Abstract
Glioblastoma (GBM) is the most prevalent and deadliest subtype of glioma. Despite current innovations in existing therapeutic modalities, GBM remains incurable, and alternative therapies are required. Previously, we demonstrated that Olea europaea leaf extract (OLE) kills GBM cells by modulating miR-181b, miR-137, miR-153 and Let-7d expression. However, although oleuropein (OL) is the main compound in OLE, its role in the antitumour effect of OLE remains unknown. This study determined the effect of OL on GBM cell line T98G and compared the results with our previous findings regarding the effect of OLE on the same cell line. The antiproliferative activity of OL and its effect on temozolomide (TMZ) response were tested inT98G cells using WST-1 assay. OL inhibition was evaluated using one-way analysis of variance with Tukey’s post hoc test. The effect of OL on miR-181b, miR-137, miR-153 and Let-7d expression was assessed using quantitative reverse transcription polymerase chain reaction. Fold differences in expression between untreated, OL or OL + TMZ-treated samples were calculated using 2−ΔCt method. Significance was evaluated using an independent sample t-test. Treatment with 277.5 and 555 µM OL resulted in 39.51% and 75.40% reductions in T98G cells within 24 h. Coadministration of 325 µM TMZ and 277.5 or 555 µM, OL caused 2.08- and 2.83-fold increases, respectively, in the therapeutic effect of TMZ. OL + TMZ significantly increased microRNA expression, particularly Let-7d, than OLE. In conclusion, OL has an antitumour effect on GBM cells mainly via regulation of Let-7d expression. The present results also indicate other minor compounds in OLE play important anticancer roles.
Introduction
Glioblastoma multiforme (GBM; grade IV astrocytoma, World Health Organization) is the most prevalent and deadliest subtype of glioma. 1 Despite current innovations in existing therapeutic modalities, GBM remains incurable, and only a small subgroup of patients with GBM survive for >5 years. 2 GBM tumours demonstrate cellular heterogeneity and grow rapidly, diffusely and infiltratively. 3 Temozolomide (TMZ) is the only anticancer drug that has been shown in a phase III study to provide modest survival benefits in GBM. 4,5 TMZ kills tumour cells by causing double-strand breaks. 6 However, because of unmethylated O-6-methylguanine-DNA methyltransferase (MGMT) promoters, most GBM tumours are inherently resistant to TMZ. 7 Therefore, alternative therapeutic approaches for the treatment of such patients with GBM tumours are promptly required. In search of alternative therapies, several investigators have been focused on the anticancer effect of medicinal herbs, dietary plants and certain phytochemical derivatives therein.
During more than half a decade, we have focused on the effect of Olea europaea leaf extract (OLE) on GBM tumours. So far, we have demonstrated that OLE kills GBM tumour and stem-like GBM cells by modulating epigenetic mechanisms, such as microRNA (miRNA) expression and MGMT promoter methylation. 8 –11 In particular, we demonstrated that OLE reduces invasion and angiogenesis and leads to apoptosis of GBM cell lines and stem-like GBM tumour cells via regulation of miR-181b, miR-137, miR-153 and Let-7d expression. 10,11 OLE contains several potentially bioactive compounds that may have antitumour properties. 12 Biochemistry-based studies and our previous high-performance liquid chromatography findings have shown that the major compound in OLE is oleuropein (OL). 11,13 Additionally, OLE comprises several additional bioactive components, such as secoiridoids, triterpenes and flavonoids, in trace amounts. 14 Because of the high amount of OL in OLE, it may be the most effective anticancer compound within OLE. However, similarities or differences between antitumour effects of OLE and OL alone remain unknown. Therefore, the present study aimed to determine the effect of OL alone on GBM cells compared with our previous findings on the effect of OLE on the same cells.
