Abstract
Mercury is a widespread pollutant. Mercuric ions uptake into tubular cells is supported by the Organic anion transporter 1 (Oat1) and 3 (Oat3) and its elimination into urine is through the Multidrug resistance-associated protein 2 (Mrp2). We investigated the effect of recombinant human erythropoietin (Epo) on renal function and on renal expression of Oat1, Oat3, and Mrp2 in a model of mercuric chloride (HgCl2)-induced renal damage. Four experimental groups of adult male Wistar rats were used: Control, Epo, HgCl2, and Epo + HgCl2. Epo (3000 IU/kg, b.w., i.p.) was administered 24 h before HgCl2 (4 mg/kg, b.w., i.p.). Experiments were performed 18 h after the HgCl2 dose. Parameters of renal function and structure were evaluated. The protein expression of Oat1, Oat3 and Mrp2 in renal tissue was assessed by immunoblotting techniques. Mercury levels were determined by cold vapor atomic absorption spectrometry. Pretreatment with Epo ameliorated the HgCl2-induced tubular injury as assessed by histopathology and urinary biomarkers. Immunoblotting showed that pretreatment with Epo regulated the renal expression of mercury transporters in a way to decrease mercury content in the kidney. Epo pretreatment ameliorates HgCl2-induced renal tubular injury by modulation of mercury transporters expression in the kidneys.
Introduction
Mercury is a prominent environmental contaminant that causes detrimental effects to human health. Potential sources of mercury exposure include dental amalgam and various commercial products, such as skin creams, germicidal soups, teething powders, incandescent lights, batteries and ritualistic practices using mercury. 1,2
The health effects of mercury vary with the different chemicals forms of this metal, the dose and the rate of exposure. The more nephrotoxic species of mercury are by far, inorganic species. 1,3 Mercuric ions (Hg2+) rapidly accumulate in renal proximal tubule cells. 3 The proximal tubular cells have specific transport systems involved in the handling of Hg+2. Several in vivo and in vitro studies indicated that Hg+2 S-conjugates of small endogenous thiols are the primary transportable forms of mercury in the kidneys. 1,3,4 The Organic anion transporter 1 (Oat1) and the Organic anion transporter 3 (Oat3) participate in the basolateral uptake of S-conjugates of Hg+2 from the peritubular blood into proximal cells. 1,3 Oat1 and Oat3 are localized exclusively at the basolateral plasma membrane of renal proximal tubule cells and both mediate the transport of many compounds that include both endogenous and exogenous substances such as various anionic drugs and environmental compounds. 5 On the other hand, the renal cellular elimination of Hg+2 S-conjugates into the luminal space is mediated by the Multidrug resistance-associated protein 2 (Mrp2). 6,7 Mrp2 is localized in the luminal plasma membrane of proximal tubular epithelial cells. A broad variety of physiologic and xenobiotic metabolites and also various unmodified drugs such as doxorubicin and cisplatin are substrates of Mrp2. 8 It has been described that the modulation of Oat1, Oat3, and Mrp2 expressions are relevant in the degree of mercury-induced toxicity. 9 –12
Mercury can cause cell injury through different mechanisms such as the formation of reactive oxygen species, lipid peroxidation, alteration of protein synthesis and metabolic disorders by binding and interacting with complexes of protein and non-protein sulfhydryl, mitochondrial dysfunction, cell apoptosis and necrosis. 3,13,14 It had also been described that Hg+2 is involved in the accumulation of inflammatory components and in the induction of autoimmunity. 1,3
There is growing evidence that erythropoietin (Epo) is a potential therapeutic agent in acute renal injury. It has been described that treatment with Epo has a nephroprotective action in several experimental models of renal injury, due to its anti-oxidative, anti-apoptotic and anti-inflammatory effects. 15 –18
Since mercury is a toxic persistent pollutant, widely present in the environment, it is important to reduce its impact on human health. Moreover, there are no available treatments (other than dialysis) for kidney damage and acute renal failure. Thus, the clinical handling of these nephropathies is difficult and expensive, and the study of new strategies to prevent nephrotoxicity constitutes an active area of investigation and a public health concern.
Based on the preceding, the central aim of this study was to evaluate the impact of the pre-administration of Epo in the renal damage induced by HgCl2.
