Abstract
We aimed to explore the possible neurotoxicity and infertility mechanisms of prolonged metronidazole (MTZ) use and the effects of antioxidant grapefruit (GP) co-therapy on MTZ-induced complications. Sixty male albino Wistar rats were divided into four groups (n = 15 each). Group I (control group) received 1% dimethyl sulfoxide (27 ml/ kg/day), group II (MTZ group) received MTZ (400 mg/kg/day), group III (MTZ + GP) received MTZ (400 mg/kg/ day) plus GP juice (27 ml/kg/ day) and group IV (GP group) received GP juice (27 ml/kg) for 60 days. Semen analyses were performed. Free testosterone, gonadotrophin (follicle-stimulating hormone (FSH) and luteinizing hormone) and thiamine levels were measured. Samples of cerebellar, testicular and epididymal tissues were used for both colorimetric assays of oxidative stress markers and histopathological examinations. Significant decreases in the sperm count, percent total sperm motility, serum thiamine levels, free testosterone levels and FSH levels were observed in the MTZ and MTZ + GP groups (p < 0.05 for all parameters). Significantly higher oxidative stress levels (p < 0.05) were observed in the cerebellar and testicular tissue homogenates of these groups than in those of the control group, and associated disruptions in the cerebellar, testicular and epididymal structures were apparent compared to those of the control group. Although the GP group showed a significantly higher sperm count and significantly lower oxidative stress than the control group (p < 0.05), with histological similarity to the control group, the GP group exhibited significantly higher prolactin levels and lower free testosterone and FSH levels than the control group (p < 0.05). Oxidative stress and decreased thiamine levels could explain the MTZ-induced neurotoxicity and infertility side effects that aggravated by GP co-administration.
Introduction
Metronidazole (MTZ) is a commonly used antimicrobial agent in the management of many anaerobic and protozoal infections. MTZ is a relatively safe drug; however, its prolonged use can be hazardous, which is evidenced by several reports that describe neurotoxicity and infertility as side effects of MTZ that limit its chronic use. 1,2 Few publications can be found in the literature explaining the mechanisms of such side effects.
Previously, prolonged MTZ therapy was shown to induce reversible, unwanted effects on male fertility, reproductive hormone levels and sperm motility and/or the sperm count. 2 –4 This MTZ-induced infertility has been shown to be prevented by supplementation with natural compounds that are known to have antioxidant activity. 3,4
MTZ-associated neurotoxicity has been reported in both humans and veterinary species, including rats. 5,6 The mechanism of MTZ-induced neurotoxicity is still unclear, but several hypotheses have been presented: one suggests that MTZ binds to RNA with subsequent inhibition of protein synthesis in neurons 7 ; another proposes that MTZ forms toxic reactive oxygen species through interactions with neurotransmitters 8 ; and yet another suggests that MTZ antagonizes the effect of thiamine. 9
Thiamine (vitamin B1) is an important coenzyme for α-ketoacids and transketolase, which are involved in carbohydrate metabolism, and a dietary deficiency of thiamine results in the ü development of Wernicke - Korsakoff syndrome and beriberi syndrome 10 The precursors of both MTZ and thiamine show structural similarity, which might indicate that MTZ is metabolized into a thiamine analogue that counteracts the effect of thiamine. 9 Although thiamine is involved in oxidative metabolism during spermatogenesis, its role in male infertility has not been clarified. 10
Grapefruit (GP) is a citrus fruit with the botanical name Citrus paradisi. GP contains bioactive compounds that are thought to possess antioxidative and anti-inflammatory activities. 11 On the other hand, GP components may have pharmacokinetic interactions that are partially attributed to the furanocoumarin content of GP, resulting in a subsequent increase in the bioavailability of certain drugs. 12
MTZ is considered a safe and widely used human and veterinarian agent, so it is essential to explore its potential biological hazards, especially its histopathological effects on organs. 13 In the last decade, several cases with MTZ encephalopathy have been reported, which has subsequently increased awareness among physicians; however, this issue still needs further clarification and study. 1 Therefore, the aim of the present study was to assess the neurotoxic and reproductive adverse effects of MTZ and to report possible mechanisms that could explain these adverse effects, using experimental male albino rats, on the basis of both biochemical alterations (including assessments of oxidative stress, thiamine status, reproductive hormones and semen analyses) and histopathological changes. Additionally, this study aimed to evaluate the effect of cotreatment with GP, an antioxidant, on MTZ-induced neurological and infertility side effects.
Methods
Drugs and chemicals
MTZ was purchased from Sigma-Aldrich Chemical Co. (UK) as analytical grade with the highest purity. The CAS number is 443-48-1.
