Abstract
Cyclophosphamide (CYP) and methotrexate (MTX) have been evaluated for their ability to induce toxicity in human peripheral blood lymphocytes (PBLs) and the protective role of mitochondrial and lysosomal stabilizing agents. The potential toxicity effects of CYP and MTX were measured in vitro by cellular parameters assays such as cellular viability, reactive oxygen species (ROS) formation, mitochondrial membrane permeability transition (mitochondrial membrane potential (MMP)) collapse, lysosomal membrane damage, intracellular reduced glutathione (GSH), extracellular oxidized glutathione (GSSG), and lipid peroxidation. Separately, human lymphocytes were treated with concentrations of 0.1, 0.2, 0.4, 0.8, and 1.6 ng/mL for CYP and 1, 2, 5, and 10 µg/mL for MTX for 6 h. Statistical evaluations showed that CYP and MTX significantly decreased the cell viability at the three highest concentrations when compared with both the negative and solvent controls. In addition, CYP and MTX were significantly induced ROS formation, MMP collapse, lysosomal membrane damage, lipid peroxidation, and GSH depletion compared with the controls. Mitochondrial and lysosomal protective agents like cyclosporine A and chloroquine, respectively, decreased cytotoxicity and oxidative stress induced by CYP and MTX. The present results indicate that CYP and MTX are toxic to human PBLs and their toxicity could be ameliorated by mitochondrial and lysosomal protective agents.
Introduction
Cyclophosphamide (CYP) as a prodrug is an extensively used drug to treat wide range of various neoplastic diseases and as an immunosuppressive agent. 1,2 The mechanism of action for CYP is well established. This drug is a nonactive cyclic phosphamide ester of mechlorethamine that induces cross-linking of DNA and RNA strands which leads to an increase in inhibition of DNA polymerase activity and thus prevents cell division. 3 CYP also acts on B cells and T cells and causes suppression of humoral and cell-mediated immunity. 4 CYP has been indicated to produce sister chromatid exchange, chromosomal aberrations, micronuclei, DNA damage, and gene mutations in cells and microorganisms. 5,6 Therefore, the International Agency for Research on Cancer classified it as carcinogenic to humans. 7 It also induces apoptosis and cell toxicity. Previous studies have shown that CYP induces an increase in free oxygen radicals and induced apoptosis. 8 –11
Methotrexate (MTX) is developed for treatment of several certain types of malignancies. MTX is widely used with higher doses in cancer therapy since 1990, and it is also used at much lower doses in treatment of rheumatic diseases. 12 Adverse effects of MTX at high dose may be life-threatening. Plasma concentrations of MTX is an essential part of its toxicity. Therefore, MTX levels should be followed carefully. To avoid MTX toxicity, concentrations should not be above 10 μM at 24 h. 13 Several investigations have showed that MTX-induced cytotoxicity is related with reactive oxygen species (ROS) generation and depletion of cellular and mitochondrial GSH which finally lead to oxidative stress and cell death. 14,15 In vitro and in vivo animal research also indicated that treatment with MTX leads to alteration in the activity of antioxidant enzymes such as catalase, glutathione peroxidase, glutathione reductase, and superoxide dismutase in spinal cord, intestinal mucosa, and liver tissues. 15,16 A recent study from Al Maruf et al. 15 showed that MTX induce mitochondrial injury and cytochrome c release in rat liver hepatocytes.
Mitochondrial oxidative damage has been known to contribute to damage in cell toxicity induced by MTX and CYP. Over the past years, investigators have developed a series of mitochondria-targeted compounds for the amelioration of mitochondrial toxicity and oxidative stress. 17 Also, lysosomes as ubiquitous membrane-bound intracellular organelles with an acidic interior are place for degradation and recycling of macromolecules. In contrast to the rather simplified view of lysosomes as waste bags, they are involvement in cholesterol homeostasis, plasma membrane repair, and cell death. 18 Therefore, protection of lysosomes against cytotoxic agents like MTX and CYP can be a good approach for inhibition of their toxicity. Due to wider clinical use of MTX and CYP in treatment of diseases, the current study is aimed at analyzing cytotoxicity effects of these drugs on human blood lymphocytes and investigating their toxicity inhibition by antioxidants butylatedhydroxy toluene (BHT), mitochondrial (cyclosporine, A), and lysosomal (chloroquine) protective agents.
