Abstract
A significant health risk exists within a section of health workers that are exposed to anaesthetic gas and vapours, found in the atmosphere of treatment or operating rooms. These compounds are classified as waste anaesthetic gases (WAG). The present study aimed at identifying alterations in hepatic and haematological parameters occurring as a result of chronic exposure to WAG potentially affecting the health of team members working in hospitals. Therefore, operating room operatives, vulnerable to long-standing WAG exposure, were recruited for this study. Sevoflurane anaesthesia metabolites (inorganic fluoride and hexafluoroisopropanol (HFIP)), haematological indices and liver toxicity markers (alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, gamma-glutamyl transferase and osteopontin) were measured. The collected results showed increased plasma inorganic fluoride, HFIP and liver toxicity markers, as well as disturbances in haematological parameters. In conclusion, exposure to halogenated inhalational anaesthetics, in general, and Sevoflurane, in particular, induces alterations in hepatic markers and haematological indices.
Introduction
A common procedure occurring in the operating room is the administration of anaesthesia. This is a necessary procedure routinely used for suppressing pain during surgery, by inducing a state of unconsciousness and temporary sensation loss in a patient. 1 Appropriate anaesthesia must be selected according to numerous factors such as availability, ease of administration, cost and acceptability of its effects. 2 During administration by inhalation, it has been reported that small quantities of vapour and waste anaesthetic gases (WAG) leak from the patient’s breathing apparatus into the atmosphere of the operating room. 3 Consequently, all surgical and support staff (anaesthetists, nurses, obstetricians and surgeons) are exposed to these volatile chemicals during anaesthetic administration. 4
Waste nitrous oxide and halogenated anaesthetics, including Sevoflurane, Enflurane, Halothane, Isoflurane and Desflurane, are frequently used during anaesthesia by inhalation. These are clear, colourless and volatile liquids at room temperature and normal pressure. 5
Anaesthesia by inhalation is achieved by using compounds that at room temperature and normal pressure appear clear, colourless and are highly volatile. Usually these are waste nitrous oxide and halogenated anaesthetics, such as Sevoflurane, Enflurane, Halothane, Isoflurane and Desflurane. 5
Inhalational anaesthetic volatiles enter the body through the respiratory system and are disseminated to the various tissues via the bloodstream. Consequently, healthcare workers risk developing occupational illnesses arising from chronic exposure to WAG. 6
Chronic exposure of inhalational anaesthetics is associated with reproductive problems, along with fatigue, headache, irritability and nausea. 7,8 In patients, inhalational anaesthetics toxicity includes raised levels of serum bilirubin and hepatic enzymes after repeated exposure. 9
The second revised edition of the Ministry of Health Formulary drug list (2014) cites Isoflurane and Sevoflurane as the most widely used inhalation anaesthetics.
In fact, within hepatic cells, Sevoflurane becomes very quickly transformed into the metabolites inorganic fluoride and hexafluoroisopropanol (HFIP) that can be detected in the bloodstream within minutes of admistration. 10 Exposure to fluoride induces hepatotoxicity and haematotoxicity in rats. 11 On the other hand, HFIP has a low binding affinity for liver proteins; therefore, it is rapidly changed to glucuronidate that is excreted in the urine. 12 To date, no investigations have been carried out to investigate the potential hepatotoxic and haemotoxic effects following chronic exposure to WAG on health hospital staff members. The current study objective was to identify alterations in hepatic markers and haematological indexes resulting from chronic exposure to WAG on hospital healthcare workers.
Subjects and methods
Study populations
Using a comparative cross-sectional protocol, this study took place between October 2018 and January 2019 in several hospitals throughout the Qassim region of Saudi Arabia. For this study, a total of 180 volunteers were recruited. They were divided into control and exposed groups. The control group was made of 60 healthy male individuals who were never susceptible to volatile anaesthetics vapours exposure (G1). The other 120 participants were all males of similar age to the control group and included operating room specialist employees, therefore, with significant long-term exposure to WAG. The latter cohort was subdivided into further six subgroups: surgeons (G2), surgeon’s assistants (SA) (G3), anaesthesiologists (G4), anaesthesiologist assistants (AA) (G5), nurses (G6) and workers (G7).
