Abstract
Electrolyzed water (EW) is a widely used disinfectant agent with high oxidation–reduction potential (ORP). Although EW has been used in many areas, such as food hygiene, agriculture, and animal husbandry, the studies presented in the literature are not enough to clarify the toxic effects of EW. The aim of this study is, therefore, to produce EWs at different pH, ORP, and chlorine concentrations and to assess their safety in terms of toxicology. At the beginning of the study, the antimicrobial activity of the EW types with respect to bacteria and fungus was investigated. EWs below pH 7 were all effective in inactivating
Keywords
Introduction
The electrochemical (unipolar) activation of water consists of the possibility to significantly change speed and selectivity of many chemical reactions, as well as properties and state of the liquid itself. This is achieved by using the energy of the metastable state of substances after a nonequilibrium electrochemical effect. The emergence and preservation of additional potential energy of water are due to the activation of atoms, ions, and molecules in the liquid itself that leads to the appearance of anomalous values of physicochemical parameters. The excited, metastable state of the solution after electrochemical exposure is called activated state. Electrochemically activated solutions are referred to in the literature as electrolyzed water (EW).
The EW is a clean technology that has been highly favored in recent days. The technology is based on electrolysis of water containing sodium chloride or potassium chloride in an electrolysis chamber, where anode and cathode electrodes are separated by an ion-permeable diaphragm. 1 Positive charge forms at the anode surface through an oxidation process, whereas a reduction process takes place at the cathode surface. Herewith, two new products form in the cathode and anode zone, known as catholyte and anolyte, respectively. The oxygen gas and many active species, such as chlorine, chlorine dioxide, and free radicals, are being formed in the anolyte zone, while hydrogen gas and sodium hydroxide are being formed in the catholyte zone. 2,3
The catholyte and anolyte have peculiar characteristics: the former has high alkalinity (pH > 11) with low oxidation–reduction potential (ORP < −800 mV) and the other has high acidity (pH < 2.5) with strong ORP value (ORP > 1100 mV) and free chlorine concentration (FCC) that can be between 40 and 1500 ppm. 4,5 Saturated with reducing agents, the catholyte (mentioned as CEW hereafter) acquires a high adsorption-chemical activity, as well as properties of strong detergent. On the other hand, the presence of a sufficient number of strong oxidants and free radicals in the anolyte turns it into a solution with strongly pronounced biocidal properties. The anolyte, namely EW, has been used as a disinfectant agent because of its high oxidation potential and the active species content. 6 EW produced in a variety of forms is widely used in many areas such as food hygiene, agriculture and animal husbandry, medicine and medical field, restaurants, schools, and hotels. 7,8 Depending on the intended use, two different types of water as strong acidic EW (StAEW) and slightly acidic EW (SAEW) can be produced. 9 For example, StAEW is known to have a strong bactericidal effect on the most known pathogenic bacteria with its low pH, high ORP, and FCC. 10 On the other hand, SAEW has a nearly neutral pH, lower ORP value, and FCC. The SAEW has attracted much interest, because it contains low free chlorine, has less Cl2 gas emissions, has low corrosive effects on surfaces, and is a highly effective antimicrobial agent with limited phytotoxic effects compared to hypochlorous acid derivatives. 11 Therefore, SAEW has been considered as a medical disinfectant due to the corrosive effects of StAEW. 12 Mixed electrolyzed water (MEW) is the name of a preparation that obtained mixing anolyte and catholyte (in an equal volume of each in our case). It is expected that the solutions do not lose their superior features after mixing due to the unipolar (i.e. irreversible) reaction during electrolysis.
The use of EW as a biocide has emerged recently. 13,14 However, all biocidal products require authorization before they are released into the market, according to European Chemicals Agency Guidance on the Biocidal Products Regulation published in 2018, and it is necessary to show their safety profile. One of the mandatory test methods to evaluate the toxicological profile for human dermal exposure is to examine the skin irritation potential of the products. 15 Skin irritation is the production of reversible damage of the skin following the application of a test substance (TS) for up to 4 h. The assessment of skin irritation has typically performed using laboratory animals. However, to reduce the use of in vivo testing, alternative in vitro methods for assessing skin irritation potential have been developed using a reconstructed human epidermis (RhE) model. 16 The RhE model is highly preferred for assessing the biocompatibility of disinfectants. 17,18 According to the same guideline, the other test method that needs to be performed is the evaluation of eye irritation. 15 Although it has not validated yet, the Interagency Coordinating Committee on the validation of alternative methods (ICCVAM) recommended hen’s egg test-chorioallantoic membrane (HET-CAM) test method is highly preferred to predict eye injury hazard potential of chemicals. As an alternative to in vivo Draize Rabbit eye test, HET-CAM is mimicking the vascular changes in the CAM. 19,20,21 As well, products may come within the remit of the regulations concerning medical devices for human use, 22 depending on the disinfectants’ intended uses. For surface medical devices on the skin, the standard recommends evaluating also their cytotoxicity, besides sensitization and irritation capacity. 23
The antimicrobial effect on various microorganisms’ of the EWs has been presented for years. 13,14 Accordingly, the usage of the EWs has been recognized in many areas such as food hygiene, agriculture, and animal husbandry. However, the studies presented in the literature are not enough to clarify the toxic effects of EW. In this study, therefore, the purpose of this work was to expose the safety profile of EWs, which is widely used as a disinfectant and/or as a medical device. Therefore, EWs at different pH and chlorine concentrations were produced, and their dermal biocompatibility properties were investigated by testing cytotoxicity, eye irritation, and skin irritation assays in vitro.