Methods
Cells, cultures and reagents
The human GBM cell lineT98G was provided by the American Type Culture Collection (Rockville, MD, USA). Cells were grown in Dulbecco’s modified Eagle’s medium-F12 (HyClone, Logan, Utah, USA) containing
Determination of OL concentration
To compare the effect of OLE and OL on T98G cells, the same concentration of OL was used that we had predetermined in OLE (15%). In our previous study, 1 and 2 mg/mL doses of OLE were effective against T98G cells and contain 277.5 and 555 µM OL, respectively (OL molecular weight, 540.51 g/mol). 10 Therefore, the effect of 277.5 and 555 µM OL on T98G cells was evaluated in the present study.
Determination of effect of OL on tumour cell proliferation and impact on response to TMZ
The inhibitory effect of 277.5 and 555 µM OL and their synergistic or antagonistic effect on T98G cells when coadministered with TMZ were assayed using a WST-1 cell proliferation kit (Roche Applied Sciences, Mannheim, Germany) according to the manufacturer’s protocol. Briefly, T98G cells were seeded at a density of 2 × 104 cells/well in 96-well plates. Twenty-four hours after plating, 277.5 µM OL, 555 µM OL, 350 µM TMZ or a combination of the two drugs were administered to cells in quadruplicate and incubated in a humidified atmosphere with 5% CO2 at 37°C for 24 h. Then, 10 µL of WST-1 reagent was added to each well of the plate and incubated for 1 h in a humidified incubator with 5% CO2 at 37°C. After thorough shaking, the absorbance of samples was measured at 450 versus 620 nm (reference wavelength) using a microplate (enzyme-linked immunosorbent assay) reader, and results were calculated as described previously. 10,11
Evaluation of the effect of OL on miRNA expression levels
To evaluate the ability of OL to modify miRNA expression, T98G cells were seeded at a density of 3 × 105 cells/well in six-well plates. Twenty-four hours after plating, 277.5 µM OL, 555 µM OL, 350 µM TMZ or a combination of the two drugs were administered to cells in quadruplicate and incubated in humidified atmosphere with 5% CO2 at 37°C for 24 h. Cells were then subjected to total RNA extraction using RNeasy kits (Qiagen, Germantown, Maryland, USA). The total RNA concentration and protein or chemical contamination were determined by measuring 260:280 and 260:230 ratios using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Wilmington, Delaware, USA). Total RNA (5 ng) was reverse transcribed using an RT 2 miRNA First Strand kit (Qiagen). Quantitative reverse transcription polymerase chain reaction (RT-qPCR) was performed for miR-181b (MIMAT0000257), miR-153 (MIMAT0000439), miR-137 (MIMAT0000429) and Let-7d (MIMAT000-0065) in triplicate using the RT 2 QPCR Primer Assay (Qiagen) standard protocol on a StepOnePlus Real-Time PCR System (Applied Biosystems, Thermo Fisher Scientific). The 2−ΔCt method was used to calculate fold differences in miRNA expression. 15
Statistical analyses
The statistical significance of WST-1 data was evaluated using one-way analysis of variance (ANOVA) with Tukey’s post hoc test using SPSS software (release 20.0; SPSS Inc., IBM, Chicago, Illinois, USA). The statistical significance of changes in miRNA expression was determined with an independent sample t-test using the Web-based Qiagen Data Analyses Centre. The value of p < 0.05 was considered statistically significant.
Results
OL decreased GBM cell viability and increased the therapeutic effect of TMZ
In the WST-1 assay, both 277.5 and 555 µM OL reduced T98G cell numbers within 24 h by 39.51% and 75.40%, respectively. When T98G cells were treated with 277.5 or 555 µM OL in combination with 325 µM TMZ, T98G cell proliferation was reduced to58.00% and 78.75%, respectively (p = 0.193 and p = 0.007, respectively, Figure 1). According to one-way ANOVA with Tukey’s post hoc test, the inhibitory effect of both OL doses alone was significantly compared with untreated samples (277.5 µM, p = 0.048 and 555 µM, p < 0.0001, Table 1). In addition, 277.5 and 555 µM OL increased the therapeutic effect of 325 µM TMZ by 2.08- and 2.83-fold, respectively (p = 0.193 and p = 0.007, respectively, Figure 1, Table 1).
