Abstract
Introduction:
Our objective was to describe the characteristics of liquid laundry detergent packet (LDP) exposures and to develop referral and treatment recommendations.
Methods:
This retrospective cohort study investigated LDP exposures reported to the National Poison Data System from January 1, 2013 through June 30, 2014. Three medical toxicologists reviewed the most significant exposures (n = 450).
Results:
Of 17,857 reported LDP exposures, 13,307 involved only an LDP (no other substance) and were followed to a known medical outcome. The median age was 2 years (range 12 days to 100 years). Approximately 10% of exposures reported a major or moderate effect. The most common symptom was vomiting (51.7%; n = 6875), but stridor or aspiration pneumonia and respiratory depression secondary to central nervous system effects also occurred. Two pediatric and two adult deaths occurred, but no causal mechanism leading to death could be identified in any of the deaths.
Conclusions:
LDPs occasionally produce a toxidrome of vomiting, stridor, hypoxia, and sedation with metabolic acidosis and respiratory failure. These symptoms and the availability of LDPs highlight the need for referral and treatment recommendations and efforts to minimize unintentional exposures. Review of data from US poison centers may provide referral and treatment recommendations that improve patient outcomes.
Keywords
Introduction
Liquid laundry detergent packets (LDPs) were introduced to the United States (US) market in 2010 and became widely available by 2012. 1 LDPs contain a combination of concentrated liquid detergents enclosed in a polyvinyl alcohol film designed to dissolve upon contact with water. The exact formulas and ingredient concentrations of LDPs vary, with as many as 50 chemicals reported for a single product. 2 –5 Despite the chemical variety, anionic and nonionic surfactants, propylene glycol, and glycerin are common LDP ingredients. LDPs are further unique because they contain up to 90% less water than traditional liquid laundry detergents.
LDP exposures reported to US poison centers (PCs) increased after their release, suggesting unintended safety risks in a pattern similar to the European release of LDPs a decade earlier. In separate statements, the American Association of Poison Control Centers (AAPCC) in May 2012 and the Centers for Disease Control and Prevention (CDC) in October 2012 warned about the risks associated with LDP exposures. 6,7 In particular, the CDC highlighted that, when compared to regular laundry detergent exposure, outcomes in younger children (<5 years) were more severe. The report also noted gastrointestinal and respiratory effects and mental status changes among LDP exposures.
Despite continued reports of LDP exposures to US PCs, formal referral and treatment recommendations to manage such exposures do not exist. 8 –20 The goals of this article are to characterize the medical presentation of patients exposed to LDPs and to propose referral and treatment recommendations to serve as a reference for the best practices in the management of exposures. The recommendations presented in this article are intended to be applicable to first responders, emergency physicians, PCs and other medical management or triage service providers, pediatricians and other pediatric clinicians, and other health-care professionals who may be consulted following an exposure to LDPs. Current scientific and clinical information are the basis for these recommendations. The authors recognize that specific patient care decisions may be at variance with these recommendations and are at the discretion of the patient and health professionals providing care, considering all of the circumstances involved; these recommendations do not substitute for clinical judgment or informed patient choice.
Methods
The National Poison Data System (NPDS) captures data from 55 regional AAPCC-accredited PCs throughout the US and its territories. PC staff are trained health-care professionals that take calls from the public and health-care providers to manage patients potentially exposed to pharmaceutical and non-pharmaceutical substances. Using standardized data collection systems, each regional PC records information, and PC staff follow up on cases that require medical management. Medical outcome is recorded after follow-up is complete, if follow-up is determined to not be required due to the low risk of the exposure, or if follow-up cannot be completed. Exposure data are collected into local databases as descriptive narratives and coded categorical variables (e.g. age, weight) using a standardized coding system. Automatic uploads of categorical data fields from local databases to NPDS occur every 8 min.