Materials and methods
Production and characterization of electrolyzed water
The EWs were produced by the electrolysis of tap water containing 5 g/L of sodium chloride in a divided, circular electrochemical reactor. The reactor comprises a steel cathode, a carbon-based anode, and a semipermeable membrane separator. It has 250 mL of the EW production capacity through a single batch run. A power supply (TTechnic, Istanbul, Turkey) was used to provide direct current, and the electric potential is given to the anode and cathode surface. A scheme of the EW production is given in Figure 1. To produce EW, the brine solution was added into the reactor, and the power supply was operated during the desired reaction time. The brine water was separated into ions because of the unipolar electrochemical process in the reactor. The brine water has a very weak current within it (i.e. approximately 350 mV), and the change of ORP value in the anolyte and catholyte zone in the opposite direction started with the onset of electrolysis. The EWs below pH 7 were collected from the anodic side of the reactor after electrolyzing was finished. CEW was collected from the cathodic side during the same run for StAEW. MEW was an equal amount of StAEW and CEW produced under the same conditions. SAEW was obtained by producing lower applied current and longer electrolysis time than StAEW produced. The production conditions and properties of EWs are shown in Table 1. The EWs were freshly prepared on the day of each experiment.

The scheme of the electrolyzed water production.
Production details and characteristics of EWs.
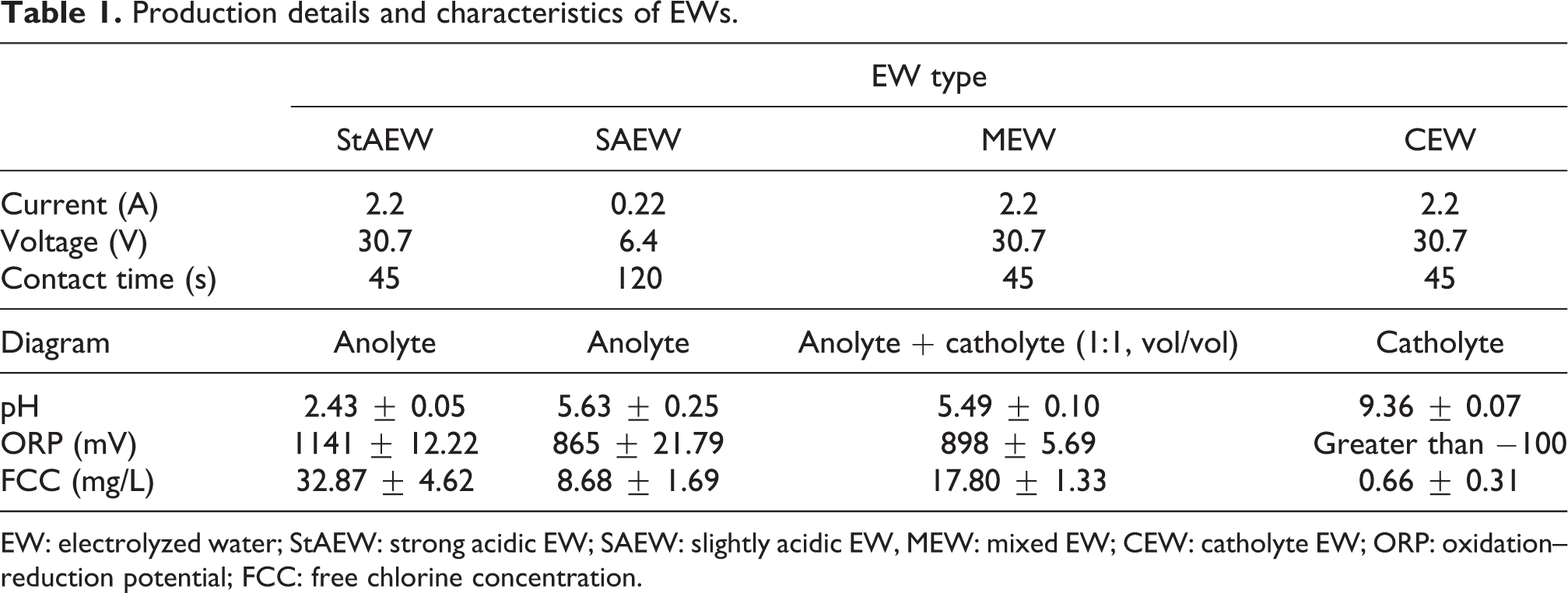
EW: electrolyzed water; StAEW: strong acidic EW; SAEW: slightly acidic EW, MEW: mixed EW; CEW: catholyte EW; ORP: oxidation–reduction potential; FCC: free chlorine concentration.
