Abstract
Our previous studies on cannabinoid type1 receptor (CB1R) activation on Methamphetamine (METH)-induced neurodegeneration and locomotion impairments in male rats suggest an interaction between CB1Rs and METH. However, the role of these receptors in METH-neurotoxicity has not been fully identified. Therefore, the purpose of the present study is to investigate the involvement of CB1Rs in these effects. We conducted an electrophysiological study to evaluate functional interactions between METH and CB1Rs using whole-cell patch current clamp recording. Furthermore, we designed the Nissl staining protocol to assess the effect of METH on the basic cerebellar Purkinje cell structure. Our findings revealed that METH significantly increased the action potential half-width, spontaneous interspike intervals, first spike latency, and decreased the rebound action potential and spontaneous firing frequency. Using CB1R agonist and antagonist, our results showed a significant interaction with some of the electrophysiological alterations induced by METH. Further, Nissl staining revealed that the exposure to the combination of METH and SR141716A resulted in the necrotic cell death. Results of the current study raises the possibility that METH consumption profoundly affect the intrinsic membrane properties of cerebellar Purkinje neurons and cannabinoid system manipulations may counteract some of these effects. In summary, our findings provide further insights into the modulatory role of the endocannabinoid system in METH-induced neurologic changes, which can be used in the development of potential therapeutic interventions for METH dependence.
Introduction
Methamphetamine (METH), an amphetamine analog, is a drug of abuse that causes psychiatric disorders in humans. 1 Psychiatric disorders reported among METH abusers can, in part, be associated with neurotoxicity induced by METH. METH is commonly abused worldwide and has become an international common health concern, which affects many aspects of social and economic life. Several studies have reported that the endocannabinoid system (ECBs) plays meaningful roles in the susceptibility to psychiatric disorders including drug abuse. 2,3 Increasing recent evidence points toward the neuromodulatory role of ECBs. 4,5 This system especially through interaction with mesolimbic dopaminergic and opioidergic systems modulate brain reward function. 3 Recent studies have raised hope about the therapeutic potential of cannabinoids in neurodegenerative disorders. 2,6 –8 Cannabinoids exert most of their functions in the brain by binding to G-protein coupled receptors (GPCRs): the cannabinoid CB1 receptor. 9 –11 The major intracellular signaling pathway mediated by CB1 receptors is through coupling to inhibitory G proteins (Gi). 12 Localization of the CB1Rs in central nervous system (CNS) has led to various studies on the neuromodulatory role of these receptors in neuronal processes associated with substance use disorder (SUD). 13 Cannabinoid CB1 receptors are mainly located in the brain and particularly expressed at high density in brain regions that have been assigned in drug addiction, including cerebellum. 6 The axons of Purkinje cells constitute the only efferent pathway to the cerebellar nuclei, and thus Purkinje cells provide the sole output neurons from the cerebellar cortex. 10 These cells are the sole neurons in cerebellar cortex that synthesize, release, and degrade endocannabinoids. 14 Majority of CB1Rs in the cerebellum are placed on axonal terminals that form synapses onto Purkinje cells. 14 Even though the cerebellum has not been included in the reward pathway and overlooked in addiction research, there is increasing evidence indicating its involvement in the behavioral patterns associated with drug dependence. 15 Although the cerebellum has low dopaminergic innervations, 16 accumulated information supports that ventral tegmental area (VTA) sends dopaminergic innervations to the vermis of the cerebellum. 17,18
Several lines of evidence indicate that change in the intrinsic electrical properties of Purkinje cells may be involved in the physiological and behavioral alterations induced by METH. 19,20 In these neurons, any variation in the electrophysiological properties such as firing rate and spike discharge pattern may affect the output of the neurons. Specifically, firing patterns as a unit of neural information processing has been postulated to play a considerable role in neuronal correlations and processing. Thus, persistent changes in firing patterns may affect synaptic excitability and neuronal function and thereby result to modify behavior. 21 This proof-of-concept study tested whether cannabinoid system manipulation using its agonist and antagonist could attenuate impairments induced by METH. Therefore, in this study, we examined the role of CB1Rs in electrophysiological alterations of the Purkinje cells exposed to METH using the whole-cell patch-clamp recording.
Experimental procedure
Subjects
Male Wistar rats (weight: 90–100 gram; age: 5-weeks-old) were bred and kept in Neuroscience Institute’s Animal House (Kerman, Iran) and were used at the beginning of the study. Animals were housed in standard plastic laboratory cages in a humidity and temperature-controlled (22 ± 1°C) colony room that was maintained on a 12:12-h light/dark cycle with ad libitum access to food and water. Animal care and experiments were conducted according to the National Institute of Health Guidelines and approved by the Research and Ethics Committee (IR.KMU.REC.1395.733) of the Faculty of Science, Kerman University.
