Abstract
To investigate the effect of baohuoside-I against nasopharyngeal carcinoma (NPC) and its underlying mechanism, baohuoside-I was employed to treat NPC cell lines CNE1 and CNE2 in vitro, followed by attachment and detachment assays to evalute the epithelial-mesenchymal transition (EMT) phenotype markers. Baohuoside-I was also administered to experimental mice to assess its effect on xenograft tumor growth and NPC cell metastasis. A microRNA (miRNA, miR) microarray was performed to screen for miRNA altered by baohuoside-I in NPC cells. Bioinformatic tools and luciferase activity assay was conducted to identify the downstream molecules mediating the anti-tumor property of baohuoside-I. Baohuoside-I inhibited EMT and metastasis and upregulated miR-370-3p in NPC cells, which was shown to directly recognize and inhibit expression of Hedgehog pathway component Smoothened (SMO). Baohuoside-I suppresses metastasis as well as EMT of NPC cells through targeting the Hedgehog pathway component SMO, and may serve as a potent anti-tumor agent in the clinical management of NPC.
Introduction
Among cancers of the nasopharynx orgin, nasopharyngeal carcinoma (NPC) is most common. 1 While NPC-resulted morbidity is relatively low in most regions around the world, it is fairly common in southern China, 2 particularly in Guangdong province. In China, NPC accounts for 18% of all cancer incidences. Globally, an estimated 65,000 fatal cases were reportedly associated with NPC in 2010. Various risk factors, such as virus infection, heredities, and dietary habits, are often linked to the occurrence of NPC, among which the Epstein-Barr virus infection is an unambigous causal factor. 3 It is believed that traditional dietary habits in southern China, for instance mass consumption of salted vegetable, fish and hot tea, critically contribute to NPC-induced morbidity among Cantonese population. 4 Additional, a number of mutations over-activating nuclear factor κ-light-chain-enhancer of activated B signaling pathway were identified in around 50% of NPC cases. Clinical managements of NPC include surgery, chemotherapy and radiotherapy, 5 all of which heavily rely on the overall health status and the stage of the tumor. Recently, a potential target for NPC immunotherapy emerged, which is the unique expression of Epstein-Barr virus latent protein in undifferentiated NPC.
Traditional Chinese medicine is an extraordinarily valuable and provides benefit for the human health around the globe. 6 Nowadays, herbs are more and more recognized for its therapeutic potential against numerous human illnesses including cancer. In China, Herba Epimedii (Chinese pinyin: Yin Yang Huo) has a history of being utilized for kidney and bone health, 7 with many other potential health benefits being extensively explored both in vivo and in vitro. Composition analysis of Herba Epimedii revealed baohuoside-I (also known as icariside II) as the main active flavonoid in Epimedii, which is known to exert a variety of beneficial activities including anti-inflammatory, anti-osteoporosis, neuroprotective and antioxidant properties. 8 More recently, baohuoside-I reportedly elicited apoptosis through reactive oxygen species (ROS) related mitochondrial signaling 9 and inhibition of U266 multiple myeloma growth 10 in human non-small-cell lung cancer cells, indicating the therapeutic potential of baohuoside-I in cancer management.
Prior reports have demonstrated that epithelial-mesenchymal transition (EMT) is intimated associated with tumor metastasis. EMT takes place in fibrosis, embryogenesis, and tumor invasion, sharing common features including the loss of epithelial cell phenotypes, increased mesenchymal markers, as well as enhanced cell migration capacity. 11 EMT is involved in the metastasis and invasion of NPC cells as well. Repression of the EMT of NPC cells could dramatically inhibit NPC metastasis. 12 –14
In the current study, we aimed to investigate the anti-tumor property of baohuoside-I in NPC and demonstrate the molecular mechanisms that underlie the effect of baohuoside-I. We focused on the microRNAome alteration elicited by baohuoside-I and attempted to elucidate its correlation with the therapeutic benefits of baohuoside-I.
