Abstract
Aim:
The present study aimed to examine the effect of paricalcitol (PRC) and vitamin D3 (vit D3) on doxorubicin (DOX)-induced nephrotoxicity in rats.
Materials and Methods:
Forty-two Wistar rats were randomly categorized into six groups: control; 2) PRC(0.5 µg/kg) and 3) vit D3(5.000 IU/kg) administered for 14 days; 4) DOX, 18 mg/kg administered on the 12th, 13th and 14th days of the study; 5) PRC (0.5 µg/kg, +DOX(18 mg/kg); vit D3(5.000 IU)+DOX(18 mg/kg). On the 15th day of the experiment, 99mTc-DMSA uptake level and biochemical parameter in serum and tissue were assay.
Results:
Activities of 99mTechnetium-Dimercaptosuccinic Acid (99mTc-DMSA) were lower in groups receiving DOX and/or PRC+DOX, vit D3+DOX than in control groups. The 99mTc-DMSA level in the group PRC+DOX and vit D3+DOX were importantly higher than DOX group. DOX caused an important increase in blood urea nitrogen (BUN), creatinine, Tumor Necrosis Factor-α(TNF- α), interleukin-6(IL-6) and nitric oxide(NO) levels compared to control groups. However, PRC and vit D3 pretreatments lowered them. Uptake of 99mTc-DMSA level was higher in groups PRC+DOX than in vit D3+DOX group. Administration of PRC and vit D3 alone did not change alterations all of parameters.
Conclusion:
The results indicated that PRC administration protects kidney in DOX-induced nephrotoxic rats. In addition, PRC has a stronger nephroprotective effect than vit D3.
Introduction
Anthracyclines are broad-spectrum antineoplastics used for the treatment of many tumor types. One of the most commonly used anthracycline derivatives is doxorubicin (DOX), an antibiotic with anti-tumor activity as a result of its oxidative stress enhancing effect. The clinical efficacy of DOX in treatment of malignancy is limited by its dose-limiting side effects such as nephrotoxicity, hepatotoxicity, skin toxicity and cardiomyopathy. 1 Kidneys are among the most susceptible organs to toxic effects of antineoplastics.
Though the mechanism of the renal toxicity of doxorubicin remains unclear, some findings have suggested that inflammation and oxidative stress play a significant role in the pathogenesis of doxorubicin-induced kidney damage. 2,3 Doxorubicin triggers the generation of inflammatory mediators such as chemokines and cytokines in endothelial and tubular cells which contribute to the initiation of nephrotoxicity. 4 Doxorubicin may exacerbate the production of free radicals, membrane lipid peroxidation, protein oxidation which eventually leads to the cell apoptosis. 5 Thus, DOX decrease in glomerular number, vacuolation and accumulation of tubular protein. 6 Although there have been studies with various agents to reduce the nephrotoxic effect of DOX, no method has been found to completely prevent its nephrotoxic effects. 7 –9
Vitamin D is a fat-soluble vitamin and essentially plays a critical role in bone mineralization and mobilization by regulation of calcium and phosphorus balance. 10 Vitamin D deficiency has been associated with many diseases including cardiovascular diseases, chronic kidney disease, acute renal injury, acute lung injury and neurological diseases. 11,12 Also, vitamin D deficiency has been reported by 80% of prevalence in patients with chronic kidney disease. 13 It is shown in clinical and experimental studies that vitamin D could decrease oxidative stress and inflammation. 14 –16 Vitamin D and its receptor plays a crucial role in the modulation of inflammatory processes and immune function. Many studies both in vivo and in vitro demostrated that Vitamin D inhibits kidney tissue inflammation by reduced generation of proinflammatory cytokines such as interleukin 6 (IL-6), IL-8, IL-12, Tumor Necrosis Factor-α(TNF-α), interferon γ (IFN-γ). 15,17,18