Drugs
In this study, we used methamphetamine hydrochloride (Sigma Aldrich, St Louis, MO, USA), WIN 55,212-2 (Tocris, Cookson, UK) and SR141716A [N-piperidino-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxamide hydrochloride] (Tocris, Cookson, UK). For in vitro studies, METH (10 µM) 22,23 was dissolved in 0.9% saline solution, WIN (10 µM) and SR (10 µM) 24 were dissolved in anhydrous dimethyl sulfoxide (DMSO), and aliquots were stored 25 given experiment. For histology study, METH (5 mg/kg) was dissolved in isotonic (0.9% NaCl) saline. WIN (3 mg/kg) 26 and SR (10 mg/kg) were dissolved in a mixture of sterile isotonic saline and anhydrous dimethyl sulfoxide (DMSO, 9:1 v/v) added one drop of 0.4% Tween oil-80 as dispersing or emulsifier agent.
Slice preparation and solutions
Male Wistar rats were decapitated under diethyl ether anesthesia, 27 the brain was quickly removed from the skull and placed into chilled (4°C) artificial cerebrospinal fluid (aCSF) of the following combination (in mM): 125 NaCl, 25 NaHCO3, 10 D-glucose, 2.5 KCl, 2 MgCl2, 1.25 NaH2PO4, 0.4 Ascorbic acid and 2.4 CaCl2, which was oxygenated (95% O2 & 5% CO2) in order to adjust the pH at7.35–7.45. The osmolality was adjusted to 295–310 mOsm. To study the intrinsic firing properties of Purkinje cells of the vermis, 100 μM picrotoxin (antagonist of GABAA receptors) and 1 mM kynurenic acid (non-selective antagonist of ionotropic glutamate receptors [N-methyl-D-aspartate (NMDA), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) and kainate receptors] and also of the nicotine cholinergic subtype alpha 7 receptors), 21,28 were added to the recording aCSF. The vermis, separating the two hemispheres of the cerebellum, was dissected out. It was then glued onto the stage of the vibroslicer and immersed in ice-cold aCSF. Parasagittal slices of 250–300 µm thickness were cut by vibroslicer and transferred to a holding chamber. The slices were incubated at least 60 min in the chamber and perfused with aCSF oxygenated with 95% O2, 5% CO2 with pH of 7.35–7.45 and temperature kept at 32–36°C.
Whole-cell patch clamp recording
Whole-cell patch clamp recording in current clamp mode from Purkinje cells was made, as described previously by Razavinasab et al. 28 After recovery, slices were mounted in a submerged chamber on the stage of an upright microscope (BX 51WI; Olympus, Tokyo, Japan). The slices were continuously superfused at the rate of 1–2 ml/min with oxygenated aCSF at room temperature (22–25°C). Purkinje cells were then visualized using infrared video microscopy (Hamamatsu, ORSA, Japan) with a 40 × water immersion objective. From each animal, only a single Purkinje cell from a single brain slice was recorded. Whole-cell current clamp recordings were made from Purkinje neurons using Multiclamp 700B amplifiers (Axon Instruments, Foster City, California, USA) and digitized with a Digidata computer interface. Electrophysiological responses were filtered at 5 kHz, sampled at 10 kHz and saved on a personal computer for offline analysis. Patch pipettes were pulled with an electrode puller from thick-walled filament borosilicate glass. The tip resistance of the electrodes was 3–10 MΩ when filled with internal solution containing 135 mM potassium methylsulfate (KMeSO4), 10 mM KCl, 10 mM Hepes, 1 mM MgCl2, 2 mM adenosine triphosphate disodium salt (Na2ATP) and 0.4 mM guanosine triphosphate sodium salt (Na2GTP). The pH of the internal solution was set to 7.3 by potassium hydroxide, and osmolality was adjusted to 280–290 mOsm. After the establishment of a gigaohm (GΩ) seal, the whole-cell configuration was achieved simply by applying a brief suction. Before rupture of the membrane, cells with a seal <1 GΩ were discarded and the test seal function was continuously monitored across the recording to be sure of the seal was stable. Series resistance (typically <15 MΩ) was carefully controlled for stability during the protocols. Signals were filtered at 10 kHz and sampled at 20 kHz using Clampex 10.2 software and stored on computer hard disk for offline analysis. The mean basal spontaneous firing frequency from a period of 3 min was computed. Membrane properties and action potential parameters such as firing regularity, after hyperpolarization (AHP) amplitude, action potential half-width and time to peak were measured. Input resistance was measured from the change in membrane potential exited by hyperpolarizing current steps (0–0.5 nA for 1200 ms duration, in 0.1 nA increments) when the neurons were held hyperpolarized with DC current (typically at about −60 mV) to avoid spontaneous firing. To elevate the excitability of neurons, action potentials (APs) were induced in Purkinje neurons from a holding potential of −60 mV in 520 ms duration current steps ranging from −300 to +300 pA in 100 pA increments. Before positive current steps, a negative prepulse protocol current with 300 pA was identified. The AP amplitude and half-width were measured as peak distance from the resting membrane potential and the width at half amplitude. First-spike latency was determined as the time between the offset of the negative current steps and the peak of the first spike. The voltage sag current in response to hyperpolarizing current pulse (0.3 nA) was measured as the peak voltage deflection divided by the amplitude of steady-state voltage deflection (Figure 1).
