Abstract
Earlier, we demonstrated that chronic exposure to monocrotophos (MCP) elicits insulin resistance in rats along with increased white adipose tissue (WAT) weights. This study was carried out to delineate the biochemical and molecular changes in adipose tissues of rats subjected to chronic exposure to MCP (0.9 and 1.8 mg/kg bw/d for 180 days). Pesticide-treated rats exhibited increased fasting glucose and hyperinsulinemia as well as dyslipidemia. Tumor necrosis factor-alpha and leptin levels were elevated, while adiponectin level was suppressed in plasma of treated rats. MCP treatment caused discernable increase in the weights of perirenal and epididymal WAT. Acetyl coenzyme A carboxylase, fatty acid synthase, glyceraldehyde-3-phosphate dehydrogenase, lipin-1, and lipolytic activities were elevated in the WAT of MCP-treated rats. Corroborative changes were observed in the expression profile of proteins that are involved in lipogenesis and adipose tissue differentiation. Our results clearly demonstrate that long-term exposure to organophosphorus insecticides (OPIs) such as MCP has far-reaching consequences on metabolic health as evidenced by the association of adipogenic outcomes with insulin resistance, hyperinsulinemia, endocrine dysregulations, and dyslipidemia. Taken together, our results suggest that long-term exposure to OPI may be a risk factor for metabolic dysregulations.
Keywords
Introduction
The broad-spectral insecticidal effects and less persistent nature of organophosphorus insecticides (OPIs) make them highly used class of insecticides in agricultural and other domains. However, residues are found in various abiotic 1 and biotic environments including human milk. 2,3 Although primarily known for their neurotoxicities, OPIs are now coming under scrutiny for their effects on other components of physiological system. 4 Recent reports clearly indicate that OPIs are associated with metabolic abnormalities not only in experimental models 5 –16 but also in human subjects. 17 –20 Insulin resistance and hyperinsulinemia appear to be the most frequently reported outcomes of studies that aimed to study the metabolic effects of OPI. 8 –11,18 –20 Endocrine dysregulations are increasingly being reported as a result of exposure to OPI. 20 Since hormones including insulin, glucocorticoids, and adiponectin exert profound impact on metabolic homeostasis, it is increasingly becoming clear that there may be causal link between endocrine dysregulations and metabolic impairments caused by OPI.
Considerable amount of work has been done in the direction of understanding the underlying mechanism OPI-induced metabolic dysregulations. Many of these have revolved around understanding the hyperglycemic potential of OPI. Studies involving single-dose exposure (acute exposure) unequivocally establish that OPI induces rapid onset and reversible hyperglycemia. 5 –7,15,16 Our earlier studies demonstrate that repeated exposure to results in blunted responses in the early phase and glucose homeostasis gradually deteriorates as exposure period is prolonged. 9 We have also demonstrated that insulin resistance induced by monocrotophos (MCP) is associated with aggravated pancreatic β-cell response, arguably to counter the former. 10 Pancreas 10,21 and liver appear to be the most studied organs for understanding the mechanism behind OPI-induced metabolic dysregulations, 6,7,9,14 with oxidative stress, glycogenolysis, and gluconeogenesis being frequently reported as probable cause. Alterations in lipid profile appear to be consistently reported aberration in experimental animals exposed to OPI. 9,14
The role of adipose tissue in the regulation of metabolic homeostasis is fairly well understood. Adipose tissue not only serves as a storage unit for excess energy but also expresses the receptor for various hormones and secretes the number of cytokines that have a broad range of biological functions. 22 Paradoxically, both excess (obesity) and loss (adipose dystrophy) of adipose tissue are associated with metabolic syndrome. 22,23 However, the contribution of white adipose tissue (WAT) in OPI toxicity is not as well studied as their effects on liver, glucose homeostasis, and lipid profile. Chronic exposure to chlorpyrifos has been reported to elicit increased weight gain as well as increased weights of perirenal WAT, albeit the OPI lacked the ability to induce adipocyte differentiation in vitro. 24 This is suggestive of the fact that the effect of OPI on adipose tissue is probably an outcome of changes in homeostasis in vivo. Hence, this study was undertaken to obtain biochemical and molecular insights into the effect of chronic exposure to MCP on WAT functions in rats.