Methods
Reagents
Pure chemicals of analytical grades were purchased from Sigma (St. Louis, MO, USA). The polyclonal antibody against Oat1 was purchased from Alpha Diagnostic International (San Antonio, TX, USA) and the polyclonal antibody against Mrp2 from Abcam (Cambridge, MA, USA). The polyclonal antibodies against Oat3 and Oat5 were kindly given by Professor N Anzai (Department of Pharmacology, Graduate School of Medicine, Chiba University, Japan). The Kaleidoscope Prestained Standards of molecular mass were purchased from Bio Rad Laboratories (Hercules, CA, USA).
Experimental animals
Adult male Wistar rats aged from 110 to130 days were used throughout the study. All animals were allowed free access to a standard laboratory chow and housed in a constant temperature and humidity environment with regular light cycles (12 h) during the experiment. All experiments were conducted according to National Institute of Health (NIH) Guide for the Care and Use of Laboratory and were approved by the Faculty of Biochemical and Pharmaceutical Sciences (UNR) Institutional Animal Care and Use Committee (Res Nº 385/2017).
Animals were treated with a single dose of recombinant human erythropoietin (Epo) (Cassará Laboratory, Buenos Aires, Argentina) (3000 IU/kg b.w., i.p.), according to Rjiba-Touati et al. 16,17 who demonstrated that this pretreatment condition protects the kidney against cisplatin, etoposide and methotrexate, and/or with a single injection of HgCl2 (4 mg/kg b.w., i.p.) 24 h after Epo injection. Experiments were performed after 18 h of the HgCl2 injection as previously described. 3,10,12,19,20
Animals were randomly divided into four experimental groups. Control animals (Control group), rats treated with Epo (Epo group), rats receiving HgCl2 (Hg group), and animals treated with Epo and with HgCl2 (Epo-Hg group).
Control and Hg groups also received the vehicle for Epo (75 µL saline/100 g b.w., i.p.). Control and Epo groups also received the vehicle for HgCl2 (100 µL saline/100 g b.w., i.p.), 18 h before the experiments.
Different sets of experimental animals were used: (a) for biochemical determinations and preparation of plasma membranes from kidneys for immunoblotting studies and (b) for histopathological studies.
After HgCl2 or the vehicle injection, rats were individually housed in metabolic cages where water was provided ad libitum but animals were deprived of food, for time-lapse urine collection before the experiments. On the day of the experiments, fasting animals were anesthetized with sodium thiopental (70 mg/kg b.w., i.p.). Depending on the type of study performed, the collection and processing of tissue samples from kidney were different. After surgical procedures, the animals received euthanasia using anesthetic overdose and death was confirmed by bilateral thoracotomy.
Biochemical determinations
The biochemical determinations were performed in urine and plasma samples from the four experimental groups (Control group, n = 6; Epo group, n = 7; Hg group, n = 6; Epo-Hg group, n = 4). On the day of the experiments, urine volume was determined by gravimetry and urine samples were centrifuged (1,000 × g for 10 min) to remove cells and cell debris. Urine samples were used to determine alkaline phosphatase activity, osmolarity (Osmu), glucose, creatinine (Cru) and Oat5 protein abundance. Our group has postulated the urinary excretion of Oat5 (a dicarboxylate/organic anion transporter located in the apical membranes of proximal tubule S3 segment) as an early biomarker of proximal tubular damage in different experimental models of renal damage. 21 –23 Blood samples were obtained by cardiac puncture with heparinized syringes and were centrifuged at 1,000 × g (10 min) to obtain blood plasma. Plasma samples were used to measure levels of urea and creatinine (Crp) and osmolarity (Osmp). Plasma urea and creatinine levels, alkaline phosphatase activity in urine and creatinine and glucose concentrations in urine were determined with commercial kits (Wiener Laboratory, Rosario, Argentina). Osmolarity was measured by freezing point (Osmomat 300, Gonotec). Fractional excretion of osmolytes (FEosm, %) was calculated employing the following formulae: [(urine flow × Osmu)/(CrCl × Osmp)] × 100. Urine flow was expressed in mL/min/100 b.w. Creatinine clearance (CrCl) was calculated as [(Cru × urine flow)/Crp].