A series of parameters were recorded from each participant, including the identification of the work environment, working occasions, work-shifts and of name of anaesthetic gases utilised routinely. In addition, the participants provided information about their smoking habits, clinical history and body mass index, estimated as weight (kg)/height (m2). Consent was obtained before initiation of the present study. Study exclusion criteria included current smokers, hospital staff with less than 1 year of employment and any subjects receiving treatment for ongoing illnesses, since they may present with a potentially skewed immune status. Study approval was acquired from the School College Morals Advisory Group and the Research Ethics Committee at the Ministry of Health for Saudi Arabia.
Collection of blood sample
Each participant was asked to fast overnight prior to the collection of blood sample. This was done in the morning, between 8:00 and 9:00 am, by extracting a 10 mL specimen from their hand veins. Each blood sample was divided into three parts. The first fraction was collected into a silicone-coated tube and centrifuged (1000 g, 15 min, 4°C) for isolating the serum. The second fraction was either transferred to a heparin-coated or ethylenediaminetetraacetic acid (EDTA)-coated tubes for eventual isolation of plasma. The third part was utilised for whole blood examination. All isolated plasma/serum samples were placed at −20°C.
Plasma fluoride levels
Fluoride plasma levels were estimated according to the protocol of Levine et al. 13 Briefly, fluoride was assessed using an ion-selective electrode fitted on an Orion 901 Ion analyser (Thermo Fisher Scientific, Chelmsford, Massachusetts, USA), equipped with fluoride-specific and combination pH electrodes. To appropriately calculate the existing plasma levels, the production of a fluoride standard curve was required, so the assay was linear over a range of 1–48 µM fluoride, with correlation coefficients equal to 0.999. Quantification limit was 1 µM fluoride. All samples were tested in duplicate.
Plasma HFIP levels
Plasma HFIP levels were estimated according to the protocol of Kharasch et al., 14 using headspace gas chromatography-flame ionization detector (GC-FID). Briefly, total HFIP plasma levels were harvested after initial conversion of HFIP conjugates to free alcohols, following incubation of each plasma sample aliquots with glucuronidase/sulphates (2000U Type HI, Sigma, St. Louis, Missouri, USA) for 15 h at 37°C. Significant reduction of pH from 5 to 2 was a necessary step and was achieved by injection of 200 µL of 3.5 M phosphoric acid solution through septum, after the samples reached room temperature. Then, each sample was vigorously mixed and stacked into the headspace auto-sampler of a HS40 headspace system (PerkinElmer, Melbourne, Florida, USA). The materials were resolved over a capillary column (30 m × 0.53 mm × 3.0 µm film thickness) containing RESTEK RTX 1701 (Restek, Bellefonte, Pennsylvania, USA). Plasma HFIP levels were ultimately estimated by extrapolation against standard curves (based on areas under peak curves) generated with an internal standard (2,2,3,3,3-pentafluoro-1-propanol). All samples were assayed in triplicate.
Complete blood counts
An automated CELLTAC MEK-6410 K haematology cell counter (Nihon Kohden Corporation, Tokyo, Japan) was used to measure the following blood parameters: haematocrit (HCT), haemoglobin (Hgb) level, mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), mean corpuscular volume (MCV), platelet (PLT) count, red blood cell (RBC) count and white blood cell (WBC) count.
Liver function tests
The activities of aspartate aminotransferase (AST), alanine aminotransferase (ALT) 15 and alkaline phosphatase (ALP) were determined using Hitachi 912 spectrophotometer (Roche Diagnostics Co., Mannheim, Germany). 16 Serum gamma-glutamyl transferase (GGT) activity was analysed according to Beleta and Gella. 17 Total serum protein and albumin were determined following Kingsley protocol. 18 Total serum protein and albumin concentrations were measured according to Henry 18 and Doumas et al. 19 experimental guidelines.
Detection of serum osteopontin
Osteopontin (OPN) was detected utilising human OPN enzyme-linked immunosorbent assay (ELISA) kit (Catalogue number: ab100618, Abcam, Chuo-ku, Tokyo, Japan). Briefly, the sample was diluted 20 times in Assay Diluent. For the execution of an appropriate standard curve, recombinant human OPN was used at increasing concentrations, spanning between 0 ng/mL and 18 ng/mL limits. In an anti-human OPN antibody pre-coated microplate, 100 µL of diluted serum sample, or standard, was added. After sealing, the microplate was incubated overnight at 4°C on a shaker. Each well was washed four times with Wash Buffer. A Biotin conjugated secondary antibody (100 μL) was added and incubated for 1 h at room temperature on a shaker. After a wash step conducted as described above, the wells received 100 µL diluted horseradish peroxidase (HRP)-streptavidin conjugate and incubated for 45 min at room temperature on a shaker. A further five times washing step was followed by the addition of 100 µL tetramethylbenzidine and 30 min incubation in the dark with shaking. Finally, 50 µL stop solution (2 M sulphuric acid) was added to each well. Immediately, the developed colour intensity was revealed on a microplate reader at 450 nm. All samples were assayed in triplicate.
