Abstract
Bisphenol A (BPA), an imperative environmental contaminant used in polycarbonate plastics. Due to limited information concerning the effect of BPA on lungs, this study design to assess whether BPA cause alterations in histopathology and trace metal content in lungs of rats. They were divided into five groups with five rats per group. Group I was named as control group. Group L6 and L12 were received BPA (10 mg/kg body weight/day) for 6 weeks and 12 weeks respectively. Group H6 and L12 were given BPA (25 mg/kg body weight/day) for 6 weeks and 12 weeks respectively. Considerable alteration in Cu, Zn and Fe was detected in experimental groups. BPA also caused significant increase in the expression of tumor necrosis factor α that mediate the pulmonary inflammatory response. Comparative study of resolved proteins i.e. 72 KDa (matrix metalloproteinase 2 fragment) and 109 KDa (nucleolin) on SDS-PAGE showed their altered expression in experimental groups. Histopathology of experimental groups revealed altered architecture of lungs. Special staining of BPA treated groups showed significant number of mast cells in alveoli and bronchioles. Prolonged administration of BPA causes deleterious aggravating lung damage even at extremely low dose, so the use of BPA should be prohibited in plastic synthesizing industries.
Introduction
BPA is one of ubiquitous important environmental contaminants in which presence of two functional phenol groups help BPA to attain properties that are beneficial in manufacturing procedures and normally used consumer products. Principal use of BPA is to make polycarbonate plastics. 1,2 Moreover, BPA is used in epoxy resins lining food and beverage containers, dental sealants, carbonless copy thermal papers, and as originator in fire retardants. 3 BPA is a customary constituent of user products relevant to food like baby bottles, food storage containers, plastic wrap, water bottles, and the linings of metal cans. Due to extensive use of BPA containing items human highly exposed globally. 4 Whole world’s population is subjected to recurring episodes of BPA, chiefly by means of packed food. There is a thought that mainly through diet human exposure occur because under high temperature and acidic or basic conditions BPA containing polymers can be hydrolyzed that caused leaching into drink and food containers. 5
It was reported that BPA could cause lots of harmful health impacts like, atypical childhood neurobehavioral problems, 6 dwindle semen quality, 7 diabetes, 8 heart rate variability and blood pressure. 9
Trace metals are important part of human beings as they play crucial role in various biological processes. Many important functions performed by Zn, Cu and Fe elements as cofactors of some enzymes. Cytoplasmic Cu-Zn superoxide dismutase enzyme (SOD) use zinc and copper as cofactors. Iron takes part in various biochemical pathways and grants a prosthetic group to variety of enzymes principally to catalase (antioxidant enzyme). 10 Harmful oxygen species cannot be detoxified if level of these trace metals decrease significantly. Extensive damage and inflammation happened if concentration of these trace elements increase or decrease. 11
Most of the pro-inflammatory mediators are released by mast cells. Mast cells are resident leukocytes of myeloid origin that are present especially in those organs that are in contact with external environment like lungs skin and intestine etc. 12 Cytoplasmic granules containing pro-inflammatory mediators such as cytokines, histamine, proteases etc are present in mast cells. It has been documented that more than 60 different types of chemokines, cytokines and growth factors are released by mast cells. 13 It was reported that exposure of BPA activates mast cells.
Accordingly, current work was intended to assess the effects of different doses of BPA on the lungs of adult male Wistar rats.
Materials and methods
Experimental animals
Adult male Wistar rats were used as experimental model. All rats were reared at Department of Zoology, University of Punjab, Lahore. They were housed in an air conditioned room at temperature of 25–27°C. They had admittance to water ad libitum and normal chow. They were weighed daily. When rats gain a weight of 175 ± 25 g, experimental work was started. For the reduction of background exposure of BPA, polysulfone cages and glass water bottles were used.
Experimental design
They were divided into five groups with five rats per group. Group I was named as control group. Group L6 and L12 were received BPA (10 mg/kg body weight/day) for 6 weeks and 12 weeks respectively. Group H6 and H12 were given BPA (25 mg/kg body weight/day) for 6 weeks and 12 weeks respectively. Dose of BPA dissolved in 1 ml of corn oil was given orally by oral gavage to each rat and they were weighed before giving the dose daily. At the end of the experiment, rats were sacrificed by making them anesthetized. Lungs were taken out by making incision. Lungs were fixed in 10% formalin (for histopathological analysis) and were freeze (for trace metal analysis and PCR) at −80°C.