To determine the characterization of EWs, the measurements of pH (Ohaus, Parsippany, NJ/USA), conductivity (Hanna Ins., USA), and ORP (Oakton, Singapore) were performed. FCC (i.e. the sum of hypochlorous acid and hypochlorite ions concentration) of EW was determined using
Preparation of bacterial culture
The ATCC cell lines used for the microbiological tests were
Microbiological tests through EW Treatment
The microbiological tests were performed according to procedures EN 1276 25 and EN 1650 26 of the European National Standards. For each treatment with EW, 1 mL of bacteria suspension and 1 mL of bovine serum albumin were mixed in a sterile test tube. Then, this mixture was transferred to a new tube that already contained 8 mL of EW and they were mixed properly. In the case of the control sample, the mixture was added in 8 mL of sterilized dH2O instead of EW. After the desired contact time, the reaction was quenched with 9 mL of Dey–Engley neutralizing broth. This form of the mixture was accepted as 1:10 dilution and then 10-fold serial dilutions were made up to 10−7 dilutions. For bacterial counting, 1 mL of each dilution was spread over the petri dish including tryptic soy agar by using a sterile swab. In the case of fungi counting, malt extract agar was used in the same way. The incubation period was 24 h for bacteria at 37°C and 48 h for fungus at 30°C. After the incubation period, colonies were counted, and agar plates containing 30–300 colonies were considered as significant. Logarithmic reduction values for each sample were calculated upon the differences between the logarithmic value of the initial and residual number of bacteria.
Cell viability with MTT assay
Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. L929 mouse fibroblast cell line (ATCC) was maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, Fountain Drive, Pailey/UK) supplemented with 10% fetal bovine serum, streptomycin, and penicillin used from Gibco at 37°C in 5% CO2. The cells were seeded 1 × 104 per well in 96-well plate and incubated for 24 h to form a semiconfluent layer. After 24 h, the cells were exposed to four different concentrations of EW by serial dilutions with DMEM (1.25%, 2.5%, 5%, and 10%, vol/vol). After 24 h incubation, MTT was added to wells at 0.5 mg/mL of concentration and incubated for an additional 2 h at 37°C. After discarding the medium from plates, 100 μL of isopropanol was added to the wells. The absorbance of the MTT formazan was determined at 570 nm by a UV-spectrophotometric plate reader (BioTek ELx808™, Turkey). Viability was defined as the ratio (expressed as a percentage) of absorbance of the cells exposed to compounds to the cells treated with 0.5% dimethyl sulfoxide (DMSO; as control). As positive control (PC), DMSO 20% (vol/vol) (Sigma, USA) was used. All measurements were conducted in triplicate, and error bars were given in the relevant figure. The cell viability ratio was determined to be the ratio between the absorbance of the tested well and the absorbance of the control well by using the following equation:

where OD570TS is the OD of the TS and OD570NC is the OD of the negative control (NC).
Eye irritation, in vitro
To evaluate the potential eye irritancy of EWs, ICCVAM recommended HET-CAM test method was applied, and the reactions on the CAM were scored according to lysis, hemorrhage, and coagulation within 300 s of the EW exposure. 19 To summarize the method, fertile White Leghorn chicken eggs weighing between 50 and 60 g were incubated at 38.3 ± 0.2°C and 58 ± 2% relative humidity until day 9. The air cell of the egg was marked and cut with a drill and then pared it off. Scores based on test protocol if lysis occurs within 30 s is 5, within 2 min is 3, and within 5 min is 1. For scoring of hemorrhage, if the reaction occurs within 30 s score is 7, within 2 min is 5, and within 5 min is 3. Lastly, for coagulation, the reaction within 30 s is scored as 9, within 2 min is 7, and within 5 min is 5 as well. 19
The inner membrane was moistened with 0.9% (wt/vol) NaCl in the incubator for a maximum of 30 min. After decanting the 0.9% NaCl solution, the inner membrane was carefully removed with forceps; 0.3 mL of EWs, a known eye irritant, 1% (wt/vol) sodium dodecyl sulfate (SDS) as a PC, and 0.9% NaCl as a NC were applied directly onto the CAM surface according to the test protocol. Then, reactions were observed on the CAM over a period of 300 s. The time for the appearance of each of the noted end points, namely, hemorrhage, lysis, and coagulation, was monitored and recorded at fixed time intervals of 0.5, 2, and 5 min. The results were considered acceptable if the NC and PC each induced a response that falls within the classification of nonirritating and severely irritating, respectively. According to historical control studies, this indicates that using 0.9% NaCl as the NC, the irritation score (IS) value was zero and using 1% SDS as PC, the IS values ranged between 10 and 19. The classification of severe irritancy for a TS was assigned when the IS value was greater than 9.