In this retrospective study, NPDS was searched systematically from January 1, 2013 through June 30, 2014 for all human exposures involving an LDP; data were received on 10 November 2014. A panel of clinicians was assembled to review the NPDS data. Data from exposures followed to a known medical outcome were included in the review because the quality of case documentation tends to be more thorough for these exposures. Descriptive statistics were calculated to summarize demographics, exposure characteristics, and outcomes, with stratification by route of exposure and medical outcome. All analyses were performed using SAS® software, version 9.3 (SAS® Institute Inc., Cary, North Carolina, USA).
A panel of three physicians with over 50 combined years of expertise in pediatrics, intensive care, emergency medicine, toxicology, and injury prevention performed the clinical analysis. A senior scientist with nearly 15 years of experience in epidemiological studies and consumer product safety evaluation moderated the review. After initial review of the data summary, a case-level review of selected exposures was used to strengthen the understanding of exposures, therapies, and associated outcomes. Exposures with (1) a serious medical outcome (NPDS medical outcome of death or major effect the (patient experienced life-threatening symptoms or significant residual disability or disfigurement as a result of the exposure)) OR (2) exposure reason of suspected suicide were selected for case-level review. These criteria were intended to capture the most severe exposures regardless of age. Additional exposures for case-level review were selected from LDP-only (no other substance) exposures in children <6 years of age with (1) clinically significant effects (acidosis, coma, respiratory depression, burns, seizure) or treatments (intubation, ventilation, cardioversion) OR (2) a medical outcome of moderate effect with reported admission to a health-care facility (HCF; noncritical care unit, critical care unit, or psychiatric facility). The pediatric subset of cases was further explored because children <6 years of age are the most common subpopulation involved in LDP exposures and single-substance exposures allow for cleaner evaluation of the contribution of LDPs to outcomes controlling for potential contribution from other substances.
Key data elements from each exposure were independently abstracted into a single database by two trained abstractors. Abstracted data were then reviewed and reconciled by a single reviewer prior to the panel’s final review and deliberations. The case record was referenced for all data abstraction discrepancies. The clinicians reviewed the full record for each case and created recommendations based upon their shared observations. The clinicians discussed each case until they reached full consensus on each element of the recommendations. Presented here is the descriptive summary of all exposures reviewed along with observational highlights gleaned from the case-level review. Each panelist reviewed the data, contributed to the discussion of exposures and the recommendations, agreed upon the recommendations as drafted, and reviewed and approved the final recommendations.
The Colorado Multiple Institutional Review Board approved this study on January 6, 2015. Human subjects were not contacted in the execution of this study and consent was not feasible.
Results
Of 17,857 total LDP exposures reported to NPDS, 13,435 (75.2%) were followed to a known outcome. The remaining 4422 (24.8%) involved a medical outcome of not followed due to minimal clinical effects expected (n = 3228, 18.1%) or nontoxic exposure (n = 278, 1.6%) or unable to follow with potentially toxic effects (n = 916, 5.1%). A total of 13,307 (74.5%) exposures were followed to a known outcome and involved an LDP-only. Four hundred and fifty-four exposures meeting the case-level review were identified, of which 450 (99.1%) were received from the regional PCs (Figure 1). Four exposure records were not submitted because the regional PC elected not to participate in the study.

NPDS case inclusion disposition. aSelected for case-level review; 102 cases received and included in case-level summary. bChildren <6 years; selected for case-level review; 348 cases received and included in case-level summary. Disposition of cases selected for inclusion in both the exposure summary and the case-level review. NPDS: National Poison Data System.
The median patient age for LDP-only exposures followed to a known outcome was 2 years (range 12 days to 100 years). Most exposures (96.8%, n = 12,887) were in children <12 years of age, with 94.1% (n = 12,517) specifically in children <6 years of age. Gender was nearly equally divided with 51.3% of exposures involving males. Exposures primarily occurred through an oral route (88.7%) but also through ocular (15.0%) and dermal (10.6%) routes. Multiple routes (e.g. ingestion and dermal) were reported for some exposures.
