Abstract
Welders’ lung disease refers to mixed exposure to different kinds of metals and chemicals from welding fumes, which affect all parts of the respiratory tract including airways and parenchyma together. This study aimed to investigate the oxidative status in patients with welders’ lung (PWL) by means of thiol-disulfide homeostasis and ischemia-modified albumin (IMA) levels. The male welder workers diagnosed with welders’ lung disease and healthy individuals were recruited in the study. Plasma levels of disulfide, disulfide/native thiol ratio, disulfide/total thiol ratio, IMA, and catalase (CAT) were determined. Pulmonary function test parameters of both groups were compared. The thiol-disulfide homeostasis parameters of PWL and control group were as follows: disulfide (20.5 ± 6.3 vs. 16.2 ± 3.9 μmol L−1, p < 0.001), disulfide/native thiol (4.36 (1.59) vs. 4.0 (1.64), p = 0.024), and disulfide/total thiol (4.01 (1.34) vs. 3.71 (1.41), p = 0.024). IMA levels in PWL were significantly higher than the control group (1.37 (0.27) mg dL−1 vs. 0.49 (0.61) mg dL−1, p < 0.001), whereas CAT activities were significantly higher in the control group (106.6 (54.5) kU L−1 vs. 78.3 (67.8) kU L−1, p = 0.003). The findings of the present study revealed that oxidative stress plays a key role in the pathogenesis of welders’ lung disease. Plasma thiol-disulfide homeostasis and IMA levels might be indicators of oxidative stress in PWL.
Introduction
Welding is defined as any process of bonding pieces of metal at joint faces that have been made soft or liquid under high temperature or by applying high pressure. 1 More than one million workers are estimated to execute some type of welding as part of their work worldwide. 2 Although the incidence of lung disease in welders is not known, epidemiology studies have shown that one of the most common occupations associated with developing occupational lung disease is welding and a large number of welders experience some type of respiratory illness. 3 A number of hazardous agents such as fumes, gases, vapors, heat, noise, and ultraviolet and infrared radiation are brought off via welding processes. The chemical composition of welding fume includes fluoride, ozone, nitrogen oxides, and carbon monoxide particles released by the heating of several metals, such as nickel, chrome, cadmium, iron, copper, magnesium, and zinc. 4 The composition and concentration of the airborne particles determine the harmful effects of welding fume on human respiratory system. A variety of respiratory problems are shown to be associated with welding fume ranging from wheezing, dryness of the throat, coughing, tightness in the chest, difficulty in breathing, deterioration of pulmonary function to chronic bronchitis, hypersensitivity pneumonitis, pneumoconiosis, metal fume fever, and lung tumors among welders. 5 –7 Welders’ lung disease refers to an intense mixed exposure to different kinds of metals and chemicals from welding fumes, which affect all parts of the respiratory tract including airways and parenchyma together. 8
Oxidative stress, an imbalance between prooxidants and antioxidants in favor of former, may play a critical role in the pathogenesis of respiratory diseases as the large surface area for gas exchange makes the respiratory system particularly susceptible to oxidative stress-mediated injury. 9 Thiols are composed of sulfhydryl (–SH) residues and have a pivotal role in coordinating the antioxidant defense mechanism. In the case of oxidative stress, the oxidant is neutralized by thiol to a less toxic by-product, where thiol itself gets oxidized to a disulfide through reversible thiol-disulfide exchange reaction. It is possible to provide valuable information on normal or abnormal biochemical processes via determination of thiol-disulfide homeostasis. Ischemia-modified albumin (IMA), an oxidant marker, results from differentiation of albumin in cases of increased oxidative stress or ischemia. The effects of welding fume exposure on correlates of oxidative stress parameters and antioxidant capacities in welders have previously been investigated, however, as far as we are aware no studies have reported any data related to dynamic thiol-disulfide homeostasis and IMA levels in welders’ lung disease. 10 –16 In this study, we aimed to investigate the oxidative status in patients with welders’ lung (PWL) disease by means of IMA levels and thiol-disulfide homeostasis using a newly developed method. 17
Methods
This study was conducted in Occupational and Environmental Diseases Hospital, Ankara, Turkey, between June 2015 and June 2018. The male welder workers, who admitted to our clinic for periodic medical examination and have been diagnosed with welders’ lung disease, were included in the study. Healthy individuals who were admitted to the hospital for routine health check, who had no history of occupational welder fume exposure, and who did not have respiratory complaints were selected as the control group.