Statistical analysis
Data were analysed utilising SPSS v.21.0 software (SPSS, Inc., Chicago, Illinois, USA). A one-way analysis of variance (ANOVA) was used to discern significant changes among the exposed subgroups, compared with the control. Following the ANOVA statistical analysis, a further Dunnett comparison test was done to scrutinise differences between the studied groups.
Results
A study of this kind has meaning when the participants are chosen carefully sharing equivalent characteristics between cohorts. For this reason, the closest socio-demographic match was ensured within the control (G1) and anaesthetic gases-exposed groups (G2–G7). Therefore, a statistical analysis of participants’ age, gender, marital status and employment years was performed: no significant difference existed between all groups as shown in Table 1.
Socio-demographic characteristics and occupational history of the study groups.a
G1: control group; G2: surgeon assistant group; G3: surgeon group; G4: anaesthesia specialist group; G5: anaesthesia assistant group; G6: nurses group; G7: workers group; ANOVA: analysis of variance; SD: standard deviation.
a Values expressed as means ± SD. One-way ANOVA/Dunnett test. Value significantly different from corresponding control at p < 0.001.
Plasma fluoride and HFIP levels
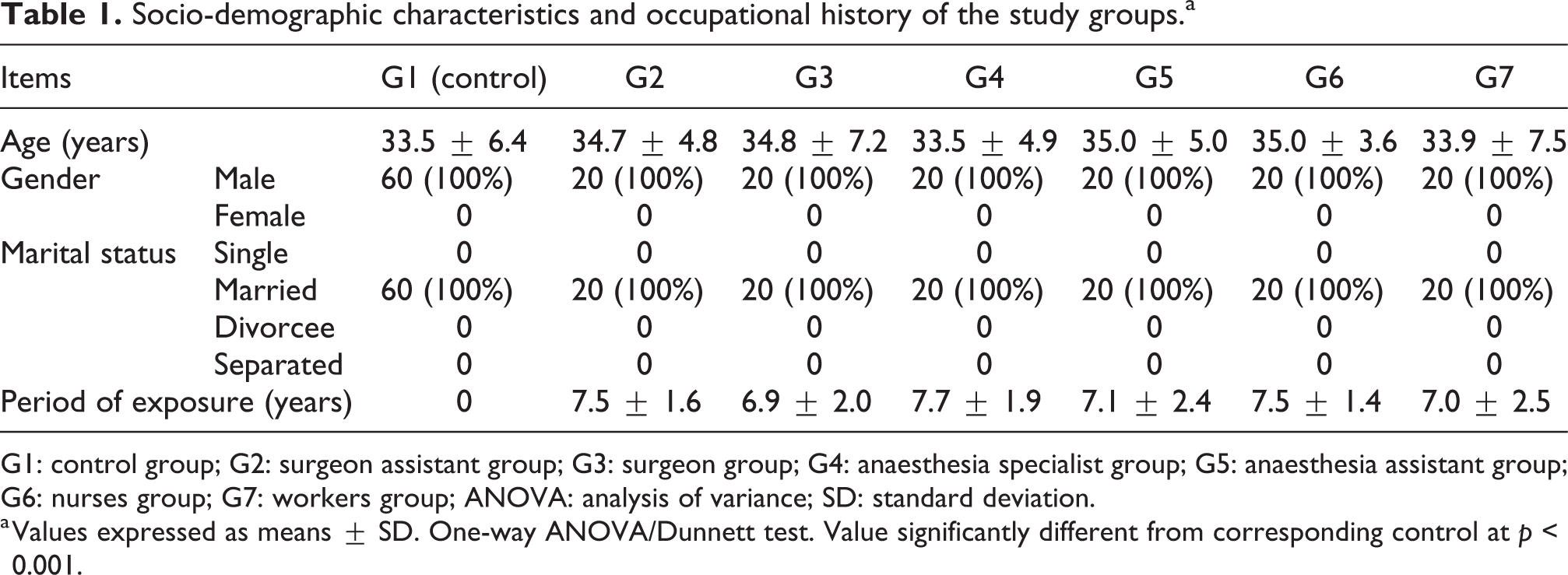
Increases in plasma fluoride levels were detected in all exposed groups (G2–G7), when compared with the control group. However, the increase was found to be significant only in the anaesthesiologists and AA (0.44 (±0.02) and 0.48 (±0.01), respectively, vs. 0.35 (±0.01) µM). In addition, these values were significantly greater than those seen in the blood of the surgeons, SA, nurses and workers (Figure 1(a)).