Materials and methods
Cell culture and drugs
The CNE1 and CNE2 cell lines, both of which are human NPC cell lines, were obtained from the Cell Bank of Chinese Academy of Sciences (Beijing, China) with certification of short tandem repeat cell identity, and regularly examined for potential mycoplasma contamination. Cells were cultured in RPMI modified medium supplemented with 12% fetal bovine serum (Hyclone, MA, USA) and 1% antibiotics cocktail (Gibco, MA, USA), and maintained in a humidified incubator with 5% CO2 and passaged at 1:4. Lipofectamine 2000 (Invitrogen, MA, USA) kit was used for transfection.
Baohuoside-I purified from Cortex periplocae (>96% purity) (New Drug Research and Development Co., Ltd., Shijiazhuang, China) was dissolved in dimethylsulfoxide (DMSO), and diluted to a working concentration of 25 μg/ml in culture medium for in vitro experiments, and a dosage of 25 mg/kg body weight per day for in vivo experiments for 30 days, as previously reported. 15 The study was conducted in accordance with the Basic & Clinical Pharmacology &Toxicology policy for experimental and clinical studies.
Cell detachment and attachment assays
For the cell attachment assay, cells were placed in 24-well plates at the density of 5 × 104 cells per well. Unattached cells were discarded after 1 h incubation, and the attached cells were then trypsinized and counted. Results were presented as the percentage of attached cells/total cells. For the detachment assay, cells were treated with 0.05% trypsin for 3 min after 24 h of regular culture, and the detached cells were collected after the trypsin was inactivated. The cells that remained were subjected to an additional incubation with 0.25% trypsin to detach and then counted. Results were presented as the percentage of the detached cells / total cells.
Real-time quantitative polymerase chain reaction (qRT-PCR)
Total RNA was extracted from tissues or cells using Trizol reagent (Invitrogen, CA, USA). Reverse transcription was then carried out using the PrimeScript RT reagent Kit (Takara, Japan) followed by qRT-PCR using SYBR Prime Script RT-PCR Kits (Takara, Japan) according to the provided protocol. The relative expression levels of target genes were calculated using the 2−ΔΔCt method, with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as the internal control and presented as the fold changes of the corresponding controls. All assays were repeated three times. Primers used in the current study were: E-cadherin Forward primer: 5′-GTC GAG GGA AAA ATA GGC TG-3′ E-cadherin Reverse primer: 5′-GCC GAG AGC TAC ACG TTC AC-3′ N-cadherin Forward primer: 5′-GGC ATA CAC CAT GCC ATC TT-3′ N-cadherin Reverse primer: 5′-GTG CAT GAA GGA CAG CCT CT-3′ Vimentin Forward primer: 5′-GCA AAG ATT CCA CTT TGC GT-3′ Vimentin Reverse primer: 5′-GAA ATT GCA GGA GAT GC-3′ Smoothened (SMO) Forward primer: 5′-GTT CTC CAT CAA GAG CAA CCA-3′ Smoothened (SMO)Reverse primer: 5′-CGA TTC TTG ATC TCA CAG TCA GG-3′ GAPDH Forward primer: 5′-GGA GCG AGA TCC CTC CAA AAT-3′ GAPDH Reverse primer: 5′-GGC TGT CAT ACT TCT CAT GG-3′
Western blot and antibodies
Cells were harvested and lysed in standard radioimmunoprecipitation assay buffer for 30 min on ice, and then cell debris were discarded. 20 µg total protein per lane were run on a 10% sodium dodecyl sulfate–polyacrylamide gel and transferred onto polyvinylidene difluoride membranes. Immunoblotting was carried out with primary antibodies as follows: anti-E-cadherin, #3195, 1:1000; anti-N-cadherin, #13116, 1:1000; anti-Vimentin, #5741, 1:1000; anti-GAPDH, #2118, 1:1000 (all from Cell Signaling Technology, MA, USA); anti-SMO, E-5, 1:1000 (Santa Cruz Biotechnology, Santa Cruz, CA, USA); anti-carcinoembryonic antigen (CEA), ab175311, 1:500 (Abcam, Cambridge, MA, USA); anti-squamous cell carcinoma antigen (SCCA), ab154971, 1:1000 (Abcam, Cambridge, MA, USA). Blots were incubated with the appropriate horse radish peroxidase-conjugated secondary antibody (anti-rabbit, #7074, 1:5000; Cell Signaling Technology, MA, USA) and visualized with the use of enhanced chemiluminescence (ECL, Millipore, CA, USA). Resulted images were then quantified using ImageJ software.