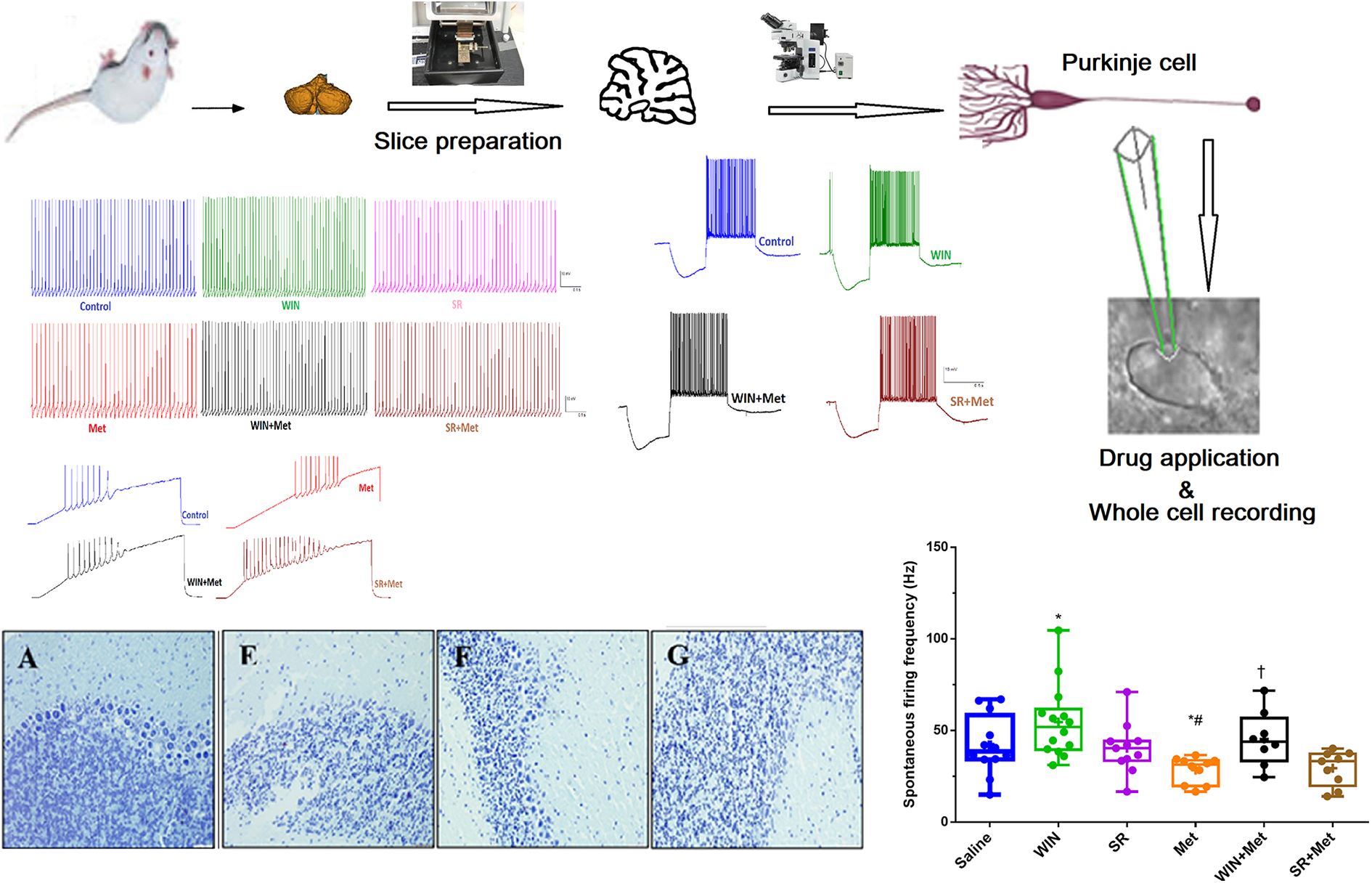
Graphical abstract.
