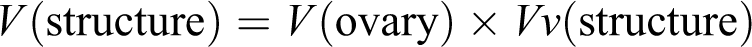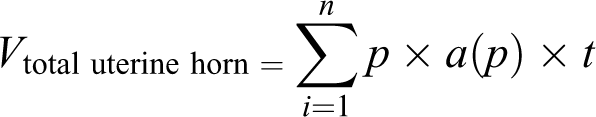Abstract
Doxorubicin (DOX) is a widely used chemotherapeutic agent with demonstrated reproductive toxicity. This study sought to determine the DOX-induced toxicity in the ovary and uterus and the preventive effects of quercetin (QCT) and vitamin E (Vit.E). Female rats were divided into six groups as follows: control, QCT (20 mg/kg), Vit.E (200 mg/kg), DOX (accumulative 15 mg/kg), DOX/QCT, and DOX/Vit.E. After 3 weeks, the toxicity of DOX in ovarian and uterine tissues and the potential palliative effects of QCT and Vit.E were evaluated by histopathological–stereological methods. The findings indicate a dramatic decline in the number of ovarian follicles (p < 0.001), ovarian and its associated structures volume, the volume of the uterus, its layers, and related structures (p < 0.05). Coadministration of QCT and Vit.E with DOX-treated rats demonstrated an alleviative effect on most of the studied parameters. Nevertheless, few adverse effects were recognized concerning these antioxidants administration (p < 0.05). In conclusion, the findings of this study support the protective role of these dietary supplements in the prevention of DOX-induced toxicity in uterine and ovarian tissues.
Introduction
Although the rate of cancer incidence has increased in recent decades, especially in young people, 1 mortality rates have declined due to advances in early detection, disease management, and treatment protocols. 2 However, the post-recovery difficulties caused by treatment strategies have reduced survivors’ quality of life. Since many long-term survivors are among women of reproductive ages, prevention of the detrimental effects of therapeutic agents on the female reproductive system has become one of the most important components of cancer management. 3
In general, chemotherapeutic agents affect cells with a high rate of division, inhibiting their growth, proliferation, and distribution. 4 Therefore, non-tumor cells that are mitotically highly active could be potentially at risk for the adverse effects of these drugs. Doxorubicin (DOX) is an antineoplastic antibiotic from the anthracyclines family as one of the most effective agents of chemotherapy in the current clinical practice. 5 Despite the dramatic antitumor activity, the application of DOX has been limited owing to induction of cytotoxicity in nontarget tissues. Undesirable effects by inhibiting the activity of topoisomerase II and damaging DNA lead to insulting tissues such as the heart, liver, kidney, testis, ovary, and uterus. 6
The mammalian ovary contains various follicles, including primordial, primary, secondary, antral, and graafian, which are at different stages of development. 7 The number of primordial follicles per female is determined during embryonic development and remains constant until puberty, known as ovarian reserve. In this regard, damage to these reserve follicles leads to a pathologic condition named premature ovarian failure (POF). 1 Since these follicles are unable to renew themselves, depletion of the ovarian reserve may lead to permanent infertility. 3 In addition to the ovary, cancer-directed therapies can have devastating effects on the natural structure of the uterus, which is followed by the complications of fertility. 8
Currently, techniques such as oocyte, embryos, and ovarian tissue cryopreservation are used to counteract these adverse effects. 9 However, these strategies have disadvantages such as being time-consuming, expensive, and invasive, increasing the risk of cancer recurrence. 10 Therefore, researchers have a strong tendency to find protective adjuvants to prevent ovarian and uterine damage, thereby improving female cancer survivors’ quality of life. It is assumed that the most important characteristic of an ideal adjuvant is to demonstrate both antitumor and cellular protective properties in cancerous and noncancerous tissues, respectively. 10
Quercetin (QCT) is a phytoestrogen belonging to the flavonoids family found in daily nutrition like onions. 11 Thus far, antitumoral, anti-inflammatory, and antioxidant activities of this compound have been suggested. 11,12 Most importantly, it is reported that QCT potentiates the antitumor effects of DOX in cancer cell lines and prevents its toxicity in nontargeted tissues. 11 Vitamin E (Vit.E) is one of the best known antioxidant compounds necessary for physiological maintenance. 13 This fat-soluble vitamin is abundant in various dietary compounds, and there are reports of its antitumor activity, potentiating DOX antitumoral activity, 14 and improving rodent’s reproductive system. 15
