Abstract
Because cadmium (Cd) is not naturally degradable by ecosystems, it interferes with many types of food chains. Cd accumulates in the kidney, liver and in the nervous tissues, especially the brain. The neurotoxicity of Cd is very high, as it alters the integrity, and increases the permeability, of the blood–brain barrier. Cd penetrates and accumulates in neurons in the brains of rats. This study reveals that Cd decreases antioxidant enzymes and increases oxidative stress in the brain. In addition, Cd increases lipid peroxidation of brain tissues. Cd increases the expression of the Cu/Zn superoxide dismutase gene. It also affects cholinergic, glutamatergic, gamma-Aminobutyric acid (GABAergic), dopamine, serotonin and acetylcholine neurotransmitters in brain tissue. Consequently, Cd increases the formation of amyloid β, a neurotoxic index, and induces apoptosis by changing the quality and the quantity of Bcl-2, Bax and p53 proteins. In conclusion, both selenium and nanoselenium show potential antioxidant activity and promote recovery from the neurotoxic action of Cd.
Introduction
Humans can be exposed to cadmium (Cd) in various ways, such as from industrial activity, cigarette smoking and its accumulation in food. 1 The total intake of Cd from food is 2.8–4.2 mg/kg body weight (b. wt.)/7 days. 2 Cd has a long lifespan (15–30 years), is not degradable and is very slowly eliminated from the human body. 3 Cd accumulates in the kidney, liver and in the nervous tissues, especially the brain. 4 Cd has a very high neurotoxicity because it alters the integrity of the blood–brain barrier (BBB) 5 ; it increases BBB permeability, allowing the chemical to penetrate and accumulate in neurons of rat brains. 6
Cd triggers oxidative stress in the nervous tissue and affects endocrine functions. 7 In addition, the cytotoxicity of Cd may be due to oxidative damage resulting from an increase in reactive oxygen species (ROS), such as super oxide anion, hydrogen and lipid peroxide. 8 The oxidative stress can be magnified by the peroxidation of unsaturated fatty acids, which leads to high levels of lipid peroxidation by-products, for example, malondialdehyde (MDA). 9 In addition, Cd depletes glutathione (GSH) 10 and other antioxidant enzymes, for example, superoxide dismutase (SOD), catalase (CAT), GSH peroxidase (GPx) and GSH reductase (GR) in vivo and in vitro 11 by ligation. 12 Cuypers 13 showed that chronic exposure to Cd is associated with a dysregulation in the gene expression of the most vital antioxidant enzymes. The most abundant isoform of SOD is Cu/Zn SOD (SOD1), and it is a good marker for the antioxidant system. 14 Several studies have demonstrated that MDA is the final by-product of the lipid peroxidation associated with Alzheimer disease (AD) 15,16 and other neurotoxicity. García-Blanco 17 investigated that mild cognitive loss and AD are directly related to a decrease in the activity of antioxidants. In accordance with Eizenberg et al., 18 the neurotoxicity of Cd is caused by its accumulation in the brain and oxidative damage induced by the impairment of cell regulation and the alteration of antioxidant gene expression.
Cd can deflect excitatory and inhibitory neurotransmission in neurons and synaptic clefts and impact the antioxidant status of the brain. 19 Cd can effect cholinergic neurotransmission by altering the activity of acetylcholinesterase (AChE). 1 The loss and blockage of cholinergic transmission could be related to the Cd-diminishing memory function 20 and motor dysfunctions. 21 Caride et al. 22 showed that the alteration of BBB integrity by Cd increases dopamine levels and decreases gamma-aminobutyric acid (GABA) levels in different brain regions. Serotonin levels increased in adenohypophysis and neurohypophysis in rats treated with Cd. 23 The high concentration of glutamate neurotransmitters becomes neurotoxic and is involved in neurodegenerative diseases, including AD. 24 The increase in glutamate levels causes enlightened depletion, reduced GSH and subsequent oxidative stress. 25
Amyloid beta protein (Aβ) is found in the brain and cerebrospinal fluid of humans with good cognitive function and memory throughout their life. 26 The pathological problems with Aβ occur when it is deposited, polymerized, entangled and misfolded into β sheets or plaques. 27 These plaques aggregate and become resistant to biodegradation. 28 Moreover, Cd activates amyloidosis and tau protein fibrillary formation, and it has been associated with unique cognitive dysfunction and the enhancement of AD, 28,29,30 Cd was found to decrease brain α-secretase 31 and increase amyloid precursor protein. 32 These metabolic changes led to an increase in Aβ plaque production and a decrease in their elimination. 30