GP juice preparation: The GP used in the current study was purchased from Kheir-Zaman Markets, Qena, Egypt. The juice was extracted with a domestic squeezer and filtered using a sieve with a mesh diameter of 1 mm. 14
Experimental design
This study included 60 male albino Wistar rats that were obtained from the Animal House of the Department of Zoology, Faculty of Science, South Valley University, Qena, Egypt. Their body weights ranged from 170 to 200 g. The rats were housed in wire-mesh cages under a controlled environment (23 ± 2°C, 55% relative humidity and a 12-h light/dark cycle) and treated according to the guidelines of the Animal House of South Valley University, Qena, where standard commercial pellets, which were used as food, and water were provided ad libitum. Other conditions pertaining to the health of the animals were maintained during the entire course of the study. All experimental protocols were performed in accordance with the local institutional guidelines and approved by the Animal Ethical Committee, South Valley University, Qena, Egypt.
The included rats were divided randomly into four groups (n = 15 each). All treatments were administered daily via the oral route using intragastric intubation. All chemicals were dissolved in 1% dimethyl sulfoxide (DMSO), and rats were treated daily for 60 days as follows:
Collection of blood and tissue samples
Twenty-four hours after treatment with the last dose, rats from various groups were euthanized by cervical decapitation. At the time of euthanization, blood samples were withdrawn from the retro-orbital veins on plain tubes and centrifuged at 4000 r/min for 10 min. The clarified serum was divided into aliquots in1-ml cryotubes and stored at −80°C until subsequent biochemical analyses. Then, the reproductive organs (testicles and epididymis) and the cerebellum were excised from each rat, thoroughly washed using chilled saline (0.9% NaCl) and blotted dry. One epididymis was used for semen analysis. One testicle, the other epididymis and part of the cerebellum from each rat were transferred rapidly into 10% buffered formalin for later histopathological processing. The other testicle and the other part of the cerebellum were frozen and stored at −80°C for subsequent oxidative stress assays.
Serum hormone assays
Serum gonadotrophin (follicle-stimulating hormone (FSH), free testosterone, prolactin and luteinizing hormone (LH)) and rat thiamine assays were performed with a microplate enzyme-linked immunosorbent assay (ELISA, Labomed, Inc. Los Angeles, USA) reader (EMR-500, Labomed, Inc. Los Angeles, USA) using commercially available ELISA kits (supplied by Chongqing Biospes Co., Ltd, China, with catalogue numbers: BYEK1093, BYEK2865, BYEK2867, BYEK2866 and BYEK2864, respectively).
Total oxidative stress and total antioxidant capacity assays of tissue homogenates
For oxidative stress and antioxidant status determinations, homogenization of frozen cerebellums and testicular tissues was performed using chilled Tris-HCl buffer (pH 7.4), a tissue homogenizer (Glas-Col, Terre Haute, Indiana, USA), and then centrifugation (4000 r/min for 30 min at 4°C). The supernatants from the tissue homogenates were used for colorimetric assays of total oxidative stress in the tissues (using commercial kits supplied by ABC Diagnostics, Egypt) and total antioxidant capacity in the tissues (using commercial kits supplied by Biodiagnostics, Egypt) with a spectrophotometer (Chem-7, Erba Diagnostics Mannheim GmbH, Germany) after spectrophotometric determination of the total protein content (using commercial kits supplied by Spectrum Diagnostics, Egypt) of each tissue homogenate sample. The oxidative stress index was calculated by dividing the oxidative stress status by the total antioxidant capacity. 17,18
Sperm collections and assessments of semen parameters
Epididymal sperm counts were performed according to the modified method by Yokoi et al., 19 whereby the cauda epididymis was minced in 5 ml of 0.9% NaCl and each sample was properly mixed using a shaker for 10 min. The epididymal sperm suspension was incubated at 20°C for 2 min. An alkaline solution containing eosin and 35% formalin was used for supernatant dilution (1:100). A Neubauer haemocytometer was used to count sperm under a light microscope (200×), and the count is expressed in millions per millilitre of the epididymal suspension. The percentage of total sperm motility was determined using a high-magnification (400×) light microscope. 20,21
Histopathological examination
Processing of the fixed cerebellum, testicle and epididymal tissues was performed using conventional techniques of paraffin embedding. Five-μm-thick sections were obtained from the prepared paraffin blocks using a microtome. Then, these sections were stained with haematoxylin and eosin (H&E),
22
for examination by light microscopy. Additionally, testicle and epididymis samples were further examined by Masson’s trichrome staining
23
for the collagenous fibres, Periodic acid–Schiff (PAS) staining
24
for general carbohydrates and immunohistochemical examination for the presence of androgen receptor detection
25
using a polyclonal antibody (catalogue number #RB-9030-R7, Thermo Fisher Scientific, Fremont, California, USA). For immunohistochemistry, 5-μm-thick sections were mounted on poly-
Statistical analysis
All data were analysed using SPSS version 22.0 (IBM, Armonk, New York, USA). The obtained data were expressed as the mean ± standard deviation (SD) and were analysed using analysis of variance followed by Tukey’s honestly significant difference post hoc test. Statistical significance was considered when p < 0.05.