Total mercury determinations
Total mercury determination in kidney, plasma and urine samples (n = 4 for each experimental group) was performed using an atomic absorption Spectrophotometer Perkin Elmer AAnalyst 300 by cold vapor Flow Injection Analysis System (FIAS) 100–Perkin Elmer as previously described. 9,11,12,20 The Hg2+ was reduced by the treatment with stannous chloride to Hg0, and the Hg0 was measured with cold vapor atomic absorption (λ = 254 nm) by a Hg monitor using argon as gas. Daily, standards for Hg2+ were prepared from a dilution of the stock solution generated by solving HgCl2 p. a. in a nitric acid solution (1.354 g/L). Detection limit of the instrument: 1 µg/L.
Preparation of homogenates and total plasma membranes from kidneys
The preparation of renal homogenates and total renal plasma membranes (n = 4 for each experimental group) was performed by differential centrifugation as previously reported by our laboratory. 10 –12 The kidneys were removed, decapsulated, washed with saline solution (9 g/L) at 4°C, dried and weighed. Then, the kidneys were cut off, minced and placed in a Dounce homogenizer in a buffer containing: sucrose (250 mM), Tris-Hepes (5 mM) at pH 7.40, and phenylmethylsulphonyl fluoride (PMSF, 0.1 mg/mL). The suspension was homogenized with four gentle strokes with the loose-fitting pestle and further, with a motor-driven Teflon pestle (600 rpm/5 strokes) and spun down at 1,200 g (15 min, 4°C). The supernatant was aspirated and an aliquot was stored at −80°C (renal homogenates). The remainder homogenates were spun at 22,000 g (15 min, 4°C). The pellet is covered by a layer called “fluffy,” which is composed of crude plasma membranes. Crude plasma membranes were resuspended in 500 µL of buffered sucrose and different aliquots were stored (−80°C).
Electrophoresis and Western blotting
Electrophoresis and Western blotting were performed as previously described by Hazelhoff et al. 10 –12 In a mix of 1% 2-mercaptoethanol and 2% sodium dodecyl sulphate (SDS), urine samples and total plasma membranes were boiled for 3 min. Proteins were separated through 8.5% (for Oat1, Oat3, and Oat5) or 5% (for Mrp2) SDS-polyacrylamide gel electrophoresis (SDS-PAGE), and then electroblotted to a pure nitrocellulose membrane (NC membrane) (Trans-Blot® Transfer Medium, Bio Rad Laboratories, Hercules, CA, USA). Ponceau Red was employed to corroborate equal protein loading and transfer between lanes as previously reported. 10,20,21,24 In this connection, Romero-Calvo et al. 24 clearly demonstrated that routine quantification of Ponceau staining is validated as an alternative to actin blotting. A commercial rabbit polyclonal antibody against rat Oat1 or a non-commercial rabbit polyclonal antibody against rat Oat3 or a non-commercial rabbit polyclonal antibody against rat Oat5 or a commercial mouse polyclonal antibody against rat Mrp2 were used to incubate membranes overnight (4°C) as previously described. 11,12,21 The specificity of Oat3 and Oat5 antibodies has been described elsewhere. 11,21 –23 The detection of blots was performed with commercial chemiluminescent reagents (PierceTM ECL Western Blotting Substrate, IL, USA). As molecular mass indicators, Kaleidoscope Prestained Standards were employed (Bio Rad Laboratories, Hercules, CA, USA). The densitometric quantification of the chemiluminescent signals intensity was performed using the Gel-Pro Analyzer (Media Cybernetics, Silver Spring, MD, USA) software.
Histopathological studies
Hematoxylin-eosin staining was employed for the histopathological studies of kidneys as previously described. 10 –12 The kidneys (n = 4 for each experimental group) were perfused with NaIO4 (0.01 M), lysine (0.075 M), phosphate buffer (0.0375 M), paraformaldehyde (2%) at pH 6.20. The histological examinations were performed at random by a blinded observer, on 10 cortical high power fields at ×400. Tubular changes such as tubular dilatation/flattening, tubular degeneration/vacuolation and acute tubular necrosis were quantified as percent area sections reflecting a given histological damage. Modifications in affected tubules were graded as follows: 0 (<5%); 1 (5–33%); 2 (34–66%) and 3 (>66%) as previously described. 12
Statistical analysis
Statistical differences between groups were analyzed by the unpaired Student’s t-test or by the multiple comparisons with ANOVA plus Newman-Keuls. A p < 0.05 was considered statistically significant. Data were expressed as the mean ± standard error (SE).