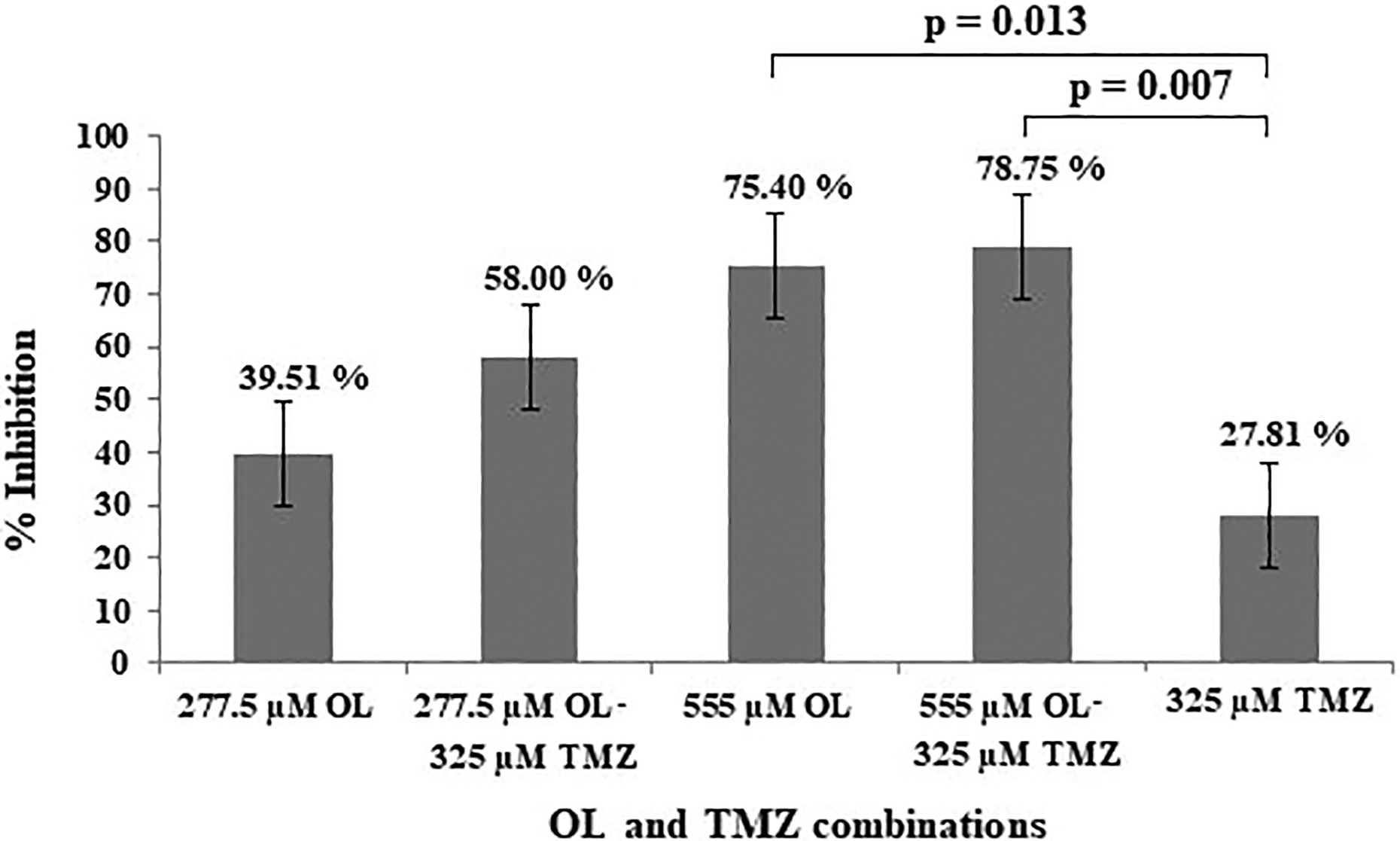
Inhibition of T98G cell viability with 277.5 and 555 µM OL. OL: oleuropein.