Vitamin D shows its effect through vitamin D receptors. Vitamin D receptor has been expressed in many tissues including kidneys, lungs, heart, nervoussystem, intestine, liver, gonad, pancreas, parathyroidgland, bone, myocardium, monocytes, macrophages and dendritic cells of the immune system. 10,15,19 Paricalcitol (PRC) (19-nor-1,25-dihydroxy vitamin D2) is a synthetic D2 analog that binds to vitamin D receptor and prevents secretion of parathyroid hormone (PTH). It is used for the treatment and prevention of the secondary hyperparathyroidism in chronic renal failure. PRC inhibits the secretion of PTH as effectively as calcitriol. 19 In addition, the anti-inflammatory and anti-oxidative properties of PRC have been shown to improve the inflammatory and cardiovascular parameters in patients with the chronic renal failure and in patients with renal damage. 20,21 Recently a study showed that activation of vitamin D receptor by paricalcitol importantly reduced TNF-α and IL-6 levels in patient with chronic kidney diseases. 22 PRC exhibits renoprotective effect in drug-induced kidney injury animal model. 20,23 PRC treatmnet protects against acute kidney injury by blocking the renal tubular epithelial cell apoptosis and reducing caspase 3 activation in lipopolysaccharide-induced acute kidney injury mice model. 23 PRC exerts a direct cell-protective effect in ipopolysaccharide-induced renal proximal tubule cell injury by significantly inhibiting nuclear factor-kappaB signaling. 24
99mTechnetium-Dimercaptosuccinic Acid (99mTc-DMSA) static renal scintigraphy is a widely used nuclear imaging method for evaluation of functional renal parenchyma. Recent studies have shown that 99mTc-DMSA static renal scintigraphy is a sensitive and reliable test for the early detection of the kidney damage. 25,26
Inflammation and oxidative stress is a therapeutic target in drug induced nephrotoxicity. Vitaminin D and its analogs of paricalcitol have showed beneficial anti-inflammatory and antioxidant effects. The purpose of this study was to demonstrate the possible effects of PRC on the prevention of nephrotoxicity caused by DOX as revealed by biochemical tests including blood urea nitrogen (BUN), creatinine, TNF- α, IL-6 and nitric oxide (NO) and DMSA static renal scintigraphy. We also aimed to compare the preventive effects of PRC and vitamin D3 (vit D3).
Materials and methods
Animals
Male Wistar albino rats weighing 220 to 280 g were used. The rats were kept under the standard laboratory conditions (23 ± 2°C, 12 h dark, 12 h light cycle (light from 06.00 to 18.00) with 65% relative humidity). Throughout the experiment, rats were provided with standard laboratory chow and water ad libitum. The experimental procedures were performed in accordance with the guidelines set out by Tokat Gaziosmanpaşa University Animal Medical and Surgical Research Center (Tokat, Turkey), (HADYEK-2019/11).
Drugs and chemicals
DOX, PRC and vit D3 were purchased from a local pharmacy. DOX was used at the dose of 18 mg/kg, while PRC dose was 0.5 µg/kg and vit D3 dose was 5.000 IU/kg. The doses of all drugs were decided based on the previous publications. 27 –30 DOX was dissolved in saline. General anesthetics ketamine and xylazine were obtained from veterinary pharma (Turkey).
Experimental design
Rats were randomly separated into the following experimental groups (seven animals in each group): Group 1: Control (sham) group (CON), treated with intraperitoneal (i.p.) injection of 0.9% saline (5 ml/kg) for 14 days. Group 2: DOX (nephrotoxic) group (DOX), treated with DOX (cumulative dose of 18 mg/kg, i.p.) at an interval of 24 h on the last three days of the study (12th, 13th and 14th days). Group 3: PRC (substance control) group (PRC), treated with PRC (0.5 µg /kg, i.p. daily for 14 days). Group 4: Vit D3 (substance control) group (vit D3), treated with vit D3 (5.000 IU/kg, i.p. daily for 14 days. Group 5: PRC + DOX group (PRC + DOX), treated with PRC (0.5 µg /kg, i.p. daily for 14 days) plus DOX (18 mg/kg, i.p. for three days before the sacrifice). Group 6: Vit D3 + DOX group (vit D3 + DOX), treated with vit D3 (5.000 IU/kg, i.p. daily for 14 days) plus DOX (18 mg/kg, i.p. for three days before the sacrifice).