Oxidative damage to the biomolecules results in metabolic changes, such as the attack of lipid bilayers and intracellular and transmembrane proteins and enzymes, leading to cell death by apoptosis and neurodegeneration. 28 Aβ induces the neurodegeneration of nervous tissues. 33 Apoptosis is known to involve intrinsic or extrinsic pathways using proteolytic caspase enzymes. The extrinsic pathway involves caspase 8 and the intrinsic pathway recruits cytochrome C, which is released into the cytosol and activates caspases 9. 32,34 Apoptosis maintains a normal balance in the brain by two phases: the initiation phase and the execution phase. Firstly, the initiation may involve ligation of a death receptor by tumour necrosis factor (TNF) or in the intrinsic pathway by nuclear transcriptional factor kappa B (NF-κB). 33,35 Apoptosis seems to be adjusted through the balance between the Bcl-2 family proteins, such as pro-apoptotic Bax and pro-survival anti-apoptotic Bcl-2 proteins and their effect on outer mitochondrial membrane permeability. 36 Cd decreased Bcl-2 and Bax in cell line experiments, 37 and Cd induces ROS and increases NF-κB and Bcl-2 to prevent apoptosis. 38 Secondly, p53 tumour suppressor protein regulates the DNA damage-induced cell cycle arrested at the G0. 39 Cd interferes with p53 function by changing its architecture. 40 The increase of p53 and Bax and a decrease in Bcl-2, in oligodendrocytes infected with a virus, result in apoptosis. 41 All of these cytosolic factors, Bcl-2, Bax and p53, are involved in cellular apoptosis 41 ; and oligodendrocytic apoptosis is also enhanced by these cytokines. 42 The p53 protein helps to upregulate Bax and downregulate Bcl-2, 42 which leads to apoptosis of oligodendrocytes. 41,42 Together, Bax, p53, ROS, caspases (9&3) and cytochrome C release leads to Cd-induced apoptosis in vivo 43 and in vitro. 44
Vekariya et al. 42 showed that the action of selenium (Se) involves the selenoenzymes, such as GPx, so it is considered as an antioxidant. Sodium selenite, the main dietary source of Se, is recommended for daily intake and has a very important role in animal and human nutrition. 45 This form is used to treat Se shortage. 46 It has been documented that Se is able to restore the oxidant/antioxidant imbalance induced by heavy metals. 47
GPx isoenzymes reduce hydrogen peroxide, using reduced GSH as a co-substrate. 48 GPx isoenzymes catalyse the reactions that could defend the cellular membrane biomolecules and prevent oxidative stress. 49 It was proposed that the toxicity of Cd is reduced by Se in neuronal cells. 9 It was shown recently that nanoselenium (SeN) has high bioavailability and antioxidant activity, 50 low toxicity and therapeutic and anticancer properties. 51 Many studies recommended the introduction of SeN into the diet, rather than Se, due to its lower toxicity. 52,53 At the nanoscale, SeN has a large surface area to volume ratio, is more efficient and easily absorbed. 53 Therefore, this study aims to investigate the effectiveness of Se and SeN in ameliorating the physiological, molecular and histological changes caused by Cd-induced neurotoxicity in rats.
Materials and methods
Chemicals
Cd sulphate powder and Se supplement were obtained from Sigma Co. (St Louis, Missouri, USA) and Sigma, INC. (New York, New York, USA), respectively. All other chemicals and kits were of high chemical grade and were purchased from standard confirmed companies.
SeN preparation
SeN were prepared in equal mixtures of 25 ml with Milli-Q water (0.22 µm membrane). The last concentration of SeN was 5 mM, and rats were administered intraperitoneal (i.p.) with 222 µg kg− 1 b. wt. for 4 weeks.
Scanning electron microscopy
The scanning electron microscope (SEM) imaging of the SeN was performed using a Joel 6360LA scanning electron microscope (JEOL Ltd, Tokyo, Japan). All samples were coated with a thin layer of gold to improve conductivity, using a JFC-1100E sputter (JOEL Ltd.). The images were captured using an acceleration voltage of 10 kV.
Fourier transforms infrared spectroscopy
The aqueous suspensions of SeN particles were placed as thin films on clean flat Zn Se discs (diameter 2.5 cm, thickness 0.2 cm) and dried in desiccators at 45°C. Infrared spectroscopic measurements were performed on the Fourier transforms infrared (FTIR) spectrometer (Shimadzu FTIR-8400 S, Japan). Spectra were collected in the transmission mode with a total of 64 scans (resolution 2 cm− 1), against the Zn Se disc background and manipulated using the software supplied by the manufacturer (IR Solution software, Version 1.21).
SEM analysis
SEM images of SeN particles (Figure 1) revealed that the obtained SeN are clearly visible as spherical particles throughout the surface of the powder. The average size of the SeN nanoparticles ranged from 70 nm to 300 nm. The surfaces of the obtained particles were waved, not smooth, and could be used for drug delivery.

Scanning electron microscope of nanoselenium particles configuration.
FTIR analysis
Figure 2 shows wave numbers of the FTIR spectra of the synthesized bio-SeN beads obtained in the range of 241–3614 cm− 1. It is noticeable that the most characteristic bands are 3614 cm− 1, suggesting the presence of N-H amide stretching. A band at 3421 cm− 1 corresponds to N–H amine, 2943 to alkane, 1649 to amide C=O stretch, 1535 to C=O, 1429 to CH–H, 1031 to alkaline amine, 526 to CF2 binding and the band 455 to CF2 wagging, 337.
