Abstract
The transient receptor potential ankyrin 1 (TRPA1) ion channel is a sensor for irritant chemicals, has ancient lineage, and is distributed across animal species including humans, where it features in many organs. Its activation by a diverse panel of electrophilic molecules (TRPA1 agonists) through electrostatic binding and/or covalent attachment to the protein causes the sensation of pain. This article reviews the species differences between TRPA1 channels and their responses, to assess the suitability of different animals to model the effects of TRPA1-activating electrophiles in humans, referring to common TRPA1 activators (exogenous and endogenous) and possible mechanisms of action relating to their toxicology. It concludes that close matching of in vitro and in vivo models will help optimise the identification of relevant biochemical and physiological responses to benchmark the efficacy of potential therapeutic drugs, including TRPA1 antagonists, to counter the toxic effects of those electrophiles capable of harming humans. The analysis of the species issue provided should aid the development of medical treatments to counter poisoning by such chemicals.
Introduction
The transient receptor potential ankyrin 1 (TRPA1) ion channel is present in primary afferent nociceptors. TRPA1 ion channels are comprised of homotetramers which detect the presence of a wide range of agonists that produce respiratory irritation, neurogenic inflammation and pain in humans and rodents. 1 –13
TRPA1 is a noxious chemosensor which mediates irritant effects of a large number of compounds in different tissues. 14 –17 Its protective role in detecting irritants in very low concentrations is important in the triggering of protective avoidance responses following exposure, thus reducing the risk of injury. 18 However, while this sensory function associated with TRPA1 is highly conserved across many species, 19 there are important species differences in the way mammals respond to different agonists and antagonists. This has a bearing on the appropriateness of the choice of animal models for assessing the hazards that various TRPA1 agonists pose to humans, particularly when attempting to extrapolate observations to humans. The choice of animal model also impacts on attempts to identify candidate antagonists as potential medical countermeasures against agonist exposure, 1,20 as responses to both agonists and antagonists can have species-specific determinants. There are also important read-across issues associated with the use of in vitro systems for modelling human TRPA1 responses (to both agonists and antagonists). These affect the relevance of observations made using human-based in vitro systems to determine the mechanistic basis of agonist interactions with TRPA1 ion channels and the identification of potential candidate therapeutic antagonists for pull-through to frequently used in vivo models. 21,22 This is because TRPA1 structure and function may differ in important respects to the human TRPA1 counterpart. 23 These points will be explored herein.
TRPA1 is a multifunctional transducer of stimuli and in this context, 24 aside from responding to noxious chemicals of both exogenous and endogenous origin, TRPA1 receptors have a role in mechanotransmission and allow numerous classes of primary afferent fibres to sense and transmit mechanical stimuli of varying intensities. 25 –28 TRPA1 receptors also transduce thermal stimuli, 29 –31 but TRPA1-mediated thermal sensitivity varies substantially between species. 32 The role of TRPA1 in mechanotransmission and thermal sensing is beyond the scope of this review which concentrates on noxious chemical sensing. Another TRP ion channel, the transient receptor potential vanilloid 4 (TRPV4), is gaining prominence in pulmonary pathology. 33 However, this channel is not included in this review for reasons now discussed.
The TRPV4 ion channel is expressed at a high level in most cells associated with the lung and respiratory tract and is implicated in numerous pathological states such as pulmonary oedema and pulmonary responses to various inflammatory mediators, and in maintenance functions, such as the regulation of vascular tone in the lung and associated structures. 34 TRPV4 has a widespread distribution throughout the lung and is most highly expressed in such areas as the septal walls of the alveoli and associated arterial vasculature to the epithelium which lines the airways. 35 Its activation causes contraction of the smooth muscle cells of the bronchi. 36 This ion channel may have a central role in bronchoconstriction responses associated with many respiratory conditions including asthma. 37 TRPV4 agonists which activate this ion channel increase the permeability of the pulmonary endothelium by generating breaks in this barrier. 38 The activation of the TRPV4 ion channel can result in acute circulatory collapse as a result of such injury, because the barrier function of the pulmonary microvascular bed fails resulting in vascular leakage. 35 TRPV4 activation can also disrupt alveolar type 1 epithelium causing alveolar oedema. 38,39 TRPV4 ion channels have also been implicated in a mouse model of ventilator-induced acute lung injury. 40
The TRPV4 ion channel can be activated by agonists such as arachidonic acid and its P450 metabolites, by mechanical stresses, acidic pH, hypotonic environments and heat. 41 The nature of the stimulus influences the TRPV4-related activation of immune cells, particularly pulmonary macrophages (in which this ion channel is highly expressed) and the subsequent injurious effects on lung tissue. 42,43 The effects associated with chlorine exposure have raised the TRPV4 ion channel to prominence. In particular, studies by Balakrishna et al. 44 in which mice were exposed to chlorine gas and then treated either immediately or 8 h post-chlorine exposure with TRPV4 inhibitors GSK2220691 or GSK2337429A demonstrated that airway hyper-reactivity and vascular and epithelial leakage were inhibited. Additionally, lung inflammatory responses such as the neutrophil and macrophage content of bronchoalveolar lavage fluid (BALF) were attenuated. It was also reported that pro-inflammatory chemokine and cytokine levels were significantly reduced and that this was accompanied by improvements in blood oxygen saturation levels and by significant reductions in the protein content of BALF. 44
While the high level of sequence homology of TRPV4 between human and rodent species (Table 1) would tend to lend confidence in the aforementioned findings, there are a number of confounding issues that may limit the value of approaches aimed at inhibiting the activation of TRPV4 in the lung. These have been summarised by Morty and Kuebler,
47
who have noted that using methods to inhibit pharmacologically the TRPV4 ion channel as a potential therapeutic approach has two principal caveats: The use of TRPV4 antagonists may result in the inhibition of the von Euler-Liljestrand system. Inhibition of this system was postulated to be potentially problematic in situations where the acute lung injury was of a heterogeneous nature. TRPV4 antagonists may compromise the role of the TRPV4 ion channel in inflammatory disease. TRPV4 antagonists could effectively reduce a subject’s capacity to neutralise pathogens associated with systemic and/or pulmonary infections found in most humans suffering acute respiratory distress syndrome. TRPV4 antagonists could also compromise the injury recovery phase, as TRPV4 also has a role in pulmonary vascular remodelling associated with chronic hypoxia.
48
Sequence homology between TRP ion channels.