Results
Kidney and body/kidney weight ratio, and plasma urea levels
Hg-treated rats presented an increase in the kidney weight and the ratio of kidney weight to body weight with respect to control rats (Table 1), as previously described. 10,20 The increase in kidney weight and kidney weight/body weight ratio was partially prevented by the previous administration of Epo. An increase in plasma urea levels was observed for Hg group with respect to Control group (Table 1) as well as in Epo-Hg group.
Kidney weight, kidney weight/body weight ratio and plasma urea levels.

Kidney weight, kidney weight/body weight ratio and plasma urea levels for the experimental groups: Control (n = 6), Hg treated (Hg, n = 6), Epo treated (Epo, n = 7) and Epo and Hg treated (Epo-Hg, n = 4). Results are expressed as mean ± SE.
a p < 0.01 versus Control.
b p < 0.01 versus Hg.
c p < 0.01 versus Epo.
d p < 0.01 versus Epo-Hg.
Biomarkers of tubular injury
Figure 1 shows the effect of Epo on the HgCl2-induced tubular injury. The urinary alkaline phosphatase activity, glucose urine concentration, urinary excretion of Organic anion transporter 5 (Oat5) and the fractional excretion of osmolytes were studied. The concentrations of the urinary parameters were related to urinary creatinine in order to correct variations in urine production as previously described. 10,12,21,23 HgCl2 induced an increase in the urine alkaline phosphatase activity, the glucose levels, the fractional excretion of osmolytes and the Oat5 abundance in urine. No significant (p < 0.05) alterations in the urinary parameters of tubular injury studied were observed in Epo group respect to Control one (Figure 1). The preadministration of Epo partially prevented the increase in the fractional excretion of osmolytes and the Oat5 protein abundance in urine but not in the urinary activity of alkaline phosphatase. Nevertheless, the increase in the urinary glucose levels induced by HgCl2 was completely prevented by the pretreatment with Epo.

Biomarkers of tubular injury. Alkaline phosphatase urinary activity (A), urinary glucose (B) Oat5 abundance in urine (C) and fractional excretion of osmolytes (D) in Control (n = 6), Hg treated (Hg, n = 6), Epo treated (Epo, n = 7) and Epo and Hg treated (Epo-Hg, n = 4) rats. Results are expressed as mean values ± SE. In Figure 1(C), the results are expressed as percentages and the mean of Control levels was set as 100%. ap < 0.05 versus Control; bp < 0.05 versus Hg; cp < 0.05 versus Epo; dp < 0.05 versus Epo-Hg.
Histopathological studies
Light microscopy examination of renal slides stained with hematoxylin/eosin is shown in Figure 2(A). No alterations in the tubular cell structure were observed in Control group or Epo group. Kidney sections from Hg group presented vacuolated cells, cellular detachment, disrupted brush border membranes, necrosis and interstitial inflammatory cell infiltration as previously described. 10,12,20,22,25 Epo prevented most of the HgCl2-induced tubular injury and only few alterations were observed, such as cellular detachment and disrupted brush border membranes as indicated by cortical tubular injury scores obtained (Figure 2(B)).

Histopathological studies in kidney tissue. (A) Representative micrographs of hematoxylin/eosin-stained sections of Control, Hg, Epo and Epo-Hg rat kidneys. Pictures are representative of samples obtained from four rats from each experimental group. In group Hg: vacuolated cells (yellow arrow), cellular detachment and disrupted brush border membranes were observed (arrow head), necrosis (star) and interstitial inflammatory cell infiltration (black arrow). Group Epo-Hg showed lesser microscopic damage than group Hg. Bars: 40 µm. (B) Tubular injury score. The scores range from 0 (completely normal hystology) to 3 (maximal and widespread injury). ap < 0.001versus Control; bp < 0.001 versus Hg; cp < 0.001 versus Epo; dp < 0.001 versus Epo-Hg.