In vitro skin irritation test
To predict skin irritation potential of EWs, the reconstructed human epidermal model EpiDerm (EPI-200, MatTek, Ashland, MA/USA) validated by European Centre For the Validation of Alternative Methods (ECVAM) was used. 16 According to manufacturer instructions, tissues were topically exposed to 30 µL of the undiluted tested EWs, NC, or PC to three single tissues each for 60 min; 5% SDS (in H2O) solution was used as PC and sterile Dulbecco’s phosphate-buffered saline (DPBS), pH 7.4, was used as NC and tested concurrently with EW samples. Tissue was thoroughly rinsed with sterile DPBS and excess of DPBS was removed by gentle shaking as well as blotted on sterile blotting paper. Tissue inserts were transferred to new well plates prefilled with fresh assay medium. After a 24-h incubation period, the medium was collected for analysis of inflammatory cytokine interleukin (IL)-1α, which is a complementary measurement of response to physical or chemical stress of keratinocytes according to EpiDerm protocol. After 3-h MTT incubation, the blue formazan salt formed by cellular mitochondria was extracted with 2 mL/tissue of isopropanol. Then, the optical density of the extracted formazan was determined at 570 nm using a spectrophotometer. The reduction of the viability of the tissues exposed to EWs in comparison to NC was used to predict the skin irritation potential. The remaining relative cell viability below 50% was considered as the irritant according to EpiDerm protocol:
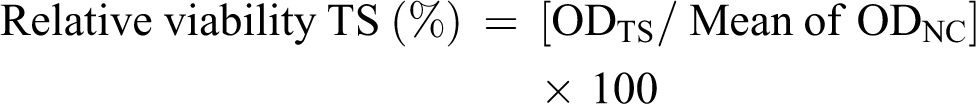
where ODTS is the OD of the TS and ODNC is the OD of the NC.
IL-1α analysis
The concentration of IL-1α released from the RhE tissues into the assay medium during the exposure period was measured by commercially available quantitative enzyme-linked immunosorbent assay (ELISA) system according to the manufacturer’s instructions (Abbkine Human IL-1α ELISA Kit, Wuhan, China). The concentration of IL-1α for each sample was calculated and a two-fold increase or greater, compared to the NC, was considered a positive irritation response according to EpiDerm-skin irritation test (SIT) protocol. 27 Results represent the mean ± SD of three individual IL-1α released from the RhE tissues.
Statistical analysis
GraphPad Prism Software version 6.0 (San Diego, California, USA) was used for all the statistical analyses. Data related to cell viability were analyzed by using one-way analysis of variance following the post hoc tests by Turkey.
Results
Characterization of electrolyzed water
Figure 2 shows the change of pH, FCC, and ORP value against electrolysis time. The increment of chlorine concentration caused the decrement of pH value during EW production, as shown in Figure 2(a). The reason for the pH decrease in the media is the formation of chlorine gas, hypochlorous acid, and hydrochloric acid in aqueous media. 5 The form of chlorine compounds (OCl−, Cl2, or HOCl) present in a disinfectant is majorly affected by the pH of the solution. Hypochlorous acid is the most active form of chlorine, and its activity is the maximum at a pH of 5.0–6.5. At a higher pH ≥ 7.5, the inactive hypochlorite ion (OCl−) predominates, and very little of the active hypochlorous acid is available (HOCl).

Characteristic parameters of EW during electrolysis: (a) pH and FCC change and (b) ORP change (5 g/L of sodium chloride concentration, 1 ampere of applied current). EW: electrolyzed water; ORP: oxidation–reduction potential; FCC: free chlorine concentration.
In our cases, the sum of the HOCl and OCl− (i.e. FCC) increased up to 378 mg/L at the end of the 20 min of electrolysis time. As another parameter for monitoring EW, ORP is a measure of the intensity of the oxidation–reduction processes in the system and is determined by the ratio of concentrations for oxidation and reduction sorts of ions forming during the reaction. As seen from Figure 2(b), the original ORP value of brine water (i.e. 351 mV) changed toward the opposite directions during electrolysis. The ORP value of EW in the anodic side of the reactor reached 1000 mV in a short time, and over time, it increased to around 1200 mV. The increment of the ORP value generally remains at about 1200 mV. 14,28
In the case of the cathode zone, the ORP value started to decrease toward minus values and reached to −960 mV after 30 min, as seen from Figure 2(b). Lower ORP values of a solution indicate a high tendency to donate electrons. The CEW with these ORP values acts as a reducing agent and can take pH value from 7 to 12.8. 5,29 As it can be seen from the maximum values of the pH and ORP parameters, the pH of the CEW corresponds to a practically concentrated alkali solution, and the pH of the anolyte corresponds to the concentration of the acid solution.
Microbiological test
Microbiological tests were conducted on
Logarithmic reduction during StAEW treatment.