99mTc-DMSA renal scintigraphy and biochemical parameters were evaluated on the 15th day of the study.
Scintigraphic imaging and analysis
DMSA scintigraphy was performed in rats two hours after 37 Mbq 99mTc-DMSA injection via a tail vein under general anesthesia. Imaging was performed with a double-detector gamma camera (E CAM, Siemens, Germany). Radiopharmaceutical uptake of each kidney was calculated on 10-min static images with the help of areas of interest drawn around both kidneys. The two values were then summed to obtain a total count for two kidneys.
Blood sample analyses (BUN and creatinine)
Blood samples (5 mL each sample) were taken from the heart after scintigraphic imaging. They were kept at room temperature for 30 minutes. These samples were centrifuged for 5 min at 3000 rpm and stored at -80°C until analysis. Serum creatinine and blood urea nitrogen levels were measured using commercial enzyme-linked immunosorbent assay (ELISA) kits (Bechman by Bechman Coulter LX-2000, Brea, CA, USA).
Tissue preparation
Kidney tissue analyses (NO, TNF- α and IL-6)
Twenty-four hours after the administration of last dose, under anesthesia, rats were sacrificed by cervical decapitation. Then kidneys were removed. All tissue samples were washed with cold isotonic saline solution (0.9%). After that, wet tissue weights were determined. Tissues were then cut into the small pieces with a scalpel into a tube and homogenized in cold PBS (pH 7.4) with stainless steel balls. Tissues were centrifuged at 2500 rpm for 20 minutes and the supernatant was used. Tumor Necrosis Factor-α (TNF-α), nitric oxide (NO) and interleukin-6 (IL-6) levels were studied using quantitative sandwich enzyme immunoassay (ELISA) kits (Bioassay Technology Laboratory in accordance with manufacturer’s instructions. The results were normalized to wet tissue weights and expressed as ng/L for TNF-α and IL-6 and µmol/L for NO.
Statistical analyses
All experimental data were presented as mean ± SEM. Group means were compared using one-way analysis of variance (ANOVA) followed by the Tukey’s post hoc test for the multiple comparisons. All statistical analyses were performed using GraphPad Prism software (ver. 7.00; San Diego, CA, USA). The probability values less than 0.05 were considered statistically important.
Results
Biochemical assays in serum (BUN, creatinine)
As shown in Figure 1, BUN and creatinine levels were 19.46 ± 0.83 mg/dL and 0.35 ± 0.03 mg/dL in control group, 127 ± 14.83 mg/dL and 2.47 ± 0.30 mg/dL in DOX (nephrotoxic) group, 53.17 ± 6.26 mg/dL and 0.64 ± 0.06 mg/dL PRC+DOX, and 73.51 ± 5.51 mg/dL and 0.66 ± 0.06 mg/dL in vit D3 + DOX, respectively. Compared to control group, BUN level was higher in DOX, PRC+DOX, vit D3 + DOX groups by 652, 273 and 377% (p < 0.001), respectively, while plasma creatinine level was higher by 705 (p < 0.001), 182 and 188% (p < 0.01), respectively. PRC and vit D3 alone did not change BUN (89 and 103%, respectively) and creatinine levels (94 and 104%, respectively) compared to control group (p > 0.05) (Figure 1).