Materials and methods
Reagents
Corticosterone, glucose-6-phosphate dehydrogenase (G6PDH), acetyl coenzyme A (CoA), malonyl-CoA and acetoacetyl-CoA, dithiotreitol, tween 20, Triton X-100, ammonia, 1,2-dipalmitoyl-sn-glycero-3-phosphate sodium salt, dihydroxyacetone phosphate dilithium salt, ethanol, isopropanol, TRIzol, bovine serum albumin (BSA), and free fatty acid (FFA) kit were purchased from Sigma Chemicals Co. (St Louis, Missouri, USA). Glucose-6-phospahte, malic acid, nicotinamide adenine dinucleotide phosphate (NADP), magnesium chloride, n-ethylmaleimide, nicotinamide adenine dinucleotide phosphate reduced (NADPH), malate, and ethylenediaminetetraacetic acid (EDTA) were procured from Sisco Research Laboratories Pvt Ltd (Mumbai, Maharashtra, India). Blood glucose, triglyceride, cholesterol, and high-density lipoprotein cholesterol (HDL-c) kits were procured from Span Diagnostics (Ahmadabad, Gujarat, India). Insulin kit was procured from Crystal Chem (Elk Grove Village, USA). Adiponectin, leptin, and tumor necrosis factor-alpha (TNF-α) kit were procured from Invitrogen (Bangalore, Karnataka, India). Polymerase chain reaction (PCR) master mix was procured from Biorad (California, USA). Orlistat was procured from Centurion Laboratories (Vadodara, Gujarat, India). All other reagents used were of analytical grade.
Animals and experimental protocol
Adult male rats (CFT, Wistar strain, approximately 150 ± 5 g) drawn from the Institute Animal House Facility were divided into three groups. This study was approved by the Institute Animal Ethical Committee, regulated by the CPCSEA, India. Rats of the first group served as control, while rats of second and third groups were administered MCP at 0.9 and 1.8 mg/kg bw/d for 180 days. The experimental regimen was based on our previous study demonstrating manifestation of insulin resistance in rats subjected to chronic MCP exposure. 9 Three rats from each group were fasted overnight to obtain fasting glucose and insulin values. The rest of the rats from each group were euthanized by ether anesthesia, and blood, perirenal, and epididymal adipose tissues were collected, weighed, and stored for biochemical and molecular studies.
Biochemical parameters in blood
Blood glucose, lipid profile, and NEFA were quantified using the commercial kit. Plasma insulin, leptin, adiponectin, and TNF-α were quantified using ELISA kits. Plasma corticosterone was estimated by the fluorimetric method. 25 Triglyceride was estimated with correction for free glycerol using orlistat, 26 and cholinesterase activity was analyzed using microtiter plate as described. 27
LPL activity and lipolysis in adipose tissue
Lipoprotein lipase (LPL) activity was measured in both perirenal and epidydimal adipose tissue using triacylglycerol (intralipid, artificial fat emulsion) as a substrate. Suitable volume of homogenate (50 μl, 5%) was incubated with substrate buffer consisting of 1.2 vol. of 10% intralipid, 4.8 vol. of rat serum (preprandial, source of apolipoprotein CII), 10.5 vol. of 18% BSA, and 35 vol. of 1.6 M Tris–hydrochloride (pH 8.1) and incubated at 37°C for 30 min. The released FFA was measured using copper sulfate complex, and interference from free FFA was omitted using 1 M sodium chloride (potent inhibitor of LPL) as a blank for individual samples. The results were expressed as nmoles of FFA released/min/mg protein. 28,29
Total lipolytic activity in adipose tissue is the sum of activities of adipose triglyceride lipase, hormone-sensitive lipase, and monoacyl glycerol lipase and was quantified by employing triacyl glycerol (intralipid) as a substrate along with BSA (2%) as a stabilizing agent in phosphate buffer (pH 7.0) as a suitable medium. The reaction was conducted at 37°C for 1 h, and FFA released was extracted using chloroform and measured using cuprizone reagent as described. The results were expressed as nmoles of FFA released/min/mg protein. 30,31
Adipose biochemical parameters
For lipogenic enzymes activity, adipose tissues were homogenized in the suitable buffer as mentioned in the protocol and supernatant was collected for measuring the activities of enzymes involved in lipid metabolism. Enzymes, namely, acetyl-CoA carboxylase (ACC), 32 fatty acid synthase (FAS), 33 lipin-1, 34 glycerol-3-phosphate dehydrogenase, 35 malic enzyme, 36 and G6PDH 37 activities were measured spectrophotometrically.