Materials and methods
Chemicals
Ficoll-paque PLUS was obtained from GE Healthcare Bio-Science Company (Freiburg, Gemany). RPMI1640 and fetal bovine serum (FBS) were taken from Gibco (Carlsbad, California USA). Trypan blue, 2′,7′-dichlorodihydrofluorescein diacetate (2′,7′-dichlorofuorescin diacetate; DCFH-DA), Rhodamine123, bovine serum albumin, N-(2-hydroxyethyl) piperazine-N′-(2-ethanesulfonic acid), acridine orange, MTX, and CYP were obtained from the Sigma Chemical Co (Sigma-Aldrich, St Louis, MO, USA).
Isolation and treatment of human lymphocytes
Ethical approval for this research study was approved by the ethics committee of Shahid Beheshti University of Medical Sciences. Volunteers were asked to fill out the informed consent form. Then, blood samples were obtained from 20 healthy volunteers in the age range of 18–30 years, with no any disease. Lymphocytes were isolated using Ficoll standard method. Diluted blood was layered on a 3-ml Ficoll–Paque, centrifuged for 20 min at 2500 r/min and lymphocytes layer was collected, suspended in erythrocyte lysis buffer (150 mM NH4Cl, 10 mM NaHCO3, 1 mM ethylenediaminetetraacetic acid (EDTA), pH 7.4), and incubated for 5 min at 37°C. Then, phosphate-buffered saline (PBS) was added immediately, and the cells were centrifuged at 1500 × g for 10 min at 20°C. The supernatant was eliminated, and the cells were washed twice with RPMI with
Cell viability assay
The cell viability of the isolated human lymphocyte was assessed from the intactness of the plasma membrane as determined by the MTT (3-(4,5-dimethylthiazol-2-yl)) assay. Lymphocytes were plated onto a 96-well plate (1 × 104 cells/ml) and incubated with different concentration of MTX and CYP for 6 h. After the incubation time, the cell viability was measured with MMT (5-diphenyltetrazolium bromide) assay. 19
Measurement of ROS
The rate of human lymphocyte ROS generation was evaluated using the probe DCFH-DA. In the presence of ROS, DCFH is oxidized to highly fluorescent dichlorofluorescein (DCF). Human lymphocytes were treated for 1, 2, and 3 h with different concentration of MTX and CYP. Also, protective effects of BHT, cyclosporine A, and chloroquine were tested against oxidative stress. After the incubation time, medium was replaced by 10 µmol DCFH-DA containing medium, after 15 min incubation, the fluorescence intensity was measured by fluorescence spectrophotometer (Shimadzu RF5000U, Shimadzu RF5000, Kyoto, Japan) at the excitation wavelength of 495 nm and the emission wavelength of 530 nm. 20
Measurement of MMP
The change in the mitochondrial membrane potential (MMP) in the human lymphocyte was measured using the cationic fluorescent dye Rhodamine123. Human lymphocytes were exposed for 1, 2, and 3 h with different concentration of MTX and CYP. Also, protective effects of BHT, cyclosporine A, and chloroquine were tested against MMP collapse. After the incubation time, medium was replaced by 1 µmol Rhodamine123 containing medium, after 15 min incubation, the medium was removed, and the fluorescence intensity was measured by fluorescence spectrophotometer (Shimadzu RF5000U) at the excitation wavelength of 470 nm and the emission wavelength of 540 nm. 21
Measurement of lysosomal membrane destabilization
Human lymphocyte lysosomal membrane integrity was assessed from the redistribution of the lipophilic dye acridine orange. The cells were exposed for 1, 2, and 3 h with different concentrations of MTX and CYP. Also, protective effects of BHT, cyclosporine A, and chloroquine were tested against lysosomal damages. After the incubation time, medium was replaced by 5 µM acridine orange containing medium. After 10 min incubation, the fluorescence intensity was measured by fluorescence spectrophotometer (Shimadzu RF5000U) at the excitation wavelength of 470 nm and the emission wavelength of 540 nm. 22
Measurement of lipid peroxidation
Lipid peroxidation was measured using the thiobarbituric acid assay and malondialdehyde (MDA) formation. Human lymphocytes were exposed for 1, 2, and 3 h with different of MTX and CYP. Also, protective effects of BHT, cyclosporine A, and chloroquine were tested against lipid peroxidation. After the incubation, cells were washed with PBS, and then lysed with PBS containing 2% triton. Then, 100 µl of cell lysate was mixed with 200 µl of thiobarbituric acid (TBA) reagent (containing 3.75% trichloroacetic acid (TCA) and 0.0925% TBA), and the mixture was incubated at 90°C for 60 min. After cooling, the mixture was centrifuged at 1000 × g for 10 min. Calorimetric absorption was measured at 530 nm. 23
Measurement of GSH and GSSG
GSH and GSSG levels in MTX/CYP-treated human lymphocytes were measured by Hissin and Hilf method. 24 After treatment of human lymphocytes with MTX and CYP, cells were lysed with 0.5 ml of TCA 10% and centrifuged at 11,000 × g for 2 min. For assessment of GSH, supernatant was diluted with phosphate-EDTA buffer and incubated with 100 µl of the o-phthalaldehyde (OPT) solution for 15 min at room temperature. For determination of GSSG, supernatant of cells was diluted with NaOH 0.1 N solution and before incubation with OPT, 200 µl of N-ethylmaleimide solution was incubated with supernatant for 30 min. The fluorescence intensity was measured by fluorescence spectrophotometer (Shimadzu RF5000U) at the excitation wavelength of 350 nm and the emission wavelength of 420 nm.