TRP: transient receptor potential; TRPA1: transient receptor potential ankyrin 1; TRPM8: transient receptor potential cation channel subfamily M (melastatin) member 8; TRPV1: transient receptor potential vanilloid 1; TRPV3: transient receptor potential vanilloid 3; TRPV4: transient receptor potential vanilloid 4.
aChen and Kym. 19
bJabba et al. 45
cBianchi et al. 46
In addition, the ability of TRPV4 monomers to form heterodimeric complexes with the transient receptor potential canonical 1 monomer (TRPC1), which can undergo assembly into functional Ca2+ ion channels, 49 further complicates the understanding of the role of TRPV4 as a target of agonists and the ability to evaluate the potential usefulness of TRPV4 antagonists in a therapeutic context. This is compounded by more recent reports which indicate that TRPV4 is involved in regulating a variety of processes in the central nervous system, including temperature, water volume and cation concentrations. 50 Intracerebral haemorrhage has been found to activate TRPV4 ion channels resulting in Ca2+ homeostasis being compromised, triggering endoplasmic reticulum-unfolded protein response and subsequent apoptotic cell death of neural cells. 51
The TRPA1 ion channel and some common agonists
TRPA1 agonists vary substantially in structure but are often powerful electrophilic agents, 52 which interact with the human TRPA1 ion channel via reversible covalent modification of one or more of a set of cysteine residues (Cys 621, Cys 641 and Cys 665) or possibly a lysine amino acid residue (Lys 710) in the pre-S1 helix of TRPA1 at the N-terminal region. 53,54 Cys 621 is particularly acidic and ionises to give a high proportion of the thiolate anion which can react with electrophiles (Figure 1). 55 –57

Model of structure of TRPA1 showing the ECD, TMD and CYD. The lateral and vertical dimensions are also given. The important Cys residues (e.g. Cys 621/641) which interact with TRPA1 agonists are located in a cleft labelled AA. These Cys residues are located in the pre-S1 helix of TRPA1 in a region situated in the cytoplasm between the S1 TMD of TRPA1 and the cytoplasm-located linker domain of the ankyrin repeat sequences (located in the CYD). The image was constructed using the VMD programme (reference: https://www.ncbi.nlm.nih.gov/pubmed/8744570) based upon the TRPA1 cryo-EM structure of Paulsen et al. 56 with non-terminal missing regions of the protein modelled in, and with the protein inserted into a POPC lipid membrane. Figure was supplied by Dr Thomas Piggot, Microbiology and Biological Detection Group, Chemical, Biological and Radiological Division, Dstl Porton Down. ECD: extracellular domain; TMD: trans-membrane domain; CYD: cytosolic domain; TRPA1: transient receptor potential ankyrin 1; POPC: phosphatidylcholine.
α,β-Unsaturated aldehydes, such as acrolein and crotonaldehyde, are potent activators of TRPA1 and are believed to cause activation through a rapid and reversible covalent reaction with a cysteine thiol group of the ion channel to generate a 1,4-addition product (Michael addition product) 58 as described in Figure 2. The addition of thiols in this manner to unsaturated aldehydes is well known. 59 –61 However, an alternative mechanism of activation of TRPA1 via reversible 1,2-addition of the thiol to the carbonyl group has also been proposed for some unsaturated aldehydes and ketones. 62 In many instances, it is not clear yet whether one or both of these mechanisms can operate at the TRPA1 receptor to cause its activation.

Michael addition of TRPA1 to unsaturated aldehydes. TRPA1: transient receptor potential ankyrin 1.
Other agonists such as hydrogen peroxide (H2O2) and hypochlorous acid (HOCl) oxidise Cys residues to sulfenic then sulfinic acids. 58 The target Cys thiol group is presumed to oxidise sequentially to the sulfenic acid, sulfinic acid and sulfonic acid as shown in Figure 3. 63 These alterations to this amino acid are presumed to be the reason the ion channel opens and the ensuing pain response is experienced. Note that sulfenic acids are usually insufficiently stable to isolate; sulfinic and sulfonic acids are more stable and can be isolated. Sulfinic acids are analogous to carboxylic acids and can be regarded as derivatives of sulfurous acid where a hydroxyl group has been replaced by an alkyl group, while sulfonic acids can be regarded as derivatives of sulfuric acid where a hydroxyl group has been similarly replaced. The acidity of the acid increases with an increase in the oxidation state of the sulfur atom and correspondingly increases its ionisation under physiological pH (7.4). The conversion of a weakly ionised thiol group (SH) to a strongly ionised sulfinic acid group (SO2H) or sulfonic acid group (SO3H) is akin chemically to switching the cysteine residue of the protein for an aspartic acid residue (SH group → CO2H group). This analogy helps to highlight the electrostatic and conformational change that such oxidation brings to the active site of TRPA1, which must lead to ion channel opening. An exposed cysteine thiol group in another protein, human copper–zinc superoxide dismutase, has been shown to oxidise to the sulfinic and sulfonic acids sequentially; the intermediate sulfenic acid, in contrast, was not observed by mass spectrometry, suggesting a fleeting existence. 64 A thiolate (S−) group is required for Cys residues to react with H2O2. 65

Oxidation of TRPA1 cysteine by hydrogen peroxide. TRPA1: transient receptor potential ankyrin 1.
The pre-S1 helix is situated in the cytoplasm between the S1 transmembrane domain of TRPA1 and the cytoplasm-located linker domain of the ankyrin repeats (Figure 1). According to Paulsen et al. 56 the Cys amino acid residues implicated in the reaction with electrophiles (especially Cys 621 which appears to be the most acidic) are accessible to solvent. Their location in TRPA1 is believed to confer on them properties such as the ability to induce allosteric modulation of the TRPA1 structure. This allows reactions between the target amino acids and chemical agonists such as electrophiles to be transduced into a structural force which results in a conformational change. 66,67 This subsequently causes activation of the Ca2+ channel, allowing cations (predominantly Ca2+ and Na+) to move from the extracellular environment into the cell, causing the membrane potential in primary afferent neurons to depolarize. 68 This in turn results in action potential firing and an increased sensation of pain. The whole linker region is essential to the control of the ion-conducting pathway. 69 A recent report by Zimova et al. 70 has hypothesised that regulation of the intracellular gate of the TRPA1 ion channel is through a complex system which involves the integration of ligand-activated signals in the lower region of the S1–S4 domain, in association with a cavity formed by polar residues. Signals are subsequently transferred via the TRP-like domain to the gate. The proximity of the pre-S1 helix to the TRP-domain suggests that allosteric control mechanisms are responsible for the way that the intracellular gate of the TRPA1 ion channel is controlled. 71
Fundamentally, TRPA1 ion channels in sensory nerves act as a detection system for potentially noxious reactive chemicals, though these ion channels are also found in tissues of non-neuronal origin such as airway smooth muscle and airway epithelial cells. 72,73 The chemical species that activate this ion channel include irritants from man-made and plant-based sources, as well as endogenous molecular species. TRPA1 effectively senses our external environment for exogenous noxious agents, which may enter the body via the eyes, nose, respiratory tract and skin. However, this ion channel also responds to inflammation processes which occur when organs such as the lungs are compromised by chemical agents or biological pathogens. 74 Currently, a surprising number of natural and man-made environmental TRPA1 agonists have been identified. These produce nocifensive behaviour, affecting principally the eyes, airways and skin resulting in pain-associated responses, for example, tearing and blepharospasm (involuntary closing of the eyelids), coughing and sneezing and skin irritation and at higher doses, the stomach and gut may also be involved, resulting in retching, vomiting and diarrhoea. The potential exists for the treatment of affected tissues and organs with TRPA1 antagonists to alleviate signs and symptoms of exposure to TRPA1 agonists. 75 –81 These agonists fall into four categories of exogenous and endogenous compounds, which are detailed subsequently.