Medical outcomes and clinical effects
Table 1 summarizes medical outcome based on the route of exposure among the 13,307 LDP-only exposures followed to a known medical outcome. The majority of exposures followed to a known medical outcome (88.5%) resulted in no effect or minor effect. However, 1390 (10.4%) exposures resulted in moderate effect or major effect and four deaths (<0.1%) were reported. Table 2 summarizes the related clinical effects among the 13,307 LDP-only exposures followed to a known medical outcome and among the subset of 1390 exposures with clinically significant medical outcomes. Vomiting was the most common clinical effect in both categories of severity. In exposures with a clinically significant medical outcome, acidemia was reported based on respiratory depression, lactic acidosis, or both. Case-level review showed that dermal, ocular, and mucosal irritation occurred, causing erythema and some blistering of the skin. Stridor was also observed. Corneal abrasions were reported, but long-term sequelae were not. No significant damage to the esophagus or trachea was found on endoscopy. The time to symptom onset was estimated in 332 (75.1%) of the 442 case-level review cases with a medical outcome of at least minor effect (minor effect, moderate effect, major effect, or death). Seventy-nine percent (n = 261) of cases with an estimated time to symptom onset reported that symptoms developed within 1 h after exposure; symptoms developed within 5 min in 19.6% (n = 65), within 5–15 min in 16.6% (n = 55), within 15–30 min in 19.9% (n = 66), and within 30 min to 1 h in 22.6% (n = 75). Ninety-six percent (n = 319) of exposures became symptomatic within 4 h, and 13 patients reported to have developed symptoms within 4–24 h postexposure.
Medical outcomes based on route of exposure for LDP exposures reported to US PCs from January 1, 2013 to June 30, 2014.a

LDP: laundry detergent packet; US: United States; PC: poison center.
a Definitions of outcomes 21 :
b More than one route could be reported for each exposure, therefore the sum of the individual routes exceeds the total number of reported exposures (n = 13,307).
Most common related clinical effects associated with LDP exposure as reported to US PCs from January 1, 2013 to June 30, 2014.

LDP: laundry detergent packet; US: United States; PC: poison center.
Table 3 presents the most common therapies performed among the 13,307 LDP-only cases followed to a known medical outcome and among the subset of 1390 exposures with clinically significant medical outcomes. Dilute/irrigate/wash was the most commonly reported therapy overall (77.2%) and in exposures that resulted in a clinically significant medical outcome (68.0%). The nonspecific “other” therapy was the third most commonly reported therapy performed. To better understand these therapies, specific “other” therapies were summarized among the exposures included in the case-level review. Of the 104 (23.1%) exposures included in the case-level review with the NPDS “other” therapy, suctioning (41.3%, n = 43), analgesics (18.3%, n = 19), and antacids (17.3%, n = 18) were the most common specific therapy mentioned.
Most common therapies performed after LDP exposure as reported to US PCs from January 1, 2013 to June 30, 2014.

LDP: laundry detergent packet; US: United States; PC: poison center; IV: intravenous.
Ocular exposures
As presented in Table 1, 2001 (15.0%) exposures involved an ocular exposure. No effect or minor effect was reported in 78.2% of all ocular exposures. In exposures reporting only an ocular route (no other exposure route), 76.4% (n = 859/1124) reported no effect or minor effect. While corneal abrasions were reported among the 13,307 LDP-only exposures followed to a known medical outcome, no patients in the case-level review developed subsequent deep corneal abrasions or ulcers that required interventions other than topical antibiotics and follow-up.
Dermal exposures
As presented in Table 1, 1415 (10.6%) exposures involved a dermal route. No effect or minor effect was reported in 88.3% of all dermal exposures. Of these, 224 (15.8%) experienced erythema or burns associated with topical contact. Among exposures included in the case-level review, one exposure with dermal route only was associated with a more significant medical outcome (major effect). This included chemical dermatitis and partial thickness burns requiring debridement likely due to delayed removal of contaminated clothing and decontamination using a “baby wipe.”