The exclusion criteria for study group and control group were as follows: existence of a recent acute disease or known chronic diseases, such as chronic obstructive pulmonary disease, asthma, bronchitis, bronchiectasis, pneumonia, tuberculosis, diabetes mellitus, cerebrovascular and cardiovascular disease, infectious or rheumatic inflammatory disease, chronic liver or kidney disease, malignancy diagnosis, taking any lipid lowering drugs, vitamin supplements, or antioxidants. This study was conducted according to the guidelines of the Declaration of Helsinki. Ethical clearance was obtained from the local ethics committee of Dr Abdurrahman Yurtaslan Oncology Educational and Research Hospital, Ankara, Turkey (file number: 2019-2/205).
Posteroanterior chest radiographs of all cases were taken in the radiology clinic of our hospital. A short exposure time with high voltage technique was used (Trophy UFXRAY, 500 mA, TM). Chest radiographs of workers were interpreted by three experienced International Labor Office (ILO) readers according to the ILO-2000 classification of pneumoconiosis. Two of the readers were specialists in pulmonary medicine and the other was specialist in family medicine with interest in occupational diseases. Radiographic abnormalities of the welders’ lung disease were classified into four profusion categories (0, 1, 2, and 3), regarding the concentration of small opacities in affected zones of the lung. Also 12 subcategories were determined as 0 (0/–, 0/0, 0/1); 1 (1/0, 1/1, 1/2); 2 (2/1, 2/2, 2/3); or 3 (3/2, 3/3, 3/+) according to the ILO classification. 18
A standard spirometry measurement was done with a dry-seal-spirometry (Zan 100, nSpire Health Inc., Oberthulba, Germany). Pulmonary function tests were interpreted in accordance with the American Thoracic Society standards.
Peripheral blood samples (total, 4 mL) were collected from the brachial vein into heparinized tubes and were stored at 2–4°C for the measurement of thiol-disulfide hemostasis parameters, IMA level, catalase (CAT) activity, and other parameters, that is, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and complete blood count (CBC). The blood was centrifuged at 1500 × g for 10 min to obtain the plasma. The separated plasma was then stored at −80°C until analysis. The novel spectrophotometric method described by Erel and Neselioglu was used for the measurement of plasma thiol-disulfide hemostasis parameters. 17 Briefly, reducible disulfide bonds were reduced to form free functional thiol groups. Formaldehyde was used to remove unused and consumed sodium borohydride, after the reaction with 5,5′-dithiobis-(2-nitrobenzoic acid), reduced and native thiol groups were all determined. The amount of dynamic disulfide was determined by using the formula: [dynamic disulfide] = ([total thiol] – [native thiol])/2. Disulfide/native thiol, disulfide/total thiol, and native thiol/total thiol ratios were calculated after dynamic disulfide, native thiol, and total thiol levels determined. The mean percent recovery was 98–101%, precision (coefficient of variation (CV%)) was 4% for high levels of CAT, and activity was determined using a method defined by Goth. 19 Briefly, the sample (0.2 mL) was incubated in 1.0 mL substrate (65 mmol/H2O2) in 60 mmol L−1 sodium–potassium phosphate buffer (pH 7.4) at 37°C for 60 s. The enzymatic reaction was stopped with 1.0 mL of 32.4 mm ammonium molybdate, and the yellow complex of molybdate and H2O2 was measured at 405 nm against a blank. One unit of CAT decomposes 1 μmol of H2O2 min–1 under these conditions. The CAT activity unit is defined as kU L−1. IMA levels were analyzed using the rapid and colorimetric method defined by Bar-Or et al. 20 Briefly, 200 μL of patient serum was placed into glass tubes, and 50 μL of 0.1% CoCl2 6H2O (Sigma-Aldrich, St Louis, Missouri, USA) was added to the tubes. This mixture was incubated for 10 min to ensure sufficient cobalt–albumin binding. Afterward, 50 μL of 1.5 mg mL−1 dithiothreitol (DTT) was added as a coloring agent, and the reaction was quenched 2 min later by adding 1.0 mL of 0.9% NaCl. Specimen absorbencies were analyzed at 470 nm by a spectrophotometer. The color of the DTT containing specimens was compared with that of the colorimetric control tubes. The results are expressed as mg dL−1. CRP levels were studied by latex-enhanced turbidimetric immunoassay method and ESR analysis was performed by Alifax Test 1 (Padova, Italy). CBC and platelet count were analyzed on using a Beckman Coulter LH 780 Hematology Analyzer (Florida, USA).
