Abstract
Although it has been widely considered that all-trans retinoic acid (ATRA) is an efficient therapeutic agent for acute promyelocytic leukemia (APL), there is an urgent need for extending and examining new therapeutics in medicine. Dithiocarbamates (DTCs) are one of the recent important chemical synthetic compounds used in cancer therapy. The aim of this study was to evaluate the apoptosis-inducing effect of 2-nitro-1-phenylethylpiperidine-1-carbodithioate (2-NDC) as an active derivative from DTCs, in combination with ATRA on human APL NB4 cells. The viability of treated NB4 cells was measured by 3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide assay in various concentrations (10–120 µM). The proapoptotic effects of 2-NDC were investigated by acridine orange/ethidium bromide staining, DNA ladder formation, and flow cytometry. We also assessed the oxidative stress-inducing effect of 2-NDC and in combination with ATRA on the NB4 cells. The alteration in gene expression levels of Bax, Bcl2, and Survivin was measured through a real-time polymerase chain reaction. Furthermore, we redetected the interaction between 2-NDC and antiapoptotic proteins Bcl2 and Survivin via molecular docking. We found that 2-NDC induced apoptosis in NB4 cells in a time–dosage-dependent manner. Also, 2-NDC triggered apoptosis by expanding intracellular reactive oxygen species, combined with ATRA. Bax/Bcl2 ratio was modulated and Survivin was downregulated in NB4 cells upon 2-NDC treatment. Molecular docking studies indicated that 2-NDC binds to the baculovirus inhibitor of apoptosis protein repeat domain of Survivin and Bcl homology 3 domain of Bcl2 with various affinities. Based on the present observations, it seems that this derivative can be estimated as an appropriate candidate for future pharmaceutical evaluations.
Introduction
Acute myeloid leukemia (AML) is a heterogeneous type of blood cancer that infects myeloid class of blood cells and leads to over proliferation of immature ones, especially in elderly individuals. 1,2 Acute promyelocytic leukemia (APL) is one of the most curable subtypes of AML, which can affect children and younger people more and differentiates by high amounts of hyper granular promyelocytes in the bone marrow and blood. 3 It is characterized by a specific chromosome reciprocal translocation t(15,17) (q22,q21), yielding fusion of the genes PML and RARα to code the PML-RARα chimeric protein. 4 This protein has been considered to have a critical role in APL pathogenesis, while blocking apoptosis and maturation of promyelocytes. 5 Although it has been mostly confirmed that all-trans retinoic acid (ATRA) induces complete remission in near 90% of APL patients, about half of the patients treated with ATRA may develop the ATRA-related syndrome. 2,6 So, there is still intense interest in new chemicals and strategies with therapeutic potential for APL. Dithiocarbamates (DTCs) are a class of small organic metal-chelating compounds with various applications in medicine and agriculture. 7 These compounds have both prooxidant and antioxidant activities and have been used for the treatment of fungal and bacterial infections, AIDS, and recently cancer. Incredible capacity of DTCs to form stable metal complexes is most critical for the use of them as superior ligands. 8 Whether or not oxidative stress is considered as an important part of apoptosis. Debates based on studies indicate that oxidants or prooxidant agents in lower doses are capable to triggering apoptosis. DTCs consist of a group of molecules that have been recently used for the redox regulation of apoptosis. 9 It has been reported that the anticancer effect of these chemotherapeutic compounds could be due to the induction of oxidative stress and reactive oxygen species (ROS)-mediated cell death. 10 Also, it seems their capacity to form a copper complex, leading to the induction of apoptosis, can be discussed as a critical therapeutic aspect. 11 Apoptosis is a tightly programmed mode of cell death with biochemical pathways that contributes to the elimination of unwanted cells. 12 It is regulated by several molecular mechanisms. The Bcl2 family and inhibitor of apoptosis proteins (IAPs) are key regulators of cell death. 13,14 Bcl homology (BH) domains of Bcl2 family control the protein interactions and lead the family to divide into three subgroups. Antiapoptotic subgroup I such as Bcl2 and Bcl-xl suppress cell death and proapoptotic subgroups II and III such as Bax, Bak, Bid, and Bimcan lead to induce apoptosis. 15,16 The Bcl2 protein binds to the outer surface of mitochondria and separates proapoptotic members such as Bax from mitochondria, leading to prevent apoptotic cell death. 17 IAPs are defined by the presence of at least one baculovirus IAP repeat (BIR) domain and block apoptosis induced by a variety of triggers. 18,19 Survivin (BIRC5) as a member of IAPs is known to bind and inhibit caspases-3 and -7 that act as final effectors in apoptotic protease cascades. 20,21 The signals of carcinogenesis modulate the central control points of the apoptotic pathways, including two above protein families and yield suppression of apoptosis. 22,23 Cancer cells can acquire resistance to apoptosis by the overexpression of antiapoptotic proteins like Bcl2 and Survivin or by the downregulation of proapoptotic proteins such as Bax. 15 In the previous research, we reported oxidative stress-induced apoptosis in chronic myeloid leukemia K562 cells by 2-nitro-1-phenylethylpiperidine-1-carbodithioate (2-NDC) from the DTC family 24 (Figure 1). In this study, we investigated cytotoxicity, cell cycle arrest, oxidative stress, and apoptosis-inducing effects of the 2-NDC in human promyelocytic leukemia NB4 cells. It has been demonstrated that 2-NDC induces apoptosis through upregulation of Bax and downregulation of Bcl2 and Survivin. Molecular docking studies were investigated to evaluate the interaction between 2-NDC and Bcl2 and Survivin proteins.