Exogenous toxic industrial compounds
Alkyl and aryl isocyanates comprise a diverse family of compounds, of which methyl isocyanate (MIC) is the most notorious. MIC was the main agent involved in the Bhopal disaster (Madhya Pradesh, India), which accounted for some 2500 deaths and serious injuries to many thousands of people in the immediate aftermath. 82,83 The death toll rose to approximately 5000 people (out of an exposed population of some 350,000) as a result of effects on the respiratory tract following inhalation of the agent. The casualties who died immediately after the release, on the night of 2 and 3 December 1984, of some 27 tons of MIC vapour had suffered extensive damage to the bronchial tree (with pulmonary oedema, haemorrhage and extensive necrosis of the airways being reported post-mortem). 84 MIC-related compounds have also been involved in other incidents, notably in a major chemical release which resulted from spillage of some 86 tons of the pesticide sodium N-methyldithiocarbamate (MeNHC(S)SNa), a soil fumigant, from a rail road tanker into the Sacramento River in 1991. 85 This material, on contact with water, resulted in chemical reactions that released methyl isothiocyanate (MITC – the main pest killing agent) and additional chemically reactive species such as MIC, methylamine (MeNH2), carbon disulfide (CS2) and hydrogen sulfide (H2S), though the proportions of these products formed in the environment on hydrolysis varies with soil pH and other factors. 86 –88 MITC is regarded as having been the main hydrolysis product – this is fortunate as it is substantially less toxic and irritating to humans, and no deaths were caused by this incident. 89 MIC causes severe irritation of the eyes, throat and nose, with the airways being particularly sensitive. The resulting airway injuries include oedema (as already noted), as well as inflammation and airway hyperresponsiveness. 83,90 Jha et al. 72 have suggested that TRPA1 inhibition by pharmacological intervention can attenuate airway hyper-responsiveness in vivo.
Alkyl and aryl isocyanates and isothiocyanates contain a conjugated reactive carbon atom as part of the isocyanate or isothiocyanate grouping. 91 Although the mode of activation of TRPA1 has not been fully delineated, it is possible (as shown in Figure 4) that they add to a cysteine thiol of the ion channel to provide either the S-alkyl urea or the S-alkyl isothiourea product, 92 therefore activating the channel, switching it from the ‘off’ to the ‘on’ state. Such reaction products are known to be unstable in the presence of water. 93 We postulate that they would hydrolyse quickly to return inactivated TRPA1, an amine (methylamine from MIC and MITC and allylamine from allyl isothiocyanate (AITC)) and either carbon dioxide (from the isocyanates) or carbon oxysulfide (from the isothiocyanates). These hydrolysis products are not that electrophilic and would therefore not be expected to activate TRPA1 (carbon dioxide and carbon oxysulfide when pure do not register on the senses: they are both odourless and non-irritant). 94 The general mechanism provided would explain the transient sensory irritancy of atmospheres of small electrophilic alkyl or aryl isocyanates and isothiocyanates. The more permanent damage to tissues caused by these compounds most probably arises from depletion of intracellular glutathione (through reaction with the thiol group of this tripeptide) and reactions with biological amines which give addition products (e.g. RNHC(=X)NHR’ from primary amines) that are much less readily hydrolysed than those from thiols, and therefore more likely to cause irreversible damage. The general mechanism of TRPA1 activation by isocyanates and isothiocyanates provided here might also explain the inability to resolve the AITC adduct of TRPA1, 55 given its predicted transient nature, and could imply a first line of defence and catalytic detoxification function of the ion channel.

Addition of TRPA1 to alkyl iso(thio)cyanates. TRPA1: transient receptor potential ankyrin 1.
Other toxic industrial compounds (TICS) of concern include acrolein and crotonaldehyde (aldehydes present in smoke), hydrogen sulfide, industrial pollutants such as toluene diisocyanate, H2O2, HOCl, formaldehyde, 8,18 corrosive gases, hydrazines 82 and chloropicrin (a soil fumigant and war gas). 95 Trichloronitromethane – or chloropicrin as it results from chlorine or hypochlorous acid reacting with picric acid (2,4,6-trinitrophenol) – is a colourless liquid (boiling point 112°C) having a sweetish odour that is a scheduled chemical under the chemical weapons convention. 96,97 It is a powerful lachrymator and irritant of the respiratory passages and skin, 98 hence its use in battle during the First World War. 99,100 Later, its insecticidal activity was observed, and it was used as a fumigating agent particularly for grain. 101 It is stable, insoluble in water and does not hydrolyse at ordinary temperatures. 102 As can be seen in Figure 5, it contains four leaving groups displaceable by nucleophiles: three chlorine atoms and the nitro group, which is normally replaced last. The chlorine atoms are particularly susceptible to displacement by sulfur nucleophiles. 103,104

Proposed reaction of TRPA1 with chloropicrin. TRPA1: transient receptor potential ankyrin 1.