The soothing effects of QCT and Vit.E on DOX-induced toxicity in nontargeted tissues such as the heart, 16 liver, 17 and kidney 18 have been reported. Despite the beneficial effects of these antioxidants on the pathological state of reproductive organs such as the ovary and uterus, 19,20 the potential protective effect of QCT and Vit.E on DOX-induced toxicity in these tissues remains unclear. This study applied doses of DOX equipotent to those used in humans, which is 400 mg/m2, particularly in breast cancer patients, and is equivalent to 0.1–10 mg/kg for animal studies. 21 Owing to the undeniable importance of histopathological evaluations on reproductive abnormalities detection, 22 this study aimed to investigate the potential protective effects of QCT and Vit.E on DOX-induced reproductive toxicity by evaluating histopathological and stereological parameters such as number of ovarian follicles, volume of the ovarian and its structures, volume of the horns, layers, and the structures of uterus. In addition, a comparison of the efficacy of these two antioxidants, one with and the other without phytoestrogenic properties, was investigated.
Materials and methods
Type of study and ethical approval of animal use
This study is an experimental animal study based on histological and stereological methods. The approval from the Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.REC.1398.747) was received, and all of the procedures were conducted based on the guide for the handling of the laboratory animals of the National Institute of Health (NIH).
Animal care and handling
A total of 48 adult female Sprague–Dawley rats were maintained at the Center of Comparative and Experimental Medicine, Shiraz University of Medical Sciences, Shiraz, Iran. The animals exhibited three consecutive regular cycles at least and they were weighed between 170 g and 190 g. The rats were kept in 12 polypropylene cages under controlled housing of 23 ± 2°C temperature, 40 ± 5% humidity, and 12/12 h light/dark cycle. The standard pelleted food (Pars Dam, Tehran, Iran) and water were prepared ad libitum. The animals were acclimatized to the laboratory environment for 14 days prior to the start of the experiment. Vaginal smears were obtained and analyzed by an expert gynecologist from all rats prior to the experiment to determine whether they were identical in the terms of sex cycle.
Experimental chemicals and drugs
DOX was purchased from EBEWE Pharma (Unterach, Austria). QCT provided from Sigma Aldrich (St. Louis, USA) was dissolved in corn oil daily. Vit.E purchased from 21st Century HealthCare (Tempe, AZ, USA) was dissolved in corn oil daily.
Experimental design
The 48 rats were randomly divided into 6 groups including control (CON) saline/corn oil, QCT, Vit.E, DOX, DOX/QCT, and DOX/Vit.E. Normal saline (0.9% w/v) was applied by seven intraperitoneal injections, every third day, beginning from the third day of the experiment. Corn oil was given by gavage every day. QCT (20 mg/kg) and Vit.E (200 mg/kg) were administrated every day. 13,23 DOX administration started on the third day of the study, which applied by seven intraperitoneal injections in cumulative doses 15 mg/kg. 16 The treatment was performed in 21 days, and on the 22nd day, the rats were killed humanely under ketamine 10% (Bremer Pharma GmBH, Warburg, Germany) and xylazine 2% (Alfasan, Woerden, Utrecht, NL) anesthesia. 24
Tissue preparation and histopathological analysis
After killing animals, the ovary and uterine tissues removed, fixed in 10% neutral formalin, and proceed. After tissue processing, the samples were blocked in cylindrical paraffin blocks. The isotropic uniform random sections obtained by using the Orientator method. 25 In this regard, the paraffin blocks containing ovaries were placed on the ϕ clock randomly, each half of which was divided into nine equal parts. By selecting a random number from 1 to 9, an appropriate cut was made along with the selected number. After that, the cylindrical block was placed on the θ-clock, each half of which was divided into nine unequal sine-weighted parts, along its cut surface on the 0–0 axis and then the random number was chosen and the cut was made along with the selected number. Serial 5- and 20-µm thick sections were obtained using a microtome and stained with hematoxylin and eosin (Merck company, Germany) method.