Experimental design in histology study
In the current study, rats were randomly classified into seven groups: Saline, DMSO, WIN, SR, METH, WIN + METH, SR + METH groups. Eleven rats were applied in each experimental group. In Saline, DMSO and METH groups, animals received saline/DMSO (1ml/kg) 29 and METH (5 mg/kg) 30 min before saline injection. In WIN/SR groups, animals received WIN (3 mg/kg)/SR (10 mg/kg) 30 min before saline injection. Animals in WIN+METH/SR+METH groups, received WIN/SR 30 min before METH injection. All drugs were freshly prepared prior to the injection. All injections were given intraperitoneally once daily for 3 days in an injection volume of 1 ml/kg.
The animals, 24 hours after the last injection were deeply anesthetized with sodium pentobarbital (50 mg/kg, i.p.) and then transcardially perfused with saline and 4% paraformaldehyde. 30 The brains were rapidly removed, and vermis of the cerebellum was separated on an ice-cold, fixed in 10% formalin and dehydrated. Cortical coronal slices were embedded in paraffin, serially sectioned (4 μm), and then dewaxed in a 600 W microwave oven at 120°C for 10 minutes. 31 The slides were incubated at room temperature for 20 minutes and then washed in phosphate-buffered saline (PBS), exposed to hydrogen peroxide 0.03%, and washed in PBS. For Nissl staining, tissue blocks were stained with 1% cresyl violet, dehydrated through graded alcohols (70, 95, 100% 2×), located in xylene and coverslipped using DPX mountant. Tissue sections were observed by a light microscope (Olympus CX31) connected to a camera. In each rat, average neurons count was obtained by calculation of 5 cerebellar serial sections ×200 magnification. Neurons that had distinctive nuclei and cell bodies were computed. In order to observe the cerebellum tissue morphology, 10 visions were randomly selected for every section. 32
Data analysis
Statistical analysis of data was performed using the SPSS software package. All data were first assessed for normality using a Kolmogorov-Smirnov test. Results were analyzed using one-way ANOVA followed by Tukey’s test on normally distributed data (Data were expressed as the mean ± SEM.). The Kruskal-Wallis test was used to analyze the data without normal distribution (Data were expressed as median and interquartile range). For figure production, we used Graph Pad Prism 8. P < 0.05 was considered statistically significant.
Results
Electrophysiological results
Whole-cell patch clamp recording (Figure 2a–g) revealed that bath application of METH (10 µm) significantly decreased the spontaneous firing frequency and METH combined with CB1 R agonist (WIN, 10 µm) returned it to the control condition (Figure 2c, P < 0.05). Also, METH significantly increased the action potential half-width (Figure 2e, P < 0.01), and spontaneous interspike interval (Figure 2f, P < 0.05) but, combination of METH with either CB1 R agonist or CRB1 antagonist returned these alterations back to the control condition. Figure 2e and f show that the equal dose of WIN alone significantly decreased the action potential half-width and spontaneous interspike interval (P < 0.05).