Oral exposures
The most commonly reported clinical effect associated with ingestion (oral route) was vomiting (58.2%). A number of case-level review patients presented with drooling and refusing to swallow liquids; in these exposures, oral and throat irritation was also found. These symptoms were sufficiently reminiscent of ingestion of caustic materials to prompt the recommendation for endoscopy; of the exposures included in the case-level review that involved an oral route, an endoscopy was performed in 110 patients (23.3%). Erythema and mild mucosal damage were also reported in 66.4% (n = 73) of these cases, but there were no reports of circumferential or other significant injury or recommendations for more aggressive care or repetitive endoscopy. These findings may be consistent with the chemical nature of the exposure. Ranging from 6.5 to 9.0, the pH of LDPs is not in the same range as known caustic or acidic cleaning solutions.
It was observed that LDPs produce profound changes in acid-base status in 61 (13.6%) of the patients whose exposure records were reviewed. Table 4 presents blood gas data for the 38 patients with complete blood gas information who fell into one of the three categories of acidosis: respiratory depression yielding a respiratory acidosis (n = 3, 7.9%), metabolic acidosis with attempted respiratory compensation (n = 19, 50.0%), and both respiratory and metabolic acidosis contributing to acidemia (42.1%, n = 16). In another 23 patients, incomplete or insufficient blood gas data prevented categorization. Elevated serum lactate was reported in 27.9% (n = 17) of all patients with documented acidosis, suggesting that metabolic acidosis was mediated via increased production or inhibited metabolism of lactate. Of cases with elevated serum lactate, reports of hypotension occurred in 23.5% (n = 4).
Reported blood gas data for patients found to have acidosis status post-LDP ingestion.
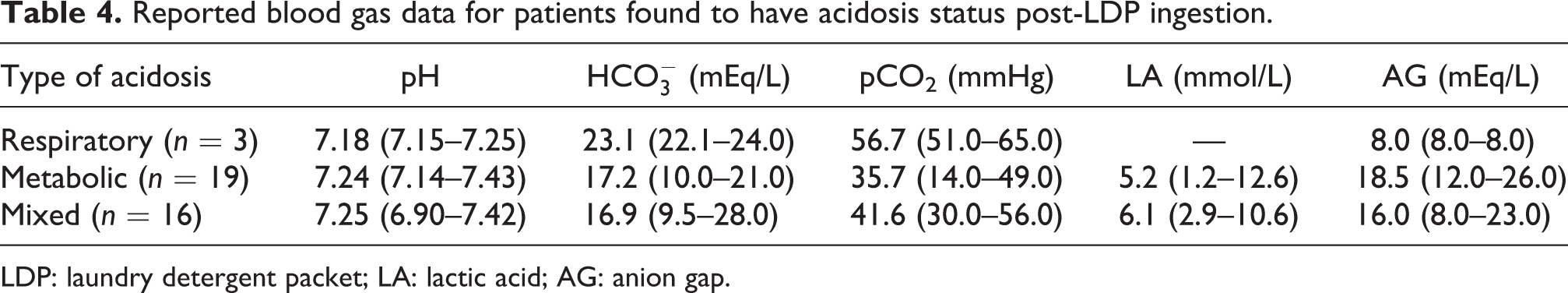
LDP: laundry detergent packet; LA: lactic acid; AG: anion gap.
Deaths
Four deaths were reported and are detailed in Table 5. Two involved elderly patients with dementia and two involved pediatric patients <2 years of age. Both adults ingested one LDP unnoticed and were taken to the emergency department (ED). One became asystolic suddenly within 24-h post-ingestion and had a do not resuscitate order in place. The other patient left the ED but returned 24 h later and was admitted to the intensive care unit (ICU) with difficulty breathing. He had a do not resuscitate order in place and later died of aspiration pneumonia and severe sepsis.
Detailed information on four deaths associated with LDP ingestion abstracted from PC case notes.

LDP: laundry detergent packet; PC: poison center; IV: intravenous; ICU: intensive care unit; AST: aspartate aminotransferase; ALT: alanine aminotransferase; DNR: do not resuscitate.