Follicles classification and follicles counting
The follicles were classified according to previous studies. 26 In the primordial follicles, primary oocytes are enclosed within single squamous or cubical epithelial cells. Unilaminar follicles contain the oocytes which are covered by cuboidal epithelial cells. Follicles that possess more than one layer of granulosa cells with no visible antrum considered as multilaminar follicles. Antral follicles demonstrate a single large antral space. Finally, the most enlarge follicles containing cumulus oophorus, corona radiate, and a large antrum considered as graafian follicles. Furthermore, corpora lutea were defined as developing from the graafian follicles following ovulation. The number of follicles was determined on 20-µm thickness sections using an optical disector method, and the following formula is given as
Estimation of ovarian volume
Cavalieri method 27 was applied to estimate the total volume of the ovary. About 8–12 random sections from each ovary were sampled. Counting probe was randomly put on the images, and then the total number of points hitting the sections was counted. The total volume of the ovary was estimated using the following formula:
Estimation of the volume density of ovarian structures
Volume density of the targeted structures, including cortex, medulla, and corpus luteum, was estimated on 5-µm thickness sections through the point-counting method and using Delesse’s formula 28
Estimation of the volume of the uterine horn
The Cavalieri method was applied to estimate the total volume of the uterine horn. In order to estimate the volume, the uterine horn was divided into 8 to 12 parallel sections with a specified distance(t). A point grid was used to calculate the slice area, which was completely randomized to the cutting surface. First, the area around each point (

An example of the point-counting method. The point counting (Cavalieri) method was used to estimate the volume of different layers of the uterine horn, lumen volume, and volume density of the uterine glands. According to this method, the tissues were serially sectioned with a particular distance between the sections. After that, a point grind randomly translated on each slab as demonstrated in the figure. Finally, the number of points landing within the object transect for each slab recorded and used in the related equation. The arrow indicates the right upper quadrant of each cross that was considered to be a point. H&E staining with magnification at ×40 performed. H&E: hematoxylin and eosin.
Estimation of the volume density of uterine structures
Volume density of the targeted structures, including perimetrium, myometrium, endometrium, lumen, and uterine glands, was estimated on 5-µm thickness sections through the point-counting method and using Delesse’s formula 28
Statistical analysis
All results are expressed as mean ± standard deviation. The normality analysis was determined by the Kolmogorov–Smirnov test, and based on that the statistical differences between groups were obtained using one-way analysis of variance and Kruskal–Wallis tests. Statistical analysis performed using SPSS software for windows version 25 and Graph-Pad Prism (version 8.0.2). A value of p < 0.05 was considered statistically significant.
Results
Histopathological analysis
The ovaries of the CON group and Vit.E received animals were healthy and normal in the terms of histo-architecture (Figure 2). The QCT-treated animals demonstrated the limited numbers of atretic follicles and a faint decrease in the volume of the ovary. Dramatic follicular atresia and decreased ovarian volume were observed in the DOX administered animals compared to the CON rats. The ovarian histo-architecture of DOX/QCT and DOX/Vit.E rats was approximately normal with the limited numbers of atretic follicles. However, a faint decrease was observed in the volume of DOX/QCT ovaries.