Statistical analysis
All data are presented as the mean ± SEM with three separate experiments. Data were analyzed by GraphPad Prism 5 (GraphPad Software, La Jolla, California, USA) using one- and two-way analysis of variance, followed by post hoc Tukey and Bonferroni test. The value of p < 0.05 was considered as statistically significant.
Results
Cell viability
The presented data at Figure 1(a) and (d) shows that BHT as an antioxidant, cyclosporine A as an MPT pore sealing, and chloroquine as a lysosomotropic agent prevent cytotoxicity induced by MTX and CYP. Also, cytotoxic effects of MTX and CYP on human lymphocytes are demonstrated in Figure 1(a) and (b). MTX and CYP caused dose-dependent cytotoxicity on human lymphocytes and significantly (p < 0.05) reduced cell viability in all used concentrations. These results confirm cytotoxic effect of MTX and CYP reported in published works. 15,25

Viability of human lymphocytes following treatment with MTX and CYP for 6 h (a) and (b). Cell viability determined by MTT assay after incubation of lymphocytes with different concentration of MTX and CYP. MTX and CYP decrease lymphocyte viability in a dose-dependent manner, and this decrease is significant at concentration higher than 0.4 ng/ml. (***p<0.001 vs. control). Protective effect of BHT, chloroquine, and cyclosporine A are tested against cytotoxicity-induced MTX and CYP (c) and (d). Presented data show that these agents significantly decreased cell death compared to treated groups with MTX and CYP. MTX: methotrexate; CYP: cyclophosphamide; BHT: butylatedhydroxy toluene.
ROS production
When the humane lymphocytes were simultaneously treated with MTX, CYP, BHT, cyclosporine A, and chloroquine, the mean fluorescence intensities were significantly decreased compared to treated groups with MTX and CYP (Figure 2). The effects of MTX and CYP on the generation of ROS in human lymphocytes are shown in Figure 2(a) and (b). Both drugs have induced dose- and time-dependent ROS generation in human lymphocytes.