We made the statistical analysis of data by SPSS (version 21.0, SPSS Inc., Chicago, Illinois, USA) package program. Coherence to normal distribution analysis was made using Kolmogorov–Smirnov test. Values were presented as mean ± standard deviation or median (interquartile range). The presence of a statistically significant difference between the groups in terms of continuous variables was examined with analysis of variance for parametric and Kruskal–Wallis test for nonparametric variables. For the significant (p < 0.05) analyses, Student’s t-test for parametric and Mann–Whitney U-test for nonparametric variables were performed; Spearman’s correlation analysis was also performed.
Results
The study included 113 subjects: 52 PWL and 61 healthy volunteers. The mean age of PWL and control group was 43.3 ± 8.5 and 32.7 ± 9.6 (p < 0.001), respectively. While 65.4% of PWL was smoking, this rate was 57.4% in the control group (p = 0.384; Table 1). The most common symptoms in the PWL group were shortness of breath (42.3%) and cough (25%; Table 2).
Basic characteristics of PWL and control group.a

PWL: patients with welders’ lung; BMI: body mass index; SD: standard deviation; IQR: interquartile range.
a Parameters were expressed as mean ± SD and median (IQR).
Symptoms and evaluation of chest X-rays according to the ILO classification in PWL.
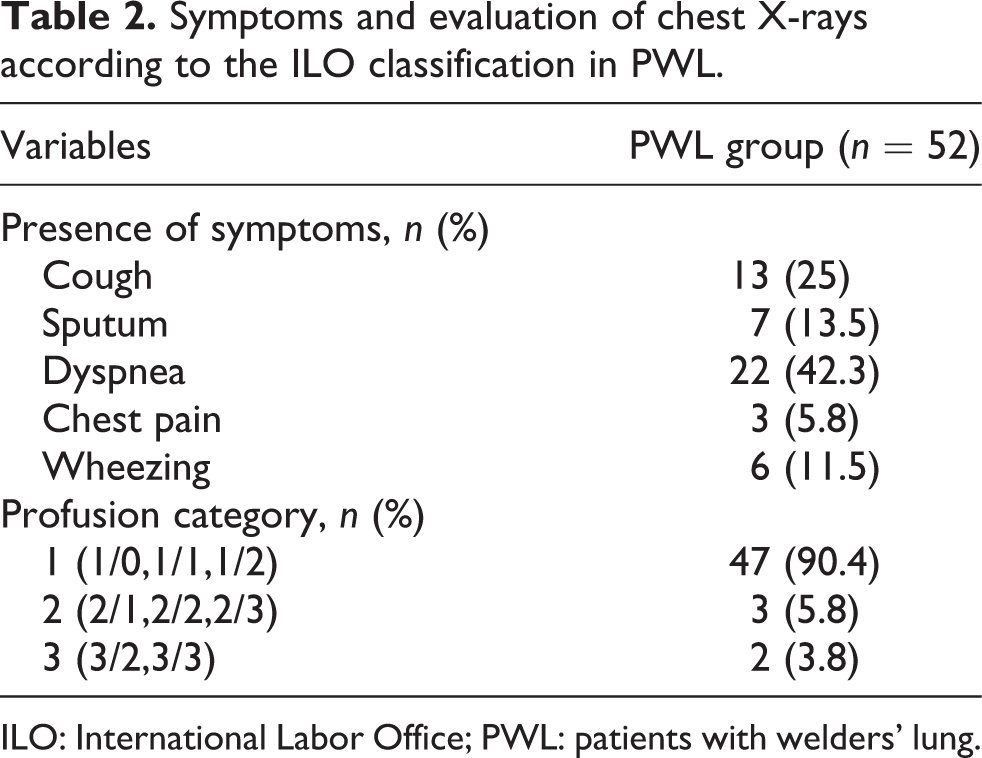
ILO: International Labor Office; PWL: patients with welders’ lung.