Ovarian histoarchitecture. A significant decrease in the number of ovarian follicles achieved after DOX administration. In this group, the photomicrographs demonstrated a significant increase in the number of atretic follicles and a remarkable reduction in ovarian volume, volume of the corpus luteum, and the number of healthy follicles. Furthermore, primordial follicles reduced in the ovaries of the QCT and Vit.E groups. DOX + Vit.E administration led to the decrease of the atretic follicle and an appropriate appearance of healthy follicles and corpus luteum. However, the number of atretic follicles in DOX + QCT group was still significant in comparison to the CON group (a1, a2, a3: CON group, b1 b2, b3: QCT group, c1, c2, c3: Vit.E group, d1, d2, d3: DOX group, e1, e2, e3: DOX/QCT group, f1, f2, f3: DOX/Vit.E group. P.F: primordial follicles, U.F: unilaminar follicles, S.F: secondary follicles (antral follicles), C.L: corpus luteum, A.F: atretic follicles, a1–f3: H&E staining with magnification at ×4, ×10, ×40). DOX: doxorubicin; QCT: quercetin; Vit.E: vitamin E; H&E: hematoxylin and eosin.
The histopathological analysis of the uterus of the rats belonging to CON and QCT groups resulted in the normal and healthy appearance in the terms of total volume, layers’ thickness, and histoarchitecture. The Vit.E administration caused a dramatic increase in the lumen volume of the uterus. The DOX-treated animals exhibited a considerable decrease in the total volume and the thickness of uterine layers. However, administration of DOX with both supplements demonstrated alleviative effects on most of the histological parameters (Figure 3).

Histoarchitecture of uterine tissue. CON animals’ uterus appeared healthy. DOX administration caused a dramatic decline in the total volume of uterine tissue and a decrease in uterine layers, glands, and vessel volume. a1–a3: CON group, b1–b3: QCT group, c1–c3: Vit.E group, d1–d3: DOX group, e1–e3: DOX/QCT group, f1–f3: DOX/Vit.E group. Arrow mark: uterine gland (a1–f3: H&E staining with magnification at ×4, ×10, ×40). CON: control; DOX: doxorubicin; QCT: quercetin; Vit.E: vitamin E; H&E: hematoxylin and eosin.
The follicular number
Generally, a significant difference was found between the groups in terms of the follicular number (Figure 4). Regarding the number of primordial, unilaminar, multilaminar, antral, graafian, and atretic follicles, DOX-treated animals demonstrated a significant difference compared to the CON group (p < 0.001). The number of primordial and unilaminar follicles of QCT-treated rats was significantly different (31.98% and 34.13% decline) from that of CON animals (p < 0.001). Treatment with Vit.E caused a 25.51% decrease in the number of unilaminar follicles compared to the CON group (p < 0.001). DOX/QCT and DOX/Vit.E groups were significantly different from the DOX-treated animals in terms of primordial, unilaminar, multilaminar, antral, graafian, and atretic follicles (p < 0.001). However, a significant difference between DOX/QCT and CON groups was obtained when the two groups were compared in the terms of unilaminar, multilaminar (p < 0.001), antral (p = 0.003), graafian (p = 0.037), and atretic follicles (p = 0.001).

A comparison of the number of ovarian follicles. Treatment with DOX chemotherapy caused a significant increase in the number of atretic follicles, as well as depleting the ovarian follicles reservation. Each bar represents the mean ± SD number of primordial (a), unilaminar (b), multilaminar (c), antral (d), graafian (e), and atretic follicles (f) in studied groups. *: the difference was significant compared to the CON group, θ: the difference was significant compared to the DOX group, p values < 0.05 were considered statistically significant. CON: control; DOX: doxorubicin; SD: standard deviation.
The total volume of the ovarian tissue
The ovarian of CON (36.70 ± 5.24 mm3), QCT (36.75 ± 4.57 mm3), and Vit.E (35.0 ± 3.53 mm3) animals demonstrated normal total volume (Figure 5(a)). The administration of DOX resulted in a significant decrease (53.53% decline compared to CON) in the total volume of ovarian tissues (p < 0.001). DOX/QCT (p = 0.005)- and DOX/Vit.E (p < 0.001)-treated animals demonstrated a significant difference with the DOX group in this regard (46.32% and 54.26% increase, respectively).