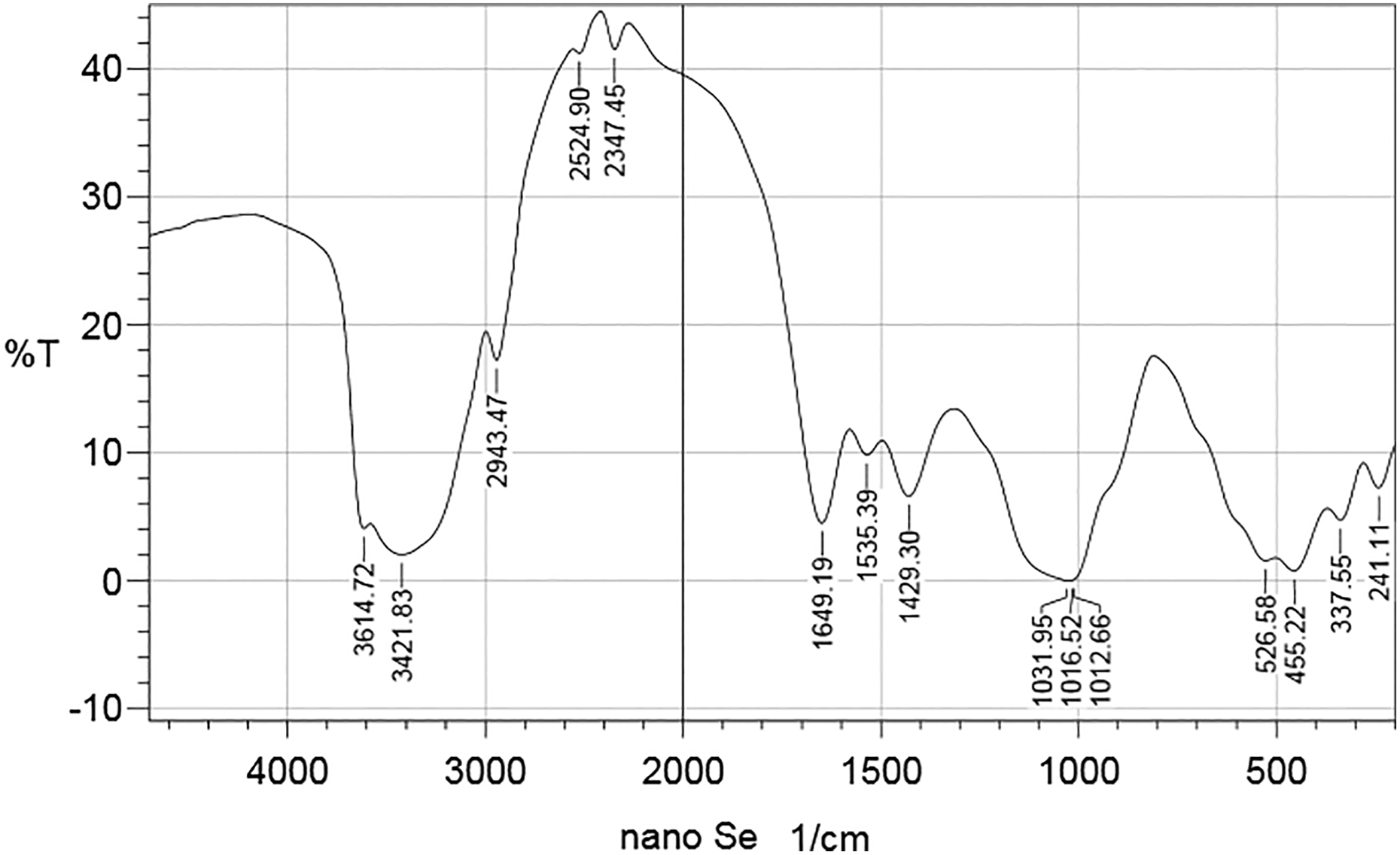
FTIR spectra for the examined organic SeN. FTIR: Fourier transforms infrared; SeN: nanoselenium particles.
Experimental animals
Male Wistar albino rats weighing 150–170 g were housed in stainless steel cages under a regulated light and dark schedule on a 12-h day/night cycle. The environment was controlled ventilation, humidity and temperature (24 ± 3°C) and fed the animals with standard laboratory rodent pellets. Food and water were provided ad libitum. Animals were examined for health status and acclimated to the laboratory environment for one week prior to use. All experiments were conducted in accordance with the standard animal ethics and the study protocol was reviewed and approved by the ethical committee of the Faculty of Science, King Khalid University. The animals were divided into four groups of six rats each.
Animal grouping
After one week of acclimatization, the rats were randomly divided into six groups, six animals in each group. Animals weighing 150–170 g were used in this study. The animals were subdivided into six groups: (1) in the control group rats received normal saline (0.9% NaCl), i.p. for 4 weeks; (2) in the Se group rats received 100 mg/kg b. wt. sodium selenite for 4 weeks 54 ; (3) in the SeN particles group rats received SeN at a dose of 222 µg kg− 1 b. wt. i.p. for 4 weeks; (4) in the Cd group rats were i.p. injected with 30 mg kg− 1 b. wt. of CdCl2 dissolved in saline, for 4 weeks; (5) in the Cd + Se-treated group rats received concomitant doses of CdCl2 (30 mg kg− 1 i.p.) and sodium selenite (100 mg kg− 1 b. wt. i.p.) for 4 weeks; (6) in the Cd + SeN-treated group rats received concomitant doses of CdCl2 (30 mg kg− 1 b. wt. i.p.) and SeN at a dose of 222 µg kg− 1 b. wt. i.p. for 4 weeks.