Xenograft mice
The immune-deficient NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ mice (3∼4-week old, 15∼20 g) were purchased from VitalRiver (Beijing, China). All animals were housed under SPF conditions with ad libitum access to rodent chow and drinking water and allowed to acclimate for at least 1 week before experiments. Xenograft tumor model was established through subcutaneous inoculation of 1 × 106 NPC cells CNE1 and CNE2, and the metastasis potential was evaluated through intravenous injection of 1 × 106 NPC cells CNE1 and CNE2 into the tail vein. Mice were randomly divided into the following experimental groups (n = 8 mice each group): (1) 25 mg/kg/day Bao-I, mice were administered through oral gavage with 25 mg/kg body weight once per day for 30 days; (2) vehicle control, mice were administered through oral gavage with the same amount of saline containing the same diluted volume of DMSO as vehicle once per day for 30 days. All animal protocols were designed in compliance with the NIH guide and approved by the Committee of Animal Use and Care of Yantai Yuhuangding Hospital (Approval number #YTYHDYY-2018-98a3).
MicroRNA (miRNA, miR) expression profiling
NPC cells were treated with baohuoside-I or vehicle control for 24 h, and then harvested for miRNA extraction using miRNeasy mini kit (Qiagen). The miRNA expression profile was established with the use of GeneChip miRNA 4.0 Array (ThermoScientific). Bioinformatics analysis was conducted using the Partek Genomics Suite software. Only changes greater than twofolds were regarded as significant. Expression of individual miRNA was verified using mature miRNA assays (Thermo Fisher, Waltham, MA, USA) following manufacturer’s protocols.
Luciferase reporter assay
The 3′-untranslated region (3′-UTR) of SMO mRNA, as well as its mutated version, cloned to the downstream of luciferase reporter (Lufr) gene of pSi-Check2 luciferase reporter plasmid (Promega, MI, USA), namely SMO-Wt and SMO-Mut, respectively. NPC cells were either co-transfected with respective luciferase constructs and miR-370-3p mimic or transfected with respective luciferase constructs followed by baohuoside-I treatment for 24 h. Then the relative luciferase activities in the cell lysates were examined using Bright-Glo Luciferase Assay System (Promega, MI, USA), and the luciferase intensity was measured with Infinite F500 Microplate Reader (Tecan, Mannedorf, Switzerland).
Statistical analysis
Data were pooled from at least three independent experiments unless otherwise noted, and GraphPad 7.0. SPSS 23.0 was used for statistical comparisons. Student t test was utilized for comparison between two groups, where p values were calculated and p < 0.05 was regarded to indicate significant difference.
Results
Baohuoside-I inhibits EMT of NPC cells in vitro
We first examined the dosage effect of 0, 5, 25 and 100 μg/ml baohuoside-I on the viability of two NPC cell lines CNE1 and CNE2, and 25 μg/ml was found to be the optimal dose of baohuoside-I in vitro as it exerted no obvious cytotoxicity, whereas 100 μg/ml baohuoside-I severely inhibited the cell viability thereby preventing further experiments (Figure S1). Next, we examined the EMT phenotypes of NPC cells in vitro, and found that 25 μg/ml baohuoside-I in culture medium was able to suppress attachment (Figure 1(A)) and detachment (Figure 1(B)) rates of both CNE1 and CNE2 NPC cell lines. Further verification of EMT suppression by baohuoside-I was achieved at the molecular level, where the transcripts (Figure 1(C)) and proteins (Figure 1(D)) of several EMT markers, namely vimentin, E-cadherin, and N-cadherin, were examined by RT-PCR and Western blot. In line with compromised attachment and detachment capabilities, we found that in NPC cells treated with baohuoside-I, E-cadherin was greatly upregulated and both N-cadherin and vimentin were markedly down-regulated (Figure 1(C) and (D)). We therefore presented evidence supporting the inhibitory activities of baohuoside-I on EMT-related events in NPC cells.