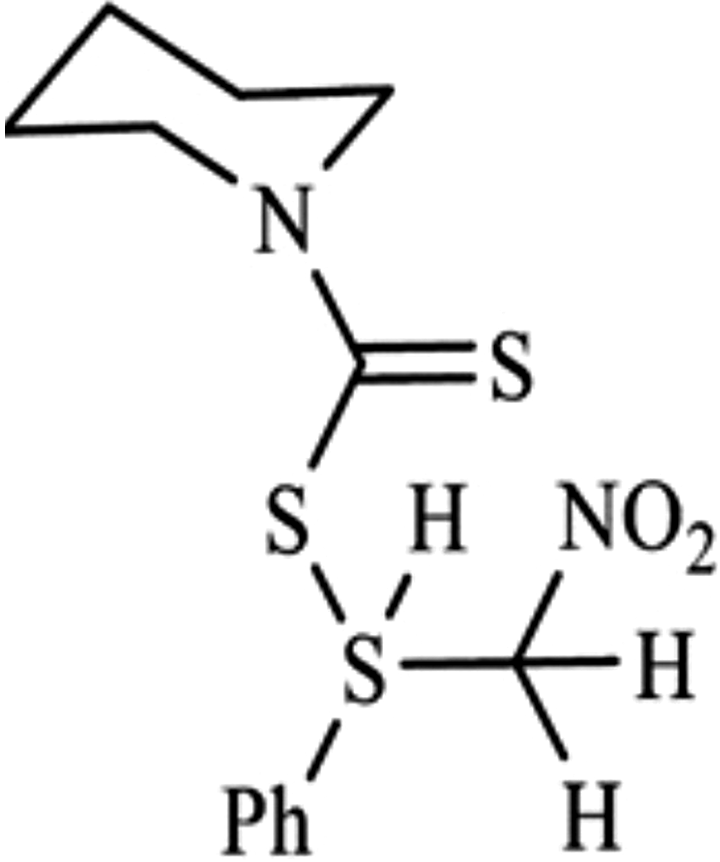
Chemical structure of 2-NDC. 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate.
Materials and methods
Cell culture and reagents
The RPMI1640 medium and fetal bovine serum (FBS) were obtained from Gibco (BRL, Life Technologies, Paisley, Scotland). Penicillin–streptomycin was obtained from Sigma Chemical Company (Germany). The NB4 APL cells were obtained from Pasture Research Institute, Tehran, Iran, and grown in RPMI1640 medium, supplemented with 10% heat-inactivated FBS (both from Gibco BRL, Life Technologies) and 1% penicillin–streptomycin, 100 U/ml penicillin, and 100 µg/ml streptomycin (from Sigma Chemical Company) and maintained at 5% CO2 and 37°C. The culture plates and flasks were purchased from SPL (Korea). The 2-NDC with the general formula of (R1R2) N–(C=S)–SX and molecular weight of 3.365 g/l was synthesized in Dr. Saeed Balalaie Laboratory (Department of Chemistry, Faculty of Science, K. N. Toosi University of Tech, Tehran, Iran). This compound was prepared according to the previously described methods. 25 MTT [3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide], ATRA, propidium iodide (PI), ethidium bromide (EtBr), acridine orange (AO), and 2′-7′-dichlorodihydrofluorescein diacetate (DCFH-DA) were obtained from Sigma-Aldrich Company (Germany). Dimethyl sulfoxide (DMSO), ethanol, isopropanol, and chloroform were purchased from Merck Company (Germany). Annexin V-FITC apoptosis kit was purchased from BD Biosciences (USA). RNA extraction kit purchased from SinaClon (Iran). Complementary DNA (cDNA) synthesis and real-time polymerase chain reaction (PCR) were performed using the PrimeScript™ RT reagent kit (Takara, Japan), and RealQ Plus 2x Master Mix Green High ROX™ was provided from AmpliQon (AmpliQon, Denmark). All gene primers including Bax, Bcl2, GAPDH, and Survivin were obtained from Tag Copenhagen A/S (Denmark).
Cell viability analysis
Cell viability was determined using the MTT, as a reagent functioning by its mitochondrial-dependent reduction to the formazan crystals. 26 For evaluation of the cytotoxicity effect of the 2-NDC, the NB4 cells were seeded at a density of 3 × 104 cells/well in a 96-well plate and incubated in RPMI 1640 with 10% FBS, then treated with different concentrations of the compound for 24–72 h. 27 Twenty microliters of MTT (5 mg/ml) were added to the medium for 3–4 h. Then, the supernatant was removed and the DMSO dissolved the formazan crystals. The optical density of the solution was measured at 570 nm using ELISA reader (Bio-Tek, USA).