Gene expression
Total RNA content was extracted from perirenal WAT using the TRIzol method and purity was estimated by determining optical density at 260/280 nm and stored at −80°C until further use. 38 The primers for real-time PCR (RT-PCR) experiment were designed using Primer Express 2 software from Applied Biosystems (Beverly, USA) (Table 1). The complementary DNA (cDNA) synthesis was performed using Thermoscientific Verso cDNA Synthesis Kit (Beverly, USA) as prescribed by the manufacturer’s instructions. The reaction mixture containing cDNA, 2× SsoFast EvaGreen Supermix PCR Master Mix (Biorad), and the specific primer pair was used for amplification on a Biorad CFX 96 system. The 2−ΔΔCt relative quantification method was used to calculate the fold change in the gene expression with β-actin as an endogenous control. Samples from five rats in each group were used, and the experiments were done in triplicates.
Details of primers used in the study.

F: forward primer; R: reverse primer; LPL: lipoprotein lipase: DGAT1: diglyceride acyltransferase 1; ACC: acetyl-CoA carboxylase; FAS: fatty acid synthase; PPARγ: peroxisome proliferator-activated receptor gamma; SREBP1c: sterol regulating binding protein 1c; ADD1: adipocyte differentiation and determination factor 1.
Statistical analysis
Mean and standard error (SE) were determined for all the parameters, and results were expressed as mean ± SE. The data were analyzed employing the analysis of variance followed by Tukey’s HSD test for comparison of means to determine the significance of differences among the groups. p Values below 0.05 were considered as significantly different.
Results
The impact of MCP on glucose homeostasis, lipid profile, adipose weights, and hormonal levels in treated rats
The MCP-treated rats exhibited an increase in fasting blood glucose and insulin levels after chronic exposure (Table 2; p < 0.05). The insulin resistance measured by HOMA-IR demonstrated declined insulin sensitivity (35–45% of control) with severe insulin resistance (2–3 fold above control) and enhanced β-cell function (40–50% above control) in treated rats (Table 2).
Effect of MCP on fasting blood glucose, insulin, and glucose homeostasis at chronic exposure.a,b,c

MCP: monocrotophos; SE: standard error; ANOVA: analysis of variance.
a Values were mean ± SE (n = 4). bRows with different alphabets are statistically different. c p < 0.05: Statistical analysis: one-way ANOVA/Tukey post hoc.
The effect of MCP on adipose weights and plasma parameters is tabulated in Table 3. Chronic exposure to MCP caused an increase in body weight gain compared to control (p < 0.05). Both absolute and relative weights of the perirenal and periepidydimal adipose fat pads were significantly increased (absolute and relative) in MCP-treated rats. Increased blood glucose (20–40% above control), hyperinsulinemia (100% above control), and increased corticosterone (30–35% above control) were observed in the plasma of MCP-treated rats. The plasma triglyceride, NEFA levels were elevated, while HDL-c (70–75% of control) levels were decreased in MCP-treated rats. The impaired adipokine levels such as declined adiponectin (55–60% of control) with an increase in plasma TNF-α and leptin levels (20–25% above control) were noticed. MCP administration caused severe inhibition of plasma cholinesterase activity.
Impact of MCP on adipose tissue weights and plasma parameters in exposed rats.