Baohuoside-I inhibits EMT of NPC cells in vitro. NPC cell lines CNE1 and CNE2 were treated in the presence of 0 and 25 μg/ml of baohuoside-I (Bao-I), followed by analyses of their (A) attachment rate, (B) detachment rate, (C) mRNA levels of epithelial marker E-cadherin, and mesenchymal markers N-cadherin and Vimentin, and (D) protein levels of these markers. Values were shown as mean ± SD. Student t test was utilized for comparison between two groups, *p < 0.05, **p < 0.01, 0 vs 25 μg/ml Bao-I.
Baohuoside-I inhibits tumorigenesis and metastasis of NPC cells in vivo
To rule out possible artifacts associated with in vitro cell culture, we further examined the anti-tumor effects of baohuoside-I in vivo with the use of xenograft mice model. Immuno-deficient mice received subcutaneous inoculation of NPC cells and tumor growth was regularly monitored. Administration of baohuoside-I to the inoculated mice substantially delayed the progression of xenograft tumor compared to the vehicle control group (Figure 2(A) and (B)). Similarly, the weight of the resected xenograft tumor from mice that received baohuoside-I was significantly less than that from the control animals (Figure 2(C) and (D)). In addition, we observed a remarkable inhibition of the lung metastasis potential of CNE1 cells in mice treated with baohuoside-I, where metastatic sites were found in only two out of six tail vein injected mice compared to six out of six in the control group (Table 1). In summary, we demonstrated the inhibitory effect of baohuoside-I on both tumorigenesis and metastasis of NPC in vivo.
Lung metastasis of tail vein injected NPCs.


Baohuoside-I inhibits tumorigenesis and metastasis of NPC cells in vivo. Xenograft tumor volumes from either (A) CNE1 or (B) CNE2 inoculated NPC cell lines were monitored in mice receiving vehicle control and 25 mg/kg/day baohuoside-I (Bao-I) treatment, respectively. (C) On day 30, mice were sacrificed to measure tumor weight. (D) Representative images of the extract tumors on day 30. Values were shown as mean ± SD (n = 8 each). Student t test was utilized for comparison between two groups, *p < 0.05, vehicle control vs 25 mg/kg/day Bao-I.
Baohuoside-I alters miRNA expression profile in NPC cells
Seeking for molecular mechanism underlying baohuoside-I inhibitory effect on EMT, we performed miRNA microarray in the CNE1 and CNE2 NPC cell lines, with or without baohuoside-I. Only miRNAs that were consistently altered by baohuoside-I in both two cell lines by at least twofolds were shortlisted (Figure 3(A)). Next, expression levels of shortlisted miRNAs were confirmed by mature miRNA assays, respectively (data not shown). A particular miRNA, namely miR-370-3p, was consistently found to be upregulated by baohuoside-I treatment in NPC cells (Figure 3(B)). The reason for us to be interested in miR-370-3p, among others, was that miR-370-3p was predicted by Targetscan to be able to target the Hedgehog pathway component SMO (Figure 3(C)).