Ovarian tissue stereological analysis. DOX administration caused a dramatic decline in ovarian, cortex, medulla, and corpus luteum volume. *: the difference was significant compared to the CON group, θ: the difference was significant compared to the DOX group, p values < 0.05 were considered statistically significant. CON: control; DOX: doxorubicin.
The volume of the ovarian layers
Figure 5(b) demonstrates the comparison of the volume of the cortex between the studied groups. According to Figure 5(b), no significant difference was revealed between CON-, QCT-, and Vit.E-treated animals. However, seven injections of DOX led to a 60.41% decrease (p < 0.001) in the cortex volume when compared to CON rats. The administration of QCT or Vit.E with DOX caused a significant increase when compared to DOX-treated animals. However, DOX/QCT animals showed a difference (22.65% decline) compared to CONs (p = 0.049).
Furthermore, the volume of medulla was significantly different (p = 0.001) between the groups (Figure 5(c)). Treatment with DOX chemotherapy (4 ± 0.63 mm3) caused a significant 23.37% decrease compared to the medulla volume of the CON-treated rats (5.22 ± 0.44mm3). In this regard, DOX/QCT and DOX/Vit.E, compared to DOX animals, revealed remarkable different results (p < 0.05).
The volume of the corpus luteum
No remarkable difference was found in the terms of the corpus luteum volume between CON (5.13 ± 0.68 mm3), QCT (4.64 ± 0.51 mm3), and Vit.E (4.30 ± 0.56 mm3) animals. The results revealed that DOX administration led to a significant decrease compared to CONs. Coadministration of QCT with DOX chemotherapy resulted in a significant difference in both DOX (p < 0.001) and CON (p = 0.001) groups (174.4% increase and 36.83% decrease when compared to DOX and CON, respectively). Nevertheless, the DOX/Vit.E animals were considerably different compared to the DOX group (Figure 5(d)).
The uterine and uterine layers volume
The comparison of the total volume of uterine revealed a significant decrease (p < 0.001) in both DOX (49.13%) and DOX/QCT (28.90%) groups when compared to the CON (Table 1). However, cotreatment of chemotherapy with Vit.E demonstrated no difference with CON animals (p = 0.776). Furthermore, DOX administration caused a significant decrease in the volume of perimetrium (50.19%), myometrium (51.19%), and endometrium (59%) layers (p < 0.001). DOX/QCT-treated animals showed a significant difference with DOX received rats in the terms of perimetrium and myometrium volume; however, a 21.94% decrease resulted in myometrium volume when compared to the CON group (p < 0.001). The coadministration of Vit.E and DOX caused a significant difference in the volume of uterine three layers compared to both CON- and DOX-treated groups (p < 0.05).
The thickness of uterine layers.a

CON: control; DOX: doxorubicin; QCT: quercetin; Vit.E: vitamin E; SD: standard deviation.
aDOX-treated animals revealed a significant decrease in the volume of the uterine horn and related layers. The alleviative effects of QCT and Vit.E coadministration with DOX achieved.
b The difference was significant compared to the CON group.
c The difference was significant compared to the DOX group, p values < 0.05 were considered statistically significant.
The volume of the uterine structures
Figure 6 shows the comparison of uterine glands, lumen, and vessels volume between the studied groups. Compared to the CONs, the QCT treatment caused a 21.34% and 17.62% decrease in the volume of uterine glands (p = 0.004) and vessels (p = 0.009), respectively. Furthermore, the Vit.E received group revealed a significant increase and decrease in the volume of the uterine lumen (p = 0.019) and vessels (p < 0.001), respectively. According to the results, seven injections of DOX chemotherapy lead to a significant decrease in the volume of uterine glands, lumen, and vessels when compared to the CON-treated group (p < 0.001). Similarly, DOX/QCT animals demonstrate a significant difference in the terms of these three structures compared to the CON-treated group. However, the volume of uterine glands and vessels of DOX/QCT coadministered animals demonstrated a 198.07% and 120.99% increase when compared to DOX group. The glands and vessels of the removed uterine tissue from DOX/Vit.E rats demonstrated a significant difference compared to both DOX and CON groups (p < 0.05), whereas no difference with CON animals was found in the terms of lumen volume (p = 0.960).