Results
Sperm parameters and reproductive hormone levels among the studied groups
The mean ± SD values of the sperm counts and total sperm motility in the MTZ and MTZ + GP groups (81.43 ± 10.51 million/ml and 42.85% ± 8.59%, respectively; 47.86 ± 14.76 million/ml and 15.0% ± 7.07%, respectively) were significantly lower than those in the control group (172.85 ± 13.87 million/ml and 77.42% ± 5.32%, respectively) (p < 0.05 for all) with a significantly lower sperm count and total sperm motility in the MTZ + GP group than in the MTZ group (p < 0.05) (Table 1).
Mean ± SD of sperm parameters and reproductive hormones among the studied groups.

SD: standard deviation; GP: grapefruit; MTZ: metronidazole; FSH: follicle-stimulating hormone; LH: luteinizing hormone.
a Significantly different from control group at p < 0. 05.
b Significantly different from MTZ group at p < 0. 05.
Additionally, the GP group exhibited a significantly higher mean ± SD value for its sperm count (204.14 ± 10.6 million/ml) than the control group (p < 0.05), with non-significant differences between the two groups regarding total sperm motility (p > 0.05) (Table 1).
The mean ± SD values of the serum-free testosterone and FSH levels in the MTZ group (10.30 ± 1.09 pg/ml and 0.06 ± 0.01 mIU/ml, respectively) and the MTZ + GP group (11.36 ± 0.77 pg/ml and 0.06 ± 0.02 mIU/ml, respectively) were significantly lower than those in the control group (18.25 ± 1.84 pg/ml and 0.07 ± 0.01 mIU/ml, respectively), with p < 0.05 for all and non-significant differences in the mean serum LH levels among the studied groups (p > 0.05) (Table 1).
The mean ± SD values of the serum-free testosterone and FSH levels (16.0 ± 1.91 pg/ml and 0.04 ± 0.37 mIU/ml, respectively) were significantly lower, and the mean ± SD values of the serum prolactin level (0.06 ± 0.07 ng/ml) were significantly higher in the GP group than in the control group (18.25 ± 1.84 pg/ml, 0.07 ± 0.01 mIU/ml and 0.01 ± 0.002 ng/ml, respectively), with non-significant differences in the mean serum levels between the MTZ and MTZ + GP groups compared to the control group (p > 0.05) (Table 1).
Serum thiamine status among the studied groups
Regarding the thiamine (vitamin B1) status, the mean ± SD serum thiamine levels of the MTZ and MTZ + GP groups (134.1 ± 3.41 mg/dl and 131.6 ± 1.91 mg/dl, respectively) were significantly lower than those of the control group (158.2 ± 4.3 mg/dl), p < 0.05, with non-significant differences in the mean thiamine serum levels between the GP group (154.5 ± 6.38 mg/dl) and the control group (p > 0.05, Table 2).
Mean ± SD of serum thiamine levels among the studied groups.

SD: standard deviation; GP: grapefruit; MTZ: metronidazole.
a Significantly different from control group at p < 0. 05.
The total oxidative stress, total antioxidant capacity and oxidative stress index in the testicular tissue homogenates of the studied groups
The testicular tissue homogenates of the MTZ group showed a significantly higher mean total oxidative stress level (0.085 ± 0.0076 mmol/g tissue protein), a higher oxidative stress index (0.023 ± 0.003, respectively) and a significantly lower total antioxidant capacity level (0.569 ± 0.10 mmol/g tissue protein) than the control group (0.005 ± 0.0009 mmol/g tissue protein, 0.003 ± 0.008 and 1.43 ± 0.111 mmol/g tissue protein, respectively, p < 0.05 for all). Additionally, the mean total oxidative stress level and oxidative stress index in the MTZ+GP group (0.117 ± 0.0288 mmol/g tissue protein and 0.12 ± 0.081, respectively) were significantly higher than those of both the MTZ and control groups, with a significantly lower mean total antioxidant capacity level (0.447 ± 0.079 mmol/g tissue protein) than those of the MTZ and control groups (p < 0.05 for all).
Additionally, in the testicular homogenate, the total antioxidant capacity level was significantly higher in the GP group (1.914 ± 0.245 mmol/g tissue protein) than in the control group (p < 0.05), with a non-significant difference in the mean testicular homogenate level of total oxidative stress or the oxidative stress index compared with the control groups (p > 0.05 for all) (Table 3).
Mean ± SD of the total oxidative stress, total antioxidant capacity and oxidative stress index in the testicular tissue homogenates of the studied groups.

SD: standard deviation; GP: grapefruit; MTZ: metronidazole.
a Significantly different from control group at p < 0. 05.
b Significantly different from MTZ group at p < 0. 05.