Trace metal analysis
Atomic absorption spectrophotometer was used for acid digestion of lungs to determine trace metal level. Preserved frozen samples (at −80°C) of lunge were taken in digestion flasks. Acid digestion was done by adding 5 ml of nitric acid in 0.5 g of lungs samples of each group. On hot plate, digestion flasks were heated at temperature of 90–95°C. For 10–15 minutes they were refluxed without allowing them to boil. Digested samples were allowed to cool. Then 1 ml of deionized water (DI) was added to the samples. Acid digested samples were received 1.5 ml of hydrogen peroxide (30%). Again, samples were heated to stop effervescence. In parts of 1 ml hydrogen peroxide was added. To stop effervescence samples were heated. Quantity of hydrogen peroxide did not go beyond the limit of 5 ml. DI water was added to leveled the digested flasks up to 50 ml. Trace metal level in each digested sample was measured by using atomic absorption spectrophotometer.
Gene expression analysis by reverse transcription polymerase chain reaction (RT-PCR)
Preserved frozen samples (at −80°C) of Lungs were used to isolate the total RNA manually by using TRIzol. To estimate the quantity and purity of the extracted RNA, Nano drop UV/VIS Spectrophotometer was used, then RNA was reverse transcribed to corresponding cDNA using cDNA Synthesis kit. Genomic PCR amplification of TNF-α was performed using the previously described primers; forward: 5′-ACA AGG CTG CCC CGA CTA T-3′ and reverse: 5′- CTC CTG GTA TGA AGT GGC AAA TC-3′. To ensure the equal loading of sample β-actin was used as housekeeping gene by using the previously reported primers forward: 5′-TGT CAC CAA CTG GGA CGA TA-3′ and reverse: 5′-AAC ACA GCC TGG ATG GCT AC-3′. 14 PCR was performed on a thermocycler (Labnet, Model: MultiGeneOptiMax) using the following thermal cycling conditions: initial denaturation at 94°C for 5 minutes, denaturation for 30 seconds at 94°C, annealing for 30 seconds at 60°C, Extension for 30 seconds at 72°C (All steps i.e. denaturation, annealing and extension were run consecutively for 40 cycles) and final extension for 10 minutes at 72°C. To find the relationship of different genes further analysis of gel was done by the help of imageJ and GraphPad Prism 5 software.
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE)
Preparation of working sample
0.21 g of frozen lung sample was incubated in 0.7% of SDS for 30 minutes at 37°C. Then these samples were homogenized with the help of homogenizer. After that, homogenized samples were centrifuged at 7000 × g. After centrifugation, supernatant was taken out with the help of micro-pipette and pellet was discarded. Then in Eppendorf tube, 10 µl of lung sample was mixed with 10 µl of loading dye with the help of vortex. To denature proteins of sample, it was heated for 10 minutes at boiling temperature. At the end, in separate wells 10 µl of each sample was loaded. 12% gel was used to resolve proteins from lung tissue samples.
Proteins which are separated by electrophoresis were quantified by TotalLab Quant v11.5. Database information of each protein band is provided by TotalLab Quant v11.5 on the basis of molecular density of each band and was analyzed to find the changes in level of protein of control and experimental groups.
Histopathological study
Formalin fixed tissues were dehydrated (by passing them through ascending concentrations of ethanol) and embedded in paraffin wax to prepare 5 µm thick sections. Paraffin embedded lung sections were stained with H&E and toluidine blue.
Statistical analysis
Statistical analysis was performed by using GraphPad Prism 5 software. Data of trace metals content were statistically evaluated by using one-way ANOVA followed by Tukey’s Post hoc test. Data were presented as Mean ± SEM. Statistical significance was established at P < 0.05.
Results
Trace metal estimation
In BPA treated groups Cu content found to be significantly varied from control group. Significant increase in Cu concentration was observed in BPA exposed groups. As dose increased, Cu level was also increased. Significant difference in Cu level between low dose groups (L6 and L12) and high dose groups (H6 and H12) was also noticed. Increase in Cu level in H12 was highly significant as compared to H6 group making a BPA’s exposure duration a crucial factor to elevate Cu level.