Mercury levels
The administration of Epo before HgCl2 reduced both kidney accumulation (Figure 3(A)) and urinary excretion of mercury (Figure 3(B)). On the other hand, the content of mercury in plasma was significantly greater in Epo pretreated rats (Figure 3(C)). The content of mercury in kidney, urine, and plasma from rats not exposed to HgCl2 was below the detection limits of the method.

Mercury content in kidney tissue, urine and plasma. Content of mercury in kidney (expressed in µg of total kidney tissue) (A), urine excreted load of mercury (B) and plasma (C) mercury levels in Hg and Epo-Hg rats. Results are expressed as mean values ± SE, n = 4 for each experimental group. *p < 0.05.
Oat1, Oat3, and Mrp2 renal expression
The effect of the pretreatment with Epo on the HgCl2-variation of the renal expression of Oat1, Oat3, and Mrp2 was evaluated. The treatment with HgCl2 increased the renal abundance of Oat1 and the pretreatment with Epo in rats exposed to HgCl2 induced a greater increase in the renal expression of Oat1 compared with not pretreated rats (Figure 4). Oat3 abundance was increased by the HgCl2 administration in renal homogenates but not in renal plasma membranes. On the contrary, in rats pretreated with Epo before to the dose of HgCl2, an increase in the abundance of renal Oat3 was observed in both homogenates and renal plasma membranes (Figure 5). Finally, HgCl2 induced an increase in renal expression of Mrp2, but that increase in Mrp2 abundance induced by HgCl2 was smaller in Epo pretreated rats in comparison with the not pretreated ones (Figure 6).
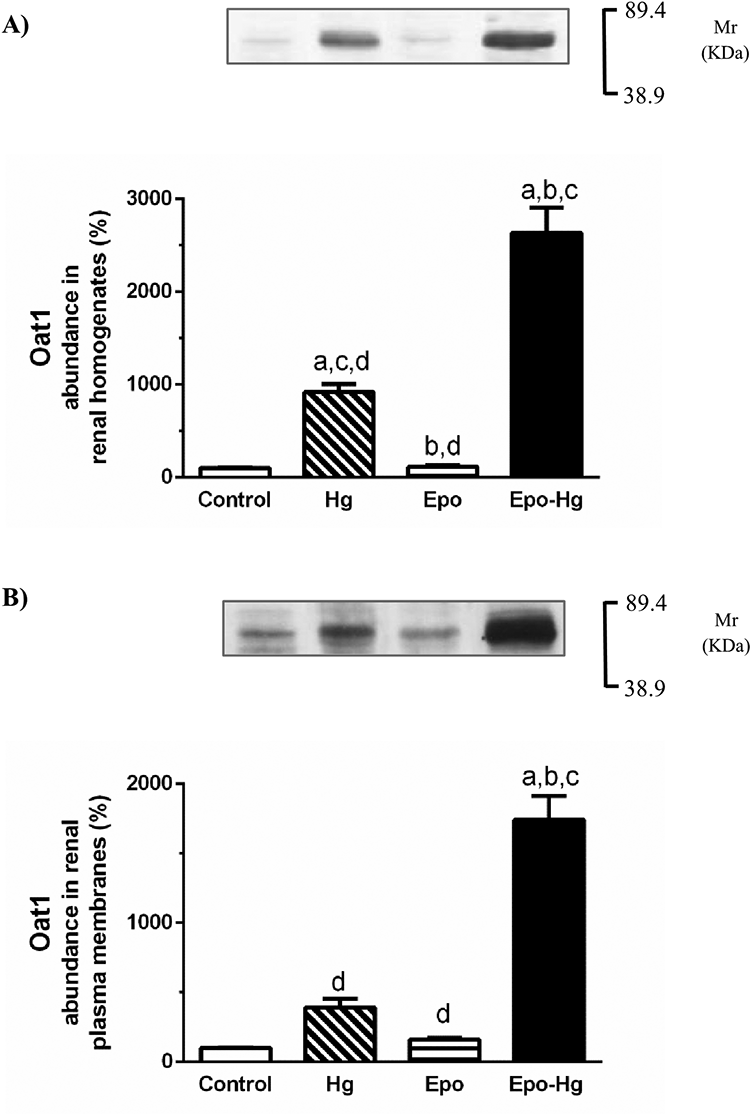
Oat1 renal protein expression. Western blotting for Oat1 in renal homogenates (A) and renal plasma membranes (B) from kidneys of Control, Hg, Epo and Epo-Hg groups. Proteins were separated by SDS-PAGE and blotted to nitrocellulose membranes. The results are expressed as percentages. The mean of Control levels was set as 100%. Results are expressed as mean values ± SE, n = 4 for each experimental group. (A): Control: 100 ± 3, Hg: 918 ± 86a,c,d, Epo: 113 ± 15b,d, Epo-Hg: 2636 ± 279a,b,c. (B): Control: 100 ± 4, Hg: 390 ± 63d, Epo: 160 ± 14d, Epo-Hg: 1742 ± 171a,b,c. ap < 0.01 versus Control; bp < 0.01 versus Hg; cp < 0.01 versus Epo; dp < 0.01 versus Epo-Hg. Kaleidoscope Prestained Standards of molecular mass (Molecular rule, Mr) corresponding to bovine serum albumin (89.4 kDa) and to carbonic anhydrase (38.9 kDa) are indicated in the right of the figure.