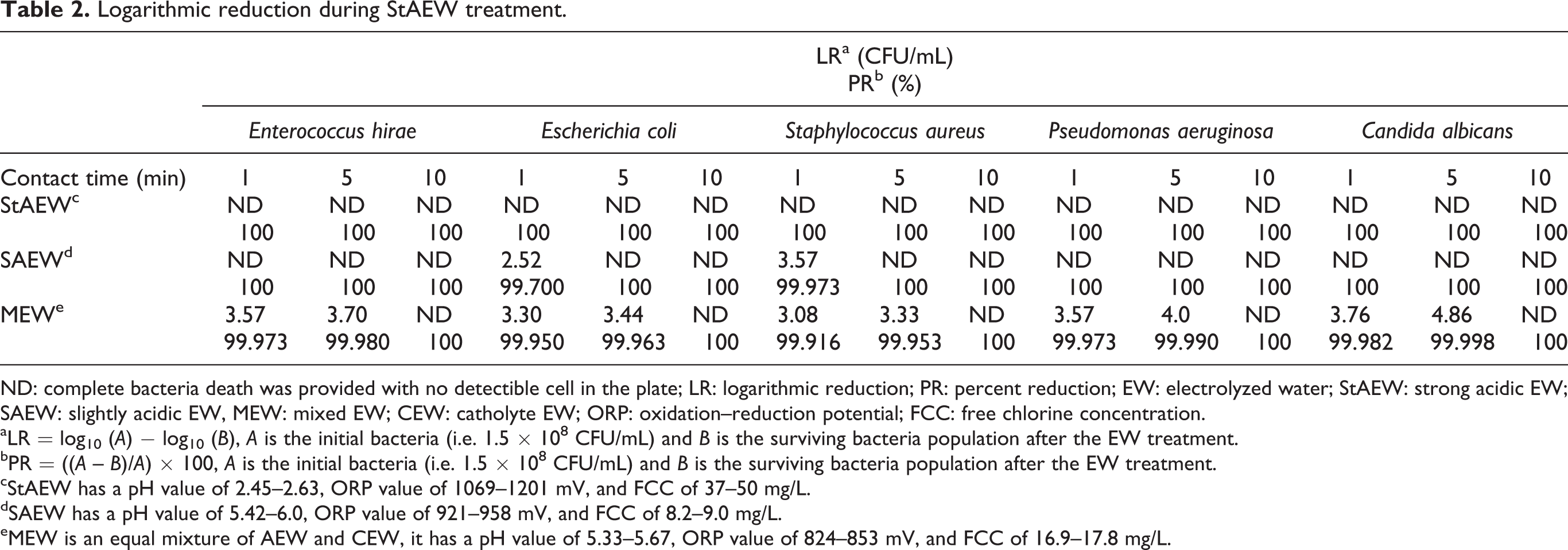
ND: complete bacteria death was provided with no detectible cell in the plate; LR: logarithmic reduction; PR: percent reduction; EW: electrolyzed water; StAEW: strong acidic EW; SAEW: slightly acidic EW, MEW: mixed EW; CEW: catholyte EW; ORP: oxidation–reduction potential; FCC: free chlorine concentration.
aLR = log10 (
bPR = ((
cStAEW has a pH value of 2.45–2.63, ORP value of 1069–1201 mV, and FCC of 37–50 mg/L.
dSAEW has a pH value of 5.42–6.0, ORP value of 921–958 mV, and FCC of 8.2–9.0 mg/L.
eMEW is an equal mixture of AEW and CEW, it has a pH value of 5.33–5.67, ORP value of 824–853 mV, and FCC of 16.9–17.8 mg/L.
Cell viability
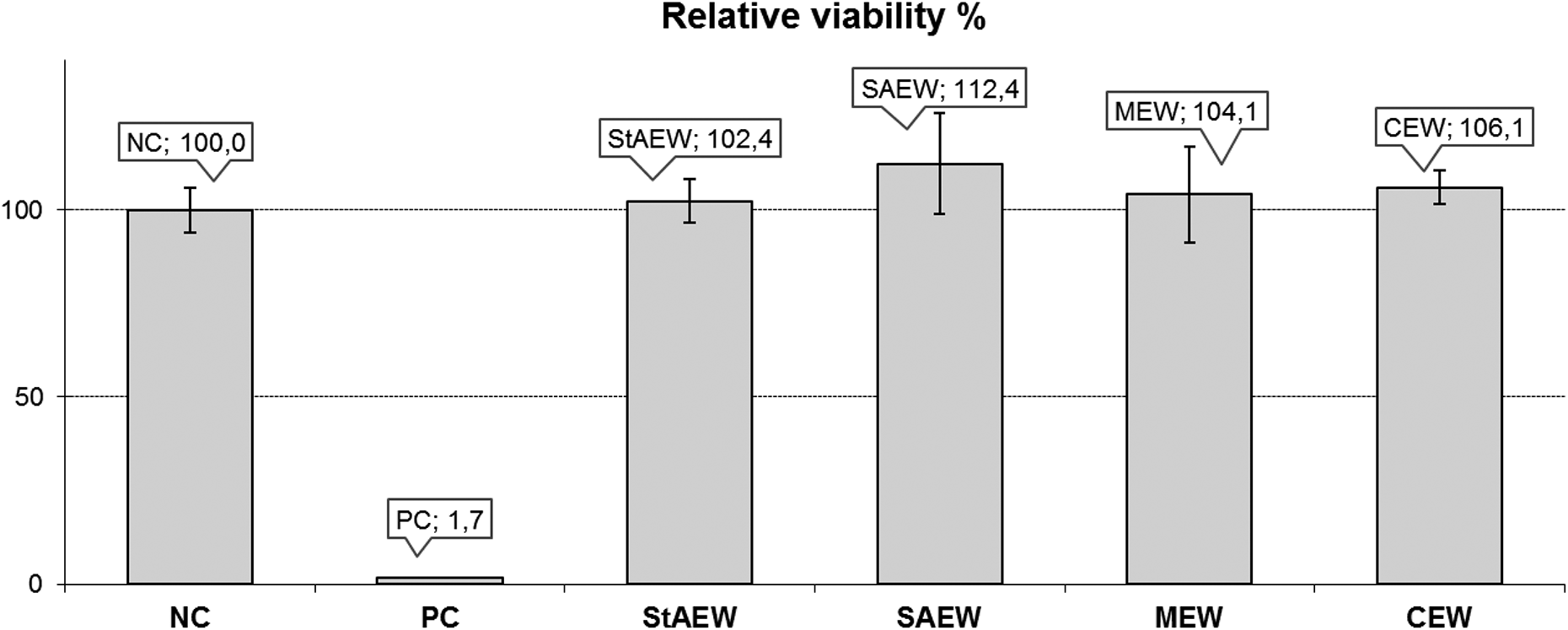
The effect of the 24-h exposures to four different concentrations of EW (1.25%, 2.5%, 5%, and 10%, vol/vol) on the cell viability is shown in Figure 3. In all experimental doses of four different EW samples, cell viability was above 70%, which was evaluated, as the samples were not cytotoxic on L929 mouse fibroblast cell line. Cell viabilities did not show a dose-dependent response in StAEW- and SAEW-treated group, while MEW and CEW exposure resulted in a dose-dependent decrease in cell viability. The cell proliferation rate was significantly increased (116.05 ± 2.38%) at the lowest dose (1.25%) of CEW. In addition, PC (i.e. DMSO 20% (vol/vol)) significantly reduced cell viability of L929 cell line.

Relative cell viability on L929 cell line after the 24-h exposure of four different concentrations of EWs. Results represent the mean ± SD of three independent experiments, which were repeated at 3 weeks. Negative control: medium (DMEM), positive control: 20% DMSO. Significant difference compared to control: *
Hen’s egg test-chorioallantoic membrane
In HET-CAM screening test for eye irritancy, after moistening, 0.3 mL of each EW was applied directly onto the CAM surface. Then, reactions on the CAM were scored over a period of 300 s. According to the results, the IS value of PC was scored as 12, which is in the acceptable range according to the ICCVAM protocol. The tested EWs did not cause lysis, hemorrhage, and coagulation according to HET-CAM scoring (Figure 4). The IS values (max 1) for the tested EWs indicate that there was no eye irritancy potential for the produced EWs (Table 3).