The effect of OL and TMZ on T98G cell proliferation.
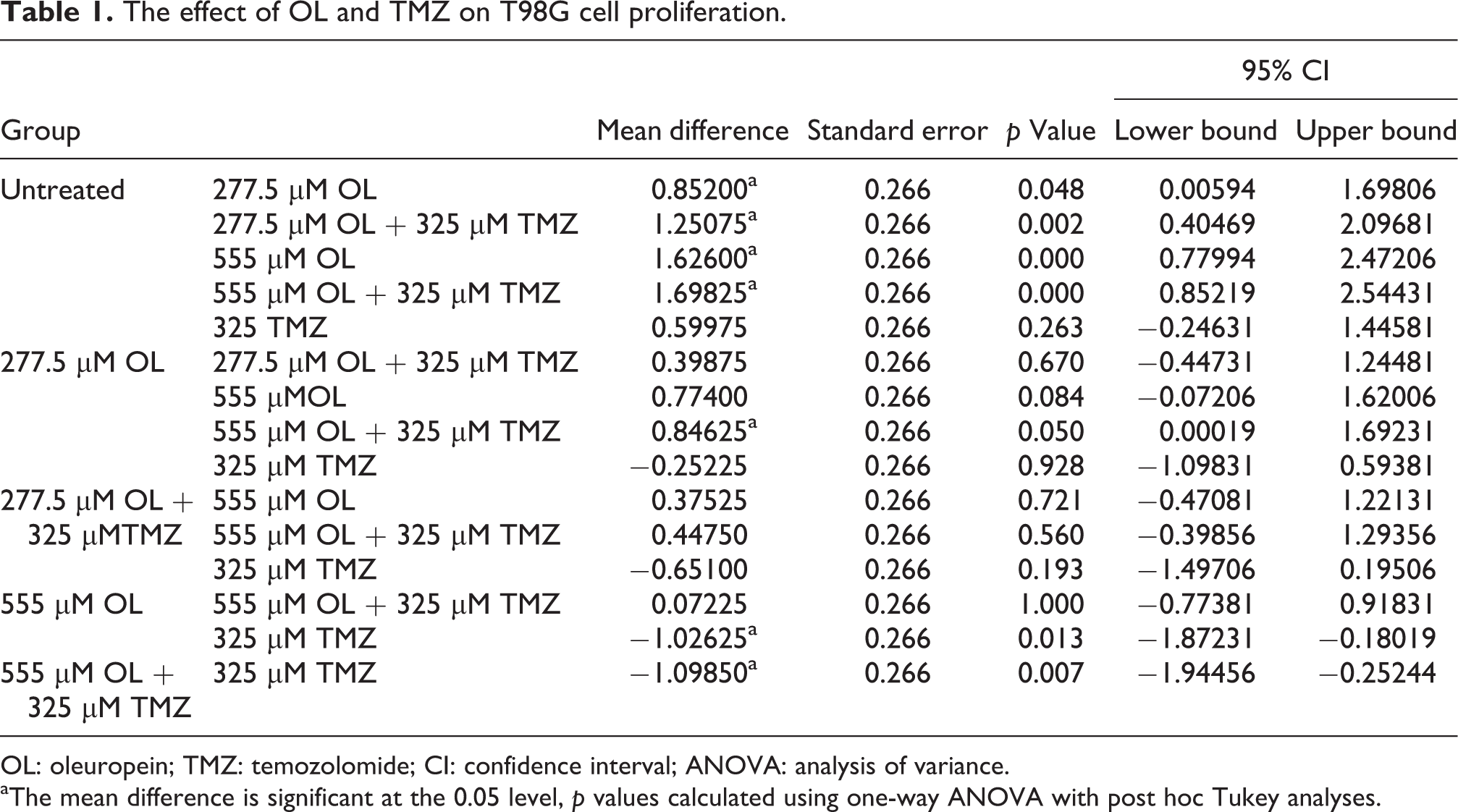
OL: oleuropein; TMZ: temozolomide; CI: confidence interval; ANOVA: analysis of variance.
aThe mean difference is significant at the 0.05 level, p values calculated using one-way ANOVA with post hoc Tukey analyses.