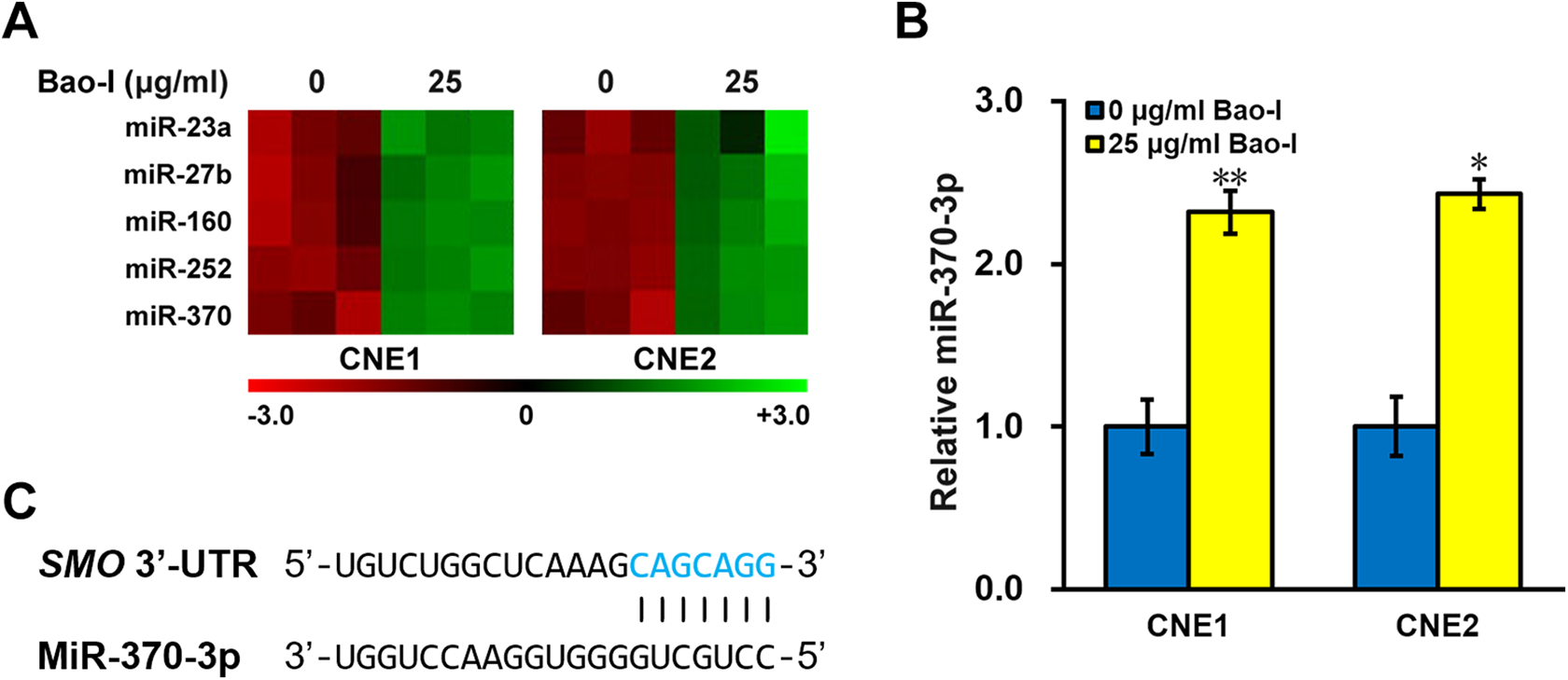
Baohuoside-I alters miRNA expression profile in NPC cells. (A) MiRNA microarray was performed in NPC cell lines CNE1 and CNE2 in the presence of 0 and 25 μg/ml of baohuoside-I (Bao-I) (three independent repeats). (B) Mature miRNA assay for miR-370-3p was performed in NPC cell lines CNE1 and CNE2 in the presence of 0 and 25 μg/ml of baohuoside-I (Bao-I). Values were shown as mean ± SD. Student t test was utilized for comparison between two groups, *p < 0.05, **p < 0.01, 0 vs 25 μg/ml Bao-I. (C) Sequence alignment between miR-370-3p and putative targeting site (red nucleotides) on the 3′-UTR of SMO mRNA.