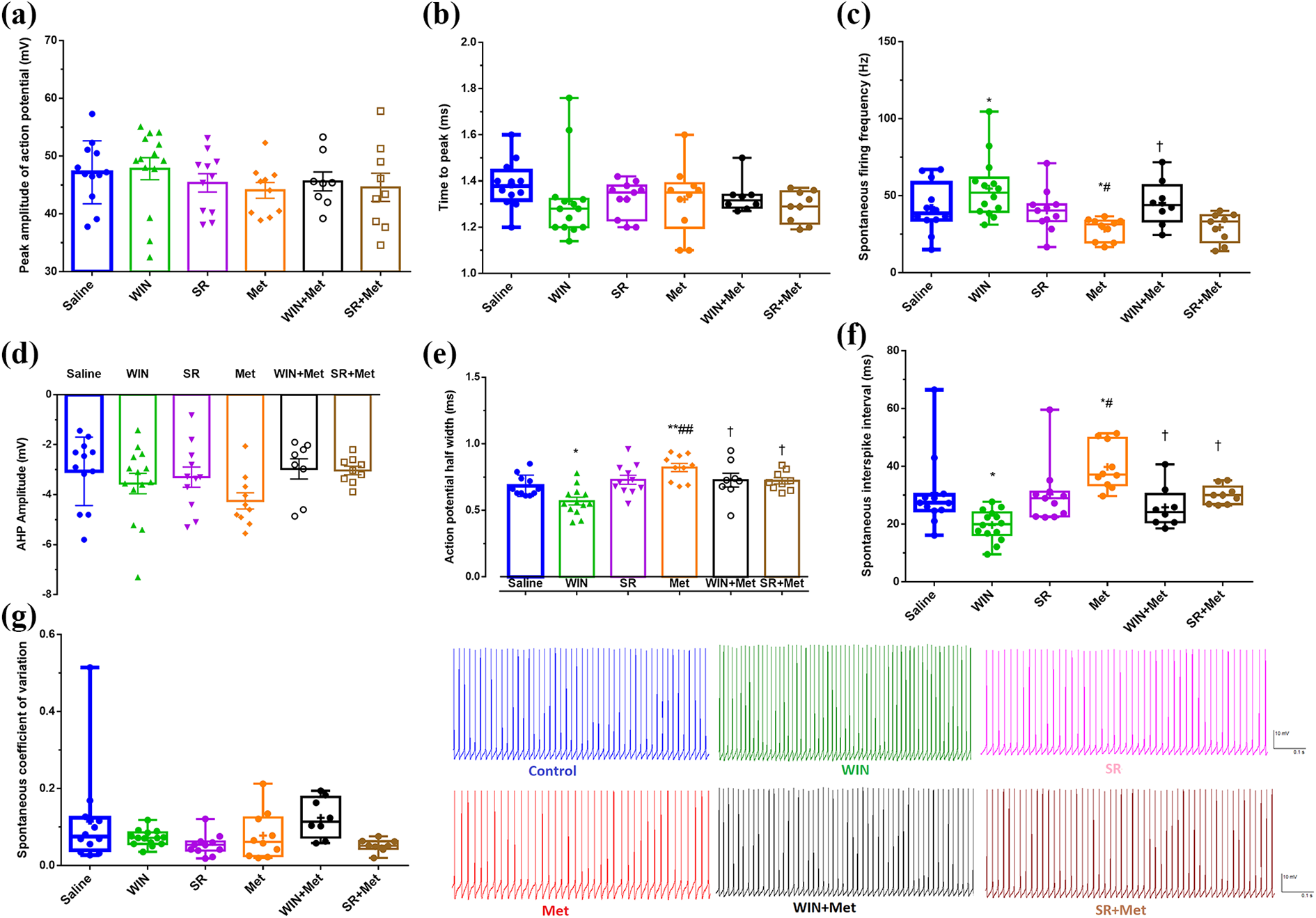
The effect of METH alone or METH combined with CB1Rs agonist (WIN)/antagonist (SR) on the electrophysiological properties of Purkinje neurons. Electrophysiological properties including, the peak amplitude of action potential (a), time to peak (b), spontaneous firing frequency (c), AHP amplitude (d), action potential half-width (e), spontaneous interspike interval (f), and spontaneous coefficient of variation (g). The data are expressed as mean ± SEM. *(P < 0.05), and **(P < 0.01) represent the significant difference with saline group. #(P < 0.05), and ##(P < 0.01) represent the significant difference with WIN group. †P < 0.05 represents the significant difference with the METH group. The number of symbols on each whisker plot represents the number of cells in each group. The color traces show the spontaneous spike activity of Purkinje cells in response to the METH alone or METH combined with CB1Rs agonist/antagonist.
Our findings showed that METH-exposed Purkinje neurons exhibited a significant decrease in the rebound action potential and METH combined with cannabinoid CB1 receptor agonist/antagonist had no effect on METH-electrophysiological changes (Figure 3a, P < 0.01). Figure 3 (b) shows that in the presence of METH the first spike latency was significantly increased in Purkinje neurons compared to control neurons and exposed to METH combined with WIN/SR reduced first spike latency increased by METH (P < 0.001). Meanwhile, exposure to METH and CB1Rs agonist/antagonist did not significantly affect the depolarizing “sag” voltage (Figure 3c).

The effect of METH alone or METH combined with CB1Rs agonist (WIN)/antagonist (SR) on rebound action potential (a), first spike latency (b) and depolarizing “sag” voltage (c) in response to different negative current prepulse injection (−0.3 nA) following 0.3 nA test pulse injection. Data represent as mean ± S.E.M. **(P < 0.01), and ***(P < 0.001) represent the significant difference with the saline group. ##(P < 0.01), and ###(P < 0.001) represent the significant difference with WIN group. †††(P < 0.001) represent the significant difference with the METH group.