Blood and brain sample collection
At the end of the experimental period (30 days), fasted rats were anaesthetized with 10 ml kg− 1 b. wt. ketamine/xylazine i.p., 55 and blood was collected by cardiac puncture into non-heparinized tubes and centrifuged at 3000 r min−1 for 15 min. Sera were carefully separated and each sample was put in a clean cup tube, labelled and kept at −80°C until biochemical analysis. The brain samples were separated, cleaned, weighed and homogenized in normal saline to form 10% (w/v) samples, then the homogenate was centrifuged (1000 r min−1) for 10 min in a refrigerated centrifuge. The resulting supernatant was separated and, after labelling, samples were kept at −80°C until biochemical analysis.
Biochemical assays
MDA concentration in brain tissue was determined using a Biodiagnostic Kit (Biodiagnostic, Giza, Egypt). 56 The activity of SOD in brain tissue was determined using a Biodiagnostic Kit (Biodiagnostic). 57 The activity of CAT in brain tissue was determined using Biodiagnostic Kit (Biodiagnostic). 58 The concentration of reduced GSH in brain tissue was determined using a Biodiagnostic Kit (Biodiagnostic). 59 The activity of GPx in brain tissue was determined using Biodiagnostic Kit (Biodiagnostic). 60 The activity of GR in brain tissue was determined using Biodiagnostic Kit (Biodiagnostic). 61 CAT activity was assayed according to the method of Goth. 62
To measure rat TNF-α and NF-κB lymphoma, a specific monoclonal antibody was used for the quantitative sandwich enzyme immunoassay technique, which was performed according to the kit instructions (R&D Systems Inc., Minneapolis, Minnesota, USA). Values of the samples were expressed as pg ml−1. The specific monoclonal antibody was used for the quantitative sandwich enzyme immunoassay technique to determine the Bax and Bcl-2 levels in rats and was used according to the kit instructions (Cloud-Clone Corp., Houston, Texas, USA). The level of Bax was expressed as pg g−1 and Bcl-2 as ng g−1. The level of p53 was determined using a specific monoclonal antibody for rat p53 using the quantitative sandwich enzyme immunoassay technique, according to the kit instructions (RayBio, Peachtree Corners, Georgia, USA). The level of p53 was expressed as ng g−1. The quantitative determination of Aβ in tissue was performed using the sandwich enzyme immunoassay technique with a specific anti-rat monoclonal antibody, according to the kit instructions (Cloud-Clone Corp). The value of Aβ 1–42 in plasma was expressed as pg ml−1. Serotonin was determined using a rat ELISA kit (Bio-source, San Diego, California, USA) by using a method based on biotin double antibody sandwich technology. The shades of a solution and the concentration of rat serotonin are positively correlated. The level of serotonin was expressed as ng ml−1. Quantitative determination of rat dopamine was performed using an ELISA kit (Mybiosource, San Diego, California, USA). The level of dopamine was expressed as ng ml−1. The immunoassay kit (EIAab, Wuhan, China) was used, according to kit instructions, to determine rat GABA concentrations in the samples, by comparing the optical density of the samples to the standard curve; the value was expressed as ng ml−1. An enzyme immunoassay was used for the quantitative determination of glutamate, according to kit instructions (Abnova, Taipei, Taiwan). The quantification of samples was measured by comparing their absorbance with a standard curve prepared with known standards. The value was expressed as μg ml−1. The activity of acetylcholine esterase in brain tissue was determined using the Abnova kit. AChE converts acetylcholine substrate to choline, which is then oxidized by choline oxidase to produce an intermediate. The intermediate reacts with a highly specific probe to generate colour, measured at 570 nm.
Statistical analysis
All grouped data were statistically performed with Prism (GraphPad Prism, 6.01) software. Differences among groups were evaluated by one-way analysis of variance followed by Duncan’s multiple range tests. All values were expressed as the mean ± standard error of six animals per group. Percent of changes in treated groups were calculated.
Histological processing
Fresh portions of the brain from each rat were rapidly cut, fixed in neutral buffered formalin (10%), then dehydrated with grades of ethanol (70, 80, 90, 95 and 100%). Dehydration was followed by clearing the samples in two changes of xylene, impregnated with two changes of molten paraffin wax, then embedded and blocked out; 5-µm thick sections were cut with a microtome (Leica RM 2025, Germany). Paraffin sections were stained and examined for alterations in the renal tissues of each rat, using an optical microscope (Olympus Microscope BP73 with Digital Camera, Japan).