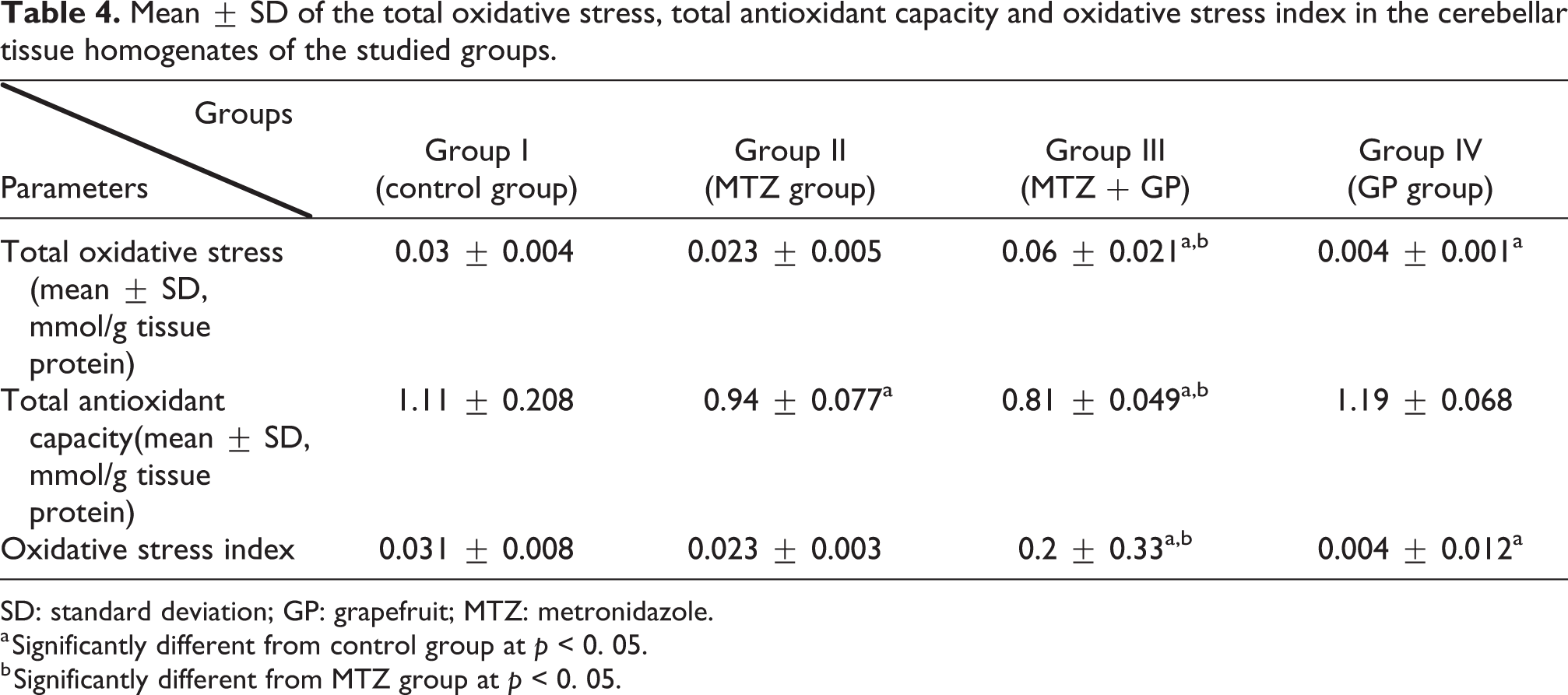
The total oxidative stress, total antioxidant capacity and oxidative stress index in the cerebellar tissue homogenates of the studied groups
The cerebellar tissue homogenates of the MTZ + GP group exhibited a higher oxidative stress level than those of the MTZ and control groups, as evidenced by the presence of a significantly higher mean total oxidative stress level and oxidative stress index (0.06 ± 0.021 mmol/g tissue protein and 0.2 ± 0.33, respectively) and a significantly lower total antioxidant capacity level (0.81 ± 0.049 mmol/g tissue protein) than those of the control group (0.03 ± 0.004 mmol/g tissue protein, 0.031 ± 0.008 and 1.11 ± 0.208 mmol/g tissue protein, respectively) and MTZ group (0.023 ± 0.005 mmol/g tissue protein, 0.023 ± 0.003 and 0.94 ± 0.077 mmol/g tissue protein, respectively, p < 0.05 for all) (Table 4).
Mean ± SD of the total oxidative stress, total antioxidant capacity and oxidative stress index in the cerebellar tissue homogenates of the studied groups.
SD: standard deviation; GP: grapefruit; MTZ: metronidazole.
a Significantly different from control group at p < 0. 05.
b Significantly different from MTZ group at p < 0. 05.
The mean total antioxidant capacity level was significantly lower in the MTZ group than in the control group (p < 0.05) (Table 4).
Additionally, the GP group exhibited a lower oxidative stress level, as evidenced by the presence of a significantly lower mean total oxidative stress level and oxidative stress index (0.004 ± 0.001 mmol/g tissue protein and 0.004 ± 0.012, respectively) than those of the control group (p < 0.05), with no other significant differences between the studied groups regarding oxidative stress markers (Table 4).
