Neuroexplorer, Matlab (MathWorks) and SPSS 17.0 software. One-way ANOVA was used to compare the data from LFP recording. Two-way repeated measures ANOVA was applied for analysis of escape latencies and swimming speeds. Independent Sample T-test was used to analyze melamine levels and blotting tests. The significant level was set at 0.05.
Results
The levels of melamine and AMPA receptor subunits in hippocampal CA1 region
After receiving 300 mg/kg body weight of melamine for 28 consecutive days, the concentration of melamine in the hippocampal CA1 region of melamine-treated rats was significantly higher than that of controls (32.82 ± 0.80 μg/g vs. 0.16 ± 0.06 μg/g, T-test, P < 0.05). Meanwhile, significant differences in the body weight were observed on 14, 21 and 28 days following melamine exposure (Table S1; all P < 0.05). The expression of the AMPA receptor subunit GluR2/3 (Figure 1b; T-test, control vs. melamine, P < 0.05), but not GluR1 (Figure 1a; T-test, control vs. melamine, P > 0.05), was significantly decreased after subacute melamine exposure.
Melamine-induced learning deficit was associated with down-regulation of GluR2/3 expression in the CA1 region
The mean escape latencies decreased remarkably as the trials continued, which indicated that rats gradually and efficiently learned the locating of the platform (Figure 1c; repeated measures ANOVA, effect of trial, F (11, 275) = 52.37, P < 0.001). There was a marked effect of melamine on average escape latency, which was prolonged significantly in melamine group compared to that in control group from Trial 5 to Trial 12 (effect of treatment, F (3, 25) = 18.51, P < 0.001). Although systemic injection of the GluR2/3 agonist, LY379268, did not affect rats’ performance, melamine-induced learning disability was markedly mitigated (Trial 5 to Trial 12: melamine vs. melamine + LY379268(SYS), all P < 0.05). This effect was specific to rats with CA1 infusion (Figure 1d; repeated measures ANOVA, effect of treatment, F (3, 25) = 1.97, P > 0.05), as agonist infusion into the CA3 produced behavioral effects not different from melamine rats (Figure 1e; repeated measures ANOVA, effect of treatment, F (3, 22) = 28.90, P < 0.001; Melamine vs. Melamine + LY379268(CA3) or Melamine + AMPA(CA3), both P > 0.05). Furthermore, the reversible effects of the systemic injection could not be duplicated by microinjection of AMPA into the CA3 region. Additionally, swimming speeds of these two groups remained constant throughout testing, with no difference were found (data not shown), indicating that motor function was not the underlying cause of the prolonged latencies as previous reported. 10,30 Together all, our results clearly indicate that the reduction in the expression of GluR2/3 subunit of the hippocampal CA1 region involved in melamine-induced learning deficits.
Subacute melamine exposure weakened the strength of phase synchronization
Although the values of PLV were comparable before behavioral test (Figure 2a; two-way ANOVA, effect of treatment, F ( 3, 25) = 0.86, P > 0.05), they were significantly reduced in low frequency bands of melamine group compared with that of control group (Figure 2b; two-way ANOVA, effect of treatment, F ( 3, 25) = 20.21, P < 0.001; post hoc, delta, theta, and alpha bands, all P < 0.05). Microinjection of LY379268 into CA1 region markedly enhanced the values of PLV (delta, theta, and alpha bands, melamine + LY379268(CA1) vs. melamine, all P < 0.05), which were closely to the value of control group (delta, theta, and alpha bands, melamine + LY379268(CA1) vs. control, all P > 0.05). No differences were found at beta, gamma or HF band.
Melamine diminished the strength of NIF from CA3 to CA1 regions
Directional index d was measured by gPDC approach between two time series obtained from the CA3 and CA1 sub-regions of the hippocampus. As shown in the Figure 2c, the directionality index d of NIF in CA3–CA1 pathway were significantly lower in melamine-treated rats than that in control rats at delta, theta and alpha frequency bands (one-way ANOVA, effect of treatment, F (3, 25) = 12.69, P < 0.001; post hoc, delta, theta, and alpha bands, all P < 0.05), while activation of GluR2/3 subunit could rescue the connection between CA1 and CA3 regions (delta, theta, and alpha bands, melamine + LY379268(CA1) vs. melamine, all P < 0.05).