It is possible that a cysteine thiol group of TRPA1 might undergo rapid nucleophilic substitution of chloropicrin to yield the S-dichloronitromethyl derivative,
104
activating or turning ‘on’ the ion channel. This product, which is likely to be unstable, might be degraded through reaction with an intramolecular thiol,
Chlorine has numerous industrial uses and is of particular concern as it is transported, stored and used in bulk quantities as a liquid under pressure. This situation, and its high toxicity, 102 has the potential to cause mass casualties. 106 Up to 14 million tons of chlorine are manufactured on a yearly basis in the United States and transported by rail to many thousands of locations. This often requires it to be moved through areas of high population density. On 6 January 2005, a train pulling three 90-ton chlorine tankers was involved in a collision in the town of Graniteville (South Carolina, USA), resulting in the release of up to 60 tons of the chemical. Eight people died from asphyxia at the scene. A further 597 people were identified as requiring medical attention in private clinics or hospitals, of which 71 were hospitalized. Of this group, one person died the same day they were admitted. 107
Chlorine is a TRPA1 ion channel agonist, like many other TICS such as formaldehyde, acrolein (also a pulmonary oedemagen), ammonia, hydrazines and various mineral acids 108 and corrosive gases. TRPA1, mostly expressed in small nociceptive neurons, contributes through its activation to the perception of noxious stimuli such as the aforementioned materials. 58,95,109,110 The intensely irritating effects of chlorine on the respiratory tract are due to the TRPA1-activating products it generates in aqueous solution (hypochlorous acid (HOCl), the hypochlorite ion (OCl−) and reactive oxygen species (ROS, e.g. superoxide (O2 −·)). 58,111,112 These products activate TRPA1 ion channels in sensory nerve endings and thus initiate an immediate irritant/pain response. When irritants such as chlorine interact with vagal sensory fibres, which terminate in the glottis and larynx, a cough reflex is triggered. On reaching the lower airways of the lung, sensory neuronal activity initiated by respiratory irritants triggers physiological responses such as tracheal and bronchial constriction, bronchospasm, mucus secretion and neurogenic inflammation. 58 Neurogenic inflammation, resulting from the release of inflammatory tachykinins from nerve endings of the sensory nervous system, results in vasodilation, extravasation of plasma and subsequent pulmonary oedema. 13,113,114 Chlorine-induced morbidity or mortality is due to non-cardiogenic pulmonary oedema which can occur within 1 h following exposure to high concentrations. 115 According to Evans, 116 domestic accidents involving various cleaning products, transportation incidents and industrial exposure to chlorine (and its related bioactive products) remain a significant cause of irritation and injury to the respiratory tract of exposed subjects, and these frequently require medical attention.
Exogenous military and law-enforcement compounds
Riot control agents (RCAs) are some of the most potent TRPA1 agonists known. The high sensitivity of TRPA1 to these agents is used to generate a variety of avoidance responses in humans. Such compounds include 2-chloroacetophenone (CN), dibenz[b,f][1,4]oxazepine (CR) and 2-chlorobenzylidenemalononitrile (CS) (Figure 6). The half maximal effective concentration (EC50) values of these agonists are in the picomolar to nanomolar range. 117,118 Their effects include pain, respiratory distress, coughing and lachrymation. 18,81,117 RCAs such as CN, CR and CS mostly affect the eyes, nose, respiratory tract and skin, and these effects have been described as being instantaneous and profound. 119 RCAs are also called ‘tear gases’, irritants or lachrymators whose main function is to cause closure of the eyes and substantial but transient discomfort. 120 These effects are used by law enforcement personnel to prevent fighting and to facilitate arrests. 121 According to Rothenberg et al., 122 CS and oleoresin capsicum (OC – a waxy resinous extract of chilli peppers, comprised of a mixture of compounds (capsaicinoids) such as capsaicin, dihydrocapsaicin and norhydrocapsaicin, of which capsaicin is the most potent) 95,123 are the predominant RCAs in use today. Consideration of OC is beyond the scope of this article, as its active components are agonists of the human transient receptor potential vanilloid 1 (hTRPV1) ion channel. 124

Some riot control agents that potently activate TRPA1. TRPA1: transient receptor potential ankyrin 1.
At concentrations which may be encountered when deployed for riot control, CS causes immediate irritation to the respiratory tract, the nose, eyes, mouth and skin. 125 The most important areas affected are the eyes and respiratory tract. In the case of CS exposure of the eyes, commonly experienced symptoms include itching and burning sensations, as well as lachrymation and blepharospasm. 126 Inhalation exposure to CS can result in choking, coughing, salivation and a feeling of chest tightness. 127
The most frequently used RCAs have large margins of safety between their effective dose (as may be generated in a riot control situation) and their potentially toxic dose. 125 An example of the size of this margin is evident when considering that the threshold for eye irritation in humans for CS is 4 μg/m3, whereas the intolerable concentration for eye irritation is 3 mg/m3. This compares with the minimum lethal concentration of 2500 mg/m3 (for a 10 min exposure) and an estimated human lethal dose of 25,000–100,000 mg min/m3. 95,128
In a later review, Schep et al. 129 confirmed that in most instances where RCAs are used, clinically significant effects would not be likely to arise in the majority of cases. Indeed, the most important characteristics of RCAs relate to their ability to cause rapid onset effects, combined with a short duration of action (once the subject has left the environment where the RCAs were deployed) and a very low toxicity. 95 In general, RCA-induced irritation of the respiratory tract and the eyes can occur within 20–60 s. 129 RCA-induced effects generally last 15–45 min after the affected individual has departed from the contaminated area. 120 If supportive therapy was required, this would take the form of directing the subject away from the RCA-contaminated area, removing contaminated clothing and decontaminating the subject by irrigating the skin and eyes. There is usually no need for further treatment, though in severe cases symptoms may need to be treated with antipruritics and analgesics. 115
Concerns relating to the toxicity of some RCAs have led to cessation of the use of many RCAs such as CN – the most frequently employed RCA till the 1950s. 119 CS has since become the most frequently deployed RCA worldwide and is less toxic, but more potent, than CN. The relative irritant potency of CS and its analogue compounds and associated structure–function relationships have been reported by Lindsay et al. 130 A detailed consideration of how RCAs are defined under the chemical weapons convention may be found in Timperley et al. 131
Exogenous plant-derived compounds
Numerous TRPA1 agonists have been identified from various plant sources. Of these, the following are important – the plant sources are in brackets: cinnamaldehyde (cinnamon), 132,133 allicin and diallyl disulfide (garlic, onion), AITC (horseradish, mustard, wasabi), carvacrol (oregano, marjoram), eugenol (cloves), oleocanthol (olive oil), methyl salicylate (wintergreen), gingerol (ginger), Δ 9 -tetrahydrocannabinol (marijuana-derived psychoactive compound) and thymol (thyme). 18,134 –136 Thiols react with dialkyl disulfide-S-oxides and dialkyl disulfides to give mixed disulfides. 137 As can be seen in Figure 7, it is probable that a cysteine thiol of TRPA1 might react with allicin and diallyl disulfide to yield the same S-allyl product. The S-oxide allicin is more reactive than diallyl disulfide as it contains a better, more electronegative, leaving group (allyl thioaldehyde compared to allyl thiol).