Both pediatric exposures occurred in children <2 years of age who bit into LDPs (Table 5). One deteriorated quickly in the ED and the other was transferred to the ICU and put on a ventilator. Both went into cardiac arrest and could not be resuscitated. The clinicians conversed with the regional PCs that managed these exposures and reviewed the autopsy data; a common causal mechanism associated with the LDP exposure leading to cardiac arrest and death could not be identified in these cases.
Discussion
Ocular exposures and outcomes
Ocular introduction of LDP ingredients due to product rupturing was one of the first problems associated with LDPs. 24 The rather ominous concerns of corneal injury were somewhat ameliorated by the failure to find any long-term adverse outcomes or need for extraordinary interventions. Given the limited severity of outcomes in ocular exposures and the inconsistency in the ability of EDs to adequately perform ocular decontamination and fluorescein slit lamp evaluation in pediatric patients, the clinicians did not feel that rigid recommendations involving these types of diagnostic tests were warranted or realistic. While it is still important to be respectful of any injury to the eye, conservative management seemed to result in good outcomes for most cases. Table 6 presents suggested HCF referral and treatment recommendations for ocular exposure to LDPs.
Suggested HCF referral and treatment recommendations for ocular exposures to LDP products.

LDP: laundry detergent packet; HCF: health-care facility.
a If caregiver is unable to perform any of the following recommended therapies, refer patient to HCF immediately.
Dermal exposures and outcomes
Similarly, the possibility of severe dermal injury may still exist, but tissue inflammation manifesting as erythema and skin blistering seemed to be the most prevalent effects. The case described earlier in this article that involved prolonged skin contact and use of a detergent-like diaper wipe seemed to result in a more serious injury. The value of this single observation is limited but can provide a cautionary note and a reasonable recommendation that early removal of clothing and decontamination with water only is optimal. Table 7 presents the HCF referral and treatment recommendations for dermal exposure to LDPs.
Suggested HCF referral and treatment recommendations for dermal exposures to LDP products.

LDP: laundry detergent packet; HCF: health-care facility.
a If caregiver is unable to perform any of the following recommended therapies, refer patient to HCF immediately.
Oral exposures and outcomes
Ingestion of LDPs may produce drooling, vomiting, bronchospasm, and stridor. Oral exposures seemed to evolve into multiple scenarios that, while they may overlap, must be considered individually for management. As in a previous case series, respiratory depression occurred in less than 1% of the patients reviewed. 13 Despite low frequency, the seriousness of this outcome merits discussion.
Respiratory failure seemed to evolve in one or more of three scenarios. The first was obtundation and loss of airway reflexes prompting intubation and mechanical ventilation of patients. However, a specific biochemical or pharmacologic etiology of this change in mental status is unknown. The neuropharmacology of nonionic surfactants, which possess general anesthetic properties, has been previously described, and the authors hypothesize that as one possible etiology of respiratory failure. 25 Obtundation with loss of airway is the most likely scenario of respiratory failure for the 16-month-old patient whose death is described in Table 5.
The second scenario of respiratory failure appeared to involve upper airway obstruction described as stridor in a number of cases and as an etiology of the respiratory failure in some patients. The term “croup-like illness” was also employed. These symptoms are presumably related to the irritant effect of chemicals on the airway. The most frequent treatment approach was racemic epinephrine or corticosteroids, although there are no prospective data to determine their effectiveness. In some cases, concerns over damage to the airway prompted fiber-optic laryngoscopy. In no case was a lesion identified that required intervention or specific treatment.