OL modulated miRNA expression in GBM cells
According to RT-qPCR, both OL doses differentially upregulated miR-181b, miR-137 and Let-7d expression and OL synergistically enhanced the effect of TMZ (Table 2). After treatment with 277.5 and 555 µM OL, Let-7d expression was increased by 125.94- and 269.96-fold (p = 0.01640 and p = 0.00171, respectively). Combination treatment of TMZ and 277.5 or 555 µM OL also increased Let-7d expression by 318- and 2.28-fold (p = 0.00013), which were 252.48- and 230.18-fold higher than the effect of TMZ alone (p = 0.0001, Figure 2). miR-181b and miR-137 expression was also significantly upregulated with 555 µM OL by 17.34- and 10.33-fold, respectively (p = 0.00059 and p = 0.00004, respectively, Table 2). Combination treatment of TMZ and 555 µM OL increased miR-181b and miR-137 expression up to 7.65- and 22.26-fold, respectively, compared with OL alone (Figure 2). Moreover, miR-181b and miR-137 expression levels were 132.82- and 615.75-fold higher than those induced by TMZ alone (Figure 2).
The effect of OL and TMZ on miRNA expression in T98G cells.a
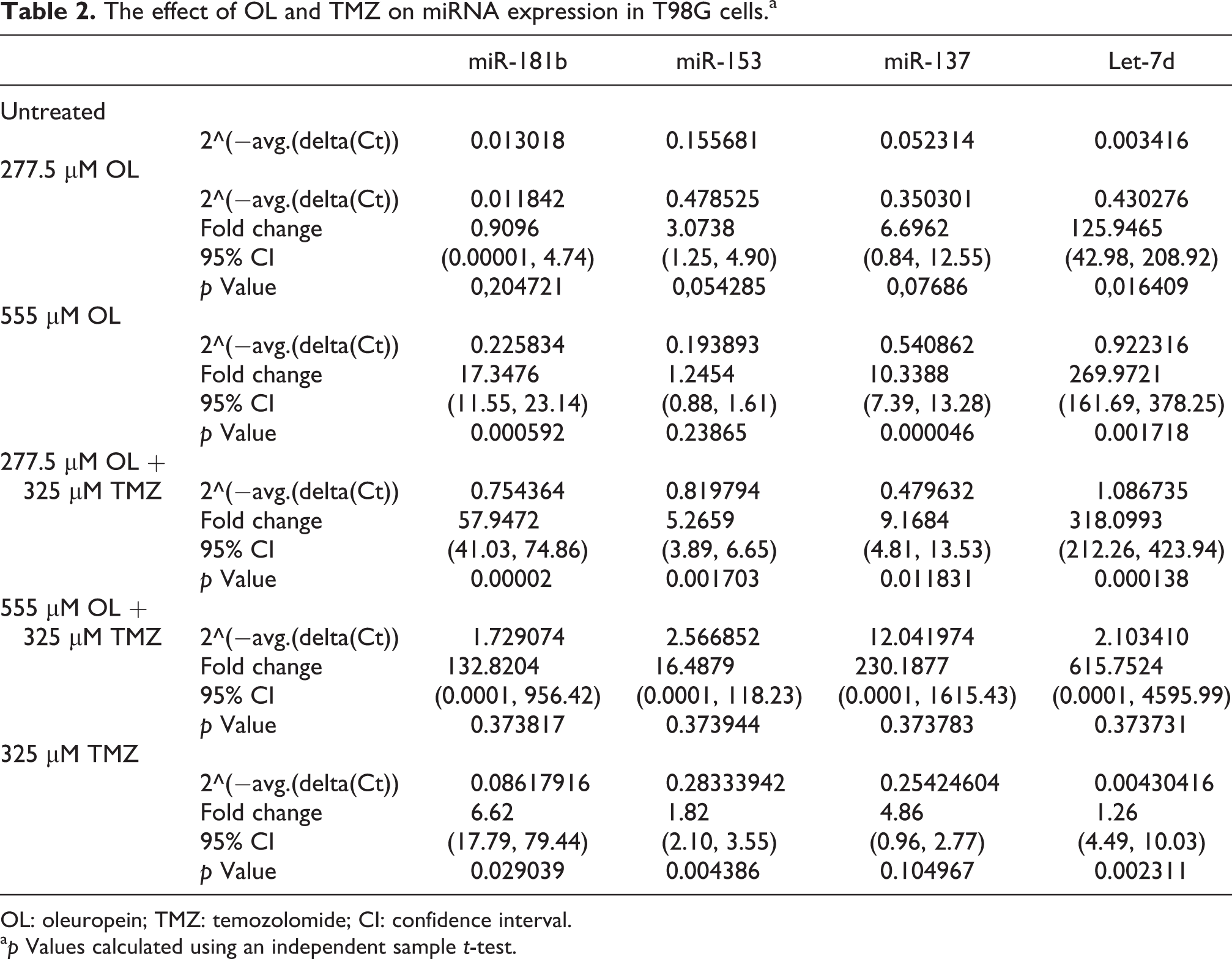
OL: oleuropein; TMZ: temozolomide; CI: confidence interval.
ap Values calculated using an independent sample t-test.

Fold differences in miRNA expression levels in T98G cells after OL or TMZ treatment. The effect of OL and TMZ was demonstrated in (a) and (b) on expression level of miR-181b, in (c) and (d) on expression level of miR-153, in (e) and (f) on the expression level of miR-137 and in (g) and (h) on the expression level of Let-7. miRNA: microRNA; OL: oleuropein; TMZ: temozolomide.
Discussion