Histopathological findings of the cerebellum, testicles and epididymis among the various study groups
Cerebellum
H&E staining and microscopic examinations of the cerebellum of the GP group (Figure 1(b)) revealed a normal histological structure that was similar to that of the control group (Figure 1(a)). Administration of MTZ induced several alterations in the cerebellum of the MTZ-treated group (Figure 1(c)) and the MTZ + GP-treated group (Figure 1(d)). These alterations included degenerative changes with some necrotic areas in the molecular layer and disruption in the Purkinje cell and granular cell layers. The Purkinje cells became shrunken and reduced in size with the loss of their specific ‘flask-shaped’ appearance. The granular cells appeared aggregated compared to the control group.

Photomicrographs of rat cerebellum stained with H&E (bar = 50 μm). (a) and (b) Cerebellum section of control and GP groups, respectively. (c) Section of MTZ-treated group showing the effects of thiamine deficiency induced by MTZ on cerebellum. (d) Cerebellum section of MTZ + GP-treated group. Outer molecular layer (M), middle Purkinje cell layer (thick arrow), inner granular layer (G), white matter (wm), necrotic areas (thin arrows) and disrupted and shrunken Purkinje cells (arrow head). H&E: haematoxylin and eosin; GP: grapefruit; MTZ: metronidazole.
The histopathological cerebellar changes in all studied groups were evaluated and are summarized in Table 5.
Histopathological findings and their scores among various study groups.

PAS: Periodic acid–Schiff; GP: grapefruit; MTZ: metronidazole.
a The change was very often found in all animals.
b The change was relatively common in all animals.
c The change was found in a few animals.
Testicles
The histopathological changes in the testes in all studied groups were evaluated and are summarized in Table 5.
Examination of H&E staining of GP-treated group revealed a normal histological structure of the testicles, with well-organized seminiferous tubules and complete spermatogenesis as well as normal interstitial connective tissue (Figure 2(b)), similar to the control group (Figure 2(a)). There were several histological changes in the testes of the MTZ-treated group (Figure 2(c)) and the MTZ + GP-treated group (Figure 2(d)). These changes included disorganization and degeneration of the seminiferous tubules. The seminiferous tubules had a single-cell layer, vacuolated germinal epithelium and widening of the intertubular spaces. Clear shrinkage of some seminiferous tubules and the presence of interstitial tissue oedema were observed in the testes of the MTZ + GP-treated group (Figure 2(d)).
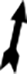
(a) to (p) Photomicrographs of male albino rat’s testis (bar = 50 μm). (a) to (d) Photomicrographs of rat testis stained with H&E. (e) to (h) Photomicrographs of rat testis stained with Masson’s trichrome stain. (i) to (l) Photomicrographs of rat testis stained with PAS reaction. (m) to (p) Photomicrographs of rat testis prepared for androgen receptors immunostaining. (a), (e), (i) and (m) Testes of control group. (b), (f), (j) and (n): Testes of GP-treated group. (c), (g), (k) and (o) Testes of MTZ-treated group showing the hazardous effects of MTZ on spermatogenesis. (d), (h), (l) and (p) Testes of MTZ + GP-treated group. STs, DSTs, ISs, intraepithelial vacuolization (arrow), oedema (stars), shrinkage of the seminiferous tubules (thick arrow), distribution of collagenous fibres ( ), marked positive immunoreactivity to anti-androgen receptors (arrow head) and negative immunoreactivity to anti-androgen receptors (
), marked positive immunoreactivity to anti-androgen receptors (arrow head) and negative immunoreactivity to anti-androgen receptors ( ). H&E: haematoxylin and eosin; PAS: Periodic acid–Schiff; GP: grapefruit; MTZ: metronidazole; ST: Seminiferous tubule; DST: degenerated seminiferous tubule; IS: intertubular space.
). H&E: haematoxylin and eosin; PAS: Periodic acid–Schiff; GP: grapefruit; MTZ: metronidazole; ST: Seminiferous tubule; DST: degenerated seminiferous tubule; IS: intertubular space.
Examination using Masson’s trichrome staining revealed that the sections of the testes of the GP-treated group exhibited a normal distribution of collagen fibres as blue-coloured stripes at the boundary tissue of the seminiferous tubules (Figure 2(f)), similar to what appeared in the normal group of rats (Figure 2(e)). The examination of the testis sections of the MTZ group (Figure 2(g)) and the MTZ + GP group (Figure 2(h)) revealed thickened collagen fibres present around degenerated seminiferous tubules.
Examination using PAS staining revealed a strong intense PAS-positive reaction in the capsule, especially in the tunica albuginea; the basement membranes surrounding seminiferous tubules; the spermatogenic epithelium and the interstitial connective tissue, as appeared in the testes of both the control and GP groups (Figure 2(i) and (j), respectively). PAS-stained sections of the testes from the MTZ-treated groups displayed faint reactions in the deteriorated seminiferous tubules (Figure 2(k) and (l)).