Oat3 renal protein expression. Western blotting for Oat3 in renal homogenates (A) and renal plasma membranes (B) from kidneys of Control, Hg, Epo and Epo-Hg groups. Proteins were separated by SDS-PAGE and blotted to nitrocellulose membranes. The results are expressed as percentages. The mean of Control levels was set as 100%. Results are expressed as mean values ± SE, n = 4 for each experimental group. (A): Control: 100 ± 4, Hg: 132 ± 14a,c,d, Epo: 93 ± 5b,d, Epo-Hg: 235 ± 10a,b,c. (B): Control: 100 ± 4, Hg: 84 ± 10c,d, Epo: 161 ± 16a,b,d, Epo-Hg: 217 ± 22a,b,c. ap < 0.001 versus Control; bp < 0.001 versus Hg; cp < 0.001 versus Epo; dp < 0.001 versus Epo-Hg. Kaleidoscope Prestained Standards of molecular mass (Molecular rule, Mr) corresponding to bovine serum albumin (89.4 kDa) and to carbonic anhydrase (38.9 kDa) are indicated in the right of the figure.

Mrp2 renal protein expression. Western blotting for Mrp2 in renal homogenates (A) and renal plasma membranes (B) from kidneys of Control, Hg, Epo and Epo-Hg groups. Proteins were separated by SDS-PAGE and blotted to nitrocellulose membranes. The results are expressed as percentages. The mean of Control levels was set as 100%. Results are expressed as mean values ± SE, n = 4 for each experimental group. (A): Control: 100 ± 2, Hg: 270 ± 25a,c,d, Epo: 142 ± 1b, Epo-Hg: 165 ± 11a,b. (B): Control: 100 ± 3, Hg: 333 ± 33a,c,d, Epo: 141 ± 12b,d, Epo-Hg: 207 ± 20a,b,c. ap < 0.001 versus Control; bp < 0.001 versus Hg; cp < 0.001 versus Epo; dp < 0.001 versus Epo-Hg. Kaleidoscope Prestained Standards of molecular mass (Molecular rule, Mr) corresponding to bovine serum myosin (206.4 kDa) and to β-galactosidase (127.5 kDa) are indicated in the right of the figure.
Discussion
The present study has evaluated the effects of Epo pretreatment on HgCl2-induced nephrotoxicity.
Epo administration 24 h before the HgCl2 injection decreased the rise in renal weight compared to animals treated only with HgCl2. It has been reported that the increase in kidney weight in HgCl2 rats is due to tissue water accumulation probably caused by the mercury inhibitory effects on renal aquaporins. 10,20,26 On the contrary, Epo has been known to up-regulate different renal aquaporins, 27 which may explain the Epo improvement on the mercury-induced renal edema.
In this work, it was demonstrated that Epo pretreatment ameliorated the HgCl2-induced tubular injury as assessed by histopathology and urinary biomarkers (such as glucose, osmolytes and Oat5).