Several studies have demonstrated that OLE polyphenols inhibit the proliferation of several cancer cell lines, including pancreatic, leukaemic and breast. 16 –19 In addition, our previous research has confirmed the anticancer effects of OLE against GBM. 8 –11 We previously demonstrated that OLE enhances the expression of tumour suppressor miRNAs, such as miR-181b, miR-153, miR-137a and Let-7d, as well as increases the response of GBM cell lines (T98G, U138MG and stem-like GBM tumour cells) to TMZ. 10,11 However, OLE comprises five different groups of compounds, including oleuropeosides, flavones, flavan-3-ols, flavonols and substituted phenols. 13 The bioactive substance within OLE with the highest concentration is the oleuropeside OL. 13 OL has consistently been reported to discriminate between cancer and normal cells, reducing cell growth and stimulating cell death only in tumour cells. 20 Recent evidence suggests that both OLE and OL protect against cancer via the modulation of miRNA expression. Abtin et al. demonstrated that OL induces MCF7 breast cancer cell apoptosis and retards migration and invasion via the downregulation of miR-21 and miR-155 expression. 21 According to recent findings of Xing et al., OL represses the radiation resistance of ovarian tumours via the suppression of miR-299 expression. 22 Similarly, OL overcomes radiation resistance in nasopharyngeal carcinoma by reducing the activity of the HIF1α–miR-519d–PDRG1 pathway. 23 In our previous studies, however, we did not determine whether the observed antitumour effects of OLE were due to OL. Therefore, the present study evaluated the therapeutic effect of OL alone on T98G cells. To compare the efficacies of OLE and OL in vitro, OL concentration used in the present study was equal to that present in the half maximal inhibitory concentrations of OLE used in our previous study (1 and 2 mg/mL). 10 OLE used in our previous study was standardized to comprise 15% OL; therefore, 1 and 2 mg/mL OLE contained 277.5 and 555 µM OL, respectively, and these two OL concentrations were used alone or in combination with TMZ to assess anticancer effects.