Moreover, the group values of the unidirectional influence
Discussions
In this study, we have successfully established the animal model, which exhibited spatial learning impairment as previously described. 31,48 It is well-known that the hippocampus formation, which is a part of the brain linked with learning and memory, is extremely vulnerable to melamine. 5,6,8,49 Based on the values of phase synchronization between CA3 and CA1 regions, the gPDC algorithm was applied to measuring the directional index of information flow on CA3–CA1 pathway. Moreover, we undertook pharmacological microinjection of AMPA subunit agonist to rescue melamine-impaired neuronal activity and the potential mechanism was investigated and clarified.
To evaluate whether melamine affect the cognitive functions of rats, MWM task, which is a well-validated method for examining spatial learning and memory in rodents, 50 was carried out. Actually, the beginning days of the training, such as the first four trials, serve to acclimatize them to swimming in the maze 36,50 and animals progressively recognize that they can escape by finding the platform. The prolonged escape latency in the trial 5–8 and 9–12 indicated that melamine-treated rats’ learning ability was considerably impaired after acclimation of the environment. Gene-targeted mice lacking the AMPA receptor subunit have deficits in hippocampal long-term potentiation (LTP) and have profoundly impaired hippocampus-dependent spatial behavioral performance, 51 which are consistent with our previous finding that the decreased expression of postsynaptic glutamate receptors involved in melamine effects on synaptic plasticity, including LTP and long-term depression (LTD). 11,31,52 Furthermore, pharmacological and genetic evidence suggests that glutamatergic transmission specifically at the AMPA receptor may play a key role in the cognitive disorders. 53 Recent studies showed that LY379268 could recover the impairments on a rewarded Y-maze test of spatial working memory. 54 This supports human studies with LY354740, which recovers impairments in spatial learning in healthy volunteers. 55 Furthermore, the measurement of the neural excitability in hippocampal CA1 pyramidal neurons found that melamine could altered the action potential of hippocampal CA1 neurons by impairing the functional properties of voltage-gated sodium channels and potassium currents. 56,57 Consistently, amplitude of both spontaneous and miniature excitatory postsynaptic currents (EPSCs) in the CA1 region of melamine-treated rats were decreased, suggesting the effects on postsynaptic receptors kinetic. 7 It is possible that such deficits in neural activity of the CA1 region may underlie the mild impairment in spatial learning seen in melamine-induced animals. In our study, microinjection of AMPA or LY379268 into CA3 was ineffectual, supporting that the residual melamine in the CA1 but not CA3 was associated with the behavioral impairment. As previous reports, 4,6,31 melamine could induce oxidative stress (Figure S1), which may involve in cognitive impairments. The above results imply that activation of GluR2/3 subunit can mitigate melamine-induced spatial learning deficits.
Synchronous oscillations in different frequency bands are considered as an important mechanism linking single-neuron activity to cognitive behavior. 58 The PLV data showed that there was a decreased amount of synchronization of intracranial electric activity. The lower synchronization at delta, theta and alpha rhythms was generally found in EEG of cognitive defective states. 59,60 These observations from PLV analysis suggested that the connection strength between CA3 neurons and CA1 neurons in hippocampus was significantly reduced, which implied that there was a disturbance in neural synchronization.
The gPDC algorithm, which performed well up to relative stronger coupling because it extracted some information from amplitudes, not only from phases, 61 was applied to measure the directional index in CA3–CA1 pathway. Obviously, the direction of information flow is mainly from CA3 to CA1, which is the main projection direction between two regions. 62 Hippocampal CA1 is proposed to play a role in matching of CA3 output with afferent input from entorhinal cortex. 63,64 The results from gPDC measurement showed that there was a more predominant driving occurred from CA3 to CA1 with directional index d > 0 in all frequency bands, which were consistent with previous finding that the topographic organization of CA3 projected heavily to levels of CA1. 65,66 Furthermore, the d values in melamine group were lower than that in control group, suggesting that the functional impairment might result from the decreased information transmission in the CA3–CA1 pathway in hippocampus, as was shown by the reduction of directional index of NIF. In addition, the non-negative index c 2 showed that the strength of CA3 driving CA1 was significantly reduced in melamine-treated rats, suggested that the cognitive dysfunction could be partly caused by the reduction of information transfer along the connections between CA3 and CA1. Taken together, the results support our hypothesis that the alteration of cognitive functions, typified by reduced directional index of NIF in the CA3–CA1 pathway in hippocampus, could be detected by using LFP recordings.