Amplification of the SOD1 gene using RT-PCR
The RNeasy kit was used to isolate RNA according to the manufacturer’s guidelines (QIAGEN, Germany). Hundreds µl of brain homogenate was used to extract RNA. RNAs inhibitors were added to the samples during the RNA extraction procedure in accordance with the kit manual. The first-strand cDNA was synthesized using Moloney Murine Leukemia Virus Reverse Transcriptase (Fermentas, Vilnius, Lithuania, USA), in accordance with the kit manual. SOD 1 primer was used. The sequence of forward primer: 5′ GTCCAGAAAGCCCCAGATACC′3 and reverse primer: 5′GTGACATCCTCAGTTCCTCTTAG′3; 1 µl (100 ug) of the synthesized cDNA was added to 2.5 µl Taq polymerase buffer 10× (Promega, Madison, Wisconsin, USA) containing a final concentration of 1 mM MgCl2, 0.2 mM dNTPs, 0.2 mM each specific primer (20 pmol) and 0.2 µl Taq polymerase (5 U µl− 1) in a final reaction volume of 25µl. The PCR reaction conditions were as follows: initial denaturation at 95°C for 10 min, followed by 27 cycles of 30 s at 94°C, 62°C for 30 min, and 72°C for 1 min. The final extension was at 72°C for 10 min, and finally the reaction was stored at 4°C overnight. Amplification products were electrophoresed in 1% agarose gel run in 0.5× TBE buffer. The gel ran for 60 min and was visualized and photographed using an ultraviolet Trans Illuminator gel documentation system.
Results
Biochemical study
Brain malondialdehyde (MDA) concentration increased and the activity of superoxide dismutase (SOD) and catalase (CAT) was diminished in Cd group (Figure 3). The concentration of MDA and the activity of SOD and CAT became around the control readings.

Brain MDA concentration increases and the activity of SOD and CAT diminishes in Cd group. The concentration of MDA and the activity of SOD and CAT become around the control readings. The values are expressed as the means ± SEM, (n = 6). Significant at *p < 0.05, **p < 0.01, ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. MDA: malondialdehyde; SOD: superoxide dismutase; CAT: catalase; Cd: cadmium.
Brain reduced glutathione (GSH) content and glutathione peroxidase (GPx) and GSH reductase (GR) activity was high decreased in their activity in the Cd group when compared with control groups (Figure 4). GPx and GR activity and GSH level increased in both Cd+Se and Cd+SeN groups related to Cd group.

Brain reduced GSH content and GPx and GR highly decreased in their activity in the Cd group when compared with control groups. GPx, GR and GSH levels increased in both Cd + Se and Cd + SeN groups related to Cd group. Effect of Se and SeN particles on GSH, GPx, and GR activity in rats in different groups during the experimental period. The values are expressed as the means ± SEM (n = 6). Significant at *p < 0.05, **p < 0.01, and ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. GSH: glutathione; GPx: GSH peroxidase; GR: GSH reductase; Cd: cadmium; Se: selenium; SeN: nanoselenium.
Cadmium increased the level of dopamine and glutamate and decreased the level of serotonin and GABA as compared with control groups (Figure 5). Both selenium and nanoselenium particles were able to ameliorate the changes of glutamate, serotonin, GABA and glutamate when compared with Cd group.

Cd increases the level of dopamine and glutamate and decreases the level of serotonin and GABA as compared with control groups. Both Se and SeN ameliorate the changes of glutamate, serotonin, GABA and glutamate when compared with Cd group. The values are expressed as the means ± SEM (n = 6). Significant at *p < 0.05, **p < 0.01, and ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. Cd: cadmium; GABA: gamma-aminobutyric acid; Se: selenium; SeN: nanoselenium.
Serum acetylcholinesterase activity increased in Cd group and the action of Se and SeN was able to restore the enzyme activity to the normal level (Figure 6).

Serum acetylcholinesterase activity increased in Cd group and the action of Se and SeN was able to restore the enzyme activity to the normal level. Effect of Se and SeN particles on serotonin, GABA, and glutamate level in rats in different groups during the experimental period. The values are expressed as the means ± SEM (n = 6). Significant at *p < 0.05, **p < 0.01, and ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. Cd: cadmium; Se: selenium; SeN: nanoselenium.
Brain amyloid β highly increased in Cd group when compared with control groups but the concentration of Aβ decreased in Cd + Se and Cd + SeN groups in comparison with Cd group (Figure 7).