The volume of uterine-related structures. QCT- and Vit.E-treated animals revealed some toxic effects on uterine related structures. DOX administration caused a remarkable decrease in the volume of uterine glands (a), the lumen (b), and vessels (c). *: the difference was significant compared to the CON group, θ: the difference was significant compared to the DOX group, p values < 0.05 were considered statistically significant. CON: control; DOX: doxorubicin; QCT: quercetin; Vit.E: vitamin E.
The thickness of the uterine layers
Figure 7 shows the comparison of the thickness of perimetrium, myometrium, and endometrium layers. According to results, the perimetrium thickness of Vit.E (p = 0.003), DOX (p < 0.001), DOX/QCT (p = 0.023), and DOX/Vit.E (p = 0.001) animals had a significant decrease (18.99%, 28.54%, 14.49%, and 20.86% decline, respectively) compared to CONs. Administration of DOX alone, as well as its coadministration with QCT, caused a significant decrease in the thickness of myometrium and endometrium layers compared to CON; however, the difference between DOX and DOX/QCT animals was significant in the terms of myometrium thickness (p < 0.05). Vit.E, when administrated with DOX, significantly increased the thickness of myometrium (p < 0.001) and endometrium (p = 0.017) compared to DOX-treated animals. However, the thickness of the uterine endometrium from DOX/Vit.E animals revealed a remarkable difference (21.63% decrease) with CONs (p < 0.001).
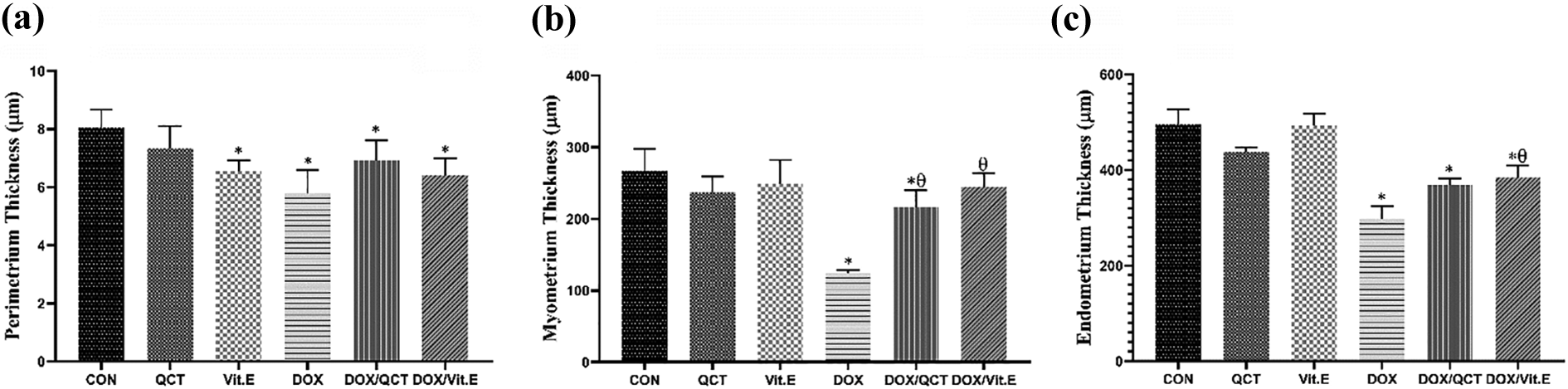
The thickness of uterine layers. Vit.E administration caused a significant decrease in the thickness of the perimetrium. The deleterious effects of DOX injection on the thickness of perimetrium, myometrium, and endometrium are presented. *: the difference was significant compared to the CON group, θ: the difference was significant compared to the DOX group, p values < 0.05 were considered statistically significant. CON: control; DOX: doxorubicin; Vit.E: vitamin E.