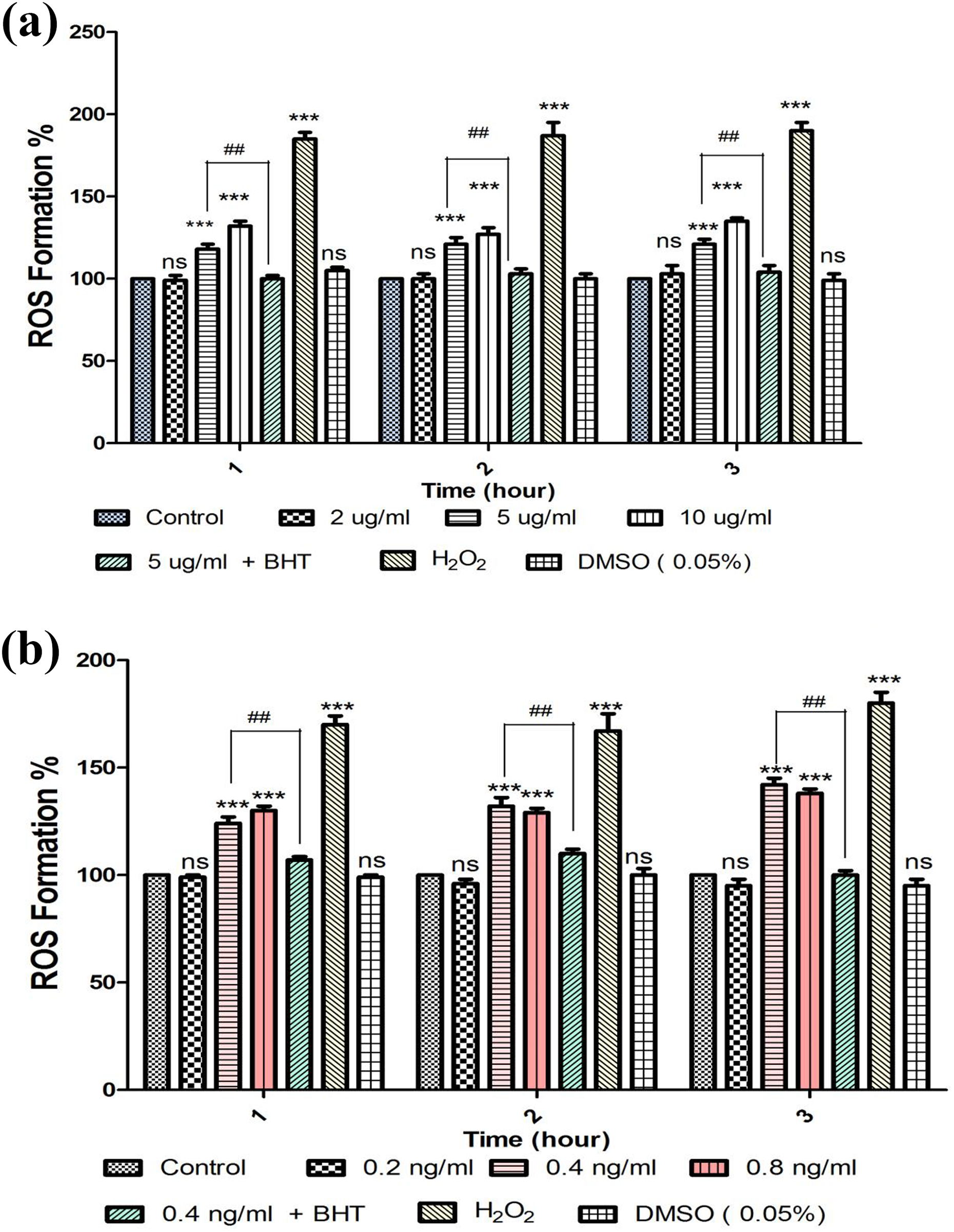
ROS generation in human lymphocyte after incubation with MTX and CYP for different concentrations and time (1, 2, and 3 h) intervals. (a) MTX and (b) CYP have induced dose- and time-dependent ROS production. BHT, an antioxidant, inhibited MTX- and CYP-induced generation of ROS in human lymphocytes. (***p < 0.001 vs. control, ###p < 0.001 vs. treated groups with drugs). ROS: reactive oxygen species; MTX: methotrexate; CYP: cyclophosphamide; BHT: butylatedhydroxy toluene.
MMP collapse
As shown in Figure 3, collapse of mitochondrial membrane potential was inhibited after treatment of lymphocytes with MTX and CYP by BHT, cyclosporine A, and chloroquine at toxic doses for each drug. Also, the effects of MTX and CYP on the MMP of human lymphocytes are presented in Figure 3(a) and (b). MTX- and CYP-induced statistically MMP collapse in dose- and time-dependent manner.

Collapse of MMP in human lymphocytes following incubation with (a) MTX and (b) CYP for 1, 2, and 3 h. Collapse in MMP started 1 h after the treatment of human lymphocytes with MTX and CYP at three concentrations. MTX- and CYP-induced mitochondrial membrane potential collapse was time and concentration dependent (p < 0.001). BHT, cyclosporine A, and chloroquine prevented MTX- and CYP-induced collapse in MMP. (***p < 0.001 vs. control, ###p < 0.001 vs. treated groups with drugs). MMP: mitochondrial membrane potential; MTX: methotrexate; CYP: cyclophosphamide; BHT: butylatedhydroxy toluene.