MiR-370-3p directly suppresses Hedgehog pathway component SMO
Next, to further verify the correlation between miR-370-3p and SMO, luciferase report assay was employed, with the 3′-UTR of SMO mRNA, as well as its mutated version, cloned to the downstream of luciferase reporter gene (Figure 4(A)). Co-transfection with miR-370-3p dramatically suppressed the relative luciferase activity, an effect that was readily negated by the mutation introduced to the seed region (Figure 4(B)). Further, we assessed the endogenous abundance of SMO transcript as well as protein following forced expression of miR-370-3p in NPC cells. As demonstrated in Figure 4(C) and (D), both SMO mRNA and protein were remarkably decreased in the miR-370-3p-transfected cells. Altogether, we demonstrated that Hedgehog pathway component SMO appeared to be the direct target of miR-370-3p in NPC cells.

MiR-370-3p directly suppresses Hedgehog pathway component SMO. (A) Wildtype (SMO-Wt, blue nucleotides) or mutated (SMO-Mut, red nucleotides) putative targeting sequences from the 3′-UTR of SMO mRNA, respectively, were cloned to the downstream of a luciferase reporter open reading frame (Lufr). (B) Luciferase activities of SMO-Wt and SMO-Mut constructs were analyzed in CNE1 cells co-transfected with either negative control miR (miR-NC) or miR-370-3p mimic (miR-370). (C) mRNA and (D) protein levels of SMO were analyzed in CNE1 and CNE2 cells co-transfected with either miR-NC or miR-370-3p. Student t test was utilized for comparison between two groups, *p < 0.05, **p < 0.01, miR-NC vs miR-370-3p.
Baohuoside-I targets SMO to suppress EMT and metastasis of NPC cells
Naturally, we next examined the effect of baohuoside-I treatment on SMO expression in NPC cell culture. As expected, both the mRNA and protein levels of SMO were significantly inhibited by baohuoside-I (Figure 5(A) and (B)). In order to establish causality between SMO and baohuoside-I inhibited EMT phenotypes, we overexpressed SMO in NPC cells, and confirmed that both SMO transcript and protein levels in baohuoside-I-treated cells could be brought back to levels comparable to the parental cells in the absence of baohuoside-I treatment (Figure 5(A) and (B)). Next, we performed attachment and detachment assays in the parental NPC cells without baohuoside-I treatment (Control + 0 μg/ml Bao-I), parental NPC cells with baohuoside-I treatment (Control + 25 μg/ml Bao-I), and SMO-overexpressed cells with baohuoside-I treatment (SMO + 25 μg/ml Bao-I), respectively. Indeed, SMO overexpression could readily rescue the baohuoside-I-suppressed attachment and detachment rates of NPC cells (Figure 5(C) and (D)). Moreover, expression profiles of EMT-related molecular markers were consistent with the observed phenotypic rescue, in terms of transcriptional (Figure 5(E)) and protein levels (Figure 5(B)) of E-cadherin, N-cadherin and Vimentin.

Baohuoside-I targets SMO to suppress EMT phenotypes and markers in NPC cells in vitro. NPC cell lines CNE1 and CNE2, with or without SMO overexpression (O/E) were treated in the presence of 0 and 25 μg/ml of baohuoside-I (Bao-I), respectively, followed by analyses of their (A) SMO mRNA levels, (B) protein levels of SMO, E-cadherin, N-cadherin and Vimentin, (C) attachment rate, (D) detachment rate, (E) mRNA levels of epithelial marker E-cadherin, and mesenchymal markers N-cadherin and Vimentin. Values were shown as mean ± SD. Student t test was utilized for comparison between two groups, *p < 0.05, **p < 0.01, Control + 25 μg/ml Bao-I vs both Control + 0 μg/ml Bao-I and SMO + 25 μg/ml Bao-I.