Discussion
Insults on ovarian reserve and induction of POF, and causing uterine toxicity by chemotherapies have led to increased rates of infertility in young female cancer survivors. 29 Owing to the inefficiency of current strategies in preventing reproductive toxicity, the use of cytoprotective adjuvants is considered an important component of cancer care studies. 30
This study evaluated the prevention of DOX-induced reproductive toxicity using two dietary antioxidants in rat models, QCT and Vit.E, to suggest adjuvants in order to enhance cancer survivors’ quality of life. QCT and Vit.E have shown favorable properties in previous studies such as prevention of DNA damage, anti-tumor, anti-inflammatory, and antioxidant activity, 31 as well as augmentation of DOX function in cancerous tissues. 32 These features, along with some reports of preventing DOX-induced toxicity in other nontargeted tissues, 33,34 led the study to the choice of these dietary compounds as a preventive adjuvant for DOX-induced cytotoxicity in the ovary and uterus.
A number of previous studies identified DOX as a cause of ovarian failure with low to intermediate risk. 21 On the contrary, the findings of this study indicated the severe damage to the ovarian reserve and a decrease in follicles with different stages of puberty following chemotherapy administration. In agreement with the present study findings, Nishi et al. 26 reported a dramatic loss of follicular pool and abnormal development of the mammary glands in rats, which may result in permanent infertility due to damaging the follicles. Interestingly, administered antioxidants with chemotherapy prevented the loss of the follicular pool. Previous studies attributed the DOX-induced ovarian failure to increased levels of DNA damage and oxidative stress, in which Vit.E and QCT showed the potential for the prevention of these mechanisms in similar states. 26,33,35 In addition, we determined the anti-apoptotic properties of these compounds against chemotherapy-induced cell death in our laboratory (unpublished data). Importantly, the administration of these two dietary supplements in rats not exposed to DOX accelerated the primordial and primary follicles atresia. Similarly, Chen et al. 36 revealed an increase in the number of atretic follicles in aging rats that had received QCT for a long period. On the contrary, the majority of studies suggested Vit.E and QCT supplementation to increase ovarian follicular reserve. 37 Although the underlying cause of these inconsistencies remains unclear, the beneficial role of reactive oxygen species in the development and maturation of follicles by the involvement of inflammatory, autophagic, and angiogenic mechanisms has been elucidated. 38 Therefore, it appears that the chronic application of antioxidants in therapeutic doses may lead to abnormal follicular development by inhibiting the production or reducing the physiologic level of free radicals. However, the administration of these antioxidants with DOX not only demonstrated no adverse effects but also prevented the reproductive toxicity of DOX. It could be extrapolated to the function of these antioxidants in restoring the excess amounts of free radicals produced after DOX administration to normal levels. 12 However, this study encourages further studies to clarify and identify the involved mechanisms.
This study demonstrated that DOX reduced the ovarian, cortical, and medullary layers volume as well as the volume of corpus luteum. Other researchers have presented similar findings. Ben-Aharon et al. showed the acute effect of DOX on ovarian size reduction using the magnetic resonance imaging method. 39 Decreased ovarian volume and its associated structures have been linked to the induction of oxidative stress in this tissue by cancer-directed therapies. 40 The disrupted tissue perfusion due to arteries damage could also be considered the cause of ovarian atrophy. 41 It is suggested that DOX could affect the ovarian vascular system, by narrowing blood vessels, inducing stromal cell damage, and causing stromal fibrosis. 42 Furthermore, reduced corpus luteum volume is involved in reducing the chance of successful conception due to its necessity for the progesterone production preparing the uterus for implantation and regulating the sexual cycle. 43 Importantly, both QCT and Vit.E demonstrated a protective role in this regard, probably due to elimination of excess levels of tissue free radicals. In addition, both of these compounds are regarded to stimulate the secretion of gonadotropins, playing a vital role in the development of the reproductive system and maintaining its functional and structural physiology. 44 Finally, the contribution of the studied nutrients in modulating the production, secretion, and function of hormones involved in tissue development such as estradiol and progesterone 45,46 as well as their depicted antiapoptotic activity could be attributed to the prevention of DOX-induced ovarian atrophy.