HET-CAM in vitro irritation test for detecting ocular irritancy. NC: negative control (0.9% NaCl); PC: positive control (1% SDS); EW: electrolyzed water; StAEW: strong acidic EW; SAEW: slightly acidic EW; MEW: mixed EW; CEW: catholyte EW; HET-CAM: hen’s egg test-chorioallantoic membrane; SDS: sodium dodecyl sulfate.
Irritation score of tested EWs according to HET-CAM protocol.

EW: electrolyzed water; StAEW: strong acidic EW; SAEW: slightly acidic EW, MEW: mixed EW; CEW: catholyte EW; NC: negative control; PC: positive control.
aThe severe irritancy classification for a test substance is assigned when the value is greater than 9.
Skin irritation
According to the manufacturer’s protocol, the assay met the acceptance criterion as far as the mean viability of PC (5% SDS) tissues expressed as a percentage of the NC tissues is ≤20% (Figure 5). None of the tested EWs was assessed as causing any skin irritation because the mean relative viability of three individual tissues exposed to the EAWs was not reduced below 50% of the mean viability of the NCs.
Percentage of relative cell viability after Epiderm SIT in vitro irritation test. Results represent the mean ± SD of three individual tissues. NC: negative control (DPBS); PC: positive control (5% SDS); EW: electrolyzed water; StAEW: strong acidic EW; SAEW: slightly acidic EW; MEW: mixed EW; CEW: catholyte EW; SDS: sodium dodecyl sulfate; SIT: skin irritation test; SD: standard deviation.
IL-1α analysis
The concentration of IL-1α released from the RhE tissues into the assay medium was calculated according to the manufacturer’s instructions. The concentration of IL-1α for StAEW, SAEW, and CEW was significantly higher compared to NC. Since the two-fold increase or greater, compared to the NC, was considered a positive irritation response, the EAWs samples were considered to have no potential for irritation. 27 The PC was found to be significantly higher compared to all tested EAWs and NC (Figure 6).