The third scenario involved aspiration of the LDP product into the airways with X-ray changes and bronchospasm leading to respiratory failure. In the majority of cases, radiographic changes as well as the bronchospasm and respiratory failure appeared to be short-lived and did not generate the serious inflammatory events associated with aliphatic hydrocarbons. However, the clinicians reached consensus that this was the most likely cause of respiratory failure in the deaths involving the 7-month-old, >89-year-old, and 72-year-old described in our data (Table 5). Treatments varied across cases and included inhaled bronchodilator therapy, antibiotics (for presumed aspiration pneumonia), and oxygen, with intubation and mechanical ventilation as clinically indicated. No specific recommendations on the effectiveness of these treatments can be made from the present data. In fact, strict recommendations for HCF referral in these instances may result in over-referral to EDs.
Oral exposures did not appear to produce long-term tracheal or epiglottic damage. Although pH is not the only determinant of injury, LDPs do not fall in a pH category associated with severe esophageal and gastrointestinal damage. This is consistent with the lack of circumferential damage or specific interventions associated with the endoscopic findings observed among the cases reviewed here. The need for endoscopy has been questioned previously after a small series of patients who underwent endoscopy required no additional interventions based on their endoscopic results. 12 The clinicians were unable to identify changes in case management associated with findings from endoscopy. This led to the (perhaps controversial) conclusion that routine endoscopy, with its own inherent risks, is an unnecessary hazard to the child.
In a number of other cases, a metabolic acidosis was observed, and, in many cases, it was documented that lactic acid was elevated. In some cases, lactate was elevated in the absence of decreased pH. Previous authors have suggested that the metabolism of propylene glycol, starting with the enzyme alcohol dehydrogenase and ultimately yielding lactate may be the cause of the metabolic derangement in these patients. 11,26
Severe human exposure to propylene glycol usually occurs from iatrogenic exposure while being used as a preservative in other drugs. A recent study discussed the pathophysiology of this process and suggested that treatment of LDP ingestions includes an early intervention with fomepizole which might block the conversion of this chemical to avoid the lactic acidosis. 27 It is impossible from the present case series to estimate the LDP quantities ingested or the amounts of propylene glycol involved and whether this acidosis is plausible on a stoichiometric basis. In discussing this fact, the clinicians felt that the rapid onset of the acidosis in these cases would likely preclude intervention with fomepizole in a timely fashion. As there is no evidence to support the efficacy of such an approach, any treatment with fomepizole must be considered experimental. Table 8 presents the HCF referral and treatment recommendations for oral exposures.
Suggested HCF referral and treatment recommendations for oral exposures (with or without aspiration) to LDP products.

LDP: laundry detergent packet; CNS: central nervous system; HCF: health-care facility; RAD: reactive airway disease; CXR: chest X-ray; PPI: proton pump inhibitor.
a If caregiver is unable to perform any of the following recommended therapies, refer patient to HCF immediately.
This study provides a review of clinical effects associated with over 13,000 LDP exposures reported to US regional PCs. Of the 13,307 LDP-only cases with a known medical outcome, most involved an oral route of exposure (approximately 89%) and no single, early-onset symptom was predictive of outcome. Most exposures (88.6%) resulted in no effect or minor effect. A relatively consistent number of symptoms forming a basic exposure toxidrome emerged, including vomiting, stridor, hypoxia, and sedation. Review of the cases revealed that health-care providers typically managed acute presenting symptoms and delayed effects did not occur. The most commonly performed therapy among all patients was dilute/irrigate/wash. These data should provide some comfort to providers managing these patients.
Several severe adverse events also occurred, but the incidence in comparison to the relatively more minor effects was low. Nevertheless, severe reactions such as aspiration pneumonia, stridor, respiratory failure, and altered level of consciousness did occur. What was most surprising in reviewing the cases was the duration of events. In comparison to a hydrocarbon aspiration, which typically progresses over 72 h and may take up to a week to improve, the aspiration events were relatively short-lived with respect to time on the ventilator. Similarly, the respiratory or metabolic acidosis and altered sensorium lasted relatively short periods of time allowing for extubation generally within 24 h of the event. Stridor in most cases was treated with standard therapy such as corticosteroids and racemic epinephrine (we have no idea whether that improved outcome or not) and also seemed to resolve fairly quickly. Such treatments should be considered when respiratory or metabolic acidosis and altered sensorium develop. More serious events like these underscore the need for preventive measures, and a careful examination of the ingredients of these products is necessary to prevent these infrequent, but severe events.