CON: control group, DOX: Doxorubicin (nephrotoxic) group, PRC: Paricalcitol substance control group, vit D3: vitamin D substance control group, PRC + DOX: Paricalcitol + doxorubicin group, vit D3 + DOX: vitamin D3 + doxorubicin group. Data were given as mean ± SEM. BUN and creatinine level in serum were higher in DOX, PRC+ DOX and vit D3 + DOX group compared to the CON group (**p < 0.01; ***p < 0.001). BUN and creatinine levels in serum were attenuated in PRC+ DOX and vit D3 + DOX groups compared to DOX group p < 0.01;p < 0.001). In addition, BUN level in serum was lower in PRC+ DOX group compared to vit D3 + DOX group (p < 0.05).
PRC + DOX and vit D3 + DOX groups significantly reduced the levels of creatinine (p < 0.001, respectively), and BUN (p < 0.001, p < 0.01 respectively), compared to the DOX nephrotoxic group. PRC+DOX group had importantly lower BUN level compared to vit D3 + DOX group (p < 0.05), (Figure 1).
Biochemical assays in tissue (TNF-α, IL-6, NO)
Wet tissue weight of kidney parameters were 0.486 ± 0.012 gr in control group, 0.477 ± 0.010 gr in DOX (nephrotoxic) group, 0,503 ± 0,015 gr in PRC, 0,493 ± 0,013 gr in vit D3, 0.487 ± 0.013 gr in PRC+DOX, 0.480 ± 0.010 gr in vit D3 + DOX, respectively. There was no change in wet tissue weight of kidney parameters compared to control group and each group (p > 0.05).
TNF-α, IL-6 and NO levels in kidney were 98.94 ± 6.88 ng/L/g, 4.70 ± 0.47 ng/L/g and 83.07 ± 6.15 mmol/L/g in the control group; 211.1 ± 6.97 ng/L/g, 8.57 ± 0.28 ng/L/g and 120.1 ± 7.28 mmol/L/g in DOX (nephrotoxic) group; 121.1 ± 5.47 ng/L/g, 5.34 ± 0.28 ng/L/g and 78.62 ± 4.44 mmol/L/g in PRC + DOX group; 134 ± 2.13 ng/L/g; 5.73 ± 0.13 ng/L/g and 81.46 ± 1.49 mmol/L/g in vit D3 + DOX group, respectively. DOX administration significantly increased TNF-α, IL-6 and NO levels 213 (p < 0.001), 182 (p < 0.001) and 144% (p < 0.01) respectively compared to control group. PRC + DOX and vit D3 + DOX groups had significantly higher TNF-α levels 122 (p < 0.05) and 135% (p < 0.001) respectively, but IL-6 113 and 121% ((p > 0.05), respectively and NO 94 and 98% (p > 0.05), respectively levels were not different from the control group. PRC alone and vit D3 alone injections did not change TNF-α 103 and 93% (p > 0.05), respectively, IL-6 89 and 101% (p > 0.05), respectively and NO levels 98 and 101% (p > 0.05), respectively compared to control group (Figure 2).

CON: control group, DOX: Doxorubicin (nephrotoxic) group, PRC: Paricalcitol substance control group, vit D3: vitamin D substance control group, PRC + DOX: Paricalcitol + doxorubicin group, vit D3 + DOX: vitamin D3 + doxorubicin group. Data were given as mean ± SEM. A) TNF-a levels in kidney were significantly higher in DOX, PRC + DOX and vit D3 + DOX groups compared to control group (*: p < 0.05; ***: p < 0.001). Pre-treatment groups of PRC + DOX and vit D3 + DOX groups significantly decreased the TNF-α levels in kidney compared to DOX group (p < 0.001). In addition, TNF-α levels in kidney was lower in PRC+ DOX group compared to vit D3 + DOX group (p < 0.05). IL-6 (B) and NO (C) levels in kidney were significantly higher in DOX groups compared to control group (**: p < 0.01; ***: p < 0.001). Pre-treatment groups of PRC + DOX and vit D3 + DOX groups had significantly lower TNF-α levels in kidney compared to DOX group (p < 0.001).