At last, xenograft mouse model was again employed to test the above results in vivo. We observed significantly restored growth in SMO-overexpressing xenograft tumors, even in the presence of baohuoside-I administration (Figure 6(A) and (B)). At the endpoints of experiments, the average weight of SMO-overexpressing xenograft tumors from baohuoside-I administrated mice were also restored to the same as parental xenograft without baohuoside-I administration (Figure 6(C) and (D)). In addition, we also examined the protein levels of metastasis markers in the extracted tumor samples, including CEA and SCCA, which were both reduced by baohuoside-I treatment and restored by SMO overexpression (Figure 6(E)). Furthermore, the lung metastasis potential of injected NPC cells was also restored by SMO overexpression in mice with baohuoside-I treatment (Table 2).
Lung metastasis of tail vein injected NPCs.


Baohuoside-I targets SMO to suppress tumorigenesis and metastasis of injected NPC cells in xenograft mouse model in vivo. Xenograft tumor volumes from either (A) CNE1 or (B) CNE2 inoculated NPC cell lines, with or without SMO overexpression (O/E), were monitored in mice receiving vehicle control and 25 mg/kg/day baohuoside-I (Bao-I) treatment, respectively. (C) On day 30, mice were sacrificed to measure tumor weight. (D) Representative images of the extract tumors on day 30. (E) Protein levels of carcinoembryonic antigen (CEA), squamous cell carcinoma antigen (SCC) were analyzed by Western blot. Values were shown as mean ± SD (n = 8 each). Student t test was utilized for comparison between two groups, *p < 0.05, Control + 25 mg/kg/day Bao-I vs both Control + 0 mg/kg/day Bao-I and SMO O/E + 25 mg/kg/day Bao-I.
Discussion
EMT refers to the process in which a polarized epithelial cell that normally interacts with basement membrane via the basal surface undergoes several biochemical alterations to resume a mesenchymal cell phenotype, including enhanced invasiveness, migratory capacity, resistance to apoptosis, and elevated production of extra cellular matrix (ECM) components. 16 EMT is completed when the underlying basement membrane is degraded and a mesenchymal cell is generated, which can migrate away from its originating epithelial layer. Meanwhile, the signature EMT marker expression profiles are altered, for example, the downregulation of epithelial marker E-cadherin and the upregulation of mesenchymal markers N-cadherin and vimentin. 11 Multiple distinct molecular processes are involved in the initiation and completion of EMT, such as transcription factors activation, reorganization of cytoskeletal proteins, expression of particular cell-surface proteins and ECM-degrading enzymes, as well as altered expression of specific miRNAs. MicroRNAs is a class of widely expressed small oligonucleotide that exerts pivotal physiological functions in mammalsm. 17 Via post-transcriptional modulations of target genes, miRNAs could fine-tune the complicated signaling network involved in numerous cellular events. Accumulating evidence suggests that miRNAs are actively involved in many aspects of tumor biology including incidence, resistance, recurrence, and metastasis. 18
MiR-370 was previously implicated in the invasive behavior of many human cancer types. For example, in osteosarcoma cells, miR-370 could inhibit cell growth and metastasis by directly targeting forkhead box protein M1. 19 In human glioma cells, miR-370-3p was found to target β-catenin to inhibit proliferation and induce cell cycle arrest. 20 Similarly by targeting β-catenin, miR-370 could also suppress the proliferation as well as progression of human astrocytoma and glioblastoma. 21 Interestingly, downregulation of miR-370 correlated with cancer progression and proliferation in esophageal squamous-cell carcinoma cells. 22 While on the other hand, upregulation of miR-370 was reported to inhibit proliferation and promote apoptosis of gastric cancer cells by targeting PTEN (phosphatase and tensin homolog), 23 as well as inhibit the proliferation, invasion and EMT by targeting PAQR4 (Progestin and AdipoQ Receptor 4). 24 Furthermore, miR-370 was consistently reported to inhibit lung cancer metastasis and liver cancer EMT. 25,26 However, to date, the involvement of miR-370 in NPC still remains a mystery. In our current study, through an miRNA microarray, we have identified miR-370-3p as a critical component in the baohuoside-I-induced suppression of EMT and metastatic phenotypes of NPC cells, for the first time.