It was reported that there was an association between the neural excitability and the low frequency oscillations of the frontal scalp areas in study on hippocampo-cortical feedback loops during signal processing. 58 Following the theories that glutamate was the major excitatory neurotransmitter in the hippocampus and mediating excitatory postsynaptic potentials was essential for EEG oscillations and evoked potentials, 58 it implies that low frequency bands, such as theta oscillations, may be related to the optimal induction of LTP when the time interval between stimuli is approximately 5 Hz. 67
Previously, the dentate gyrus (DG) of aged rats developed an increased glutamate uptake rate compared to the DG of young animals, indicating a possible age-related change in glutamate regulation to deal with increased glutamate release that occurred in late-middle age. 68 However, no age-related changes in resting levels of glutamate were observed in the DG, CA3 and CA1. The role of mGluR1 in the induction of 3,5-dihydroxyphenylglycine-LTD was increased with advanced age and, in contrast to young adults, induction involved a significant contribution of NMDA receptors and L-type Ca2+ channels. 69 Furthermore, a reduction in ionotropic receptors and their constituent subunits with age has been correlated with a decline in memory function, 70,71 which can be modulated with pharmacological agents that facilitate activation of the glutamate receptors. 72,73 Therefore, it is worth noting that age-associated alterations in neurotransmission and subsequent effects on function of the hippocampal circuitry may involve in this study. Furthermore, our findings support that melamine is able to pass through the blood–brain barrier (BBB) and take up in the hippocampus. 48,74 It may be attributed to its low molecular weight and amphiphilic structure, 75,76 which could effective cross the BBB. 77,78 Molecular charge and the concentration in the blood may also impact on passing through the BBB and diffusing into the brain. 79,80 However, it is still unclear what factors are important in determining such passage and the underlying mechanism need further investigation in detail.
In summary, gPDC analysis showed that coupling directional index based on LFP reduced robustly in low frequency bands, suggested that the pattern of information flow was drastically affected in melamine-treated rats, which could partly induce cognitive dysfunction. The unidirectional index c 2 showed that CA3 driving CA1 was critically decreased, indicating that the cognitive dysfunction could be in part caused by the reduction of information transfer along CA3–CA1 pathway. Furthermore, LY379268 could effectively rescue learning and the directional coupling in the hippocampus, indicated that AMPA subunit GluR2/3 in CA1 region involved in melamine-induced cognitive and neural dysfunction. Our findings may advance the understanding melamine-induced cognitive disorders.
Supplemental material
Supplemental Material, Supplementary_information_final - Subacute melamine exposure disrupts task-based hippocampal information flow via inhibiting the subunits 2 and 3 of AMPA glutamate receptors expression
Supplemental Material, Supplementary_information_final for Subacute melamine exposure disrupts task-based hippocampal information flow via inhibiting the subunits 2 and 3 of AMPA glutamate receptors expression by Wei Sun, Xiaoliang Li, Dongxin Tang, Yuanhua Wu and Lei An in Human & Experimental Toxicology
Footnotes
Author contributions
Conceived and designed the experiments: WS, XLL, DXT, LA; performed the experiments: XLL, WS; analyzed the data: XLL, WS; wrote the manuscript: WS, DXT, LA.
Ethics statement
All procedures were done in accordance with ethical guidelines laid down by the ethics Committee on the Care and Use of Animals Committee of Guizhou University of Traditional Chinese Medicine and in accordance with the practices outlined in the NIH Guide for the Care and Use of Laboratory Animals.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants from the National Natural Science Foundation of China 31700929 to LA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.