Morphological study of apoptotic cells
AO/EtBr staining is a valid and easy technique for the detection of apoptotic and necrotic cells. 28 The cells (5 × 105 cells/well) were seeded in a cell culture plate and treated with 2-NDC (at IC50 value) for 24–72 h. Then, the cultures were centrifuged, supernatants were eliminated, and the cells were stained by 20 μl mixture of AO/EB (1:1, 100 μg/ml) for 5 min. Finally, the photos were imaged by a fluorescence microscope (Olympus BX41, Hamburg, Germany). The viable cells appear uniformly green with AO and EtBr staining. Cells in the early stages of apoptosis are observed light green and orange to red color were detected in the late-stage apoptotic cells and necrotic cells.
DNA fragmentation assay
DNA ladder assay is a simple sensitive and rapid method for estimating apoptosis in single cells. NB4 cells (5 × 105 cells/well) treated with a specific dose (IC50 value) of 2-NDC for 24–72 h were collected and centrifuged. Afterward, the samples were resuspended in 500 µl of lysis buffer (CL buffer, including Tris-HCl, sodium chloride, Triton X-100, and H2O) and incubated at room temperature for 10 min. Then, the DNA was extracted with chloroform/isoamyl alcohol/isopropanol and dissolved in distilled water. An aliquot of each sample was subjected to electrophoresis in a 1.5% agarose gel containing EtBr.
Cell cycle analysis
This method in cell biology employs flow cytometry to distinguish cells in four phases of the cycle (Sub G1, G1, S, and G2/M). Before analysis, the cells were permeabilized and treated with PI that stains DNA quantitatively. 29 NB4 cells (5 × 105 cells/well) were cultured in a 96-well plate and treated with 10, 20 (IC50 value), and 90 µM of 2-NDC for 72 h. Then, the cells were washed with PBS. Afterward, 50 µg/ml PI containing 20 µg/ml ribonuclease A and 1 µg/ml Triton X100 was added and incubated for 1 h at room temperature. The cells were then analyzed by flow cytometry (PI has excitation/emission maxima of 493/632 nm). The results determine the percentage of cells in each phase, and increasing in the Sub G1 cell population of the treated cells confirms the inducing of apoptosis.
Annexin V/PI double staining assay
This assay is based on the ability of the protein Annexin V to bind to phosphatidylserine (PS) exposed on the outer membrane in apoptotic cells. 30 The NB4 cells (1 × 106 cells/well) after treatment with 2-NDC (IC50 value) for 24–72 h were harvested and washed with PBS and resuspended in the prediluted binding buffer (1×). Then, 5 µl of Annexin V-FITC and 5 µl of PI were added. The mixture was incubated in the dark at room temperature for 15 min and measured by flow cytometry at excitation/ emission of 535/617 and 494/518 nm, respectively, for PI and FITC.
ROS level detection
Biological systems generate ROS during aerobic metabolism. 31 For the measurement of ROS level in this study, flow cytometric analysis was performed using DCFH-DA. This reagent is a stable dye that permeates into the cells and hydrolyzes by intracellular esterase to DCFH, which is trapped within the cells. DCFH2 would be oxidized to 2′-7′-dichlorofluorescein (DCF), in the presence of H2O2. 32 NB4 cells were cultured in a six-well plate and treated with 20 µM of 2-NDC and 2 µM of ATRA, in addition to a combined concentration of both for 24–72 h. The cells were incubated with 50 µl of DCFH-DA (10 µM) for 20 min. The general amount of produced peroxide was determined by an increase in DCF using flow cytometry at 485 nm excitation and 530 nm emission.
Real-time PCR analysis
The real-time PCR is a laboratory method to amplify and detect targeted fragments in a DNA molecule by a fluorescent dye. After the treatment of the NB4 cells with 2-NDC (at IC50 value), total RNAs were extracted from cells using RNX-plus reagent. Then, 1 µg of RNA was reverse transcribed into cDNA using Reverta-L kit (Amplisens, Russia). 33 Specific primers with the following sequences were used: GAPDH forward, 5′-CAAGGTCATCCATGACAACTTTG-3′, reverse, 5′-GTCCACCACCCTGTTGCTGTAG-3′; Bax forward, 5′-GCAAACTGGTGCTCAAGG-3′, reverse, 5′-ACTCCCGCCACAAAGA-3′; Bcl2 forward, 5′-TGGGAAGTTTCAAATCAGC-3′, reverse, 5′-GCATTCTTGGACGAGGG-3′; and Survivin forward, 5′-CAGATTTGAATCGCGGGACCC-3′, reverse, 5′-CCAAGTCTGGCTCGTTCTCAG-3′.
The real-time PCR condition was as follows: 1 cycle of initial denaturation at 95°C for 15 min, 40 cycles of amplification at 95°C for 30 s, 63°C for 30 s, and 72°C for 30 s, with a single fluorescence acquisition. GAPDH gene was used as an internal control.
Molecular docking studies of Bcl2 and Survivin proteins
For docking studies, three-dimensional structures of antiapoptotic proteins such as Bcl2 and Survivin were retrieved from the RCSB protein data bank with their respective PDB id’s 2W3L and 1E31. Studies were performed with these proteins and the 2-NDC using the automated docking tool, AutoDock 4.2.6. All calculations dockings were performed using the Lamarckian Genetic Algorithm method. Then, affinity of the compound to interact with Survivin and Bcl2 was assessed. The interactions of complex Survivin and Bcl2 with ligand conformation were analyzed using UCSF Chimera. 34,35
Statistical analysis
The data were analyzed using a general linear model procedure by SPSS software (version 24), and Tukey’s multiple range tests were used for mean comparison at 5% probability level.