The immunohistochemical findings of the testes from the GP group (Figure 2(n)) and the control group (Figure 2(m)) revealed that the spermatogenic epithelium showed slightly positive immunoreactivity for androgen receptors, while Leydig cells showed strong positive immunoreactivity. On the other hand, the immunohistochemical findings of the testes obtained from the MTZ-treated groups displayed negative immunoreactivity for androgen receptors in all testicular tissues (Figure 2(o) and (p)).
Epididymis
Regarding H&E examination, the epididymis of the GP group (Figure 3(b)) and the control group (Figure 3(a)) showed a normal histological structure with a normal sperm density. Again, administration of MTZ induced several adverse histological changes in the epididymis of the MTZ group (Figure 3(c)) and the MTZ + GP group (Figure 3(d)). These changes included the existence of vacuolated basal cells in the epididymal epithelium beside the majority of epididymal ducts with no or low numbers of spermatozoa in their lumina.
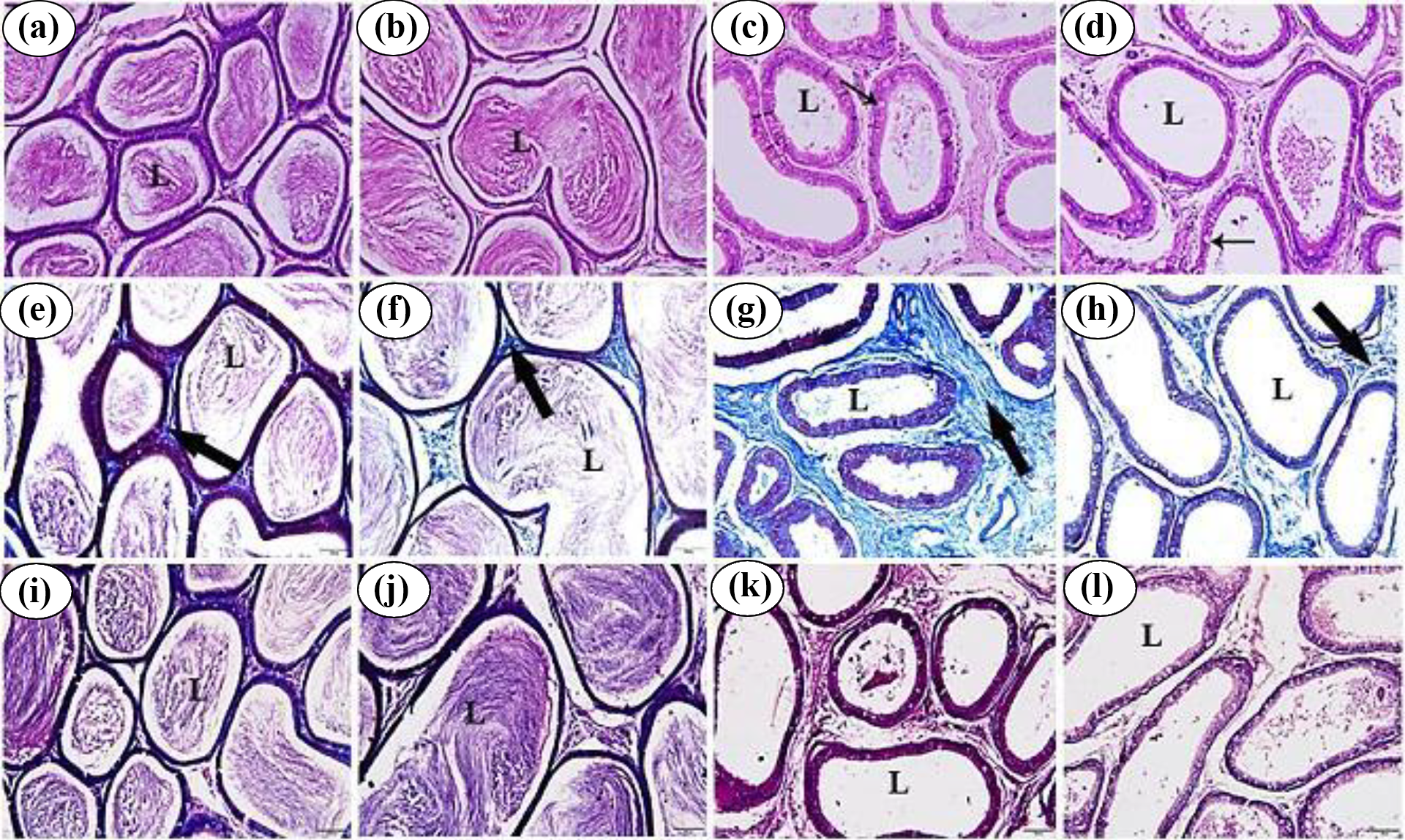
(a) to (l) Photomicrographs of epididymis sections of male albino rats (bar = 50 μm). (a) to (d) Rat epididymis sections stained with H&E. (e) to (h) Rat epididymis sections stained with Masson’s trichrome stain. (i) to (l) Rat epididymis sections stained with PAS reaction. (a), (e) and (i) Epididymis of control group. (b), (f) and (j) Epididymis of GP-treated group. (c), (g) and (k) Epididymis of MTZ-treated group showing effects of thiamine deficiency induced by MTZ. (d), (h) and (l) Epididymis of MTZ + GP-treated group. Epididymal ducts (L), vacuolated basal cells (thin arrows) and distribution of collagenous fibres (thick arrow). H&E: haematoxylin and eosin; PAS: Periodic acid–Schiff; GP: grapefruit; MTZ: metronidazole.