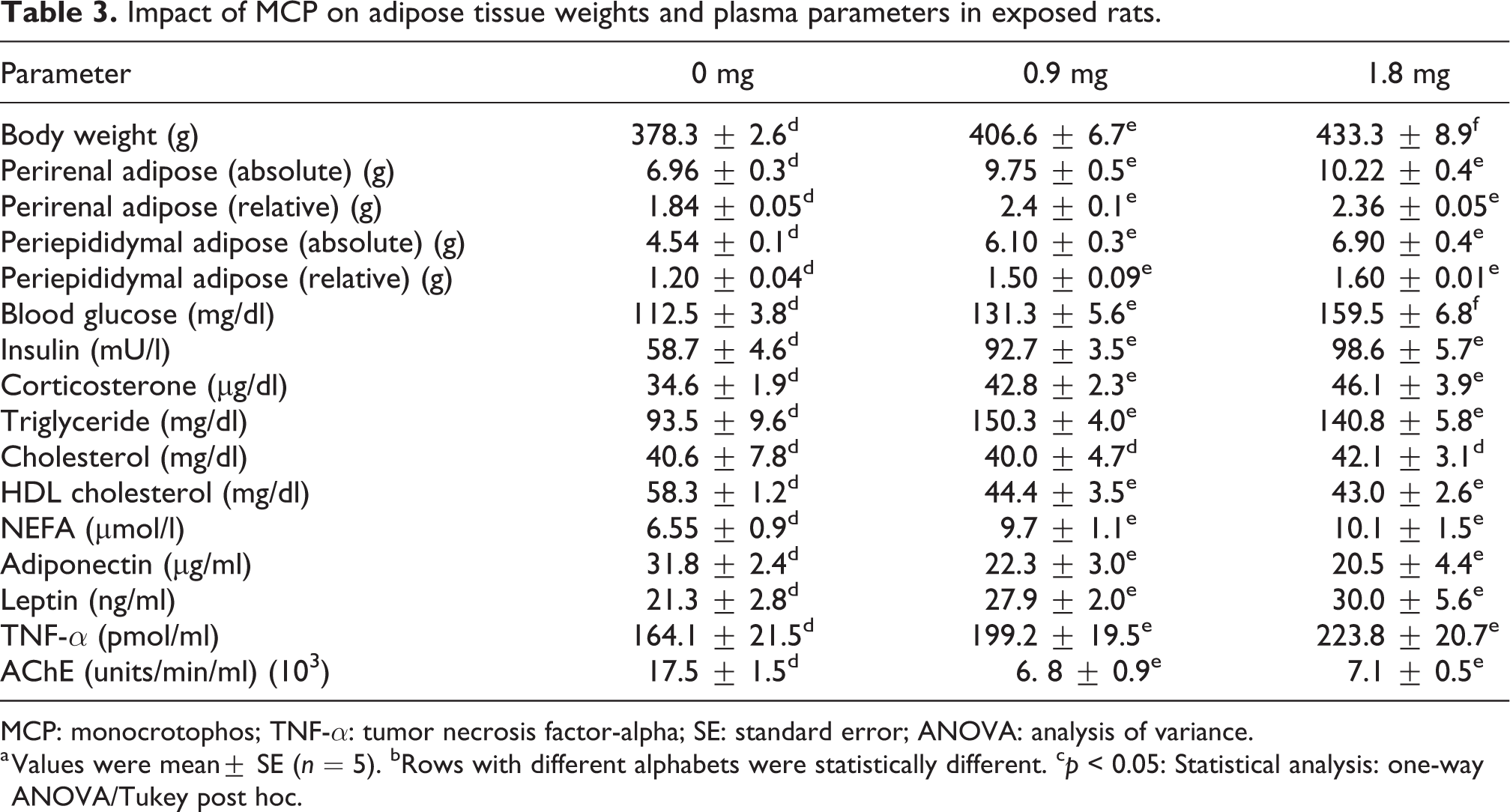
MCP: monocrotophos; TNF-α: tumor necrosis factor-alpha; SE: standard error; ANOVA: analysis of variance.
a Values were mean± SE (n = 5). bRows with different alphabets were statistically different. c p < 0.05: Statistical analysis: one-way ANOVA/Tukey post hoc.
Impact of MCP on genes expression in adipose tissue
The expressions of various transcription factors and enzymes which regulate lipid synthesis and differentiation of perirenal adipose tissue in treated rats are represented in Figure 1. The sterol regulating binding protein 1c (SREBP1c), a key transcription factor that regulates denovo fatty acid synthesis in adipose tissue, was upregulated (1.5–2 fold above control), which may account for enhanced ACC and FAS expression (1–3 fold above control) in treated rats. The expression of lipin-1 and diglyceride acyltransferase 1 (DGAT1), key factors that regulate triglyceride synthesis in adipose tissue, was considerably elevated. The adipocyte differentiation and determination factor 1 (ADD1 and peroxisome proliferator-activated receptor gamma (PPARγ) (1.5–2 fold above control) were significantly enhanced in WAT of pesticide-treated rats (Figure 2).

Effect of MCP on the expression of key transcription factors and enzymes which regulate adipose mass and triglyceride content in rats exposed for 180 days. Values are mean ± SE (n = 5) and represent mean fold change in mRNA levels in treated rats compared to control. MCP: monocrotophos; SE: standard error; mRNA: messenger RNA.