Results
2-NDC inhibits growth and viability of NB4 cells
The effects of different concentrations of the 2-NDC (10–120 µM) on growth and viability of NB4 cells (3 × 104 cells/well) were studied for 24–72 h, using MTT assay as mentioned in materials and methods. 2-NDC decreased the viability of the cells in a dose and time-dependent manner. The results indicated that the cell viability was reduced in a 10–30 µM dose of the 2-NDC. In contrast, it was increased significantly at high doses (40–120 µM) and time (>72 h), in comparison to low concentrations and proved a progressively lower toxic effect. So it sounds there is an inverse relationship between concentration and viability. The IC50 value, following 24 h exposure, was found to be 20 ± 1 µM (Figure 2). According to an analysis of variance, the significance of different mean squares was confirmed at p < 0.05.
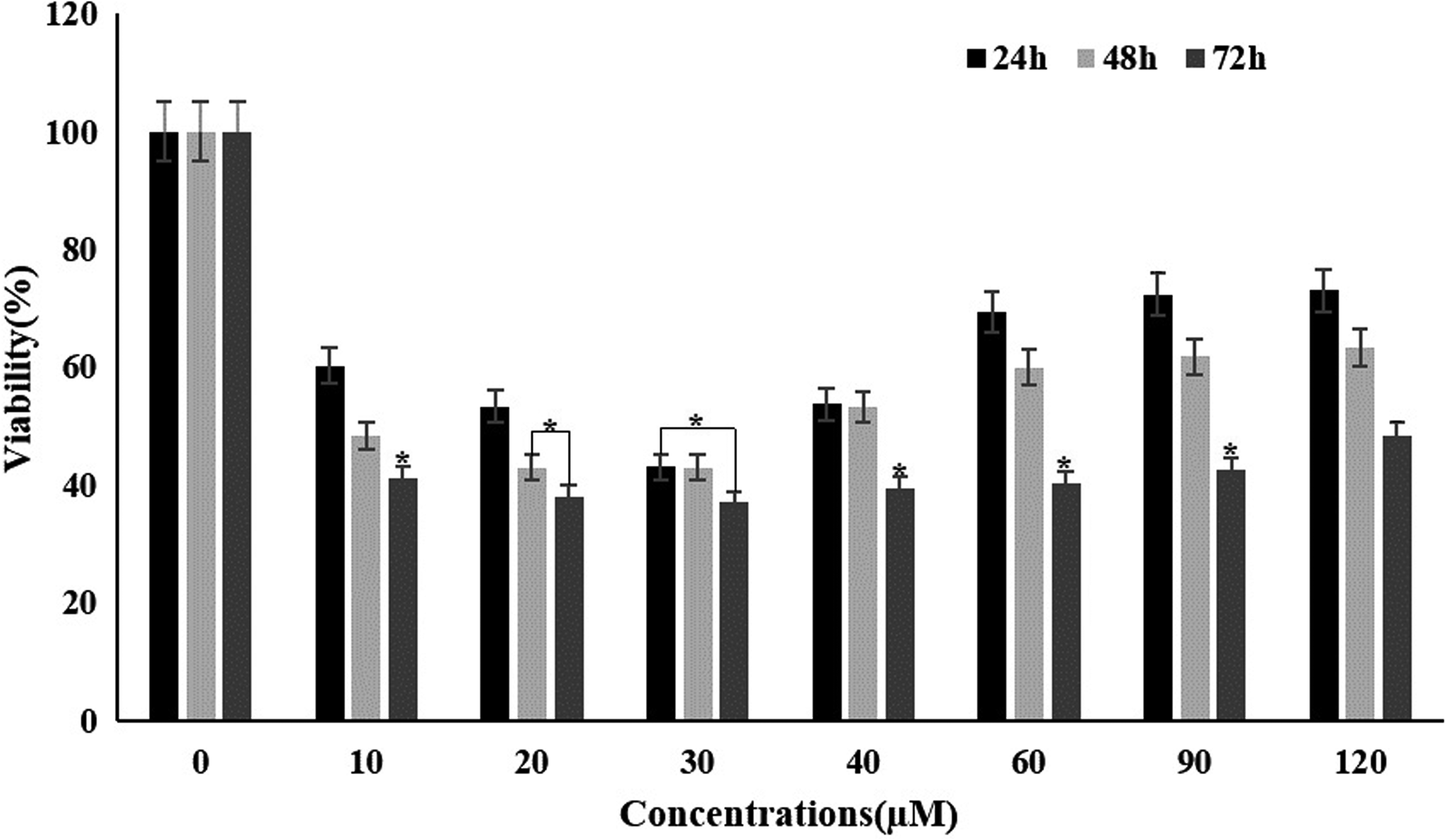
Effect of the 2-NDC on cell viability of NB4 cells. The cells were treated with various concentrations of the compound (10–120 µM) during 24–72 h. Cell viability percentage was measured by MTT assay, using corresponding controls. Data shown are the mean ± SD of three independent experiments.*p < 0.05 values were obtained by comparing each group with its control. 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; MTT: 3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide; SD: standard deviation.