Regarding sections examined by Masson’s trichrome staining, the GP group showed a normal distribution of collagen fibres in the interstitial connective tissue around the epididymal ducts (Figure 3(f)), as did the control group (Figure 3(e)). A marked increase in the thickness of collagen fibres in the interstitial connective tissue was observed in the MTZ group (Figure 3(g)) and the MTZ + GP group (Figure 3(h)).
Regarding PAS-stained sections, the examination of the rat epididymis obtained from the GP group (Figure 3(j)) showed strong intense PAS-positive material in the lumina of the epididymal ducts, as observed in the control group (Figure 3(i)). PAS-stained sections of the epididymis were obtained from the MTZ-treated groups and displayed intense reactions in the epididymal epithelium and faint reactions in sperm debris and in sloughed off germ cells in the lumina of the epididymal ducts (Figure 3(k) and (l)).
A summary of the histopathological changes in the epididymis in all studied groups is presented in Table 5.
Discussion
The potential toxicity of drugs or other chemicals in humans is an essential area of research. 26 MTZ has been chosen for the current study because it is a widely and commonly used drug with many different therapeutic indications. MTZ is used for amoebiasis and for trichomonas and giardiasis infection control. Additionally, it is used for the control of anaerobic organisms, Clostridium difficile-induced pseudomembranous colitis and Helicobacter pylori-induced gastric ulcers. 27 Both the blood–testis barrier (BTB) and blood–brain barrier have similar permeability characteristics and distributions of MTZ to all tissues, including the seminal fluid and brain tissues. 28 –30 The current study reports the possible mechanisms by which chronic MTZ therapy can lead to neurotoxicity and infertility and explores the possible interactions of GP when used in combination with MTZ.
The findings of the present study revealed significant side effects in terms of infertility that are induced by MTZ in the form of decreased sperm count and percent total sperm motility, with significant reductions in the serum levels of free testosterone and FSH. These effects were depicted by regressive histological changes in both the testicular and epididymal tissues of these groups compared with those of the control group. These findings are in line with those of many researchers. 4,13,15,26,30 –32
These spermatotoxic effects of MTZ can be partially explained by the fact that MTZ crosses the BTB and negatively affects the germ cells in the seminiferous tubules, with subsequent decreased secretion of testosterone. 15,32 However, the mechanism of the inhibitory effect of MTZ on reproductive hormones (gonadotrophins and testosterone) is still unclear. Although it is well known that some imidazole drugs (e.g. ketoconazole) inhibit the biosynthesis of testosterone by inhibiting CYP-mediated enzymes (17α-hydroxylase and 17, 20-lyase), this is not the case with MTZ. 33 Likewise, MTZ has not been shown to inhibit the activity of these enzymes in vitro nor does it inhibit the biosynthesis of testosterone in mouse Leydig cell cultures. 33,34
MTZ is a synthetic redox active prodrug. 35 Oxidative stress is a hazardous condition specifically for sperm due to the increased rate of metabolism and replication of cells in the testis; it causes damage to seminiferous tubules. Additionally, oxidative stress can dramatically reduce testicular testosterone production with subsequent effects on spermatogenesis. 36,37 In the current study, the infertility side effect of MTZ was associated with significant oxidative stress in testicular tissue homogenates in the MTZ and MTZ + GP groups compared with the control group. In agreement with our findings, Ligha and Paul 38 reported testicular oxidative stress in male rats following MTZ administration (15, 200 or 400 mg/kg) for 8 weeks. Additionally, Kumari and Singh 39 reported an association between the oral administration of MTZ at a dose of 500 mg/kg for 28 days and testicular homogenate oxidative stress in male Swiss mice.
In the present study, the GP group exhibited a significantly higher total antioxidant capacity in the testicular tissue homogenate than the control group and a significantly increased sperm count compared with the control group. Additionally, at the histopathological level, there were no effects on the cerebellar, testicular or epididymal structures of the GP group, which were similar to those of the control group. The antioxidant and testicular protective effects of GP have been confirmed by many investigators. 14,40 –44
Despite these antioxidant properties of GP, the combined use of GP with MTZ in our study resulted in aggravation of both the infertility and neurotoxicity side effects of MTZ. This adverse effect has been proven at both the biochemical and histopathological levels and can be explained by the possible pharmacokinetic interaction between MTZ and GP. GP is a known inhibitor of cytochrome P450 isoforms. 45,46 Furanocoumarins present in GP are metabolized to reactive metabolites that covalently bond to the active site of CYP3A4, resulting in irreversible inactivation (mechanism-based inhibition), 45 with a subsequent increase in the systemic MTZ concentration, enhancing the toxicity of MTZ. No pharmacokinetic interaction studies in the literature that describe the effects of the combined use of GP and MTZ have been found.