Previous studies suggested that uterine alterations induced by chemotherapeutic agents could cause fertility-related complications. Seyhan et al., for example, demonstrated that cisplatin could damage the endometrium and uterine glands. 47 The thin endometrium is associated with infertility due to impaired endometrial receptivity. 48 The present study results described that DOX administration significantly reduced the thickness of uterine layers. In addition, damage to uterus vessels has also been observed. Thus far, vascular damage has been reported after administration of various cancer therapeutic agents that may reduce blood supply to cells, resulting in atrophy and decreased uterine volume. 49 Moreover, damage to the uterine muscles and decreased myometrium volume can be involved in further problems with uterine contractions during labor. 49 Consistently, the study findings indicated a decrease in the size of uterine glands after DOX administration. The uterine glands play a pivotal role in uterine receptivity. Besides, the impact of these glands in luminal fluid homeostasis and blastocyst implantation has been suggested. 50 The present study findings suggest that the administration of either QCT or Vit.E can recover DOX-induced uterine structural abnormalities. QCT is a flavonoid with estrogen-like activities suggested in the uterus. This phytoestrogen probably works by modulating estrogen receptors’ role in uterine homeostasis. 51 The phytoestrogenic function of QCT has been implicated in improving the effects of polycystic ovaries on the uterus. 52 Furthermore, it has soothing effects on aging-related fibrosis associated with the PI3K/Akt/mTOR signaling pathway. 53 Additionally, Vit.E is one of the strongest antioxidants essential for the reproductive process. 54 The results of this study demonstrated that both of these supplements were able to alleviate the toxicity of DOX in the uterus appropriately. The most likely cause for this protective function may be related to the antioxidant activities of QCT and Vit.E. Several researchers attributed uterine damage after DOX administration to the induction of ovarian failure. Ovarian follicles are primarily responsible for the synthesis and secretion of reproductive hormones, estradiol, and progesterone. 55 These sex steroids exhibit essential roles in the management of uterine cell proliferation, the stromal-epithelial cells cross-talk, and the required differentiation for appropriate pregnancy preparation. 56,57 Therefore, based on the role of the ovary in maintaining uterine homeostasis, the prevention of toxicity in the ovary by these antioxidants may lead to healthy uterine tissue. However, further studies in this regard are necessary. Importantly, according to some of the found adverse effects related to these antioxidants consumption by the rats not exposed to DOX, this study does not support the administration of therapeutic doses of these antioxidants to healthy individuals. The potentially unexpected effects of these antioxidants in the uterus can be similar to what was mentioned concerning the ovary. However, further studies are necessary to determine the promising protective function of these antioxidants against DOX-induced reproductive toxicity.
Conclusion
The present study findings indicate that histopathological and stereological QCT and Vit.E can alleviate the DOX-induced toxicity on the histoarchitecture of ovarian and uterine tissues. Decreased ovarian follicles, ovarian and uterine atrophy, and damage to tissue layers and vessels have been the consequences of DOX administration, which antioxidants coadministration with chemotherapy can recover them. However, some of the revealed histological abnormalities related to antioxidants consumption cause the study not to support the administration of therapeutic doses of antioxidants to healthy individuals who did not experience cancer-directed therapies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been extracted from the MSc thesis of Mohammad Samare-Najaf which was supported by the Vice-chancellor for Research Affairs of Shiraz University of Medical Sciences (Grant no. 21397).