2-NDC enhances ATRA-induced viability inhibition in NB4 cells
To determine the effect of 2-NDC on the viability of NB4 cells in combination with ATRA, 3 × 104 cells/well were treated with 20 µM (IC50 value) of the 2-NDC and 1–2 µM of ATRA for 24 h. The percent of cell viability was measured by the MTT assay. The results showed that the viability of the cells was diminished by more than 85% and it was decreased by 70% upon 24 h of treatment with 2-NDC and ATRA (Table 1). As shown in Table 1, the effect of 2-NDC (at IC50 value) on NB4 cells in combination with ATRA (1–2 µM) caused a significant decrease in cell viability compared to the 2-NDC treatment. It confirmed that 2-NDC improves ATRA efficacy on growth inhibiting in NB4 cells.
The NB4 cells were treated with an IC50 value of 2-NDC, two concentrations of ATRA, and combinations of 2-NDC and ATRA for 24 h.a
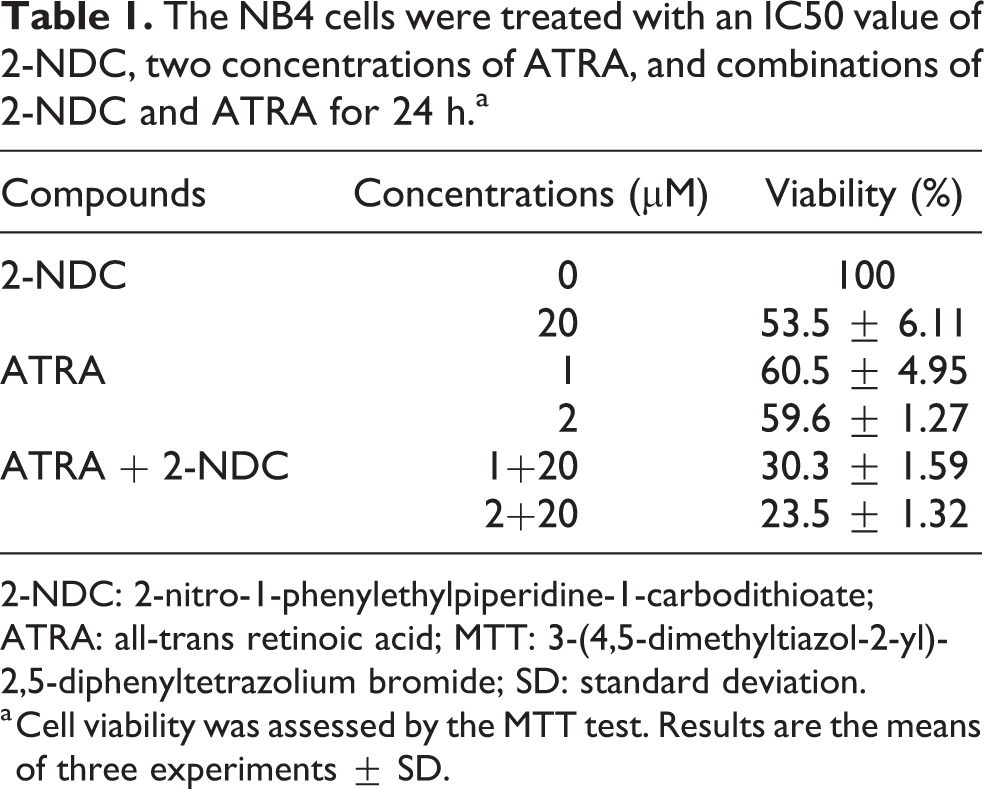
2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; ATRA: all-trans retinoic acid; MTT: 3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide; SD: standard deviation.
a Cell viability was assessed by the MTT test. Results are the means of three experiments ± SD.
Morphological evaluation of apoptotic cells
To evaluate the morphological changes, the treated NB4 cells with 2-NDC were studied by optical and fluorescence microscopy after 24 h. The control cells had a round morphology, whereas treated cells demonstrated chromatin condensation and cell shrinkage as morphological features of apoptosis. After staining the cells with AO and Et/Br, the viable cells were uniformly green. In contrast, early and late apoptotic cells had condensed bright green and orange chromatin, respectively (Figure 3(a)). Furthermore, the formation of DNA ladders on the gel electrophoresis upon the treatment of NB4 cells with 2-NDC confirmed apoptosis (Figure 3(b)).