IL-1α released into the assay medium after exposure of EpiDerm™ tissues to controls and EW samples. Results represent the mean ± SD of three individual tissues. NC: negative control (DPBS), PC: positive control (5% SDS). *
Discussion
The production of EWs according to their usage purpose was evaluated within the experimental setup, and in this context, four types of EW were produced in a special reactor divided by a membrane. The brine concentration was kept constant (i.e. 5 g/L) during the production of EWs, however, the longer electrolysis time was required to obtain SAEW.
The characteristic properties of the StAEW were pH 2.43, ORP 1140 mV, and FCC 32.87 mg/L. All test microorganisms were inactivated in a short time with the application of StAEW, as proved in the literature earlier. 10,30,31 On the other hand, nearly neutral SAEW has more application areas than the StAEW, since it does not cause the dangerous Cl2 gas formation as well as corrosion of the processing equipment. 10 In this study, SAEW and MEW were produced through 45 s of electrolysis time to obtain moderate disinfection conditions. Although both EWs had similar pH and ORP values, the inactivation effects on test microorganisms were different according to the results of the microbiological test. For example, MEW could not inactivate before 10 min, although the chlorine content of the MEW was higher than that of the SAEW. It can be said that the inactivation mechanism of EWs is not due to the content of chlorine in it or the addition of catholyte to anolyte (i.e. MEW) decreased inactivation efficacy by suppressing activities of oxidant moieties in EW.
pH and ORP values are vital for living microorganisms since they can stay alive and reproduce on nutrient media only in certain ranges of ORP values, depending on the acid-alkaline characteristics of this media. In the pH range of nutrient media from 3 to 8, ORP ranges that are compatible with the life of microorganisms are between 600 and 800 mV. Even if the bacterial resistance against the acidic media increases (i.e. pH 2–3), microorganisms do not give growth at ORP < 400 ÷ 550 mV and at ORP > 900 ÷ 1050 mV. 32 The application of EWs to the microorganisms changes the pH and ORP values of nutrient media or, in other words, the speed and the direction of oxidation–reduction processes in the system. In this way, EWs can affect the functional properties of electroactive components of biological systems. Furthermore, EW is an aqueous medium with enhanced electroacceptor properties. In the chloride-type anolytes, the greatest biocidal activity is due to the free radicals ClO·, Cl·, and HO·, whose sources are both HClO and ClO−. Moreover, the highest oxidizing ability of the anolyte in reactions on the membranes of bacterial cells is associated with the presence of both forms

In addition to this reaction, water oxidation with the release of oxygen takes place at the anode
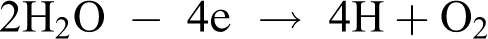
Depending on the micro conditions on the anode surface, reactions of the hydrogen peroxide and a number of its decomposition products formation also can take place

The products of these reactions, possessing high reactivity, can be stored for a long time in the anolyte, which has low pH values (2–4) and high redox potential (800–1200 mV). Electrochemical reactions occurring in the cathode zone are associated with the water reduction reaction

When an electron passes from a cathode to a hydroxonium ion, a number of highly active products arise, which provide catalyst properties to catholyte. The resulting active water for a long time keeps low values of the redox potential (from −700 to −960 mV) and high pH values (9.5–13).
Free radicals and atomic oxygen oxidize the membrane of the bacterial cell. From the thermodynamic point of view, the formation of atomic oxygen is the energetically favorable process causing the growth of the entropy factor and the decrease of the Gibbs energy. As a chemical oxidizer, “active” oxygen disrupts the work of oxidative–reductive enzymes. By oxidizing catalytic groups in the active center, it interferes with their participation in the separation of hydrogen from the substrate and in the transfer of protons and electrons to the respiratory chain.
The processes of free-radical oxidation strengthening at the tissue level are accompanied by the accumulation of either lipid peroxides or products of lipid peroxidation in cell membranes and mitochondria that leads to an increase in the oxygen utilization and influences the oxidative phosphorylation. The destructive effect of free radicals on bacteria has been reported comparing the EW with NaOCl and ClO2. 33 Although the mechanism leading to the final death of bacteria was similar for all disinfectants, the antimicrobial test with the EW resulted in faster lipid peroxidation and more release of intracellular micro- and macromolecules than that of NaOCl and ClO2.
The use of the EW is well known and becoming favorable because chemical disinfectants, such as benzalkonium chloride, formaldehyde, and glutaraldehyde, are potentially toxic to humans and corrosive to the application area. 34 Despite the increased use of EWs and evidence of their effects on microorganisms in the literature, the assessment for its toxic effects has been relinquished up to date. It is desirable for a disinfectant agent to be noncytotoxic as well as killing microorganisms adequately, and not to damage the living organisms or materials to which it is applied. Therefore, it is not sufficient to investigate the potency of EW only in terms of ability to destroy specific pathogens.