The thiol-disulfide homeostasis parameters of the PWL and the control group were as follows: disulfide (20.5 ± 6.3 vs. 16.2 ± 3.9 μmol L−1, p < 0.001), disulfide/native thiol (4.36 (1.59) vs. 4.0 (1.64), p = 0.024), and disulfide/total thiol (4.01 (1.34) vs. 3.71 (1.41), p = 0.024). IMA levels in PWL were significantly higher compared to the control group (1.37 (0.27) mg dL−1 vs. 0.49 (0.61) mg dL−1), whereas CAT activities were significantly higher in the control group (106.6 (54.5) kU L−1 vs. 78.3 (67.8) kU L−1, p = 0.003; Figure 1). Based on smoking habit, IMA levels (1.01 (1.0) mg dL−1 vs. 0.93 (0.76) mg dL−1, p = 0.383) and CAT activities (92.3 (63.3) kU L−1 vs. 97.3 (70.6) kU L−1, p = 0.383) did not differ significantly among smokers and nonsmokers. The pulmonary function test parameters—forced expiratory volume (FEV1) act (3.47 ± 0.6 vs. 4.12 ± 0.58), forced vital capacity (FVCact) (4.23 ± 0.77 vs. 4.75 ± 0.60 mL), and FEV1/FVC 83 (8.75) vs. 87 (7.5)—were significantly lower in PWL compared to the control group (p < 0.001; Table 3). Spearman’s correlation analysis showed an inverse correlation between albumin, CAT, and IMA and disulfide while CRP correlated positively with IMA and disulfide. FEV1 act, FEV1/FVC, peak expiratory flow (PEFact), and maximal expiratory flow (MEF25–75) showed inverse correlation with IMA and disulfide (Table 4).

The levels of (a) CAT, (b) IMA, (c) disulfide, and (d) disulfide/native thiol and disulfide/total thiol in both control and welders’ lung groups. IMA: ischemia-modified albumin; CAT: catalase; PWL: patients with welders’ lung.
Comparison of laboratory findings and pulmonary function test of PWL and control group.a

PWL: patients with welders’ lung; IMA: ischemia-modified albumin; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; SD: standard deviation; IQR: interquartile range.
a Parameters were expressed as mean ± SD and median (IQR).
Correlations between serum thiol-disulfide homeostasis parameters and IMA levels with other parameters in study population (n = 113).
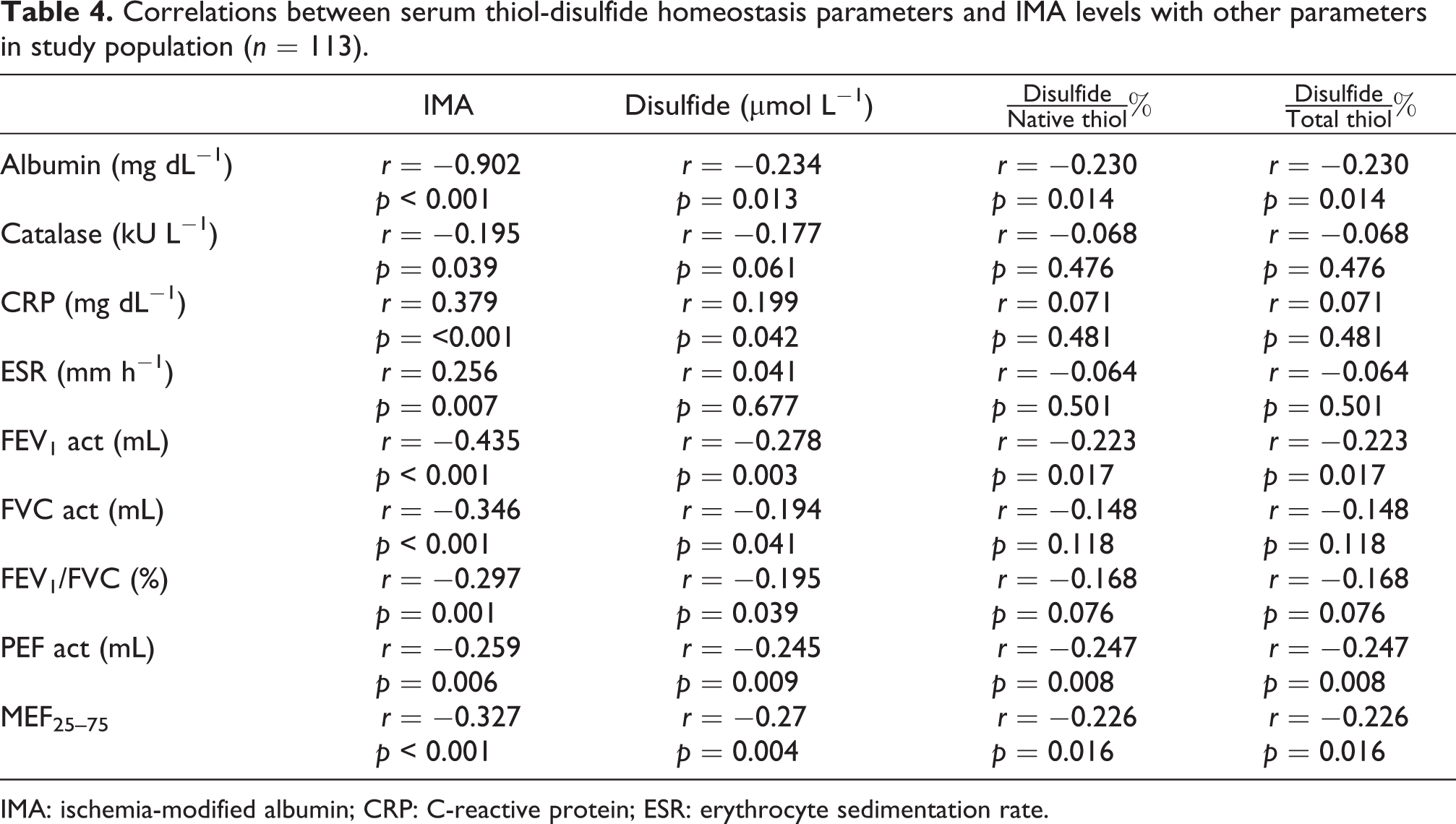
IMA: ischemia-modified albumin; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate.