There was highly considerable altered Zn concentration between control and all BPA treated groups. Significant decrease in Zn level was observed in BPA treated groups in comparison of control group. While Lungs Fe content of all experimental groups did not differ significantly as compared to control groups (Table 1) (Figure 1).
Mean level of trace metals (Fe, Cu and Zn) in control and BPA treated lungs.


Result of (a) copper profile, (b) zinc profile and (c) iron profile of rat’s lung showing altered expression in copper, zinc and iron level in BPA exposed groups in contrast of control group. *Significant at P < 0.05; **significant at P < 0.01; ***significant at P < 0.001.
PCR analysis of pro-inflammatory cytokine expression (TNF-α)
PCR analysis of TNF-α gene expression revealed that there was highly significant up regulation in TNF-α in all experimental groups i.e. L6 (0.51 ± 0.13 folds), H6 (0.61 ± 0.05 folds), L12 (1.02 ± 0.08 folds) and H12 (1.83 ± 0.03 folds) as compared to control group when analyzed by one-way ANOVA (P < 0.0001). When inter group analysis was done by using Post hoc Tukey’s test, significant difference in TNF-α level between low dose groups [L6 (0.51 ± 0.13 folds) and L12 (1.02 ± 0.08 folds)] and high dose groups [H6 (0.61 ± 0.05 folds) and H12 (1.83 ± 0.03 folds)] was also noticed. Dose duration is an imperative parameter to alter gene expression. Increase in gene expression of TNF-α in L12 and H12 was highly significant as compared to L6 and H6 group respectively making a BPA’s exposure duration a crucial factor to elevate expression of TNF-α gene as shown in Figure 2(a) and (b).

(a) Image of densitometric analysis exhibiting gene expression of beta-actin and TNF alpha in control and BPA treated groups. (b) Showing highly significant (*significant at P < 0.05; **significant at P < 0.01; ***significant at 0.001) upregulation in gene expression of TNF alpha in all BPA treated groups in contrast of control group.
Quantification of lung protein by SDS-PAGE
Lung protein of control and BPA exposed group were resolved on SDS-PAGE along with protein marker of molecular weight ranging between 10 and 220 kDa. The gel having bands of lung proteins were analyzed by using software (TotaLLab quant).
Comparative study of resolved proteins showed expression of matrix metalloproteinase 2 fragment (Type IV collagenase, 72 KDa) was enhanced in all BPA exposed groups (except H12) in comparison of control group. Its level was quiet down regulated in H12 as compared to control. Expression of nucleolin protein (109 KDa) was lower in L6 and H12 in contrast of control while in L12 and H6 group its expression was up regulated as compared to control (Figures 3 to 7).