While these recommendations are based upon a case-level review of 450 patients, limited published experience is available to provide strict evidence-based guidelines. As may be surmised, the variability in narratives did not always allow for direct conversion to structured data. However, it did enable the clinician panel to develop treatment recommendations for health-care providers (Tables 6 to 8). The recommendations were developed based on available data, which were incomplete at times. However, these remain the best data that were available at the time of analysis from NPDS, which is a large and homogenous database with standardized data collection procedures. From the end of our catchment period (June 30, 2014) through the end of 2017, an additional 48,000 exposures involving LDPs were reported to NPDS, which included three additional deaths. 28
Introduction of LDPs into the European market was also met with unique severe toxic effects almost exclusively in children, prompting product changes. In response to the increase in US exposures, a new American Society for Testing and Materials (ASTM) International standard for LDPs was issued on September 15, 2015. 29 This standard includes the addition of an adversant (bitter taste to the capsule), application of a less soluble film to delay the release of the detergent, and increased capsule integrity to withstand a higher level of pressure before breaking and release of the detergent. The goal is to make LDPs less attractive and more difficult for children to access. However, compliance with the ASTM International standard is voluntary. Given the large volume of cases of LDP exposure in such a short time period, adoption of the standard should be enforced and monitored. The present referral and treatment recommendations should also be reviewed regularly to evaluate emerging clinical effects, referral thresholds, diagnostic procedures, treatments, and related recommendations as the products continue to evolve.
Limitations
The encounters that comprise NPDS are collected from spontaneous, self-reported calls made to US PCs. Exposures in NPDS comprise a portion of the total number of incidents that occurred, which is a limitation of this type of passive reporting system. The increasing presence of LDPs in the market place and the continued report of exposures associated with these products highlights the pressing need to optimize referral and treatment recommendations to improve patient outcomes. Systematic reevaluation of emerging data will be necessary to update these recommendations as these products change over time. The volume of reports of exposures reported to NPDS underscores the usefulness of PC data in monitoring the impact of product safety interventions.
A second limitation was that this analysis included different products and formulations that might produce disparity in the clinical effects. Despite the heterogeneity of the LDP products involved in the exposures, the data were surprisingly consistent.
Conclusions
LDPs, in a small percentage of cases, produce a toxidrome of vomiting, stridor, hypoxia, and sedation with metabolic acidosis and possible respiratory failure. The increasing popularity of LDPs and the continued report of exposures to these products highlights the pressing need to minimize unintentional exposures through education and product packaging and to optimize referral and treatment recommendations to improve patient outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The American Association of Poison Control Centers (AAPCC; ![]() ) maintains the national database of information logged by the country’s poison centers (PCs). Case records in this database are from self-reported contacts. They reflect only information provided when the public or health-care professionals report an actual or potential exposure to a substance (e.g. an ingestion, inhalation, or topical exposure), or request information/educational materials. Exposures do not necessarily represent a poisoning or overdose. The AAPCC is not able to completely verify the accuracy of every report made to member centers. Additional exposures may go unreported to PCs and data referenced from the AAPCC should not be construed to represent the complete incidence of national exposures to any substance(s).
) maintains the national database of information logged by the country’s poison centers (PCs). Case records in this database are from self-reported contacts. They reflect only information provided when the public or health-care professionals report an actual or potential exposure to a substance (e.g. an ingestion, inhalation, or topical exposure), or request information/educational materials. Exposures do not necessarily represent a poisoning or overdose. The AAPCC is not able to completely verify the accuracy of every report made to member centers. Additional exposures may go unreported to PCs and data referenced from the AAPCC should not be construed to represent the complete incidence of national exposures to any substance(s).
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by an award granted to Denver Health and Hospital Authority’s Rocky Mountain Poison & Drug Safety from the American Cleaning Institute.