Impact of MCP on adipose depots in 180d treated rats. (a), (b) and (c) represents perirenal adipose depots in 0, 0.9 and 1.8mg/kg b.w treated rats. (d), (e) and (f) represents epidydimal adipose depots in 0, 0.9 and 1.8mg/kg b.w treated rats.
Biochemical analysis in adipose tissue
The biochemical parameters of perirenal and epidydimal adipose tissue are depicted in Figure 3. The considerable increase in lipogenesis and lipolytic activity was noted in the adipose tissue of treated rats. MCP treatment was associated with significant increase in ACC, FAS, lipin-1, and glyceraldehyde-3-phosphate dehydrogenase activities that were elevated in both WATs of pesticide-treated rats. Interestingly, significant modulation of G6PDH and malic enzyme was observed only in epididymal WAT after MCP treatment. MCP caused marginal decrease in LPL activity only in perirenal WAT. The degree of lipolysis was considerably higher in adipose tissue of treated rats.

Effect of MCP on adipose biochemical parameters. ACC (a), FAS (b), gly-3-p dehydrogenase (c), glu-6-p dehydrogenase (d), malic enzyme (e), lipin-1 (f), LPL (g), and total lipolysis (h) activities. Values are mean ± SE (n = 5). The bars with * represent statistical difference of p < 0.05 between the different groups, respectively. MCP: monocrotophos; ACC: acetyl-CoA carboxylase; FAS: fatty acid synthase; gly-3-p: glyceraldehyde-3-phosphate; glu-6-p: glucose-6-phosphate; LPL: lipoprotein lipase; SE: standard error.
Discussion
OPI is known to induce metabolic dyshomeostasis in animal models 5,6,9,10 and exposed humans (incidental or occupational exposure). 14,18,19 Earlier, we demonstrated the potential of MCP to induce insulin resistance following chronic exposure and it was associated with symptoms of dyslipidemia and an increase in WAT accumulation at perirenal and epidydimal regions in rats. 9 The adipose hypertrophy or obesity is strongly associated with the symptoms of metabolic syndrome in humans and experimental animals; therefore, in this study, we delineate the role of impaired adipose physiology on the onset of metabolic dyshomeostasis in MCP-treated rats.
In the current study, MCP-treated rats exhibited insulin resistance (HOMA-IR) which was associated with enhanced β-cell function to regulate euglycemia. The results correlate with our previous observations 9,10 and others who found symptoms of insulin resistance during chronic exposure to various OPI. 11 –14 Further, both perirenal and epidydimal WAT weights were significantly increased in treated rats and associated with elevated levels of plasma insulin and glucocorticoid levels. The insulin is known for its anabolic nature. It upregulates the expression and activity of key transcription factors, rate-limiting enzymes of lipid biosynthesis, 39 and LPL expression 40 in adipocytes. This results in an increase in adipose triglyceride content via enhanced de novo synthesis and excess NEFA absorption, respectively. Besides, the study also shows that glucocorticoids induce preadipocyte differentiation and adipogenesis during hyperinsulinemia. 41 This might result in an increased WAT accumulation in MCP-treated rats. Similar outcomes were observed in patients suffering from Cushing syndrome, a disease characterized by the increase in plasma glucocorticoid and insulin levels. 42 The increased plasma NEFA and triglyceride noticed in this study might be attributed to impaired insulin action on adipose tissue which in turn results in declined LPL activity with an enhanced degree of lipolysis even at hyperinsulinemia in MCP-treated rats. 43,44
Adipose tissue secretes various cytokines that have broader biological functions. These cytokines have both pro- and anti-inflammatory properties. The type and quantity of cytokine secretion are proportional to the age, origin (subcutaneous or visceral), and type of macrophages associated with it. 45 –47 The metabolic syndrome strongly correlates with the degree of visceral adiposity and pro-inflammatory cytokines secreted from it. Therefore, in this experiment, the rats treated with MCP exhibited a decline in adiponectin along with enhanced leptin and TNF-α levels compared to control animals. In the current study, the increase in perirenal and epididymal adipose depots and impaired cytokine secretion might have involved in the induction of fasting hyperglycemia and insulin resistance in MCP-treated rats.