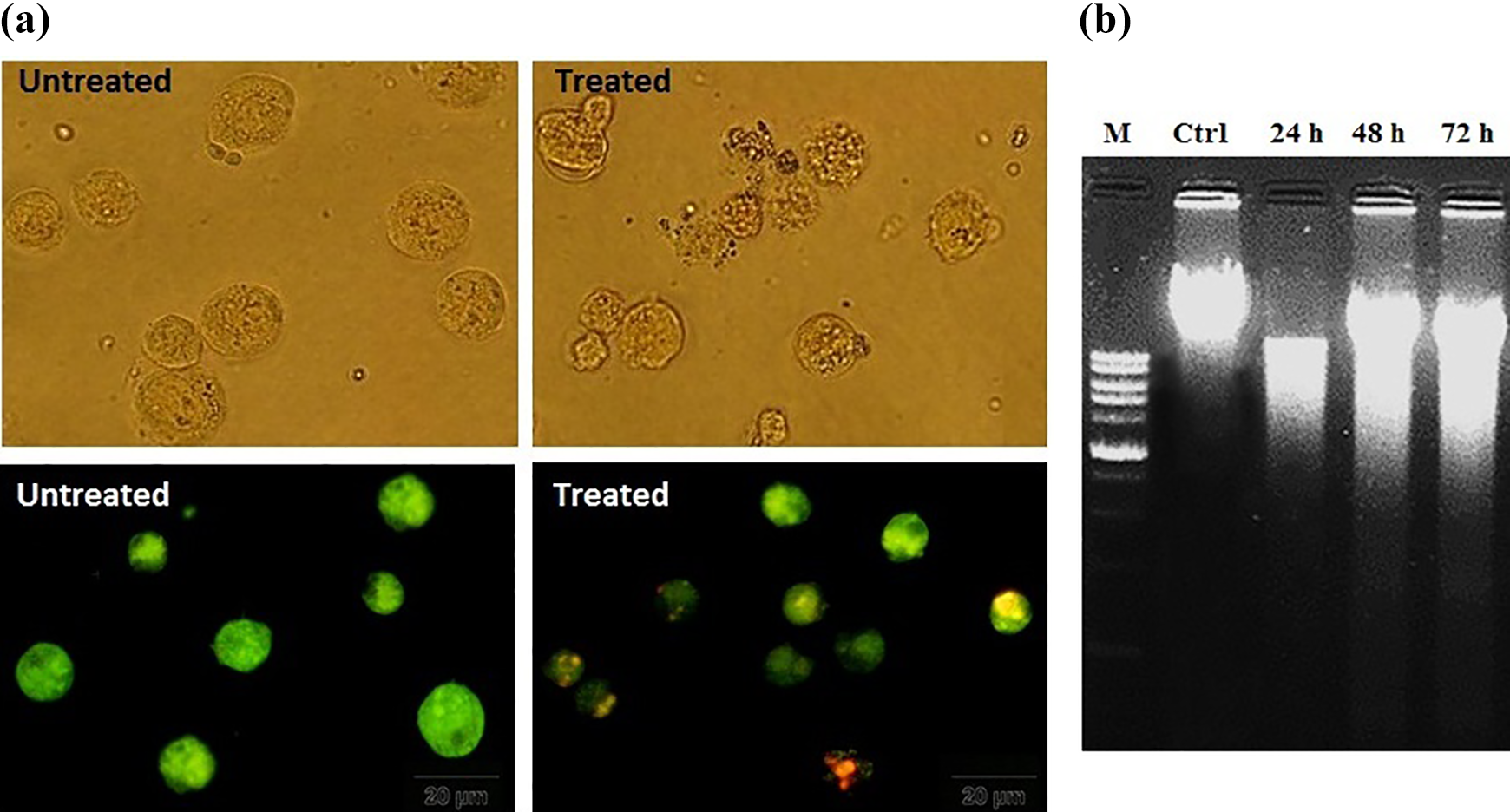
(a) Fluorescence microscopy of the control and treated NB4 cells with IC50 value of 2-NDC after staining with AO/EtBr. (b) DNA fragmentation was detected after treatment of NB4 cells with 20 µM of 2-NDC for 24–72 h. 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; AO/EtBr: acridine orange/ethidium bromide.
Flow cytometric assessment of cell cycle and apoptosis
The flow cytometric method was used to confirm apoptosis. The distribution of the cells in different phases of the cell cycle was studied. The results indicated that the number of cells in the Sub G1 phase was increased after 72 h treatment with 10, 20, and 90 µM of 2-NDC. As it is shown in Figure 4(a), the cell cycle distribution of NB4 cells was among Sub G1, G0/G1, S, and G2/M phases by almost 4%, 51%, 22.9%, and 22.1%, respectively, for the control group. The percentage of cells in the Sub G1 phase was increased to 32.5%, 40.8%, and 18% after 72 h treatment with 10, 20, and 90 µM of the compound compared to the control (untreated cells). According to our MTT assay’s results, it infers that the reduction of cell population in the Sub G1 phase after 90 µM treatment of 2-NDC could be a confirmative point for its low toxic effect in high doses. To confirmation of the 2-NDC anticancer effect in association with ATRA, evaluation of cell cycle phases was performed. After NB4 cells culturing in a six-well plate, the cells were treated with 2 µM of ATRA and a combination of two compounds for 72 h. As it is inferred from the results, treatment of the cells with ATRA shifted them 27.9% to the Sub G1 phase in comparison to the control group, whereas it was enhanced by more than 90% in combination therapy. So, this finding suggested that the 2-NDC improves Sub G1 arrest and ATRA-induced apoptosis in NB4 cells. The detection of surface-exposed PS by Annexin V-FITC/PI double staining has been shown as an early and general marker of apoptosis. According to Figure 4(b), the cells in the lower left quadrant showed viable cells. However, early apoptosis, late apoptosis, and necrosis were discriminated between lower right, upper right, and upper left quadrants, respectively. Flow cytometric analysis revealed that the treatment of the NB4 cells with 2-NDC (at IC50 value) after 72 h causes nearly 70% of cells to die through apoptosis.