Changes in (a) plasma fluoride and (b) HFIP concentration levels in different groups under study. The significance of difference was analysed by one-way ANOVA and Dunnett test (compare all vs. control group) using computer program. Values are expressed as mean ± SEM. One-way ANOVA was significant at p < 0.05. Dunnett test was significant from corresponding control group value at *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001. HFIP: hexafluoroisopropanol; ANOVA: analysis of variance; SEM: standard error of the mean.
Analysis of HFIP plasma levels illustrated that these were significantly elevated in all the exposed groups (G2–G7), when compared with the control group (Figure 1(b)). In both the anaesthesiologists and AA, the levels were significantly increased as compared with the ones detected in the surgeons, SA, nurses and workers (Figure 1(b)). This study did not show significant plasma HFIP level changes occurring within these latter four groups.
Changes in the complete blood pictures
Full blood count analysis disclosed some changes occurring between different test groups. These findings are illustrated in Table 2. When compared to the control group, the exposed groups displayed a significant reduction in the blood Hgb percentage (%Hgb), RBCs number, HCT percentage (%HCT), MCV, MCHC, MCH and blood PLTs number. Furthermore, anaesthesia specialists, nurses and workers groups suffered a significant reduction in %Hgb and %HCT, when compared with surgeons, SA and AA groups. In addition, RBC numbers were significantly lower in surgeons and AA groups, than in SA, anaesthesia specialists, nurses and workers groups. Likewise, MCV levels were considerably reduced in workers than in surgeons, SA, AA, anaesthesia specialists and nurses groups. A MCH and MCHC level drop was detectable in anaesthesia specialists, when compared with surgeon, SA, AA, nurses and workers groups. Finally, a lower PLT count was found in surgeons and anaesthesia specialists than in SA, AA, nurses and workers groups. To complete the picture, this study did not find any statistically significant change in complete blood count (CBC) parameters between the surgeon, SA, and AA groups. Likewise, no statistically significant differences appeared to exist between anaesthesia specialists, nurses and workers groups.
Changes in Hgb, RBCs, HCT, MCV, MCH, MCHC and the blood PLTs (103 × µL) in different studied groups.a

G1: control group; G2: surgeon assistant group; G3: surgeon group; G4: anaesthesia specialist group; G5: anaesthesia assistant group; G6: nurses group; G7: workers group; Hgb: haemoglobin; RBCs: red blood cells; HTC: haematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; PLT: platelet; ANOVA: analysis of variance; SEM: standard error of the mean.
a The significance of difference was analysed by one-way ANOVA and Dunnett test (compare all vs. control group) using computer program. Values are expressed as mean ± SEM. One-way ANOVA was significant at p < 0.05.
b p < 0.05: Dunnett test was significant from corresponding control group value.
c p < 0.01: Dunnett test was significant from corresponding control group value.
d p < 0.001: Dunnett test was significant from corresponding control group value.
e p < 0.0001: Dunnett test was significant from corresponding control group value.
Changes in WBCs
Different study groups experienced alterations in WBC numbers and lymphocytes, monocytes (MO) and granulocytes (GR) percentage values. These variations are illustrated in Table 3. A greater increase was detected in WBCs number and in lymphocytes and GR percentages within the exposed groups, than within the control. Conversely, MO percentage showed marked decrease in the exposed group, compared to control individuals.
Changes in total WBCs, lymphocyte, MO and GR in different studied groups.a

G1: control group; G2: surgeon assistant group; G3: surgeon group; G4: anaesthesia specialist group; G5: anaesthesia assistant group; G6: nurses group; G7: workers group; WBCs: white blood cells; MO: monocytes; GR: granulocytes; ANOVA: analysis of variance; SEM: standard error of the mean.
a The significance of difference was analysed by one-way ANOVA and Dunnett test (compare all vs. control group) using computer program. Values are expressed as means ± SEM. One-way ANOVA was significant at p < 0.05.
b p < 0.01: Dunnett test was significant from corresponding control group value.
c p < 0.05: Dunnett test was significant from corresponding control group value.