Proposed reactions of TRPA1 with garlic and onion chemicals. TRPA1: transient receptor potential ankyrin 1.
Linalool, found in many species of plants such as citrus, mints and laurels, is also a TRPA1 agonist. 138 Polygodial, found in the Tasmanian pepper and water pepper, gives these plants their spicy flavour. 139 Polygodial is also found in the medicinal tree winter’s bark and in the medicinal plant horopito. 140,141 The distribution of this agonist extends to more primitive plants such as liverworts and ferns. 142 It is even found in sea slugs, possibly as a defence against predation by fish. 143 The noxious fungus-derived deterrent isovelleral is also a TRPA1 agonist and is believed to protect fungi from being eaten by fungivores. 139 The structures of a selection of the naturally occurring exogenous TRPA1 activators from plants are given in Figure 8.

Natural exogenous TRPA1 activators from plants. Top row: Phenols in herbs and spices: carvacrol in thyme and oregano; eugenol in basil, cloves, nutmeg and bay leaves; oleocanthal in extra-virgin olive oil (thought to be responsible for the transient stinging it causes in the mouth); and thymol, an isomer of carvacrol, from thyme oil. Middle row: Methyl salicylate, a chemical with a sweet odour produced particularly by the wintergreens; gingerol, the spicy constituent of fresh ginger (and related structurally to capsaicin and piperine, the chemicals that impart spiciness to chilli peppers and black peppers, respectively) and (−)-trans-▵ 9 -tetrahydrocannabinol, the main psychoactive component of cannabis. Bottom row: Other molecules from plants that activate TRPA1: linalool from flowers and spices (the (R)-isomer is found in lavender and the (S)-isomer in coriander); polygodial, a constituent of Dorrigo pepper and water pepper used in cooking (it has a warm and spicy flavour); allicin, formed when garlic is chopped or crushed, and thought to contribute to the pungent aroma of fresh garlic and diallyl disulfide, from garlic and other plants of the genus Allium, including onions, shallots, leeks and chives. Many of the phenolic compounds shown in this figure have no obvious reactive sites and may activate TRPA1 by electrostatic non-covalent binding. Oleocanthal and polygodial contain α,β-unsaturated aldehyde features and might activate the ion channel through Michael reaction with a cysteine residue at the positions indicated by the arrows (refer to Figure 2 for further details of such chemistry). Allicin and diallyl disulfide are also thiol reactive and may activate TRPA1 through cysteine attack on the S–S hinge (see Figure 7 for more information). TRPA1: transient receptor potential ankyrin 1.
Endogenous TRPA1 agonists
Aside from the aforementioned agonists which can activate TRPA1 ion channels, there are a number of potent molecular species which are generated by the body following damage to tissues. This tissue damage can occur as a consequence of oxidative stress and inflammation.
144
These endogenous molecular species are also potent TRPA1 agonists and include ROS such as H2O2,
111,145
nitric oxide (NO),
146
9-nitrooleic acid,
147
4-hydroxynonenal (4HNE),
13
4-oxononenal (4ONE)
148
and the A- and J-series of prostaglandins (metabolites of PGE2 and PGD2, respectively) such as 15-δ PGJ2.
12,149,150
Hydrogen sulfide is also known to be a TRPA1 agonist; it acts as a cellular messenger/transmitter (as does NO) and is produced from
Endogenous TRPA1 agonists have differing origins. For example, 4HNE is produced when oxidative stress or inflammation results in liposome peroxidation, 13,153 and 9-nitrooleic acid 154 (a reactive modifier of Cys residues) is generated as a consequence of phospholipid nitration, itself a result of excess NO production in tissue inflammation processes. 147 Proposed mechanisms of ion channel activation by 9-nitrooleic acid and 4HNE, through reversible addition of a cysteine thiol to their electrophilic double bond, are provided in Figure 9.

Top row: 9-Nitrooleic acid, a component of the nitrated fatty acid pool in humans, contains a double bond activated by a nitro substituent and is able therefore to add cysteine. 154 Bottom row: 4HNE, produced by lipid peroxidation in higher animals and in higher concentration during stress, contains an α,β-unsaturated aldehyde presumably able to add cysteine as shown (see Figure 2 for further examples of such chemistry, which is quite well catalogued in the literature). 4HNE: 4-hydroxynonenal.
NO is a signalling molecule with important roles which include pain sensation and sensitisation, neurotransmission, vascular signalling and immune responses. 155,156 The noxious gas hydrogen sulfide is generated in the body by many enzymes such as cystathionine-γ-lyase 157 and has had various functions attributed to it, such as protecting cardiac muscle and neurons from the effects of oxidative stress and acting as a relaxant in smooth muscles. 158