Lysosomal membrane destabilization
Lysosomal membrane destabilization was inhibited after treatment of lymphocytes with MTX and CYP by BHT, cyclosporine A, and chloroquine at toxic doses for each drug (Figure 4). Also, the effect of MTX and CYP is shown in Figure 4(a) and (b). MTX- and CYP-induced statistically lysosomal damages in dose- and time-dependent manner.

Lysosomal membrane destabilization in human lymphocyte after incubation with (a) MTX and (b) CYP. After 1, 2, and 3 h treatment, MTX and CYP caused significant (p < 0.001) lysosomal membrane leakage. Chloroquine (100 µM) prevented MTX- and CYP-induced lysosomal membrane leakage. BHT, cyclosporine A, and chloroquine prevented MTX- and CYP-induced lysosomal damages. (***p < 0.001 vs. control, ###p < 0.001 vs. treated groups with drugs). MTX: methotrexate; CYP: cyclophosphamide; BHT: butylatedhydroxy toluene.
Lipid peroxidation
Lipid peroxidation as an indicator of oxidative damage to the lipids was measured in human lymphocyte using thiobarbituric acid reactive substances assay as a by-product of lipid peroxidation. Pretreatment of human lymphocytes with BHT, cyclosporine A, and chloroquine at toxic doses for each drug significantly decreased the lipid peroxidation level after 3 h incubation time (Figure 5). We also showed that the amount of intracellular MDA as the result of lipid peroxidation significantly increased when cells incubated with MTX and CYP at toxic dose during 1–3 h (Figure 5(a) and (b)).

Induction of lipid peroxidation in human lymphocyte and after incubation with (a) MTX and (b) CYP. Lipid peroxidation significantly increased when cells were incubated with MTX and CYP at highest time (3 h). BHT, cyclosporine A, and chloroquine prevented MTX- and CYP-induced lipid peroxidation. (***p < 0.001 vs. control, ###p < 0.001 vs. treated groups with drugs). MTX: methotrexate; CYP: cyclophosphamide; BHT: butylatedhydroxy toluene.
GSH and GSSG content
Human lymphocytes were treated with MTX and CYP and 1 h after treatment collapse in intracellular GSH and increase in extracellular GSSG levels was observed. Pretreatment of human lymphocytes with BHT, cyclosporine A, and chloroquine inhibited both collapse of intracellular GSH and increase in extracellular GSSG. Intracellular GSH levels significantly (p < 0.001) decreased at 1, 2, and 3 h following treatment of human lymphocytes with all MTX and CYP concentrations as shown in Figure 6(a) and (c). The effects of MTX and CYP on extracellular GSSG content are shown in Figure 6(b) and (d). This finding indicates a significant (p < 0.001) increase in lymphocytes extracellular GSSG level in concentration-dependent manner.

Effect of (a, b) MTX and (c, d) CYP on GSH and GSSG content As demonstrated, significant (p < 0.001) intracellular GSH decrease and raises in lymphocytes extracellular GSSG was found after treatment with MTX and CYP. BHT, cyclosporine A, and chloroquine prevented depletion of GSH (***p < 0.001 vs. control, ###p < 0.001 vs. treated groups with drugs). MTX: methotrexate; CYP: cyclophosphamide; GSH: glutathione; GSSG: oxidized glutathione; BHT: butylatedhydroxy toluene.
Discussion
It is obvious that immune system protects the body from bacteria, viruses, and tumor cells. 26 Lymphocytes are an important part of the immune system and play an outsized role in defending the body against diseases. Lymphocytes’ role is antibodies production against bacteria, viruses, fungi, parasites, and toxic chemicals, killing the invader organism and infected cells and release cytokines to warn other cells of the danger. There are two main types of lymphocytes in human blood: T cells and B cells. 27 T lymphocytes continually scan and monitor the cells for infection and kill cancer cells, which is a promising approach to cancer treatment (known as CAR-T therapy). 28 However, B cell lymphocytes do not attack and kill cells, viruses, or bacteria. Instead, they manufacture antibodies. 27 Therefore, these cells have an important role in controlling infectious conditions and removing cancerous cells in the body. Our results in this study showed that MTX and CYP as antineoplastic drugs have toxicity effect on human lymphocytes (Figure 1(a) and (b)).