We determined that 277.5 and 555 µM OL reduced GBM cell proliferation by 39.51% and 75.40%, respectively, whereas 1 and 2 mg/mL OLE reduced proliferation by 74% and 81%, respectively, according to our previous study. 10 These results support the anticancer effect of OL and agree with the results of other studies in various tumour types. 24 Because the inhibitory effect of OL alone was weaker than that of OLE, our data revealed that other trace substances present in OLE substantially contribute to its therapeutic effect on GBM cells.
Similar to OLE, OL alone also had a synergistic effect on TMZ, because a combination treatment of 277.5 or 555 µM OL and TMZ resulted in a 58.00% and 78.75% reduction in cell proliferation. The synergistic effect of OL on TMZ had a greater impact on GBM cell growth than OLE alone, as shown in our previous studies. 10,11 In 2012, we used an intravenously injectable form of TMZ, and in 2014, we used ≥98% pure TMZ dissolved in DMSO. These preparation and purity differences likely caused different levels of TMZ inhibition observed in our two previous studies. However, the experimental condition of both studies was a homogenous experimental setup, and both compared fold differences within the same experimental setup. Therefore, in the present study, we did not compare differences in the synergistic effect of OL and OLE with TMZ quantitatively but relatively. In our previous study, the combined effect of 1 mg/mL OLE and 325 µM TMZ was 1.00-fold higher than the effect of TMZ alone. In the present study, the combined effect of 277.5 or 555 µM OL and 325 µM TMZ was 2.08- and 2.71-fold higher than that of TMZ alone, respectively. Although we should consider TMZ purity and preparation differences between the studies, these results may also imply the potential inhibitory effect of other OLE substances on TMZ action.
In our previous studies and other studies, the effect of plant extracts on gene regulation through modified miRNA regulation was demonstrated. 8 –11 Therefore, we evaluated differences in miRNA expression due to OL and compared them with those due to OLE in GBM cells. A brief comparative summary of the difference between the effects of OLE and OL is presented in Table 3. Neither 1 mg/mL OLE nor 277.5 µM OL significantly altered miR-181b expression. However, 555 µM OL caused an 8.54-fold increase in miR-181b expression compared with 2 mg/mL OLE. Moreover, OL significantly increased the TMZ response (p = 0.00002) related to miRNA expression. However, the stimulatory effect of OLE on TMZ response was higher than that with OL (1 mg/mL OLE = 580.03-fold increase; 277.5 and 555 µM OL = 57.94- and 132.82-fold increases, respectively). These results suggest that although OL enhanced miR-181b expression, the stimulatory effect on TMZ response comes from another trace substance in OLE. Luteolin is a natural flavone found in OLE. 13 According to a previous study, luteolin has an antitumour effect on T98G cells and enhances miR-181a expression. 25 Moreover, de Souza et al. demonstrated the synergistic anticancer activity of luteolin extracted from Achyrocline satureioides, which is high in luteolin. 26
The comparative effect of OLE and OL on fold differences of miRNA expression.