Discussion
The current study found that thiol-disulfide homeostasis, a reliable indicator of oxidative stress, shifted in favor of oxidant, disulfide in PWL. The significant increase in IMA and decrease in antioxidant enzyme CAT in PWL also suggested welding fume-induced oxidative stress. Another important finding was the significant decline in all pulmonary function tests in PWL.
The imbalance between systemic expression of ROS and the detoxifying mechanisms of biological systems results in oxidative stress. As the cellular redox balance is vital for cell survival, dynamic thiol-disulfide homeostasis is one of the main indicators of this balance. Oxidative stress plays an important role also in the pathogenesis and progression of pulmonary diseases, that is chronic obstructive pulmonary disease, asthma, and community-acquired pneumonia, which are associated with lower total thiol levels in comparison with healthy individuals. 21 Previous studies have already reported much data on various oxidative stress markers in welder fume-exposed welders. 10 –16 In a study investigating oxidative markers in welders, Fidan et al. reported that oxidant–antioxidant status was negatively affected as serum SH (sulfhydryl) and glutathione, a known antioxidant and free-radical scavenger, levels were lower and oxidative damage markers were higher in welders than control group. 22 Han et al. investigated the effects of welding fume in welders and reported significant increases in oxidative stress markers, such as glutathione peroxidase, isoprostane, compared with control subjects. 23 Likewise, Li et al. reported that erythrocyte superoxide dismutase (SOD) activity and serum malondialdehyde levels of welders were 25% less and 78% higher, respectively, compared to nonexposed subjects. 11 Our study is the first study to determine thiol-disulfide homeostasis in welders. We found that the disulfide level, disulfide/native thiol, and disulfide/total thiol ratios in PWL were significantly higher. This result may be explained by the fact that thiol oxidation is higher in PWL than the control group, and, therefore, oxidative stress is more significant in the exposed group.