The inflammatory process in tissues and organs is accompanied by the infiltration of white blood cells such as neutrophils and macrophages. These immune system cells, and activated neutrophils in particular, produce large quantities of superoxide (O2 −·) as a consequence of NADPH oxidase activation. 159 Superoxide is a precursor molecule for H2O2 and various other ROS. As a consequence, chemical and other injuries to tissues, such as the respiratory tract, may be exacerbated if the injury also results in inflammatory responses where white blood cells such as neutrophils are present in significant numbers. 160,161 The effects of H2O2 are at least partly mediated via hydroxyl radicals (·OH), suggesting that numerous forms of ROS can act as TRPA1 agonists. 162 The most important strong oxidant produced by neutrophils is HOCl, an effective microbiocidal agent. 159 This is generated by myeloperoxidase-catalysed oxidation of Cl− by H2O2. 160 Inflammatory neutrophils can also produce OCl− endogenously. 111
In inflammatory responses in the interstitial airway, it is possible for neutrophil-derived OCl− to achieve concentrations in the millimolar range. This is much greater than the EC50 required for the activation of TRPA1. 111,163 The lipid peroxidation that occurs when plasma membranes are exposed to exogenous oxidants, or OCl− or ROS, results in the generation of TRPA1-active inflammatory mediators such as prostaglandins, isoprostanes (prostaglandin-like compounds formed in vivo from the free radical-catalysed peroxidation of essential fatty acids, primarily arachidonic acid, without the direct action of cyclooxygenase enzymes), 4HNE and 4ONE. 164,165 The latter two species are chemically similar to acrolein, 3 though only in the sense that the substructure of acrolein features in both molecules. The reactive nature of OCl− as an oxidant also results in tissue damage during inflammation processes. 166
The species issue
TRPA1 ion channels from many mammalian species (e.g. human, rat and mouse) can be activated by many types of electrophilic molecules via a common mechanism (covalent modification of a target Cys residue in TRPA1). However, there is an issue associated with species-related antagonism or activation of the ion channel which is currently impacting research into analgesia approaches which target TRPA1 ion channels. For example, the compounds CMP 1, CMP 2, CMP 3, AMG 2504, AMG 5445, AMG 7160 and AMG 9090 (Figure 10) inhibit human TRPA1 function. However, CMP 1, CMP 2, CMP 3, AMG 5445 and AMG 9090 (the latter two compounds being only partial agonists) activate rat TRPA1 ion channels. It should be noted that AMG 2504 and AMG 7160 have little effect on rat TRPA1. These differences between the two species are due to opposite gating effects. 19,167,168
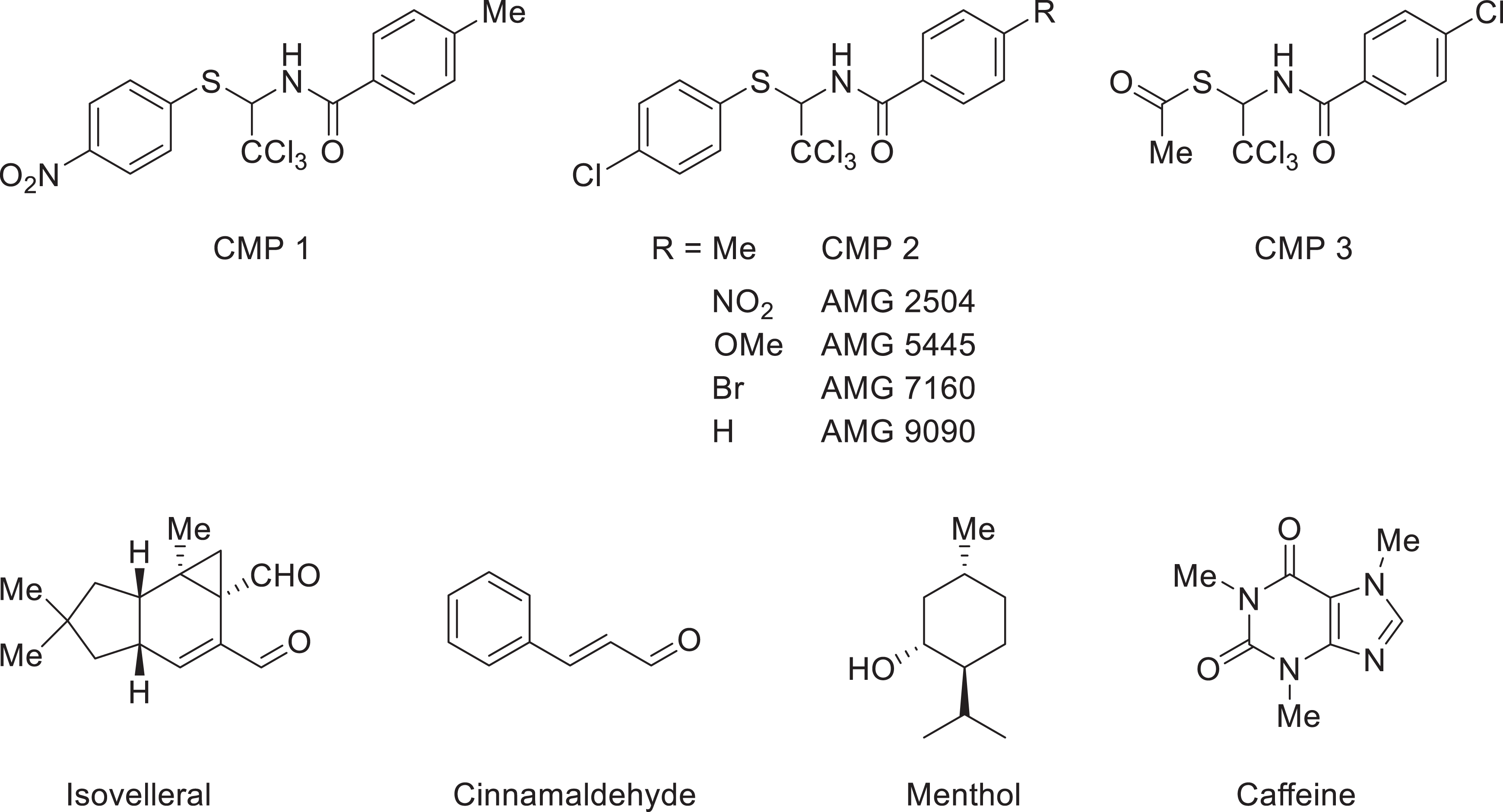
Some antagonists and agonists of TRPA1 channels of some animal species. Top row: Drugs designed as antagonists. Bottom row: The fungal TRPA1 agonist isovelleral, the plant-derived agonists cinnamaldehyde and menthol, and the hTRPA1 antagonist caffeine from vegetation from Africa, East Asia and South America and present in many beverages consumed by humans. TRPA1: transient receptor potential ankyrin 1.
Additional concern regarding species differences arises when considering responses to the commonly used TRPA1 agonist, AITC. Chen et al. 169 showed that when human, rhesus monkey, rat and mouse TRPA1 ion channels were challenged with AITC under the same conditions, all four types of ion channel were activated. However, while the non-human primate (NHP) and human versions of TRPA1 were not sensitive to changes in temperature from 8°C to 24°C, the colder temperatures did activate rat and mouse TRPA1 ion channels. This difference in thermal response was reported to be due to a single amino acid difference in the S5 transmembrane domain of TRPA1. Additionally, the TRPA1 agonist menthol (Figure 10) causes activation of human TRPA1, whereas in the mouse this agonist activates TRPA1 ion channels at low doses, but blocks these channels at high doses. 170 Human TRPA1 activity is suppressed by caffeine (Figure 10), whereas in mice this ion channel is stimulated by caffeine. 171 These species differences are judged to have hindered drug discovery research though interestingly, menthol and caffeine are considered to be members of a group of compounds considered to be non-reactive TRPA1 agonists because they do not react with the nucleophilic target Cys residues of TRPA1. 46 One report has also commented upon inter-strain differences between C57BL/6 and Balb/c mice in relation to TRPA1 agonist- and allergen-induced inflammatory responses, necessitating even more careful consideration of what constitutes an appropriate in vivo model. 72 Amino acid sequence homology among TRP ion channels varies substantially between certain mammalian species, particularly for TRPA1 ion channels (Table 1).