Figure 4 shows that exposure to METH resulted in a significant increase in the rheobase current compared to the control condition. But the application of METH combined with cannabinoids CB1 receptors antagonist significantly decreased the rheobase current compared to the METH group. Also, WIN alone significantly decreased the rheobase current compared to the control condition (P < 0.01).

The effect of METH alone or METH combined with CB1Rs agonist (WIN)/antagonist (SR) on rheobase current. The data are expressed as mean ± SEM **(P < 0.01), and ***(P < 0.001) represent significant difference with the saline group. ###(P < 0.001) represent the significant difference with the WIN group. ††(P < 0.01) represents the significant difference with the METH group.
Histological studies
In the current study, we conducted the Nissl staining protocol to evaluate possible interaction between the METH and CB1Rs agonist/antagonist on neuronal morphology features (Figure 5). The cell death pattern induced by METH includes Purkinje cell necrosis with loss of cell membrane integrity was observed. Furthermore, the Nissl’s bodies represented the chromatolysis. Figure 5 shows that necrotic cell death significantly increased in the Purkinje cells of the METH-treated rats (p < 0.001). Cannabinoid CB1 receptor antagonist alone enhanced necrotic cell death in the Purkinje cells. But Pre-treatment with CB1Rs agonist/antagonist had no effect on necrotic cell death induced by METH.
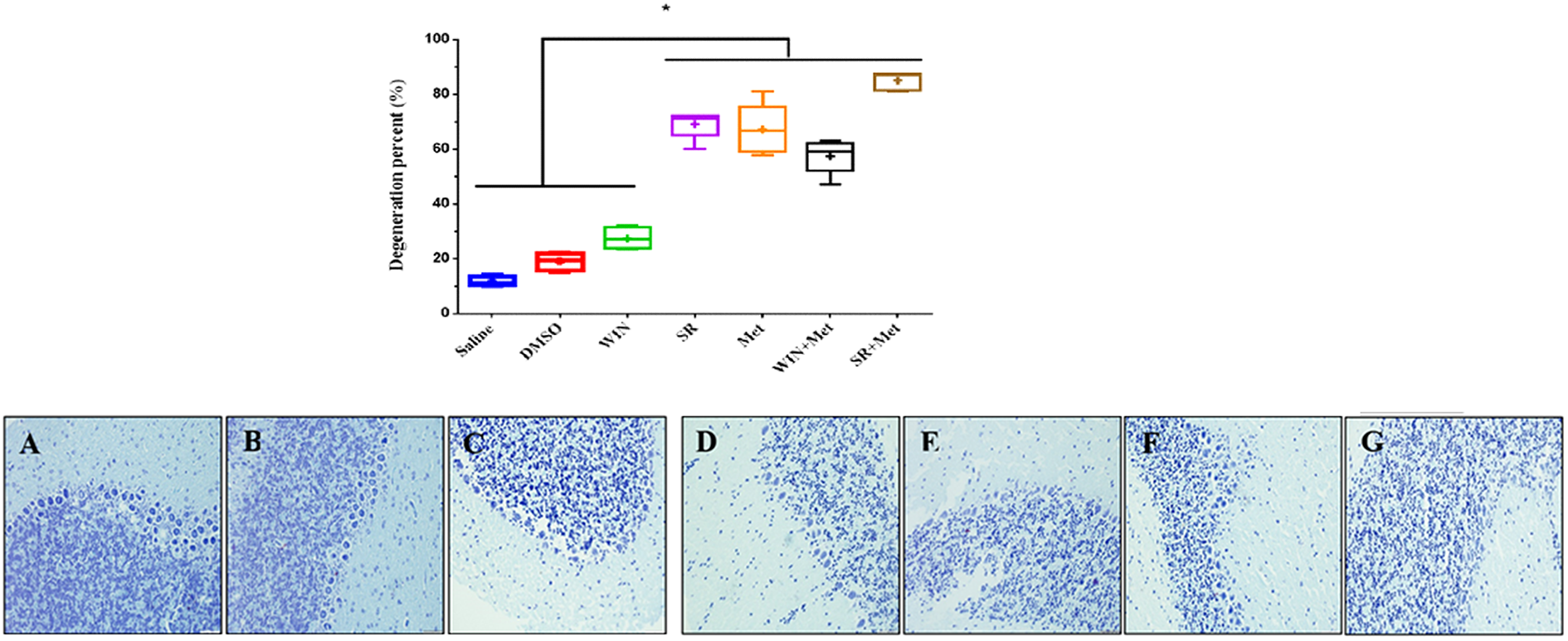
Effect of repeated exposure to methamphetamine (METH) alone or METH combined with CB1Rs agonist (WIN)/antagonist (SR) on necrotic cell death using Nissl staining. Necrotic cell death significantly increased in METH/SR-treated rats compared to the saline group (p < 0.001). Pre-treatment with CB1Rs agonist/antagonist had no effect on necrotic cell death induced by METH. (a) saline, (b) DMSO, (c) WIN, (d) SR, (e) METH, (f) WIN+METH, (g) SR+METH groups.