Showing expression of electrophoretically separated lung proteins of different molecular weight in control and BPA exposed groups along with protein ladder.
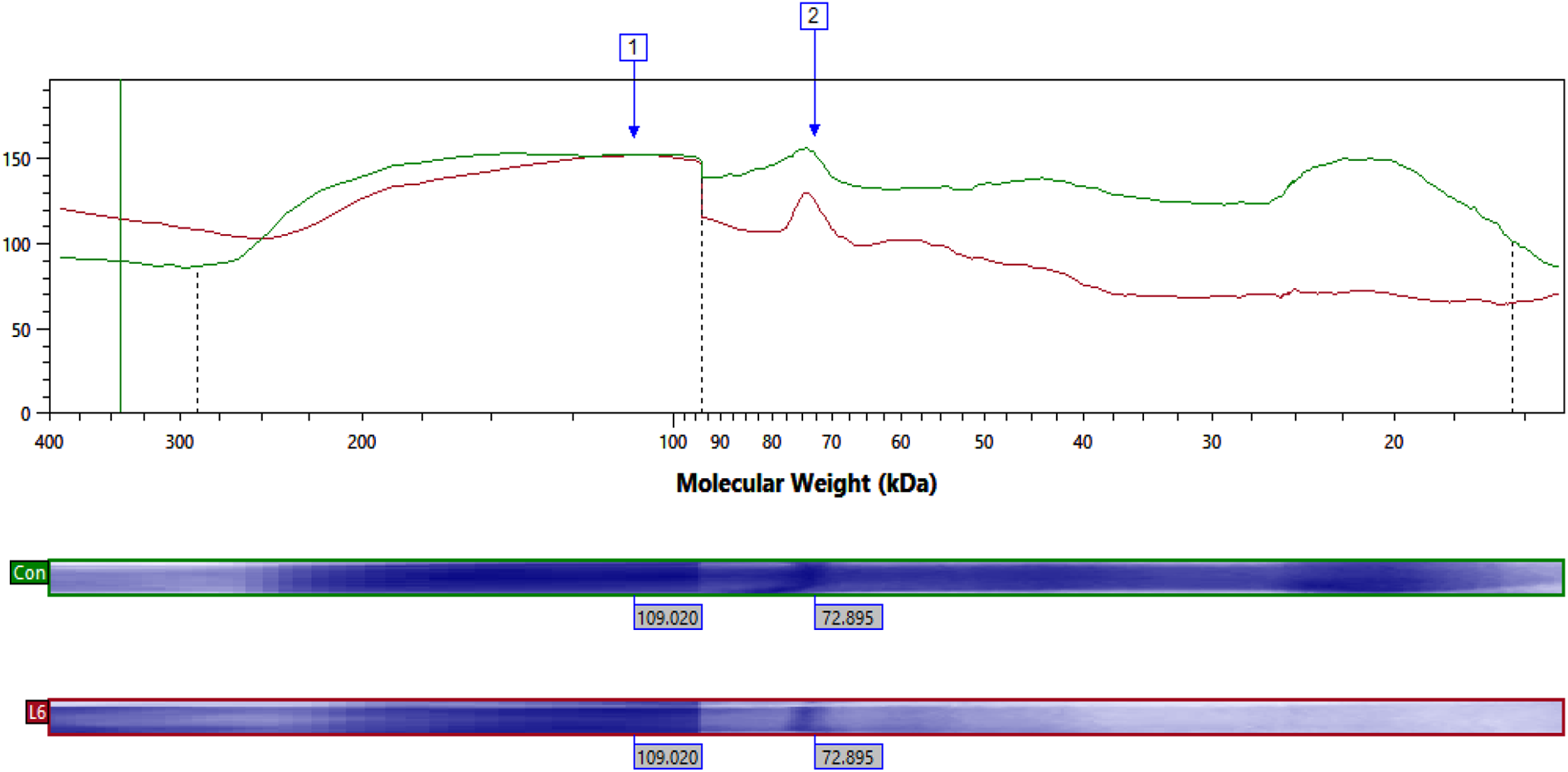
Showing expression of electrophoretically separated lung proteins of different molecular weight in control and L6 along with protein ladder.

Showing expression of electrophoretically separated lung proteins of different molecular weight in control and H6 along with protein ladder.

Showing expression of electrophoretically separated lung proteins of different molecular weight in control and L12 along with protein ladder.

Showing expression of electrophoretically separated lung proteins of different molecular weight in control and H12 along with protein ladder.
H&E staining
Lung sections of healthy control group rats showed normal structural design of lungs. They showed normal polygonal shaped alveoli with a bronchiole and blood vessel. Alveoli were spaced with thin inter alveolar septa. Histopathology of group II and group III revealed slightly inflammed alveoli and mild thick inter alveolar septa with increase in height of alveolar and bronchiolar epithelium due to restricted areas of nuclear infiltration. Replete in blood in connective tissues between alveoli was and dilated congested blood vessels were also detected. Rigorous alveolar damage and grievous nuclear infiltration with manifest thickness in inter alveolar septum was observed in group IV and V. Extravasated RBCs were also noticed in the alveolar lumen with severe dilated congested blood vessel (Figure 8).

Lung section of (A) control, (B) L6, (C) L12, (D) H6, and (E) H12 groups stained with H&E staining. Optika B-150 DBR microscope was used to capture image at the magnification of 40× and 100×.
Toluidine blue staining
Histology of control group lung tissue showed very few numbers of mast cells of smallest size. While staining of group II and group III revealed significant number of mast cells of considerable size in inflamed alveoli and bronchiole with marked increase in height of inter alveolar septum. Number and size of mast cells increased as dose increased (Figure 9).

Lung section of (A) control, (B) L6, (C) L12, (D) H6, and (E) H12 groups showing mast cells stained with toluidine blue staining. Optika B-150 DBR microscope was used to capture image at the magnification of 40× and 100×.