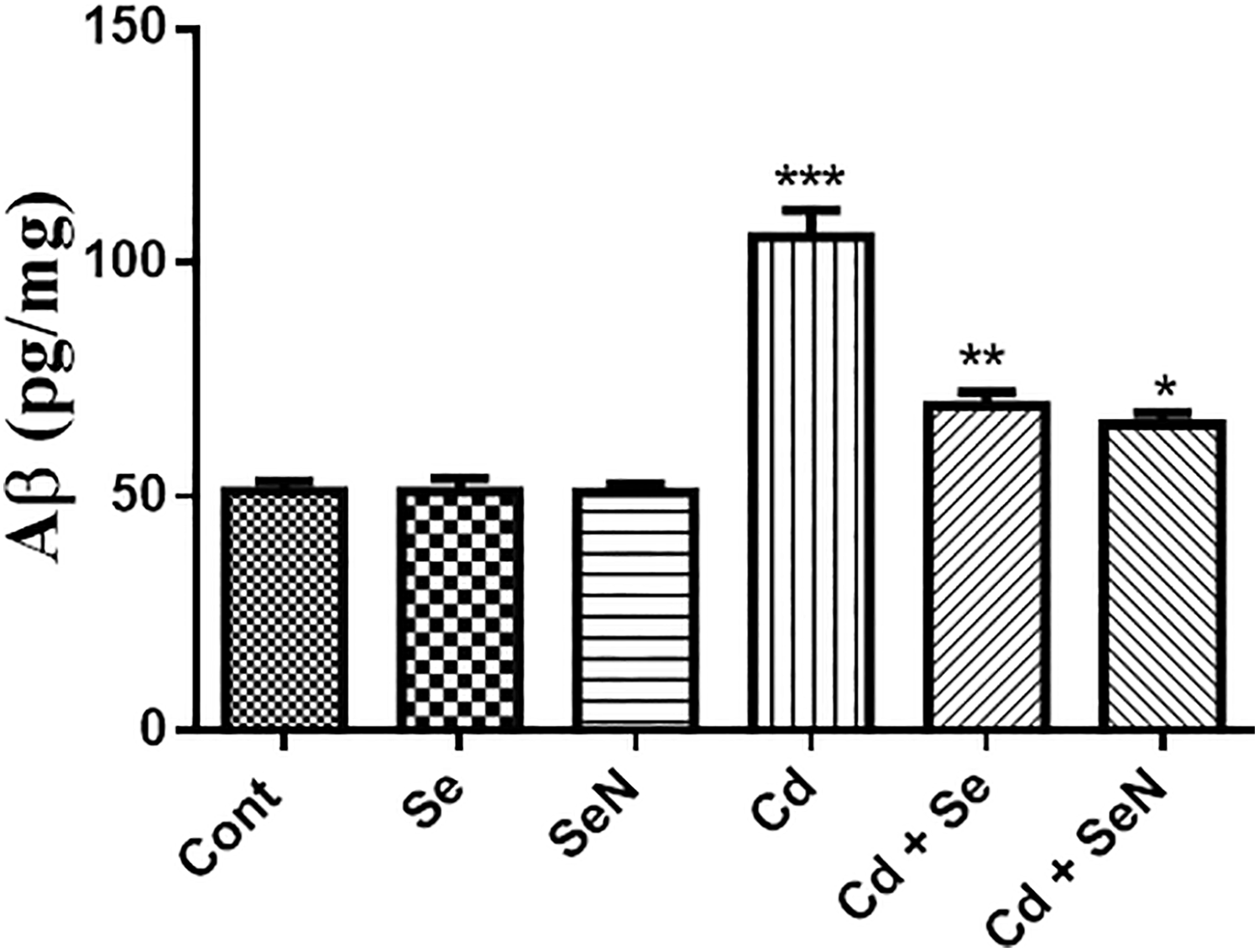
Brain Aβ highly increased in Cd group when compared with control groups but the concentration of Aβ decreased in Cd + Se and Cd + SeN groups in comparison with Cd group. The values are expressed as the means ± SEM (n = 6). Significant at *p < 0.05, **p < 0.01, and ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. Aβ: Amyloid beta protein; Cd: cadmium; Se: selenium; SeN: nanoselenium.
Brain Bax and P53 levels increased but Bcl-2 was decreased as compared with control groups (Figure 8). Both Cd + Se and Cd + SeN groups had an increase in Bcl-2 and a decrease in Bax and P53 level when compared with Cd group.

Brain Bax and p53 levels increase but Bcl-2 decreases as compared with control groups. Both Cd + Se and Cd + SeN groups have an increase in Bcl-2 and a decrease in Bax and p53 level when compared with Cd group. The values are expressed as the means ± SEM (n = 6). Significant at *p < 0.05, **p < 0.01, and ***p < 0.001. *, **, *** indicate comparisons with respect to the control group. Cd: cadmium; Se: selenium; SeN: nanoselenium.
Histological changes in hippocampus
Examination of the control group brains, stained with H&E, showed the hippocampus as a thick C-shaped structure, called cornu ammonis (CA), and another interlocking v-shaped structure, namely the dentate gyrus (DG). The CA was formed of three areas: CA1, CA2 and CA3 as in Figure 10(a). Only DG regions were examined. The DG region was formed of three layers: the polymorphic layer, granular layer and molecular layer. The granular layer contained closely packed cells with large vesicular nuclei. Few scattered glial cells could also be identified (Figure 10(b)).

The expression of SOD1 gene in different rat groups: lane M: marker 100 bp step ladder (Promega), lane 1: control group, lane 2: Se group, lane 3: SeN group, lane 4: Cd group, lane 5: Cd + Se group, and lane 6: Cd + SeN group. SOD1: Cu/Zn superoxide dismutase; Cd: cadmium; Se: selenium; SeN: nanoselenium.