In addition to our discovery of the anti-tumor activity of baohuoside-I in the NPC setting, baohuoside-I has also been reported to exert similar actions in other types of cancers. For instance, baohuoside-I could suppress cell growth and inhibit cyclin D1 and survivin in esophageal carcinoma through a β-catenin-dependent mechanism. 15 Another study in non-small cell lung cancer suggested that baohuoside-I could induce mitochondria-dependent apoptosis. 9 In cervical and breast cancer cells, baohuoside-I was found to inhibit expression of CXC chemokine receptor 4. 27 More recently, baohuoside-I was reported to repress proliferation and migration of melanoma cells by elevating expression of miR-144. 28 Since miRNAs inhibit its target gene by directly recognizing the complementary sequences in the 3′-UTR of mRNA, 17 we continued with bioinformatic tools to identify the potential downstream target of miR-370-3p, in the context of NPC cell EMT. One of the predict gene with high confidence was the Hedgehog pathway component SMO. Shh, Dhh and Ihh are members of the Hedgehog family. 29 Hedgehog ligands interact with patched homolog 1 (PTCH1) and PTCH2, both of which in the absence of ligand binding would negatively regulate SMO activity. 30 Activated PTCH1/2 dis-inhibits SMO and initiates an intracellular cascade to activate Gli family transcription factors and in turn facilitate expression of target genes. 31 The Hedgehog pathway has been frequently implicated in the EMT of various human cancers. For instance, Oncogenic forms of Shh is identified in basal cell carcinoma, and mis-regulation of Shh is observed in esophageal and stomach cancer, pancreatic adenocarcinoma, and prostate adenocarcinoma. 32 In pancreatic cancer cell lines, cyclopamine, a steroidal alkaloid that binds and deactivates SMO, 33 disrupts Shh and reduces EMT. 34 Other studies demonstrated that Hedgehog mediated suppression of Wnt signaling pathway via paracrine mechanisms involving local mesenchymal stromal cells. 35 Aberrant activation of Hedgehog pathway was also reported in NPC, and was suggested to be a novel therapeutic target. 36 However, the exact involvement of Hedgehog pathway in EMT and metastatic behavior of NPC is yet unknown. Data in our current investigation has pointed to the Hedgehog signal transducer SMO as the critical player in promoting the metastasis and EMT of NPC cells both in vivo and in vitro. Our results reveal that targeting SMO, either by miR-370-3p or baohuoside-I, may provide synergistic therapeutic efficacy against metastasis and EMT of NPC cells (Figure 7).

Schematic model of the study. Targeting SMO, either by miR-370-3p or baohuoside-I, may provide synergistic therapeutic efficacy against metastasis and EMT of NPC cells.
In conclusion, we have presented evidences that the flavonoid baohuoside-I could inhibit metastasis and EMT of NPC cells both in vivo and in vitro. Baohuoside-I treatment particularly upregulated miR-370-3p in NPC cells, which was shown to directly recognize and inhibit expression of Hedgehog signal transducer SMO, which mediated the inhibitory effect of baohuoside-I against metastasis and EMT of NPC cells. Therefore, baohuoside-I suppresses metastasis and EMT of NPC cells through targeting the Hedgehog pathway, and may serve as a potent anti-tumor agent in the clinical management of NPC.
Supplemental material
Supplementary_materials - Effects of baohuoside-I on epithelial-mesenchymal transition and metastasis in nasopharyngeal carcinoma
Supplementary_materials for Effects of baohuoside-I on epithelial-mesenchymal transition and metastasis in nasopharyngeal carcinoma by Q Wang, S Jiang, W Wang and H Jiang in Human & Experimental Toxicology
Footnotes
Author contributions
QW and SJ contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