The potential for TRPA1 antagonists to alleviate pain and acute irritation following exposure to TRPA1 agonists suggests a potential to ameliorate the acute effects of many TRPA1 activators. Such properties should be explored with a view to assessing their ability to reduce the risk of short-term morbidity or chronic health problems developing in the longer term in survivors, particularly as TRPA1 ion channels are involved in the initiation of inflammatory reactions after they have been activated. The rational selection of antagonists of TRPA1 ion channels to mitigate the effects of exposure to reactive agonists may reduce casualty levels, but this would require studies which utilise the appropriate mix of in vivo and in vitro approaches to allow more relevant extrapolation from in vitro models to in vivo models and ultimately to humans. It therefore follows that accuracy of the modelling of relevant toxicological and pathological responses to inhaled chemical agents is essential, as many of these may induce species-specific TRPA1 agonist responses. This is supported by Bianchi et al., 46 who compared the pharmacological profile of the rhesus monkey TRPA1 (rhTRPA1) channel with that of human, rat and mouse TRPA1. They found that the rhTRPA1 profile was similar to that of human TRPA1 (hTRPA1), but that while rat and mouse TRPA1 (rTRPA1 and mTRPA1) had similar profiles, their pharmacology differed from hTRPA1 and rhTRPA1. This was attributed to their amino acid sequence homologies (expressed as the percentage to which they were identical) which for hTRPA1 versus rhTRPA1 was 97%, rTRPA1 versus mTRPA1 was 97% and rhTRPA1 versus rodent was 79% (Table 1).
This led Bianchi et al. 46 to propose that the rhesus macaque might be of value as a model for TRPA1 in preclinical studies. The similarity in the TRPA1 sequence homology between hTRPA1 and chimpanzee TRPA1 is striking (99.7% identical) (Table 1). However, there are significant ethical, legal and logistical considerations involved in the use of NHPs. Alternative species to consider may include the guinea pig. Guinea pigs are frequently used as models for determining TRPA1 ion channel-mediated responses. Satoh et al. 172 reported that desflurane induced the contraction of airways in guinea pigs through tachykinin release from afferent C-fibres, a process which was due to TRPA1 activation. 173 An important point to note in the context of RCAs and war gases is that selective activation of TRPA1 by respiratory irritants in humans and guinea pigs causes a cough response. 109,174,175 The cough response is important, as it helps to clear the upper respiratory tract of foreign material, and in doing so, reduces the risk of noxious materials penetrating more deeply into the respiratory tract. An experimental system for measuring this response in guinea pigs exposed to AITC has been described by Daller et al. 176 A review by Padilla-Carlin et al. 177 indicated that this species is a valuable model of many infectious diseases and therapeutic intervention strategies. This is because humans and the guinea pig have similarities with respect to pulmonary physiology, 178 immunological (especially with respect to innate immunity) and hormonal and corticosteroid responses. 179 –183 Shiratori et al. 183 have indicated that the innate immunity system of the guinea pig is more similar to that of humans rather than that of the mouse, especially in relation to CD1 subclasses (of which guinea pigs possess eight isoforms that are homologues of human CD1e and human group 1 CD1b and CD1c subclasses).
Guinea pigs may therefore be of value as an in vivo model for assessing exposure to airborne TRPA1 agonists and the potential therapeutic benefit of TRPA1 antagonists. However, according to Romanenko et al., 184 the paucity of information about the genetics of the guinea pig as an experimental animal in comparison to other models such as the rat and the mouse is a major reason for the guinea pig falling out of favour as a model species over the past 10–15 years. Indeed, until the Romanenko et al. 184 article, there was no comparative analysis available between the karyotype of man and that of the guinea pig. In consequence, rodent models such as the mouse 185 –191 and rat 106,192 –195 and other species such as the rabbit 196,197 have featured prominently in studies on the toxicology of chlorine exposure.
According to Hoyle and Svendsen, 198 responses to chlorine are species dependent, though in general it causes persistent inflammation, tissue remodelling and airway obstruction. The distal airways, because they have insufficient basal cells to act as progenitor cells, tend to undergo less efficient repair following chlorine exposure and are also prone to subsequent chronic inflammatory and fibrotic responses. In contrast, the larger airways, with their pseudostratified epithelium, can undergo more efficient repair because the basal epithelial cells which survive the chlorine exposure act as progenitor cells to replace the multitude of differentiated cell types that comprise the surface of the upper respiratory tract. However, as noted by Lindsay, 199 the return of normal structure and function to damaged lungs depends on various factors such as the extent of the damage to lung tissue as well as the regenerative capacity of progenitor cells within the lung. Indeed, according to O’Koren et al., 189 from studies of mice exposed to chlorine gas, bronchiolitis obliterans – a significant cause of dysfunctionality in airways exposed to toxic chemicals such as chlorine – was preceded by the loss of basal epithelial cells (that act as progenitor cells for the epithelium of the large airways). This loss occurred at high concentrations of chlorine (350 ppm) but not at lower concentrations (200 ppm). An unfortunate characteristic of both alveolar and airway epithelia is that they have a low capacity for regeneration and an intrinsically lower level of cell turnover than that of the other major epithelial tissue types, such as those of the gastrointestinal tract and the skin. 200
Given that the above in vivo models (notably rat and mouse) are extensively used in TRPA1 ion channel studies, and that they all have intrinsic limitations, there are some species that are conspicuous by their absence in TRPA1-related studies. These include the domestic pig (Sus scrofa) and its various strains (e.g. the large white pig, the Göttingen miniature pig and the Yucatan miniature pig). This is surprising, given that they have been extensively used in many areas of research of direct relevance to humans. Indeed, Larsen and Rolin 201 have noted that the pig is a useful species for the in vivo modelling of pathophysiological and physiological responses to various disease states of significance to man, particularly as numerous organ systems in the pig are similar to those in humans. According to Štembírek et al., 202 physiological similarities between the pig and the human mean that the extrapolation of experimental results from the former to the latter ‘is more exact compared with other experimental animals such as the mouse, rat or rabbit’. These similarities relate to the anatomy of the cardiovascular and urinary systems and the skin as well as the function of the immune system and the gut. 203 According to Jugg et al., 204 small animals are not necessarily the optimal choice when one needs to extrapolate aspects of the toxicological assessment of the effects of lung damaging agents to humans. In particular, large animal models such as pig and dog have a breathing physiology which more resembles that of humans. Additionally, their lung size and anatomical structure together with their larger vasculature, tracheas and bronchi permits these species to be fully instrumented using equipment of the type employed in hospital intensive care units, 205 thus allowing constant monitoring of biochemical and physiological parameters during experimental procedures.