Discussion
The extensive consumption of BPA-containing products has raised concerns among scientists and regulatory agencies that human exposure to BPA may have undesirable effects on different vital organs. 15 Accordingly, current work was intended to assess the effects of different doses of BPA on the lungs and liver of adult male Wistar rats.
Trace metals are important part of human beings as they play crucial role in various biological processes. In current study, the levels of Cu, Fe and Zn were recorded in BPA treated lung tissues against control. Considerable alteration in trace metal level (Cu, Zn and Fe) was detected in experimental groups treated with BPA. Highly significant Cu overloading was observed in BPA exposed groups in contrast of control group. Cu is very imperative cofactor for variety of enzymes like IysyI oxidase, cytochrome C oxidase etc. 16 As enzymes are responsible for various metabolic activities which are requisite to sustain life so, increased level of copper in BPA exposed experimental groups might have led to various drastic changes in rat’s lungs.
Zn content was found to be significantly declined as compared to control. Zn is cofactor of various enzymes and key component of many metalloproteins like regulatory zinc finger proteins. 16 It was reported that in Zn deficient tissues, activities of several zinc dependent enzymes were affected badly. In some enzymes, zinc atoms take part in catalysis and are very crucial to maintain structure of apoenzyme. Zinc also performs a role to stabilize biomembrane structure and polynucleotide confirmation. 17 Low Zn concentration in experimental groups against control group showed less capability of enzymes against oxidative stress. 18 BPA treated groups may have less stable biomembrane structure and less capability of enzymes as compared to control. All aforementioned anomalies might be present in our BPA treated groups as zinc content in them was also decreased to highly significant level.
Increase in Fe content was exhibited in experimental groups exposed to BPA against control group but this increase was not up to significant level. Iron takes part in various biochemical pathways and grants a prosthetic group to variety of enzymes principally to catalase (antioxidant enzyme). 10 The heme group containing Fe is a cofactor of catalase. Catalase has heme group in its biding site which is responsible for its catalytic activity. 19 When control of iron gets disturbed, it is more crucial for growth of tumor cells. Increased Fe level in BPA treated groups may has negative impact on catalase activity.
Chronic lung toxicity provokes pulmonary apoptosis, necrosis and other inflammatory effects. The inflammatory response is mediated by proinflammatory cytokines, especially TNF-α. Release of TNF-α is one of the initial actions in several types of pulmonary damages which consequently alter the effect of many other cytokines. PCR analysis of TNF-α gene expression showed that there was highly significant elevation in pulmonary TNF-α of all groups i.e. L6 (0.51 ± 0.13 folds), H6 (0.61 ± 0.05 folds), L12 (1.02 ± 0.08 folds) and H12 (1.83 ± 0.03 folds) in contrast of control group when analyzed by one way ANOVA (P < 0.0001). These findings were in accordance to previous findings as a previous data have shown a significant correlation between BPA and inflammatory markers TNF-α. This previous study revealed that higher BPA levels (1.4-fold increase compared with the control) are associated with higher levels of TNF-α pro-inflammatory cytokines. 20 Similar kind of study was carried out on rats to investigate the effect of food additive (sodium nitrate) on rat liver showed significant up regulation of hepatic TNF-α in sodium nitrate exposed rats. Similar observations in the present study are observed suggesting that BPA has dose dependent effect on up regulation of TNF-α in liver tissue. 21 BPA induced the TNF-α level to increase. Our results are in accordance with previous results found in the literature as Previously it was documented that higher BPA plasma induced higher circulating levels of TNF-α. 22 Higher concentrations of TNF-α increase the number of TNF alpha receptors on the surface of cell. Due to which more excessive amount of TNF-α released. Up regulated amount of TNF-α produce exocrine and endocrine affects like hypotension, microvascular coagulation, fever and initiation of metabolic wasting. 23 High level of TNF-α is cytotoxic for endothelial cells and cause the synthesis of ROS, proteases and collagenases. 24 TNF-α perform important role in cell death by a number of mechanisms such as oxygen free radicals, protein kinases second messenger pathways, regulation of nuclear regulatory factors and DNA fragmentation. 25