Photomicrographs of a section in the hippocampus of (a) Control group showed the different areas of the hippocampal formation where the hippocampus proper formed of the CA as CA1, CA2, CA3 and CA4 regions and continued as subiculum. DG surrounded CA4 by its upper and lower limbs. Note lateral ventricle related to CA1 and CA2. (M) Dentate molecular layer inside concavity of CA and of DG (H&E, ×100). (b) Control group showed DG area composed of three layers; molecular layer (ML), five to six compact layers of granular layer (G) with dark nuclei in DG and polymorphic layer (O) with pyramidal cells (arrows) granular cells (arrow heads). The granular cells densely packed with a large open-faced vesicular nuclei (arrows). The dark vertical line represented the thickness of the granule cells layer and few scattered glial cells noticed within the tissue (irregular arrows; H&E, ×400). (c) Cd-intoxicated group showed granule cells appeared loosely packed and darkened with marked shrinkage in size of granular cells also, dystrophic changes in the form shrunken with pyknotic darkened nuclei (arrows), with an increase in the space around them, the surrounding neuropils also vacuolated (*) and associated with large vacuoles (V; H&E, ×400). (d) Cd-intoxicated group showed more marked affection of granular cells layer of DG where areas devoid of cells (large arrows), granular cells showed marked retraction of processes with vacuolation (V), decrease thickness of the granular cells layer of the DG to reach two layers in some areas as indicated by the black line, with increased apoptotic neurons with dystrophic changes in the form of shrunken hyperchromatic, irregular with chromatolysis (small arrows). They have pericellular halos (arrow heads) indicated to cell damage and the surrounding neuropils also vacuolated, molecular layer (ML) showed marked enlargement of neurons and of glial cells (H&E, ×400). (e) Cd + Se-treated group showed some protective effect in the form of normal granule cells (G) and the thickness of granule cells of DG region appeared near to the control group as indicated by the black vertical line. Molecular layer (M) mostly showed normal cells and fibers (H&E, ×400). (f) Cd- and Se-treated group showed that most of the cells have large vesicular nuclei (arrows) and normal blood vessels (arrow heads; H&E, ×400). (g) Cd + SeN-treated group showed the thickness of granule cells of DG region appeared near to the control group, as indicated by the black vertical line, molecular layer (M) mostly showed normal cells and fibers (H&E, ×400). (h) Cd + SeN-treated group showed most of the cells with large open-faced vesicular nuclei (arrows; H&E, ×200). (i) Se-treated and (j) SeN-treated groups showed the thickness of granule cells of DG region appeared near to the control group as indicated by the black vertical line. Most of the granular cells showed large open-faced vesicular nuclei, granular cells showed less vacuolation also, molecular layer showed normal size of cells with widened capillaries (arrows; H&E, ×400). CA: cornu ammonis; DG: dentate gyrus; Cd: cadmium; Se: selenium; SeN: nanoselenium.
Examination of the Cd-administered groups revealed an apparently decreased thickness of the granule cell layer in the DG region, in comparison to the control group (Figure 10(a)). The granule cell layer appeared to be shrunken, loosely packed with a decrease in cell number, showed dystrophic changes in the form of shrunken hyperchromatic, with dilated blood vessels and degenerated and vacuolated brain tissue (Figure 10(b) and (c)). Figure 9 illustrates the normal architecture of the brain. Treatment with Se or SeN particles prior to Cd intoxication reduced the incidence of these pathological changes in the brain tissue and resulted in almost normal brain architecture, similar to that of the control.
RT-PCR and the SOD1 amplification
The RT-PCR for the SOD1 gene gave positive results with the treated and non-treated animals but the band in the treated samples was in high intensity. The band intensity reflected the high expression levels for the SOD1 gene as a response for the treatment.
Discussion
Cd is a neurotoxic heavy metal that effects the BBB and accumulates in brain tissues, causing metabolic, histological, pathological and molecular changes. 63 Cd has the ability to disrupt antioxidant enzymes, such as SOD, CAT, GPx, GR, as well as non-enzymatic antioxidants, for example, GSH. 64 Moreover, cytosomal (SOD1, GPx), mitochondrial (Mn-SOD), Peroxisomes (CAT) and other extracellular SOD decrease after Cd administration as in Figures 3 and 4. 64 GSH, as a co-substrate, is involved in the elimination of different extrinsic toxicants and ROS by binding with them under the catalysing reaction of GPx, forming GSH disulphide. 64 GPx is concomitant with SOD isoforms and CAT enzymes in arresting ROS. The lipophilicity of Cd allows its accumulation in the brain and causes the aggregation of ROS, 65 such as lipid peroxides, in oligodendrocytes, as well as a decrease in GSH content and increase in oxidative stress. 13,66,67 The neurotoxicity of Cd was established in the brain of the Cd group by altering the oxidant/antioxidant status. Increased activity of SOD, CAT, GPx, and GR enzymes and an increase in the GSH content of groups treated with Se and SeN particles are indicative of the antioxidant role of Se and SeN particles. The improvement may be due to the formation of selenoproteins such as GPx, 46 which improve the antioxidant capacity and is a possible chemotherapeutic agent for the brain.