(a) Cell cycle analysis of the NB4 cells after 72 h treatment with 10, 20, and 90 µM of 2-NDC and 2 µM of ATRA. The percentage of cells in each phase was measured using flow cytometry. (b) Apoptosis was also confirmed in treated NB4 cells with 20 µM of 2-NDC by Annexin V/PI double staining assay. Flow cytometric analysis indicated a shift from low left quadrant panel (untreated cells) to low right (early apoptosis) and upright quadrant panel (late apoptosis). 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; ATRA: all-trans retinoic acid; PI: propidium iodide.
Assessment of ROS level in NB4 cells
It has been approved that most chemotherapeutic agents can trigger apoptosis by the elevation of intracellular ROS level and change in redox homeostasis of cancer cells. 10 To examine ROS production in treated NB4 cells by 2-NDC and ATRA, all steps mentioned in the methods section were performed. According to the results, 2-NDC (20 µM) increased ROS generation by 13.7% after 24 h. The amount of ROS level was reduced by 5.2% and 17.5%, respectively, during 48 and 72 h of treatment, compared to 24 h. It can be a confirmative point for the antioxidant effect of 2-NDC in longer times. 24 As it is demonstrated in Figure 5, ATRA (2 µM) raised the amount of ROS production by 10.5% in contrast to control cells, as well as the 2-NDC enhanced it by 8% during 24 h of treatment. Therefore, it could be perceived that 2-NDC in association with ATRA can extend the intracellular ROS level, leading to apoptosis.

Effect of the 2-NDC and ATRA on intracellular ROS generation. The NB4 cells were treated with 20 µM of 2-NDC and 2 µM of ATRA, in addition to a combination of both for 24–72 h. The general amount of ROS level was assessed by flow cytometry after DCFH-DA incubation. 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; ATRA: all-trans retinoic acid; ROS: reactive oxygen species; DCFH-DA: 2′-7′-dichlorodihydrofluorescein diacetate.
Evaluation of Bax, Bcl2, and Survivin genes expression in NB4 cells upon 2-NDC treatment
We used a real-time PCR technique for the examination of Bax, Bcl2, and Survivin genes expression in NB4 cells before and after treatment with 20 µM of 2-NDC. According to Figure 6(a), the expression of proapoptotic Bax gene in the treated cells was increased significantly after 24 h. Upregulation of genes occurred gradually during 48 and 72 h. As shown in Figure 6(b), the downregulation of Bcl2 as an antiapoptotic gene was observed after treatment with 2-NDC. The levels of relative expression of Bcl2 were almost equal in 48 and 72 h. Also, the expression of Survivin gene was reduced remarkably after 24–72 h treatment with the compound (Figure 6(c)). The insignificant amount of changes in the expression level of three evaluated genes during 48 and 72 h of exposure could be clarified by the results achieved from ROS detection.

Evaluation of Bax, Bcl2, and Survivin genes expression. Bax (a), Bcl2 (b), and Survivin (c) genes relative expression level by real-time PCR during 24–72 h treatment with 20 µM of 2-NDC. Data shown are representative of three experiments as mean ± SD (*p < 0.05, **p < 0.01). 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; PCR: polymerase chain reaction; SD: standard deviation.
Ligand–Bcl2 and ligand–Survivin interactions analysis
The interactions of the 2-NDC with Bcl2 and Survivin were evaluated using Auto Dock 4.2. Analysis of the docking results indicated that the BH domain binding site of Bcl2 interacts with 2-NDC. According to the results of Auto Dock 4.2 runes, its binding energy was calculated as −6.73 kcal/mol. Furthermore, the results showed that 2-NDC can interact with the BIR domain binding site of Survivin and its binding energy is −8.02 kcal/mol. It proves that the 2-NDC has a high affinity to interact with Survivin, in comparison to Bcl2, because of its lowest binding energy and inhibition constant (Ki; Table 2). The UCSF Chimera software also analyzed the complexes of two proteins with 2-NDC. The interactions of 2-NDC with Bcl2 and Survivin binding sites residues are shown in Figure 7(a) and (b). In the interaction between Bcl2 and 2-NDC and amino acids Lys22 and Arg26 with shorter distances than 2Ȧ between donor and acceptor plays an important role. However, amino acids Glu63 and Lys62 are the most important ones with shorter distances than 2.5Ȧ in Survivin binding site.
The results of inhibition constant and binding energy values of the 2-NDC compound docked with Survivin and Bcl2.

2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate.

Interaction between the 2-NDC from DTCs and the BIR domain binding site of Survivin (a) and BH domain binding site of Bcl2 (b). The important amino acids in the interaction between 2-NDC and two proteins are indicated with black arrows. 2-NDC: 2-nitro-1-phenylethylpiperidine-1-carbodithioate; DTC: dithiocarbamate; BIR: baculovirus inhibitor of apoptosis protein repeat; BH: Bcl homology.