Discussion
In the current study, whole-cell patch clamp recording in current clamp mode was performed to investigate the functional effects of METH alone or METH combined with CB1Rs agonist/antagonist on electrophysiological properties of the Purkinje cells of the cerebellum. Whole-cell patch clamp recordings revealed electrophysiological alterations in Purkinje neurons exposed to METH. The electrophysiological findings of this study demonstrated that in vitro treatment with METH led to profound changes in the intrinsic membrane properties of Purkinje neurons.
Taken together, our findings showed that METH decreases neuronal firing level. METH caused a significant reduction in the firing frequency and a significant increase in the action potential half-width as well as action potential interspike intervals. METH likely decreases firing of neurons through increasing extracellular dopamine and activating dopamine autoreceptors on nerve terminals and finally, reducing dopamine release and neurotransmission. 22
Increasing the extracellular dopamine level by METH occurs through a combination of two mechanisms. First, METH is a substrate for dopamine reuptake transporter and the effect is therefore thought to be mediated through inhibition of dopamine uptake and elevation of its reverse transport, resulting in increased extracellular dopamine. Second, METH increases release of dopamine from dopaminergic nerve terminals. 14,32 On the other hand, increased dopamine neurotransmission and reduction of the neurons firing level following exposure to METH might be due to modulation of the activity of calcium-activated potassium (BKCa) channels. METH has been shown to suppress BKCa channels activity. BKCa channels regulate neurotransmitter release and action potential. 33 Previous reports have established that BKCa channels serve as a vital link between intracellular signaling pathways and electrical signals in the neuronal cells. These channels modulate the neuronal activity through action potential termination and altering firing frequency. 34 Therefore, METH–induced reduction in spontaneous firing activity in Purkinje cells could be attributed to the inhibition of BKCa channels activity by METH. 33 Also, it is possible that METH by phosphorylation of the α/β subunits of the BK channels increase trafficking of the channels and leads to change in gating features of the channel. 33,35
Our results showed METH-electrophysiological changes in Purkinje cells that exposed to METH combined with CB1Rs agonist returned to the control condition. Cannabinoid CB1 receptors serve as retrograde messengers that are produced in the postsynaptic neurons. 36 –38 These retrograde messengers are widely expressed in the brain and distributed in numerous synapses in several brain regions, including the cerebellum. 36 –38 These diffusible lipophilic molecules have been proposed to influence the strength of the presynaptic terminals inputs and modulate synaptic transmission 39 by activating presynaptic cannabinoid CB1 receptors. In particular, in the cerebellum, eCBs inhibit synaptic transmission at granule cell to Purkinje cells synapses by regulating presynaptic calcium influx through N-, P/Q-, and R-type calcium channels. 40 CB1 receptors also exert rapid actions, including the inhibition of voltage-dependent Ca2+ channels (mainly N- and P/Q-type) 41 and the activation of K+ channels. 42 WIN 55,212–2 significantly increases the frequency of spontaneous firing of Purkinje neurons in the presence of CB1 receptor antagonist, 40 which is consistent with our results. Thus, our results revealed that endocannabinoids probably mediated retrograde signals from postsynaptic neurons to presynaptic terminals and led to the reduction of neurotransmitter release and finally increase neuron firing level. 43 METH treatment also induced a significant increase in the latency to the first spike that was accompanied by a significant decrease in the rebound spike firing at negative prepulse evoked currents. These results suggesting a likely enhancement of transient K outward channel (IA) currents in Purkinje neurons exposed to METH. IA current has been shown to have a modification effect on first-spike latencies that has an important implication for neuronal coding and synaptic integration. 44,45 METH decreased the spontaneous firing frequency and the cannabinoid agonist WIN, that by itself increases this parameter, normalized the firing frequency when combined with METH. There are several scenarios to explain the WIN action. First, that somato/dendritic CB1 receptor increases excitability. 10,46 Second, the increase in frequency by WIN can be the consequence of a reduction of the inhibitory input onto Purkinje cells compatible with the expression of CB1 receptors on the Basket cells GABAergic terminals. 46