Two types of lung proteins of molecular weight 72 KDa and 109 KDa were resolved on the SDS-PAGE in control and experimental groups when analyzed by software. Comparative study of resolved proteins showed expression of matrix metalloproteinase 2 fragment (Type IV collagenase) was enhanced in all BPA exposed groups (except H12) in comparison of control group. Surprisingly, its level was quiet down regulated than that of control. Low level of matrix metalloproteinase 2 fragment in H12 group might be attributed to high dose and prolonged exposure of BPA. Maintenance of the extracellular matrix is very important for the normal functioning of lung. Almost all resident lung cells (epithelial, endothelial, macrophages, fibroblasts etc) produce MMPs. MMPs are involved in wound healing process after lung injury. 26 Basically, it is an inflammatory protein. Matrix metalloproteinases perform a vital role in the process of inflammation of lungs 27 and their level become increase in response of chemical exposure, oxidative stress, lung disease etc. A study conducted on preterm babies who subsequently show CLD (chronic lung disease) revealed increase level of MMP in them than those who do not have CLD. Enhanced expression of MMP during oxidative stress may leads to alveolar basement membrane disruption. 28 In our study also, BPA exposed rats’ lung showed up regulated expression of matrix metalloproteinase 2 fragment except H12 group. Expression of nucleolin protein was quiet strange in all BPA treated and control group. In L6 its expression was lower in contrast of control while in L12 and H6 group its expression was up regulated as compared to control. Increase in nucleolin was reported in a previous study conducted on male Sprague-Dawley (SD) rats that were administered high dosage geniposide (300 mg/kg daily), medium dosage geniposide (100 mg/kg daily) and low dosage geniposide (30 mg/kg daily) for 3 days showed sequential increase in the level of nucleolin in all experimental groups 29 as it was observed in H6 and L12 groups in our study. But L6 and H12 showed unusual behavior. Expression of nucleolin in L6 and control group did not differ that much might be due to low dose exposure (10 mg kg) for short time period. Nucleolin performs a crucial role in cell proliferation, chromatin decondensation. 30 stability of mRNA, apoptosis, RNA/DNA binding and helicase activity. 31 Altered expression of nucleolin might cause variety of disruptions in afore mentioned mechanisms.
The lungs of BPA treated rats showed dose dependent histological changes. Histopathological analysis of lungs tissue showed extravasated RBCs, thick congested blood vessels, obstructed bronchioles by shed epithelial cells, slightly inflamed alveoli and thick alveolar septum with some restricted areas of cellular infiltration in experimental groups treated with low dose (10 mg/kg) but these results were more pronounced in high-dose BPA-treated groups (25 mg/kg). These results were in harmony with previous results like, it was reported that mild dose of BPA induced mild thickening of alveolar septa, localized focal areas of inflammatory cell infiltration, extravasated RBCs and dilated congested blood vessels and these alterations were more noticeable in groups treated with high dose. 15 Many collapsed alveoli in BPA-exposed rats could be attributed to decline synthesis of surfactant because of phospholipids’ (resident of pneumocyte type II) peroxidation. Degenerative changes (due to oxidative stress) in type II pneumocytes is another reason of decrease synthesis of surfactant. 32,33 Current study exhibited marked thickening of blood vessels which might be attributed to cyclophilin A and oxidative stress. Cyclophilin A (a chaperone protein) is released from vascular smooth muscle cells in reaction to reactive oxygen species. Cyclophilin A activates proliferation and migration of vascular smooth muscle cell and recruitment of inflammatory cell in vivo and in vitro. 34 Thickened interalveolar septa that observed in current study might be attributed to enhanced cellular infiltration (with nutrophils, lymphocytes, macrophages and eosinophils) and deposition of huge amount of interstitial collagen fibers. 35 Microanatomy of lungs exhibited mast cells. Number and size of mast cells increased as intensity and duration of dose increased. These results were similar to previously documented results. 36
Conclusion
In present study, the experimental conclusions may serve as alarm about exacerbate impacts of BPA. Prolonged administration of different doses of BPA cause deleterious aggravating lung damage, considerable alteration in trace metal level (Cu, Zn and Fe), up regulate amount of TNF-α that produces exocrine and endocrine affects like hypotension, microvascular coagulation, fever and initiation of metabolic wasting even at extremely low dose, so the use of BPA should be prohibited in plastic synthesizing industries and specious handling of plastic containers should be evaded to lessen health hazards and researchers should extend the investigation based upon the toxicity of xenoestrogens so that worldwide use of BPA and other likely xenoestrogens would be replaced by non-toxic chemicals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