In general, an increase in adipose mass is adaptive in nature, and agonists of PPARγ enhance insulin sensitivity via the upregulation of pre-adipose differentiation, maturation, and prevention of ectopic fat accumulation. 48 However, the old adipose which accumulates excess triglyceride exhibit declined insulin sensitivity, 46 characterized by severe lipolysis albeit hyperinsulinemia. In this study, rats treated with MCP for longer duration exhibited enhanced PPARγ expression along with SREBP1c, resulting in the induction of excessive fat accumulation in the perirenal and epidydimal region. The SREBP1c regulates lipogenesis by upregulation of ACC and FAS expression. This may result in enhanced lipid accumulation in adipose tissues of treated rats. 39 The upregulation of SREBP1c, SREBP/ADD1, PPARγ, and LIPIN-1 was noticed in MCP-treated rats, which are known to induce adipose differentiation and enhance triglyceride storage in many experimental animal models. 49 –52
The DGAT1 plays a major role in the regulation of triglyceride synthesis in adipose tissue. In addition to triglyceride synthesis, DGAT1 is well coupled with insulin sensitivity and energy metabolism. 53 The DGAT1-deficient mice exhibits decreased adiposity with declined tissue triglyceride content. Further, these rats exhibited increased insulin sensitivity, energy expenditure, and resistance to the development of obesity in high-fat diets. 54 In contrast to this, transgenic animals overexpressed with adipose DGAT1 exhibited glucose intolerance with obesity. 55 Therefore, any factor which enhances either expression or activity of DGAT1 is ascribed to be a risk factor for the development of obesity and glucose intolerance. In this experiment, enhanced DGAT1 expression was noticed in rats and it might be the reason for the development of various metabolic syndrome including insulin resistance, dyslipidemia, and symptoms of obesity in treated rats.
Although the doses of OPI used in experimental studies are much higher than environmental or occupational exposures, the relevant outcomes have been observed in humans during the circumstances of environmental or occupational or intentional exposures. Acute exposure to OPI in humans causes hyperglycemia, glycosuria, and acute pancreatitis a symptom similar to that of animal studies. 56 A prospective study conducted by Gifford et al. revealed impaired glucose homeostasis and changes in the action and secretion of insulin in pesticide self-poisoning exposures. 17 Similarly, symptoms of diabetes have been observed in pesticide applicators exposed to various OPI such as chlorpyrifos, diazinon, and trichlorfonin a dose-dependent manner. 18 Positive correlation between plasma malathion levels with waist circumference and insulin resistance was documented in a farmers’ cohort, 19 and parallel outcomes are noticed in animal studies also. Insulin resistance and increase in body weight have been observed in rats subjected to variable dose and duration of exposure to different OPIs. A recent study demonstrated increased incidence of diabetes among Thai farmers exposed to different OPIs. 57 Thus, it is obvious that OPI exposure is a major risk factor for metabolic dysregulations among those who are occupationally exposed. One has to take cognizance of the fact that the levels of exposure to OPI among occupationally exposed people are likely to be higher than the general population. However, a recent study revealed that glycated hemoglobin levels correlate very well with plasma levels of OPI, but not with acetylcholinesterase activity. 58 This study indicates that low levels of OPI exposure may cause metabolic deregulations. Hence, we believe that further studies are needed to evaluate the effects of low-level chronic OPI toxicity on metabolic health.
Conclusion
MCP-treated rats demonstrated impaired adipose biochemical functions exhibited by increased WAT accumulation with increased lipolysis and decreased LPL activity albeit an increase in plasma insulin levels. The fatty acid biosynthesis machinery was upregulated via an increase in the expression of SREBP1c, ADD1, and DGAT1 which are known to induce adipose differentiation and triglyceride accumulation in young adipocytes. Further, compared to controls, the MCP-treated rats exhibited altered insulin sensitivity which was associated with impairment in cytokine secretion from adipose tissue. Therefore, the impaired adipose physiology may be one of the factors responsible for the development of metabolic syndrome in MCP-treated rats.
Footnotes
Acknowledgments
The authors wish to thank the Director, CFTRI, Mysore, for his support to the study. The first author (RN) acknowledges the financial support from ICMR, India in the form of research fellowship.M/s Hyderabad chemicals supplies Ltd is greatly thanked for the gift of technical grade sample of monocrotophos.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