PRC + DOX and vit D3 + DOX groups had significantly lower TNF-α, IL-6 and NO levels (p < 0.001), compared to DOX (nephrotoxic) group. PRC + DOX group significantly reduced TNF-α level compared to vit D3 + DOX group (p < 0.05), (Figure 2).
Renal scintigraphy imaging assays
99mTc-DMSA uptake by kidney was 310235 ± 285 cpm in the control group, 72874 ± 798 cpm in DOX (nephrotoxic) group, 234714 ± 166 cpm in PRC + DOX group and 143661 ± 110 cpm in vit D3 + DOX group. DOX, PRC + DOX and vit D3 + DOX groups had significantly lower 99mTc-DMSA uptake level 23 (p < 0.001), 75 (p < 0.05), and 46% (p < 0.001), respectively compared to control group. PRC alone and vit D3 alone injections did not change 99mTc-DMSA uptake level 99 and 94% (p > 0.05), respectively) compared to control group (Figure 4). 99mTc-DMSA uptake was significantly higher in PRC + DOX and vit D3 + DOX groups than in DOX (nephrotoxic) group, (p < 0.001). PRC + DOX group had significantly higher 99mTc-DMSA uptake than vit D3 + DOX group (p < 0.001), (Figures 3 and 4).
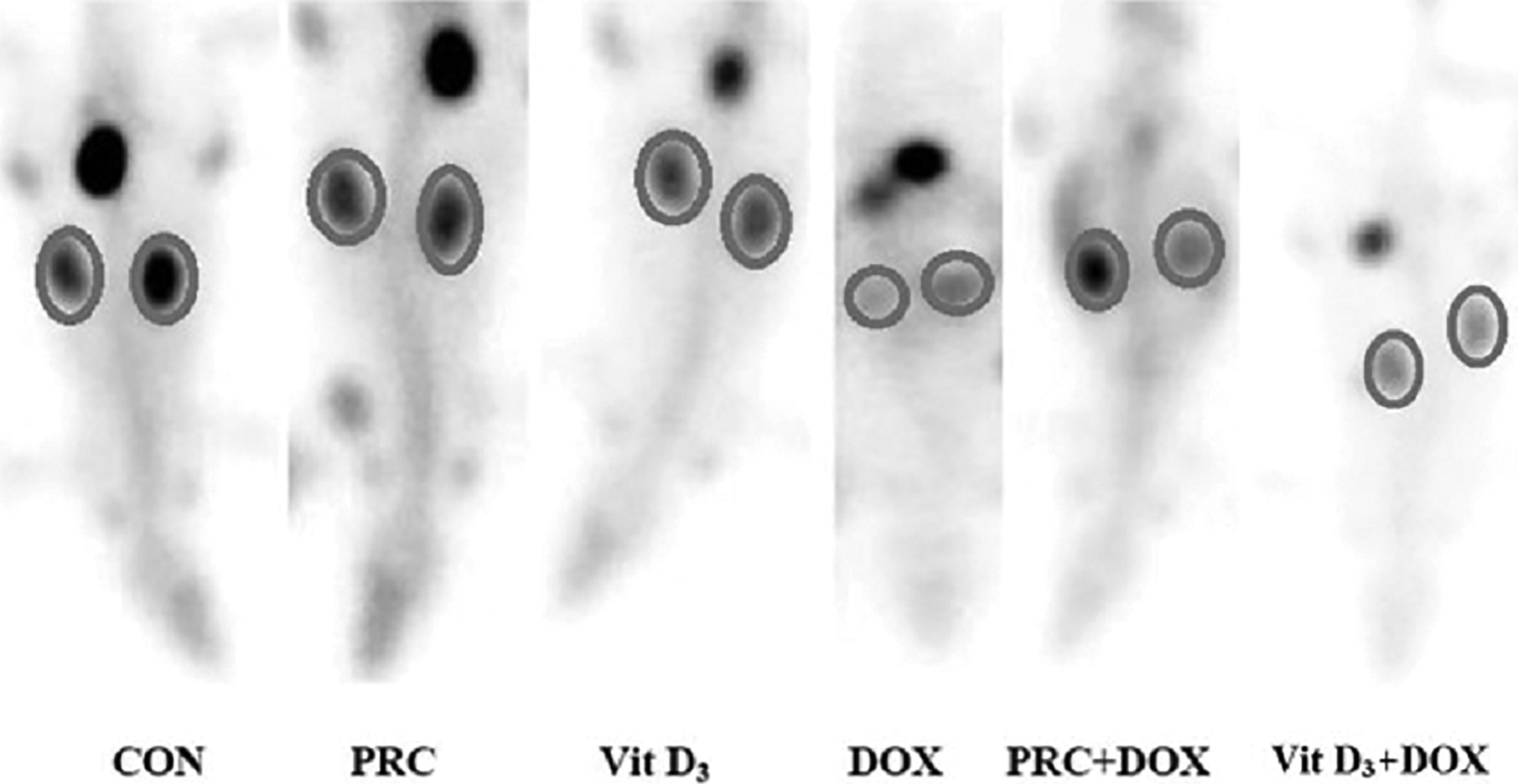
CON: control group, DOX: Doxorubicin (nephrotoxic) group, PRC: Paricalcitol substance control group, vit D3: vitamin D substance control group, PRC + DOX: Paricalcitol + doxorubicin group, vit D3 + DOX: vitamin D3 + doxorubicin group. It was shown that 99mTc-DMSA uptake level in the kidney was higher in the control group. On the other hand, in the DOX group, 99mTc-DMSA uptake level appeared to be low because of the developing damage. 99mTc-DMSA uptake was higher in PRC + DOX and vit D3 + DOX groups.

CON: control group, DOX: Doxorubicin (nephrotoxic) group, PRC: Paricalcitol substance control group, vit D3: vitamin D substance control group, PRC + DOX: Paricalcitol + doxorubicin group, vit D3 + DOX: vitamin D3 + doxorubicin group. Data were given as mean ± SEM. The DOX, PRC + DOX and vit D3 + DOX groups had significantly lower uptake of 99mTc- DMSA radiopharmaceutical compared to CON group (*p < 0.05; ***p < 0.001). On the other hand, pre-treatment groups of PRC + DOX and vit D3 + DOX significantly increased 99mTc-DMSA radiopharmaceutical uptake level compared to the DOX group (p < 0.001). In addition, 99mTc-DMSA radiopharmaceutical uptake level in kidney was higher in PRC+ DOX group than vit D3 + DOX group ( p < 0.001).