The immune cell functions especially lymphocytes are linked to ROS formation. Hence, the oxidant/antioxidant balance is very important in the immune cell activity. The antioxidant contents in lymphocytes play a fundamental role in protecting these cells against oxidative stress and therefore preserving their adequate function. 29 When a disturbance in the oxidant/antioxidant balance occurs due to chemical exposure leads to ROS formation, lipid peroxidation, and depletion of intracellular antioxidant like glutathione that finally results in dysfunction and toxicity in lymphocytes. 30 Our results here indicated that MTX and CYP caused ROS formation, lipid peroxidation, and depletion of glutathione in human lymphocytes which protected by BHT as an antioxidant agent. Several studies showed the similar effects of other antioxidant against oxidative stress induced by MTX and CYP.
Studies demonstrated that, in addition to the role of mitochondria in cell life as powerhouse, they have the main role in cell death in response to oxidative stress conditions. Accumulation of ROS along with Ca2+ overload causes a situation known as mitochondrial permeability transition (mPT) that is associated with mPT pore opening in the inner membrane of mitochondria (IMM). 31 Agents such as ions (Pi, H+, Ca2+, and Mg2+), ROS, adenine nucleotides regulate opening of the mPT. 32 Opening this pore is accompanied with the loss of the MMP and proton gradient across the IMM. Adenosine triphosphate (ATP) hydrolysis in an attempt to maintain the MMP is induced by F0F1-ATPase at low electrochemical potential, and the function of adenine nucleotide translocase (ANT) in transporting ATP becomes reverse. 33 mPT can occur at low and high conductance leading to reversible of irreversible consequences. In the reversible mPT, ions and solutes with the molecular mass <300 Da pass through the pore and do not induce notable matrix swelling. But in irreversible mPT, solutes, water, and ions with the molecular mass up to approximately 1.5 kDa pass through the pore, thus enhancing colloid–osmotic pressure in the matrix. This action leads to rupture of the outer membrane of the mitochondria resulting in cell death via apoptosis and/or necrosis depending on the ATP contents in cells. 31 Obtained results in this study showed that MTX and CYP induce mPT in human lymphocytes that inhibited by cyclosporine A as an mPT pore sealing agent and inhibitor of ANT.
Lysosomes are the acidic vesicles for degradation and recycling of defective cellular material and provide an optimal physicochemical environment for enzymatic activities, which need to be controlled. 34 Currently, documented functions like immune response, cell death, plasma membrane repair, energy and nutrient sensing, and secretion reveal the importance of lysosomes in controlling fine decisions in the life of a cell. 35 Environmental tensions such as oxidative stress and ROS formation can damage lysosomal membranes and lead to membrane permeabilization. Normal function of lysosomes depends on intraluminal acidic pH and requires stable membrane-dependent proton gradients. 36 Chloroquine as a lysosomotropic agent prevents endosomal acidification. This drug accumulates inside the acidic parts of the cell, including lysosomes and endosomes. This accumulation leads to inhibition of lysosomal enzymes that require an acidic pH and prevents fusion of endosomes and lysosomes. Moreover, chloroquine inhibits autophagy as it raises the lysosomal pH, which leads to inhibition of both fusion of autophagosome with lysosome and lysosomal protein degradation. 37 Our results showed that MTX and CYP induce lysosomal damages in human lymphocytes and that their damage is prevented by chloroquine.
Conclusion
The obtained data from this study showed that both anticancer drugs with different mechanisms in cancer therapy lead to oxidative stress and mitochondrial and lysosomal damage in human lymphocyte. Our results also demonstrated that mitochondrial and lysosomal protective agents have a promising effect for inhibition of cytotoxicity of MTX and CYP as widely used drugs in treatment of cancers and other diseases. Moreover, presented data indicated that using of mitochondrial and lysosomal protective agents can be useful for the viability of lymphocytes and their good functions in immune system.
Footnotes
Acknowledgements
The data provided in this article was extracted from the Pharm D thesis of Dr Rezvan Pirhadi. The thesis was conducted under supervision of Prof. Jalal Pourahmad at Department of Toxicology and Pharmacology, Faculty of Pharmacy, Shahid Beheshti University of Medical Sciences, Tehran, Iran. This study was supported by Shahid Beheshti University of Medical Sciences, Deputy of Research with ethical code IR.SBMU.PHNM.1397.143.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