Overall, although MTZ is well tolerated, continuous therapy for more than 4 weeks may be associated with serious reversible neurotoxicity ranging from convulsions to encephalopathy or cerebellar manifestations that can be rapidly improved with termination of MTZ usage. 47,48 Our histopathological findings revealed the occurrence of MTZ-induced neurotoxicity in the form of cerebellar cortical Purkinje and granular cell layer damage. Purkinje cells are GABAergic and principle neurons of the cerebellar cortex, while granular cells include glutamatergic neurons. 49 It has been reported that neurotoxicity in rats occurs following oral administration of MTZ at a dose of 540 mg/kg for 60 days. 13 Additionally, MTZ-induced neurotoxicity has been frequently reported in humans. Toumi et al. 50 reported cerebellar syndrome in a 27-year-old male patient treated with MTZ for a cerebral abscess. Retamal-Riquelme et al. 51 reported reversible cerebellar syndrome in a 34-year-old female who received MTZ for 49 days for a liver abscess and concluded that MTZ-induced neurotoxicity should be considered because it has a good prognosis after cessation of drug usage. Additionally, AlDhaleei et al. 52 reported such toxicity in a 38-year-old male patient who received MTZ for more than 10 weeks for a brain abscess. The exact mechanism of MTZ-induced cerebellar toxicity is not fully understood but has many hypotheses. One hypothesis suggests that MTZ and its metabolites may bind to neuronal RNA with subsequent inhibition of protein synthesis, resulting in reversible axonal swelling with symmetrical damage to the cerebellar nuclei. 7 Another hypothesis postulates that MTZ may modulate gamma-amino butyric acid (GABA; an inhibitory neurotransmitter) receptors in the vestibular and cerebellar systems. Additionally, MTZ may induce semiquinone and nitro anion neurotoxic radicals. 53
Thiamine has a critical role in spermatogenesis, as it has been demonstrated that male germ cells are more susceptible to apoptosis triggered by intracellular thiamine deficiency than any other tissue type. In addition, the hexose monophosphate pathway (HMP), rather than mitochondrial metabolism, functions in highly motile sperm, evidenced by their higher thiamine (cofactor for transketolase) content, as HMP provides NADPH, a factor required for reduced production of glutathione, which is an essential antioxidant for spermatozoa. 10,54,55
The MTZ-induced cerebellar lesions in rats are topographically and qualitatively similar to those induced by thiamine deficiency in rats and in Wernicke’s encephalopathy in humans, as thiamine deficiency induces neuronal loss. 55 –57 In the current study, the neurotoxicity side effect of MTZ was associated with significant oxidative stress in the cerebellar tissue homogenates of the MTZ and MTZ + GP groups compared with the control group and lower serum thiamine levels in the MTZ and MTZ + GP groups than in the controls. It is proposed that MTZ acts as a thiamine analogue or antagonist via inhibition of thiamine pyrophosphorylation, 9,58,59 which can partially explain MTZ-induced neurotoxicity via thiamine deficiency. Thus, the current study provides additional evidence to support the association between blood thiamine levels and not only MTZ-induced neurotoxicity but also MTZ-induced infertility side effects.
A striking observation in our study was the finding that the animals treated with GP only showed significantly lower free testosterone and FSH serum levels with significantly higher serum prolactin levels than the control animals. Although these findings cannot be explained and need to be confirmed by further studies, no report in the literature could be found regarding the effects of GP on male hormones. GP could have a stimulatory effect on aromatase, which catalyses testosterone conversion into oestrogen. Monroe et al. 60 suggested an important interaction between GP intake and endogenous oestrogen levels; additionally, GP may have an effect on the catabolism or the interconversion of sex hormones and prolactin, so carefully controlled pharmacokinetic studies are required to properly understand GP effects on MTZ, sex hormones and prolactin metabolism to confirm the findings of the current study.
Conclusions
In conclusion, our results revealed that MTZ induces side effects of neurotoxicity and infertility when used for long periods, which maybe attributable to thiamine deficiency and oxidative stress as underlying mechanisms. Patients should be monitored for MTZ adverse effects, such as central neuropathy and thiamine status, particularly if MTZ has been prescribed for long-term use. GP, despite its antioxidant effect, lowers serum testosterone and FSH levels, increases prolactin serum levels and enhances MTZ toxicity, so GP should be avoided when MTZ is prescribed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current research has been funded by the authors themselves.