Changes in liver function tests
Liver function assessment was performed by analysing ALT concentrations, as shown in Figure 2(a). Significant serum ALT concentration growth was shown for all exposed groups, when compared with the control. Furthermore, greater serum ALT levels increase was detected in the surgeons and AA groups, than in SA, anaesthesia specialists, nurses and workers groups. On the other hand, no statistically significant change was detected in the serum ALT levels between SA, anaesthesia specialists, nurses and workers groups.

Changes in the
The analysis of serum AST concentrations within all groups is displayed in Figure 2(b). Significant serum AST levels elevation was detected in the surgeons, anaesthesia specialists, AA and nurses groups, when compared with the control. Also for this parameter, greater serum AST concentration was detected in the surgeons, anaesthesia specialists, AA and nurses groups, than in SA and workers group. In addition, no statistically significant serum AST change existed between the SA and workers groups, when compared to the control.
Serum ALP analysis is illustrated in Figure 2(c) for all groups. The surgeons, anaesthesia specialists and AA groups experienced significant increase in serum ALP levels when compared with the control group. Interestingly, greater serum ALP concentration was detected in the surgeons, anaesthesia specialists and AA groups, than in nurses and workers groups. No statistically significant serum ALP levels change was detected in SA, nurses and workers groups, when compared to the control.
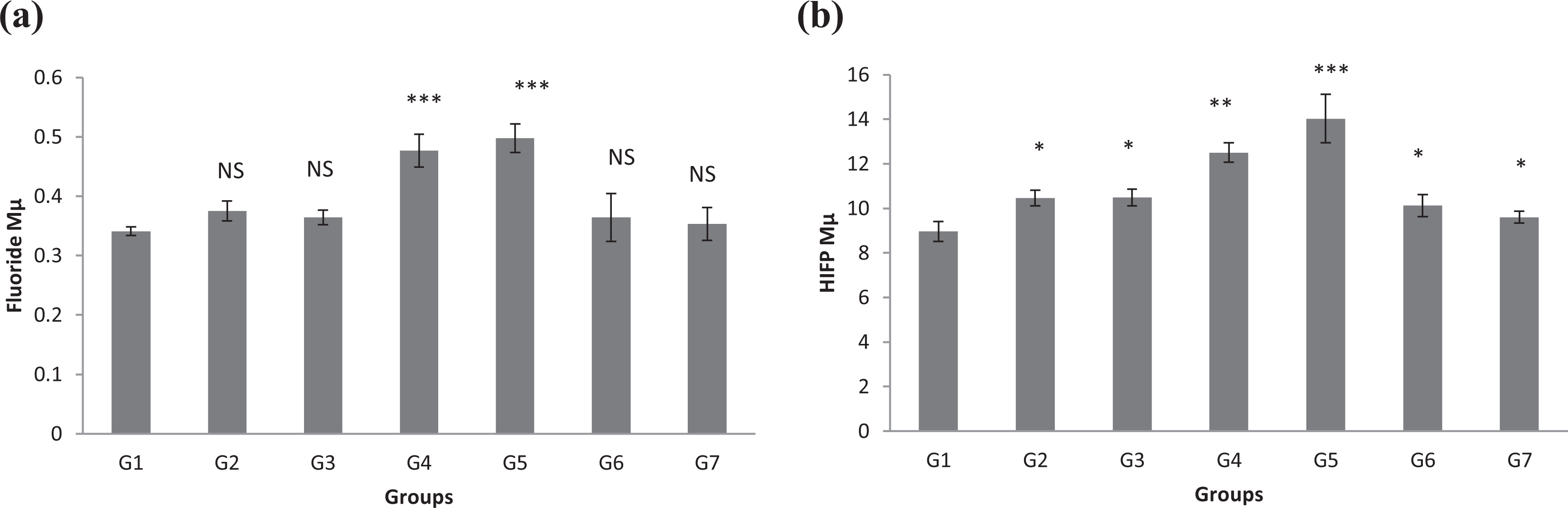
Serum OPN and GGT variations analysis between groups are illustrated in Figure 3(a) and (b). The present study demonstrated that the levels of serum OPN and GGT were significantly elevated in surgeons, anaesthesia specialists and AA groups, when compared with the control group. In addition, significant serum OPN and GGT levels increase was detected in surgeons, anaesthesia specialists and AA groups when compared with the SA, nurses and workers groups. On the other hand, this study did not show significant serum OPN and GGT concentrations change within the SA, nurses and workers groups (Figure 3(a) and (b)).