To assess the cytotoxicity of the produced EW, the MTT assay was performed in the mouse fibroblast cell line (L929) in this study. L929 cells were used due to their ease of culturing in a reproducible manner. Also, according to Swain et al., 35 this cell line is suitable for preliminary cytotoxicity evaluation for a wide range of biomaterials because of suitable proliferation rate and adherence on most of the biomaterial surface. In all experimental doses of four different EW samples, cell viability was above 70%, which was evaluated, as the samples were not cytotoxic on L929 mouse fibroblast cell line. In similar 24-h exposure model, the results indicated that EWs were not cytotoxic, since the cell viability was above 70% for all tested concentrations (1.25% ÷ 10% vol/vol) of four different types of EW. Similar to our results, Gomi et al. 36 reported that the 24-h exposure to AEW (pH 2.8) did not lead to cytotoxicity on human pulp cells, when dilutions between 10 and 1000 fold (vol/vol) were used. In another study, 37 strong alkaline EW (pH: 12.15, ORP: −1068 mV) and StAEW (pH: 2.49, ORP: 955.9 mV) were tested for 24 h on stratum corneum tissue sample of the hairless rat by MTT assay, and it was observed that both EWs are highly safe.
In addition to the pharmaceutical compatibility and industrial studies on EWs, there are several cytotoxicity studies investigating the effect of EW on the cancer cell lines due to its variable capacity to oxidation/reduction. 38,39 Tsai et al. 38 suggested that the treatment by CEW (pH: 8.10 ÷ 9.50, ORP: −160 mV ÷ −400 mV) together with glutathione combination significantly decreased the survival rate of HL-60 leukemia cells for 48- and 72-h exposure. According to the study, EW may lead to dose- and time-dependent cytotoxicity in combination with glutathione through a mitochondria-dependent pathway while this effect was not seen with 24-h exposure. 38 In a different study, 39 the effect of apple juice reconstituted with alkaline EW (pH 10.59, ORP: −139 mV) on human colon adenocarcinoma (HT-29) cells was investigated. It was suggested that HT-29 cells treated with 0.5 μL/mL of apple juice reconstituted with alkaline EW for 72-h showed a decreasing effect on the cell viability when a dose of alkaline EW increased. This effect was attributed to the antioxidant activity of polyphenol-rich apple juice and EW, which may alter cell functions, such as cell proliferation and cell cycle arrest. 39
In vitro test methods are becoming more popular as an alternative to the animal test methods. 40 In our study, we, therefore, preferred in vitro alternative testing methods for the evaluation of the irritation potential of the EW samples. In the HET-CAM assay method for the detection of eye irritants, none of the produced EWs caused lysis, hemorrhage, and coagulation, and so it was evaluated as nonirritant, according to testing score. Although it is not yet a valid method, a good correlation between the results obtained by the HET-CAM test and the eye irritation tests on rabbit was shown in previous studies. 41,42 HET-CAM is also recommended by ICCVAM to predict eye irritation potential of chemicals. 19 This method was also preferred for the evaluation of vaginal irritancy potential of chemicals. 43
Irritation potential of EW samples was evaluated with the validated EpiDerm SIT. All the EWs were assessed as nonirritant, according to the European Union (EU) and Globally Harmonized System (GHS) classification because the mean relative viability of three individual tissues exposed to the EW samples was not lower than 50% of the mean viability of the NCs. 16
EpiDerm-SIT was highly preferred for the in vitro SIT of chemicals, including cosmetics, pharmaceutical ingredients, and medical devices. 44,45 In addition to the irritation test, assay medium was collected following the exposure period, and IL-1α levels were measured as a secondary end point, which is the indicative response of keratinocytes in case of physical or chemical stress. Measurement of IL-1α was described as additional data, which helps to ensure the correct classification of the tested material. 27 EpiDerm-SIT results are consistent with that of cell viability assay. Since the two-fold increase or greater compared to the NC was considered a positive irritation response according to the test protocol, StAEW samples were considered to have no potential for irritation. 27
Conclusions
According to the microbiological assay conducted in this study, StAEW, SAEW, and MEW provided the complete bacteria inactivation in 10 min. With electroactivation, water gains high activity of ions that provide the destruction of bacteria without any harm to the living cell. In the cytotoxicity assay, no significant difference was observed between EW groups. In the HET-CAM assay for detecting the potential eye irritancy, none of the tested EWs caused eye irritation, according to the testing score. Besides, the SIT showed that none of the tested EWs caused dermal irritation, which was also confirmed by IL-1α assay.
In conclusion, it has been shown that EW samples produced in the anolyte zone (i.e. StAEW, SAEW, and MEW) have antimicrobial efficacy and are safe according to the cytotoxicity and several biocompatibility tests. Catholyte EW has no cytotoxicity effect and irritation potential. In addition, the EWs are reasonably easy to produce without using any chemicals except salt, which provides an advantage both in the technical and economical manner.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed financial support for the research, authorship, and/or publication of this article: This project is financially supported by the Scientific and Technological Research Council of Turkey (TUBITAK) 1002 program (116Z169).