Discussion
Although it has been considered that typical chemotherapeutic agents completely cure most of the patients with APL, there are still elusive factors of drug resistance in blood cancer cells. 36,37 Therefore, new therapy strategies and synthetic chemical compounds against leukemia are recent of intense interest. 2 DTCs as a family of sulfur-based metal-chelating compounds have cell growth inhibition and apoptosis-inducing effects. 38 It has been reported previously that certain members of DTCs such as diethyl dithiocarbamate (DEDTC), ffiram (DSF), and pyrrolidine dithiocarbamate (PDTC) could bind with tumor cellular copper, leading to inhibit tumor growth through the inhibition of ubiquitin-proteasome activity and induction of cancer cells apoptosis. 8,38 DSF and PDTC are also known as the inhibitors of the signaling pathway that cause to induction of apoptosis. 39 –41 Furthermore, PDTC and DEDTC are approved to initiate redox active copper-mediated apoptosis in thymocytes. 42 Since cancer cells have a higher demand of iron and copper ions for growth, di-2-pyridul hydrazone dithiocarbamate 5-acetic acid as a potent derivative of DTCs possesses an excellent metal-chelating ability. It disturbs the metal homeostasis leading to inhibit the proliferation of cancer cells through upregulation of p53. 43 In the present research, we demonstrated that 2-NDC from DTCs induced apoptosis in NB4 cells via overexpression of Bax and a concomitant downregulation of Bcl2 and Survivin. It has been previously suggested that 2-NDC can initiate oxidative stress-induced apoptosis in chronic myeloid leukemia K562 cells. 24 This compound caused a significant decrease in the viability of treated NB4 cells with low concentrations (10–30 µM). However, a proliferative inducing effect in higher doses (>30 µM) was observed. The biphasic effect of DTCs on growth inhibition and apoptosis induction in different cultured cancer cells has been reported. 42 For confirmation of prooxidant effect, PDTC in low doses (100µM>) induced apoptosis by inhibition of ERK and NF-κB pathways. Nevertheless, it appeared as an antioxidant and caused to ascend the viability and proliferation during treatment of cells with higher concentrations (100–150 µM) through overexpression of phospho-ERK and NF-κB activity. 24,44 According to previous studies and also obtained results from MTT assay and measurement of ROS level in our research, it can be inferred that the 2-NDC has an antiproliferative effect at low concentrations and times as well as an inducing proliferation property in high doses and times. The effect of 2-NDC on NB4 cells growth and viability was also evaluated in combination with ATRA. According to the results, 2-NDC enhanced ATRA-induced apoptosis in NB4 cells by 70% during 24 h of treatment. It has been proved that combination therapy, especially about APL patients, is more efficient than treatment with ATRA alone. In an evaluation of APL patients undergoing treatment with ATRA and ATRA in combination with arsenic trioxide (ATO), complete remissions were achieved in 57% with ATRA and 100% with ATO/ATRA. 45 Furthermore, we confirmed the occurrence of apoptosis in treated NB4 cells with 2-NDC, using several considerable assessments. Morphological studies and DNA fragmentation assay indicated the induction of apoptosis in treated NB4 cells during 24–72 h. In addition, due to the close relevance of apoptosis to the cell cycle, different phases of the cell cycle were analyzed in NB4 cells before and after treatment. The results revealed that there is a reciprocal association between the decrease in proliferation and Sub G1 arrest. The apoptosis was also evaluated by Annexin V/PI double staining assay. In order to complete the previous results 24 and confirm the 2-NDC redox-regulated apoptotic effect on NB4 cells, the amount of intracellular ROS was detected via flow cytometry. The real-time PCR results showed that upregulation of Bax and downregulation of Bcl2 and Survivin occur in treated NB4 cells with 2-NDC. Apoptosis induction and altered gene expression levels of Bax and Bcl2 were also reported previously by Li et al. after the treatment of the HeLa cells with an active derivative from DTCs. 46 Survivin and Bcl2 are regulated at two levels, gene expression (measured at the RNA level) and protein activity. To confirm the apoptotic-inducing effects of the 2-NDC through downregulation of Survivin and Bcl2, we analyzed the molecular docking interaction of the compound with Survivin and Bcl2. The docking studies demonstrated that 2-NDC had a higher binding affinity (ΔG: −8.02 kcal/mol) to bind to the BIR domain of Survivin, than the BH domain of Bcl2 (ΔG: −6.73 kcal/mol). These data confirmed the real-time PCR findings. Downregulation of Survivin was more impressive than Bcl2 at the gene expression level following the treatment of 2-NDC (Figure 6). However, coordination between docking study and gene expression findings cannot be sufficient alone and more experimental data (protein activity) are required for evaluating the role of 2-NDC in the Bcl2 and Survivin expression pathways.
Conclusion
In conclusion, following previous studies, we reported that the compound from the DTC family (2-NDC) has an antiproliferative effect and high apoptosis-inducing activity in the human APL NB4 cells. It was achieved using several methods. Induction of apoptosis occurred through overexpression of Bax and downregulation of Bcl2 and Survivin. Considering our findings, this compound can be a great candidate for more pharmacological researches and suggested as an efficient chemotherapeutic agent in cancer therapy.
Footnotes
Acknowledgments
The authors appreciate the support of this investigation by the research council of the University of Tabriz, Tabriz, Iran. We would like to thank Dr Saeed Balalaie for providing N-(2-nitro-1-phenylethylpiperidine-1-carbodithioate (2-NDC).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