Changes in the (a) serum OPN and (b) serum GGT levels in different studied groups. The significance of change was analysed by one-way ANOVA and Dunnett test (compare all vs. control group) using computer program. Values are expressed as mean ± SEM. One-way ANOVA was significant at p < 0.05. Dunnett test was significant from corresponding control group value at *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001. G1: control group; G2: surgeon assistants group; G3: surgeon group; G4: anaesthesia specialist group; G5: anaesthesia assistants group; G6: nurses group; G7: workers group. OPN: osteopontin; GGT: gamma-glutamyl transferase; ANOVA: analysis of variance. SEM: standard error of the mean.
Among study groups, the total serum protein concentration differences are displayed in Figure 4(a). Significant total serum protein levels reduction was shown to occur in surgeons, anaesthesia specialists and AA groups, compared to the control group. No statistically significant total serum protein concentration change existed between SA, AA, nurses, workers and control groups. However, a meaningful total serum protein levels decrease was witnessed in surgeons, anaesthesia specialists and AA groups, when compared with SA, nurses and workers groups.

Changes in the (a) serum total protein and (b) serum albumin levels in different studied groups. The significance of difference was analysed by one-way ANOVA and Dunnett test (compare all vs. control group) using computer program. Values are expressed as mean ± SEM. One-way ANOVA was significant at p < 0.05. Dunnett test was significant from corresponding control group value at *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001. G1: control group; G2: surgeon assistant group; G3: surgeon group; G4: anaesthesia specialist group; G5: anaesthesia assistant group; G6: nurses group; G7: workers group; ANOVA: analysis of variance: SEM: standard error of the mean.
Serum albumin concentrations changes are illustrated in Figure 4(b). Significant serum albumin levels decrease was experienced by the anaesthesia specialists, AA and nurses groups, compared to the control group. Furthermore, a meaningful serum albumin concentration reduction was detected in the anaesthesia specialists group, versus the surgeons, AA, nurses and workers groups. Finally, no significant serum albumin levels change was detected between surgeons, SA and workers, when compared with the control group.
Discussion
During the normal process of administering anaesthesia to patients, a mixture of nitrous oxide and halogenated anaesthetics (Sevoflurane, Desflurane, Enflurane, Halothane or Isoflurane) is used to carefully establish the required treatment levels. Anaesthesia care providers (ACPs), such as anaesthesiologists, AA and nurse anaesthetists, are responsible for the inhalational anaesthetics administration using face masks, laryngeal airway masks or tracheal tube connected to an anaesthesia machine. Because of their highly volatile nature, inhalational anaesthetics escape into surrounding environment during administration. These are defined as WAG. Thousands of ACPs are at risk of exposure to WAG and to the subsequent toxic effects development. 6
It is noteworthy that the risks from inhaling anaesthetic gases have long been recognised. Therefore, a continuous effort is being made to improve contamination control procedures, particularly in medicinal services, such as pharmacies. Nevertheless, the issue of work-related exposure to inhalational anaesthetics has not been completely resolved. 6,20
Interestingly, controversy exists on the harmful effects to health induced by long-term exposure of anaesthetic room personnel to inhalational anaesthetics. 21
This report describes a study implemented for analysing WAG exposure in hospital health personnel in the Qassim region. It is worth clarifying that, in these hospitals, Sevoflurane represents the inhalation anaesthetics compound of preference, because it appears to be less harmful to the heart and airways. All study groups were matched in socio-demographic data with no statistically significant differences existing between non-exposure control (G1) and exposure groups (G2–G7), with regard to age, gender, marital status, illness history and length of employment.
The rate of inhalational anaesthetics metabolism is calculated by the fluoride level detection in serum and urine, causing toxic effects. 22 Blood fluoride increases during short anaesthetic and longer ICU term of utilisation: Sevoflurane shows significantly higher concentrations than Isoflurane and Desflurane, due to its prominent metabolism and fluoride content. 13,23
The current study showed significant plasma fluoride levels elevation in the exposed groups (G2–G7), when compared with the control group, and in both anaesthesia specialists and AA groups when compared with surgeons, SA, nurses and workers groups.