Discussion
In the present study, using both scintigraphic and biochemical methods, we demonstrated the renoprotective effect of PRC and vit D3 on DOX-induced-nephrotoxicity on an animal model. Both substances resulted in dramatical reductions in inflammatory cytokine (TNF-α and IL-6), NO, serum BUN and creatinine levels in renal tissue. Besides, they increased 99mTc-DMSA uptake levels in kidney. We showed that renal protective effect of PRC is stronger than that of vit D3. To our best knowledge, this research is the first to demonstrate that PRC has a renoprotective effect on DOX-induced nephrotoxicity.
DOX is a highly effective chemotherapeutic agent. Yet, its use is limited because of its nephrotoxicity and toxicities to other organs. 31 –33 It has been widely agreed that its nephrotoxic action generates free radicals and oxidative stress in cell. 34 –37 A number of studies demonstrated that DOX leads to direct oxidative injury to DNA and lipid peroxidation. 32,38 Oxidative stress destructs cell membrane. Blood urea nitrogen (BUN) and serum creatinine are released into blood by injured membranes of glomerular cells. 39 These biomarkers are known to be ideal biomarkers to determine renal glomerular function. 40,41 Previous studies demonstrated that DOX injection induces renal injury revealed by higher BUN and creatinine levels. 33,42 –46 Similarly, in the present study, DOX injection increased serum creatinine and BUN levels in rats, which is an indication of nephrotoxicity and a decrease in glomerular filtration. 47
DOX-induced renal toxicity involves an inflammatory process. 43,48 Previous studies showed that DOX raises the levels of several cytokines such as TNF-α and interleukins (IL-6), leading to severe inflammation and oxidative stress. 2,49,50 In the present study, we showed that DOX administration to rats importantly increased TNF-α and IL-6 activity in renal tissue.