When HgCl2 induces a severe cellular damage, a decrease in the glomerular filtration rate is observed and as a consequence, plasma levels of urea are increased. 3,9 –12 In our experimental design, a comparable increase in plasma urea levels was observed in Hg and in Epo-Hg group. In this regard, Kasap et al. 15 have described that Epo did not show significant protective effects on renal glomerular function, but did result in a decrease in tubular damage, apoptosis, and oxidative stress in cyclosporin-induced nephrotoxicity.
In the present study, the content of mercury in kidney and the urinary excretion of mercury were lower in the rats pretreated with Epo than in rats that only received HgCl2. In order to explain these results by focusing on the role of mercury transporters, the impact of Epo administration before HgCl2 on the renal expression of Oat1 and Oat3 and Mrp2 was evaluated. The renal protein expression of Oat1 was increased following HgCl2 treatment. A larger up-regulation of Oat1 protein expression in Epo-Hg group, both in homogenates and in renal plasma membranes than the observed in Hg group was detected. The increase of Oat1 abundance in renal homogenates would indicate an increase in Oat1 protein synthesis probably induced by a synergist effect of HgCl2 and Epo. One of the potential Hg-mediated increase mechanism in Oat1 expression could be via direct substrate stimulation, as described for other Oat1 substrates. 28 Relative levels of Oat3 protein expression in homogenates and in plasma membranes were higher in Epo-Hg rats than those observed for Hg group. Takeda et al. 29 have reported that hydrogen peroxide down-regulates organic anion transport mediated by Oat1 and Oat3. More recent studies, have demonstrated that renal Oat3 function and expression are decreased in concomitance with the increased oxidative stress in rats with type 1 diabetes mellitus and gentamicin-induced nephrotoxicity. 30,31 Thus, the up-regulation of both Oat1 and Oat3 induced by Epo in HgCl2-treated rats could be linked to the reduced redox status caused by Epo, as also described for other antioxidant substances in different models of nephrotoxicity. 32 –34 Finally, HgCl2 increased the renal expression of Mrp2 in plasma membranes and in homogenates. In Epo-Hg group the renal expression of Mrp2 was also increased but this increase was significantly lower than that seen in Hg group. In this sense, it has been described that Mrp2 could be up-regulated in response to oxidative stress. 35 Thus, the decrease production of reactive oxygen species induced by Epo 15 could explain its partial abolishment effects on the renal Mrp2 up-regulation mediated by HgCl2.
In the Epo-Hg group, the greater increase in the expression of both Oat1 and Oat3 following HgCl2 injection and the lower expression of Mrp2 than Hg group would somehow explain the differences in the content of mercury in the renal tissue and in the urinary excretion, as well as in mercury plasma levels. It has been described that Oat1 and Oat3 have the capacity to transport their substrates in a bidirectional way. 5,36 Therefore, Oat1 and Oat3 could be facilitating the release of mercuric ions into the blood, which would justify the greater plasma concentration of mercury in animals treated with Epo and the lower mercury content in kidney tissue. The lower Mrp2 expression in Epo-Hg rats would explain the lower urinary excretion of mercury and would increase the gradient of mercuric ions facilitating its expulsion from cells to blood mediated by Oat1 and Oat3 (see graphical scheme in Figure 7). Thus, Epo would be protecting proximal cells against mercury cytotoxicity, which is evidenced by the lesser tubular renal damage observed in Epo-Hg group compared with Hg group. The role of other tissues in detoxifying the higher plasma levels of mercury observed after Epo pretreatment remains to be evaluated.