Fluoride induces a reduction in MCHC, MCH, Hgb, MCV and HCT counts. 24 Decreased Hgb synthesis, reduced RBC manufacturing and their increased destruction are observed in the bone marrow, resulting in the onset of anaemia. 25 These events may provide a possible explanation for the effects induced by fluoride.
The PLT counts decline is due to either bone marrow suppression or trapping of PLTs in the spleen. 26 The present study confirmed a significant %Hgb, RBC, %HCT, MCV, MCH and MCHC decline within the exposed groups, when compared to the control group. These findings suggest that the haematopoietic system is toxicologically sensitive to inhalation anaesthetics, which could provoke anaemia. An analysis of anaesthesia specialist personnel disclosed that they are subjected to a toxic working environment, since CBC parameters were affected by the continuous exposure to toxic levels of inhalation anaesthetics. 27 These observations harmonise with the present study findings.
Furthermore, this study is consistent with investigations reporting that significant alterations in all blood parameters arose from persistent exposure to high halogenated volatile anaesthetics concentrations in the workplace. 24,25 In particular, exposure to high halogenated anaesthetics concentrations triggered a significant reduction in %Hb, RBC and PLTs, 27 consistent with further findings demonstrating haematological alterations following exposure in the workplace to the same compounds. 28
Hepatic toxicity pathogenesis, related to inhalational anaesthetics, includes reduction in liver blood flow 29,30 and production of toxic metabolites. 22 As consequence of liver blood flow and oxygenation reductions, the hepatic function is altered. 23,29,30 In addition, cardiac output reduction is induced by inhalational anaesthetics and results in liver blood flow reduction and subsequent liver oxygen supply alteration. 29
It should be mentioned that surgical patients anaesthetised with Sevoflurane do not show significant hepatotoxic consequences in a small number of studies. 31,32 However, elevated liver function markers were recorded after surgery with Sevoflurane anaesthesia, 33 especially in morbidly obese patients. 34
In ACPs, Sevoflurane safety should be monitored, because long-term exposure could increase the likelihood for liver function impairment occurrence. This hepatic damage may be considered unacceptable to some physicians. The present study showed significant increase in serum ALT, AST, ALP, GGT and OPN levels when compared with the control group. Conversely, a significant serum albumin and total protein contents decrease was measured in exposed groups, when compared to the control group. These conclusions conform to findings that illustrated hepatotoxicity induction by halogenated anaesthetics in experimental animal models. 33
A relationship exists between anaesthetic gases exposure and hepatic markers impairment. 35,36 In particular, significant AST and ALT upsurge was triggered by anaesthetic gases exposure. 37 Moreover, it was discovered that Sevoflurane is metabolised into inorganic fluoride by the liver microsomal cytochrome p450 enzyme machinery, inducing hepatic toxicity. 14 These findings confirm the present study observations. Fluoride causes deposit of fat in the liver, 38 affects liver protein expression 39 and necrosis. 40 In addition, fluoride induces vacuolisation in liver cells, increased liver weight, 41 elevated oxidative stress and damage, 42 necrosis and impaired liver function. 40 However, it was interesting that in a study significant ALT and AST levels increase was not detected after Sevoflurane administration. 43 These results explanation lays in the Sevoflurane metabolism that does not give rise to the formation of trifluoroacetic acid. Hence, Sevoflurane hepatotoxic potential is considered to be low. 33,44
The overall assessment of the study data suggests that chronic exposure to WAG induces significant elevation in the mean values of blood liver markers (AST, ALT, ALP, GGT and OPN), together with a significant reduction in MO and increase in WBCs, GR and lymphocytes. At present, these results are considerably significant in light of the following: (a) no participants had confounding factors that could impair their hepatic function, and (b) no significant differences existed in participants’ age or risk duration for all exposed groups. The severity of toxicity effects depends on many factors, including the utilised anaesthetic type, its concentration, exposure time, the operating theatre environment, the target organ susceptibility and individuals’ genetic features. 45 Hence, variations are observed within different groups, which may or may not experience an effect. The present study findings motivate to explore further our fundamental proposition: more analysis is necessary for the confirmation of this investigation outcome.
Conclusions
The current study provides evidence that exposure to inhalation anaesthetics in general and Sevoflurane in particular induces alteration of haematological indices, including decreased RBC count, Hgb level, HCT value and MCHC. In addition, exposure is accompanied with alteration in liver markers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