Human albumin converts to IMA when the N terminus of albumin is altered because of oxidative stress or ischemia. Previous studies have shown that serum IMA levels were significantly higher in patients with pulmonary embolism, acute coronary syndrome, stroke and mesenteric ischemia, cerebral hemorrhage, and peripheral vascular disease. 24 To our knowledge, no studies evaluated the relationship between welding fume and IMA levels. In our study, serum IMA levels of PWL were significantly higher than controls indicating welding fume-induced oxidative stress. The observed IMA alteration could be attributed to oxidative damage denoted by the oxidation of macromolecule, that is, albumin, due to increased oxidative stress, which was also confirmed by thiol oxidation. Albumin forms the major component of plasma thiol pool; thus, it represents the predominant antioxidant in plasma. 25 As the large proportion of serum antioxidant activities can be attributed to albumin, it was not surprising to observe the significant decline in albumin levels in PWL in our study. Furthermore, the inverse correlation between albumin and IMA and disulfide also reflects the antioxidant response to welding fume insult in PWL. We also observed the CAT activity was significantly lower in PWL than controls, which suggest the decrease of the antioxidant defense system as a result of chronic welder fume exposure. Likewise, Stepniewski et al. have revealed that exposure to welding fumes had significant negative effects on the antioxidant defense system, especially on SOD and CAT enzyme activities. 26 These findings are also supported by the correlations observed between CAT activity and IMA and disulfide levels in welders in our study.
The median smoking (package/years) in PWL was significantly higher when compared to controls. We observed significant correlations between smoking (package/years) and IMA and disulfide levels. Some studies have also indicated that smoking can trigger the oxidative stress in various tissues. 15 Thus, it may be concluded that not only smoking but also exposure to welding fumes results in a more serious oxidative stress.
The findings of this study suggest that welders’ lung may be associated with significant decline in lung function, hence, all pulmonary function parameters including FEV1, FVC, and FEV1/FVC were lower than that of controls. This finding is consistent with that of Daniela Haluza et al., who reported significant decreases in FVC, FEV1, and MEF50 in welders. Similarly, Bogadi-Sare 27 and Wang et al. 28 reported significant reduction of FEV1/FVC ratio in welders. Chinn et al. reported that welding fume exposure significantly increased the rate of lung function decline. 29 However, Ozdemir et al. observed no differences between nonsmoker welders and controls by means of pulmonary function tests but smoker welders had lower FVC, FEV, and PEF values than smoker controls who smoked. 1 Likewise, in a study of 41 welders, it was reported that spirometry showed no impairment in lung function in both the welders and the nonexposed group. 7 We found no significant difference in pulmonary function between smokers and nonsmokers in PWL. Pulmonary function tests including FEV1, FVC, FEV1/FVC, PEF, and MEF25–75 showed significant negative correlations with oxidative stress indicators, that is, IMA and disulfide levels. These correlations may suggest that one of the main molecular mechanisms underlying the decrement of pulmonary functions could be welding fume-induced oxidative stress.
It is possible to protect the patient from additional complications that may occur due to oxidative stress by detecting thiol-disulfide homeostasis disorder PWL and by considering the antioxidant replacement therapy strategies by removing the patient from the exposure environment. It can be predicted that the nutritional supplements and medicinal products (such as acetylcysteine) containing sulfhydryl group may be useful for the recorrection of thiol-disulfide homeostasis disorder, which is observed in PWL and is an indicator of oxidative stress. However, randomized controlled studies are needed on this subject.
The most important limitation of this study was that welding-fume concentrations and its correlation with thiol-disulfide homeostasis parameters and other oxidative stress markers could not be determined as air samples of workplace and surrounding the breathing zones of welders could not be monitored. Since there is no cutoff value defined for the determination of thiol-disulfide homeostasis disorder, a regression analysis could not be done to determine independent factors (i.e. age, smoking, etc.) associated with the studied thiol-disulfide parameters.
We have evaluated the thiol-disulfide homeostasis in PWL lung by using a novel, fast, easy, relatively cheap, fully automated, and optionally manual spectrophotometric assay. The findings of the present study revealed that oxidative stress plays a key role in welders’ lung diseases. Our results revealed that plasma thiol-disulfide homeostasis and IMA levels might be indicators of oxidative stress in PWL. Further studies need to be carried out to understand the mechanisms underlying welders’ lung diseases.
Footnotes
Acknowledgement
We would like to thank our hospital staff for their contribution to the collection of patient data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