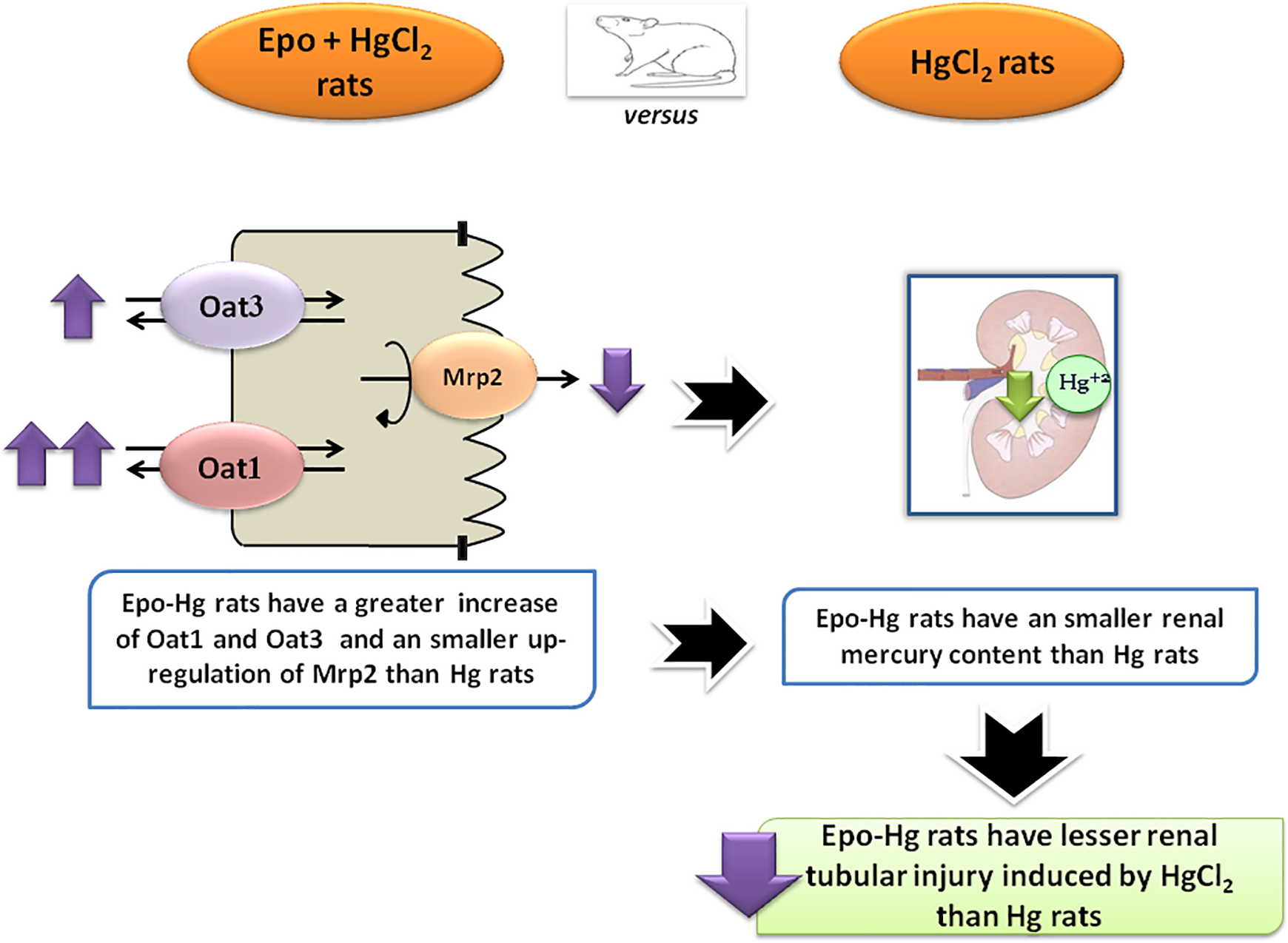
Scheme illustrating the mechanisms involved in the lesser renal tubular damage induced by mercury in Epo-Hg treated rats as compared with Hg treated rats.
Footnotes
Acknowledgments
The authors thank Professor N Anzai (Department of Pharmacology, Graduate School of Medicine, Chiba University, Japan) for kindly providing Oat3 and Oat5 specific antibodies; Professor A Chevalier (GIHON Laboratorios Químicos SRL, Facultad de Ciencias Exactas, Universidad Nacional de Mar del Plata, Argentina) for mercury determinations; and Mrs Alejandra Martínez (Área Morfología, Facultad de Ciencias Bioquímicas y Farmacéuticas, UNR) for collaborating with histopathological techniques. The authors also thank Wiener Lab Argentina for analytical kits.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) [PICT 2017: Grant Number 0936], Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) [PIP 2015-2017: Grant Number 0460], and Universidad Nacional de Rosario (UNR) [PIP 2016-2019, Grant Number BIO 479].