METH increased potential half-width and spontaneous interspike interval while the combination of METH with either agonist or CB1R antagonist returned these values to normal. The fact that the agonist and antagonist of CB1R have the same effect restoring these parameters requires more investigation. When added alone, WIN decreases these parameters and the antagonist SR141617 has no effect indicating the absence of any tonic activation of CB1 receptors. It seems logical that WIN re-establish the increase in action potential width and interspike interval produced by METH, but it is not clear at all why the antagonist SR also rescue these parameters. METH and WIN have opposite effects on potassium channel activity this can explain that a CB1 agonist corrects for the METH-induced changes but again remain no clear why a CB1 antagonist had the same effect. Maybe we can explain that SR141716A is an inverse agonist of constitutively active CB1 receptors. 47
In this study, we examined the neuroprotective effects of eCBs on cells necrotic in cerebellum rats exposed to METH via Nissl staining. Our findings showed that the METH increased necrotic cell death in the Purkinje cells of the cerebellum. The mechanism responsible for neurotoxicity induced by METH is complicated, and various mechanisms have been discussed in this phenomenon. Possible mechanisms underlying of neural cells damage induced by METH is thought to be related to the excitotoxicity induced by METH. 48 Glutamate acts as the principal excitatory neurotransmitter in the central nervous system and has been identified to play a crucial role in METH-excitotoxicity. 49 However, excessive extracellular glutamate level can lead to an influx of calcium, to trigger a rise in intracellular calcium level. Increased calcium level followed by activation of Ca2+-dependent proteases, formation of nitric oxide (NO), free radicals and consequently induction of apoptotic cascades. Accumulated evidence suggests that NO has been implicated in neurotoxicity induced by methamphetamine since the METH-induced neurotoxicity is attenuated following combined application of METH with nitric oxide synthase (NOS) inhibitors. 50
Also, our results show that antagonist of cannabinoid CB1 receptors increased necrotic cell death in the Purkinje cells of the cerebellum. CB1Rs are mainly expressed on the terminal of neurons, where they can control neurotransmitters release. 51 Especially, they are localized on glutamate and GABA terminals, thus, can control the release of these neurotransmitters. Reports also note that activation of CB1Rs inhibited presynaptic glutamate release. 52 Moreover, previous findings have reported evidence of interaction between the endocannabinoid system and NOS. Mechanism of interaction between these systems probably related to the involvement of CB1Rs in the NO synthesis. ECBs through the modulation of Nitric Oxide synthase activity and the formation of NO, protect neurons from METH-excitotoxicity. 52 –55
Conclusion
In conclusion, the electrophysiological findings of this study demonstrated that in vitro treatment with METH led to profound changes in the intrinsic membrane properties of Purkinje neurons. Firing frequency was decreased in MET group, which could also be restored to control levels upon CB1 antagonist treatment. The findings of this research suggest that observed changes in the electrophysiological properties following METH exposure are attributable, at least in part, in mechanisms underlying METH dependence and cannabinoids system is involved in mediating cerebellum behaviors induced by METH. These findings provide further insight into the development of novel pharmacotherapies for the addiction to METH. However, further studies are needed to be conducted to answer whether CB1Rs involved in attenuation of METH-induced neurotoxicity.
Highlights
METH significantly decreased the spontaneous firing frequency of Purkinje cells
CB1Rs have an interaction with electrophysiological alterations induced by METH.
Exposure to the combination of METH and SR resulted in the necrotic cell death.
Cannabinoids system is involved in mediating cerebellum behaviors induced by METH
Footnotes
Authors’ note
Chemical compounds studied in this article Methamphetamine hydrochloride (PubChem CID: 66124); Win-55212-2 (WIN) (PubChem CID: 5311501); SR141716A (PubChem CID: 54585418)
Author contributions
All authors critically reviewed content and approved final version for publication. ER has conceived and designed the concept and collected data, searched the literature, and drafted the manuscript. MSh has searched the literature, categorized the searched papers and helped design the study. SD has critically reviewed the manuscript for its content, originality, usage of English language, and accuracy of interpreted histological data. MS has critically reviewed the manuscript, designed the study, and helped in manuscript preparation. He is the archival author and attests to the integrity of the original data and the analysis reported in this manuscript. All authors have made substantive contribution and attest to approving the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Kerman Neuroscience Research Center and Kerman University of Medical Sciences as a grant (97000860) for the PhD thesis conducted by Effat Ramshini.