In contrast, rodent models have inherent limitations as there are numerous differences in physiology between primate species and rodents. This necessitates carefully considered selection of the appropriate species which model human disease states relevantly, and the pig is often selected as a clinically relevant model for various disease states, 206 as well as wound and trauma care. The pig and the dog have also been utilised for preclinical assessment of pharmaceuticals administered by the inhalation route. 205 According to Giraud et al., 207 pigs are of value in assessing preclinical technologies and constitute an appropriate alternative to primates as in vivo models for extrapolating the potential value of therapeutic intervention approaches to conditions which affect humans.
In this context, Dstl has had a long history of using the pig model, from studying aspects of the mechanism of action of blistering agents (vesicants) such as sulfur mustard and Lewisite, 208 –210 to the analysis of tissue repair processes associated with the lesions caused by these agents, 211 –214 as well as assessing ways of protecting pig skin against vesicants. 215 –217 The pig has also been used to establish the effects of blast injury 218 and haemodynamic and metabolic responses to haemorrhage, 219 with a view to establishing the pig as the preferred in vivo model to respond reproducibly to standardised blast wave energies and controlled haemorrhage. 220 This enabled the efficacy of combined hypotensive resuscitation and recombinant activated factor VII treatment in the management of arterial haemorrhage to be defined. 221 This model was also used to assess the value of prehospital resuscitation with combined packed red blood cells (PRBCs) or a combination of PRBCs and fresh frozen plasma in the attenuation of acute trauma coagulopathy. 222 Liu et al. 223 have used the pig to study the effects of non-penetrating ballistic impact on the brain, and the pig has been employed as an experimental model of intracranial hypertension. 224
The pig has also been used to model the following conditions: type 1 diabetes, 201 diabetic wound healing, 225 non-healing in bone, 226 human autosomal dominant retinitis pigmentosa, 227 glaucoma, 228 fulminant hepatic failure, 229 ischaemic heart disease, 230 congestive heart failure, 231 bariatric surgery on obesity, 232 bacterial infection, 233 –236 Trypanosoma cruzi infection, 237 viral myocarditis, 238 cystic fibrosis 239 and Duchenne muscular dystrophy. 240 The pig has also been recommended as a surgeon’s training model for human surgery (e.g. for axillary lymphadenectomy). 241
It is of potential interest that despite the very broad range of applications of the pig as an in vivo model used to assess various chemical, biological and physical hazards, and its value in medical countermeasure research, it is not used as a model to investigate the utility of TRPA1 antagonists. This is despite the use of the large white pig as a fully instrumented in vivo system in studies relating to the inhalational effects of toxic gases such as phosgene and chlorine. Studies such as that by Gunnarsson et al. 242 showed that the pig was very sensitive to chlorine exposure (a 10-min exposure to 140 ppm chlorine (100 L) causing severe lung injury and subsequent high mortality within 6 h). Wang et al. 243 explored the value of nebulised budesonide and intravenous betamethasone (both anti-inflammatory drugs) on chlorine-exposed pigs (having received a 15-min exposure to 400 ppm chlorine) and claimed ‘positive effects on recovery of lung function’ with respect to reduced lung wet-weight to lung dry-weight ratios and improved lung histology. However, failure to explore the potential value of TRPA1 antagonists against agents such as chlorine gas may be a potentially missed opportunity (though it should be noted that the pig is conspicuous by the paucity of papers on its use as a model in TRPA1 studies). Colombo et al. 136 have reported that thymol activates the TRPA1 gene in pig pyloric and oxyntic mucosa, whereas it downregulates genes associated with other TRP channels. Boudry and Perrier 244 also reported that ion secretion in the small intestine of the pig is increased by TRPA1 activators such as cinnamon and thyme. Such studies have been conducted in pigs with a view to assess the value of thymol as an orally administered antibiotic for pigs and as a means to enhance food intake by pigs through improving the flavour of animal feed. However, at high doses of added thymol, its bitter taste can cause pigs to refuse feed containing this TRPA1 agonist. The pig’s olfactory system has a sensitivity that is extremely high in relation to most other mammals and is well suited to such feedstock studies. 245 Such research however, occupies a rather narrow niche. In reviews by Bode et al. 246 and Kuzmuk and Schook, 247 the role of the TRPA1 ion channel in the pig is simply not discussed. This is interesting, given the high level of sequence homology of the pig genome to man (60%) compared to the sequence homology between rodents and man (40%). 248,249 Additionally, there is a greater similarity in chromosomal structure between the pig and the man than other species such as the mouse, rat, cat, dog, cattle and horse. 250,251 In contrast, the most extreme example of a mammalian species which has adapted to its environment by limiting sensitivity to various TRPA1 agonists such as ammonia is the naked mole-rat (Heterocephalus glaber), a burrowing mammal native to parts of East Africa. In a study by La Vinka et al., 252 it was found that mole rats do not avoid ammonia (in the form of sponges saturated with a 10% solution) but they can sense it. This may be an adaptive advantage as this mammal lives underground in tunnels in large social groups, where ammonia may accumulate in certain locations (e.g. in latrines) in the burrow complexes. Mole rats were also found to be insensitive to capsaicin (a potent TRPV1 agonist) applied to their nostrils in the form of a 1.5% solution. This case illustrates how vital it is to carefully select the relevant species for in vivo toxicological and therapeutic studies.
Summary
The selection of the optimum in vivo model system becomes critical to successful identification of relevant biochemical and physiological responses which can be used to benchmark the efficacy of potential therapeutics. This is an important point, for candidate therapeutics may themselves have species-specific characteristics in terms of the way they interact with TRPA1 ion channels (and other potential targets) to attenuate potentially irritating, debilitating or even potentially lethal responses. In effect, for agents that are known to target ion channels such as TRPA1, these models would ideally be based on NHPs, though the associated ethical, legal and logistical considerations may require initial research and efficacy testing to be conducted on another species such as the guinea pig, which has more relevant pulmonary physiology and immune equivalents to humans than rodent species. This is not necessarily mandatory for other TRP ion channels, as can be seen from Table 1, but does highlight the need to be aware of the relevant molecular target(s) for the agonist under investigation and whether the species being employed is the most appropriate one.
This notwithstanding, accurately selecting the appropriate molecular targets in the most relevant in vivo models would also have the benefit of better read-across from in vitro studies (i.e. if using NHPs as the in vivo model of choice, preliminary work should be conducted on in vitro cell systems expressing human or NHP TRPA1). This would give greater confidence in establishing the potential value of candidate antagonists and other potential therapeutic compounds (when assessed in an in vitro context) for subsequent testing using in vivo models based on NHPs. This close matching of in vitro with in vivo models clearly requires the correct selection of the latter to make best use of the properties and potential of the former and may require a more critical evaluation of the relevance of systems that are currently being used in medicinal and toxicological contexts.
Footnotes
Authors’ note
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