In accordance with these metabolic changes, Cd increased AChE activity in different brain compartments such as the striatum, hippocampus and cerebellum. 68 Cd alters the lipid bilayer of membranes and enhances AChE activity (Figure 5). 69 A high activity of AChE increases the lysis of acetylcholine, which decreases the response and stimulation of acetylcholine receptors. So Cd directly affects cholinergic neurotransmission efficacy in the synapsis, causing cognitive dysfunction. 70,71 Moreover, Cd alters gamma-Aminobutyric acid (GABAergic) and glutamatergic pathways, dopamine 45 and serotonin concentration in rats administered with Cd as in Figure 6. 23 The neurotoxic effect of Cd in experimental animals is due to its impairment of catecholaminergic and serotoninergic transmission. 5 Therefore, it has been postulated that Cd-induced blockage of cholinergic transmission in the brain is mediated by high AChE activity. 72 In addition, the increase in dopamine 22 and glutamate 24 is involved in many neurodegenerative diseases, such as Parkinson’s disease and AD. It is associated with a decrease in GSH and antioxidant enzymes and an increase in oxidative stress. 25 Hence, Cd-mediated neurotoxicity could be explained by the downregulation and upregulation of various neurotransmitters. Se has the ability to improve neurotransmitter function in the brain and cognitive function in AD. 34 This article proposes that the antioxidant properties of Se and SeN could be the reason for these activities.
Amyloidosis is the most accepted hypothesis for explaining the onset of AD, initiated by many toxicants, such as Al 73 and Cd.31,72 Aβ increases ROS and lipid peroxidation. 74 Also, oxidative stress induces Aβ in the brain (Figure 7), 28 which initiates the pathophysiology of AD 16 and may be involved in apoptosis. 33 Aβ is neurodegenerative factor and is involved in cellular cell death. The initiation phase begins with the ligation of TNF and NF-κB transcriptional factors to death receptors. 35 However, there is a negative relationship between Cd and the expression of SOD and CAT genes that scavenge ROS. 35 The results herein show that the increase in the expression of SOD1 was attenuated by SeN more than Se (Figure 9). There is a relationship between apoptosis and the activity of SOD1 in nervous tissue. 75 The increase in oxidative stress and the decrease in antioxidants induce apoptosis through an extrinsic pathway. p53 induces Bax to form channels in the outer mitochondrial membrane to export cytochrome C enzymes and Bcl-2. 76 The decrease in Bcl-2, and the increase of Bax by Cd (Figure 8), induces the cells to die by apoptosis. Therefore, Cd promotes the activation of pro-apoptotic proteins (Bax) and the decrease in anti-apoptotic proteins (Bcl-2) in the brain tissue. Moreover, Cd elevates the levels of ROS that activates TNF and NF-κB, which signal to the cells to die through the p53- dependent pathway. 41,77
The examined sections of the brain tissue did not show any differences between the Se- or SeN-treated and control groups, and they were similar to the normal structure (Figure 10). On the other hand, Cd administration induced histological changes in the hippocampus, including spongiform or vacuolization, pyknosis changes and a reduction in the granular cell layer thickness of DG. Se- and SeN-treated groups showed recovery from the Cd-induced pathological changes, which indicates that Se is capable of preventing the neuronal damage induced by Cd. Therefore, we suggest that Se might inhibit Cd-induced hippocampus damage. The histological alterations found in the present work agree with the experiments done by many researchers, 78,79 Damage induced by excessive Cd may be due to the production of free radicals in the brain and hippocampus. Recent studies have shown that the production of ROS may be associated with Cd toxicity in the lungs, liver and brain, resulting in increased lipid peroxidation, altered calcium homeostasis, impairment of antioxidant enzymes and finally DNA damage, 9,80 Lipid peroxides that accumulate due to lipid peroxidation are known to be harmful to cells and tissues. 65 An important aspect of selenium’s (Se and SeN) role in neuroprotection is the attenuation of free radical formation through its antioxidant and anti-inflammatory effects. It also efficiently interacts with various reactive oxygen and nitrogen species. The efficacy of Se in Cd-induced neurotoxicity is mainly attributed to its high lipophilicity. 80 These results illustrate the importance of Se for the inhibition of oxidative stress and in protecting the integrity and function of hippocampus tissues, which it achieves by increasing GPx activity, preventing lipid peroxidation, acting as an antioxidant and scavenging free radicals. 80 Se and SeN particles act as anti-cancer and anti-tumour agents, by causing disruption of the apoptotic pathways and cell cycle custody. 81
Conclusion
The antioxidant activities of the biogenic SeN and sodium selenite seem to be identical, except in the expression of SOD1. Their ameliorative action against Cd-induced neurotoxicity is comparable. Alterations of peroxidants and antioxidants in experimental Cd intoxication indicate definite oxidative stress in brain tissues. Also, the adverse effects of Cd on the antioxidant system were inhibited by Se and SeN particles, and Se has the ability to attenuate the neurotransmitter changes initiated by Cd intoxication. Se and SeN particles decrease Aβ and hence decrease amyloidosis in the brain; this may be due to their antioxidant action. The anti-apoptotic role was established by increasing Bcl-2 and p53 and decreasing of Bax. Cd administration induced histological changes in the hippocampus including spongiform or vacuolization and pyknosis, all of which were attenuated by Se administration in the Cd-treated groups. Therefore, the use of supplementary antioxidant substances such as Se and SeN particles may be useful in treating neurotoxicity and subsequently improving cognitive function.
Footnotes
Acknowledgement
The author would like to express their gratitude to the Research Centre of Advanced Materials of King Khalid University, Saudi Arabia for support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Deanship of Scientific Research, King Khalid University [general research project, Grant Number: G.R.P.-172-39].