Several researchers have recently showed that vit D3 and PRC are renoprotective in many experimental nephrotoxic models through their anti-fibrotic and anti-inflammatory actions. 14,51,52 A number of studies demonstrated that vit D3 inhibits the production of monocyte-derived cytokines such as TNF-α, interleukin-1 (IL-1) and IL-6. 51 Lee et al. 53 reported that vit D3 receptor agonist PRC protected kidney through significantly decreasing cytokine IL-6 and TNF-α in a mouse model of the ischemia/reperfusion-induced acute kidney injury. Park et al. 20 showed that in gentamicin-induced renal injury model of rats, levels of interleukin-1 beta (IL-1β), TNF-α and interferon gamma (INF γ) increased in kidney tissue and that these parameters were decreased after PRC treatment. In another study, it has been shown that PRC decreases chemokines, inducible nitric oxide synthase (iNOS), monocyte/macrophage infiltration and adhesion molecules. It has also an anti-inflammatory effect in cyclosporine induced nephrotoxicity model. In line with the previous studies. 20,51,53 , we demonstrated anti-inflammatory effects of PRC and vit D3.
Moreover, our data showed that DOX significantly enhanced nitrosative stress indicated by renal tissue NO level. The pretreatment with PRC and vit D3 reversed the DOX-induced nitrosative stress by reducing tissue NO level in kidney. 8 Many studies reported that NO production by up-regulation of the inducible form of NOS is one of the fundamental mechanisms of DOX-induced cytotoxicity. 54 –57 NO increases contribute to ROS and oxidative stress. 58 Oteki et al. 58 found that the level of NO increased in tubulointerstitial injury caused by DOX treatment. A number of studies confirmed the crucial role of vit D3 as a potent antioxidant. 59 –61 Additionally, many studies have displayed the role of vit D3 in the regulation of inducible form of NOS. 62,63 Garcion et al. 62 mentioned that vit D3 exerts its protecting effects through preventing free radical formation by the reactive species of nitric oxide and oxygen. Thanks to antioxidant effect of PRC and vit D3, Dox-induced oxidative stress could be reduced and kidney could be protected.
99mTc-DMSA scintigraphy is an imaging modality preferred in the evaluation of renal parenchymal cohesion. 99mTc-DMSA is absorbed by tubular absorption, binds to the plasma proteins, and is retained by renal cortex. 64 Studies have shown that DMSA uptake in the kidneys provides a quantitative assessment of renal function. 26,65 It is more sensitive than anatomic imaging methods and more effective in showing the early damage. In their study demonstrating the toxic effects of gentamycin on the kidney, Lora-Michiels et al. 66 reported that 99mTc-DMSA uptake indicated the damage before GFR decrease. The present study revealed that DOX injection decreased 99mTc-DMSA uptake level compared to the control. PRC and vit D3 treatment increased 99mTc-DMSA uptake level in the kidney compared to the DOX-groups.
Conclusion
The results of this study demonstrated that PRC and vit D3 efficiently protect the kidney tissue from the harm caused by DOX-induced nephrotoxicity. The anti-inflammatory and antioxidant activities are considered the fundamental mechanisms responsible for nephroprotective effect of PRC and vit D3. Our results also showed the usefulness of 99mTc-DMSA renal scintigraphy in the diagnosis of renal nephrotoxicity.
Footnotes
Author contributions
HA: Research concept and design. HA, FD, MD: Conducting experiments, data collecting, analysis and interpretation of data. HA, FD, MD: Preparation of article and revisions. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