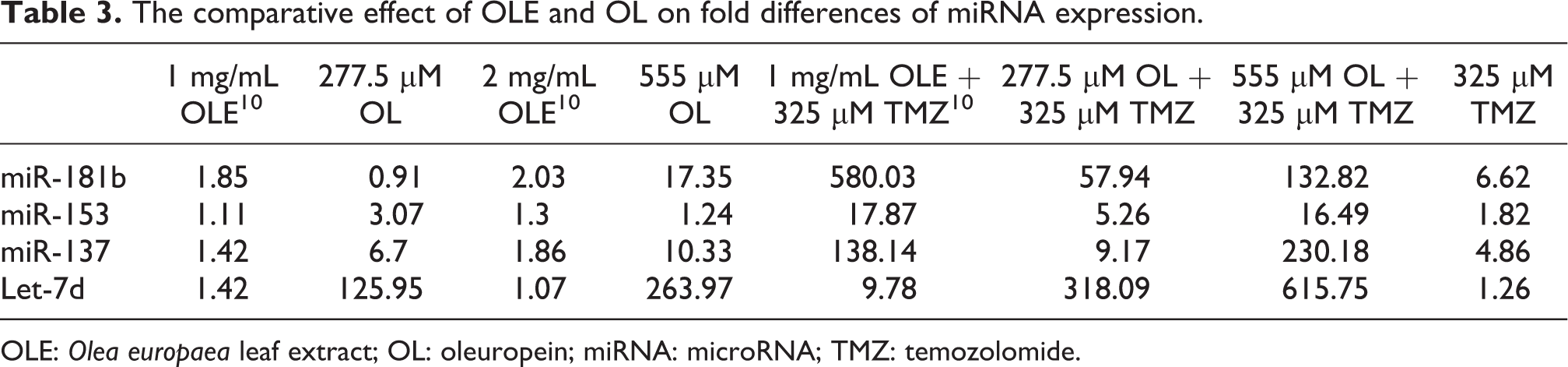
OLE: Olea europaea leaf extract; OL: oleuropein; miRNA: microRNA; TMZ: temozolomide.
The dysregulation of mammalian target of rapamycin (mTOR) signalling mediates chemoresistance in GBM. 27 A recent study demonstrated that the downregulation of miR-153 expression may cause an overactivation of mTOR signalling in GBM. 28 Similar to OLE, OL did not cause a noticeable alteration in miR-153 expression. In contrast, both OLE and OL caused a significant stimulatory effect miR-153expression in the presence of TMZ. 10,11 However, the stimulatory effect of 277.5 µM OL on TMZ response was 3.39-fold lower than that of 1 mg/mL OLE, and the difference between 1 mg/mL OLE and 555 µM OL was negligible (1.08-fold).
The chemokine CXCL12 plays a role in the regulation of migration and patterning processes in neurogenesis. However, in GBM, this protein is mostly localized in the necrotic and angiogenetic areas and mediates the proliferation of GBM progenitor cells. 29 Hatterman et al. demonstrated that sublethal doses of TMZ significantly induce CXCL12 expression, 30 and Li et al. demonstrated that miR-137 inhibits CXCL12. 31 In addition, our previous work revealed that OLE did not significantly alter miR-137 expression in T98G cells (1 mg/mL OLE: 1.41-fold; 2 mg/mL OLE: 1.86-fold), whereas a combination of OLE and TMZ stimulated miR-137 expression by 138.14-fold. 10 In contrast, the present study showed that 277.5 and 555 µM OL alone increased miR-137 expression by 6.7- and 10.33-fold, respectively. Taken together, these results clarify the role of OL in increasing TMZ response as well as prove the role of other trace amount substances of OLE besides OL on TMZ stimulation.
The expression of Let-7 family members is mostly reduced in different cancer types. 32,33 However, although Wang et al. recently revealed that Let-7 family miRNA levels are not reduced in GBM tumours, defective MYC and E-box3 binding causes an abnormal MYC-mediated transcriptional inhibition of Let-7d. 34 In a previous study, we demonstrated that c-MYC was overexpressed in stem-like GBM tumours and that the OLE reduced its expression levels. 11 Well-studied target genes of Let-7d are high mobility group A2 (HMGA2) and signal transducer and activator of transcription 3 (STAT3). 35,36 Recent evidence demonstrated that the heightened HMGA2 expression participates in glioma invasion by involving transforming growth factor-β-induced epithelial mesenchymal transition. 37,38 Stat3, which is encoded by STAT3, plays a role in the regulation of cyclin D1, Bcl2 family proteins, Rho family guanosine triphosphates, hypoxia-inducible factor 1α and vascular endothelial growth factor, thereby inducing the proliferation and invasion of tumour cells. 39 −44 Although the effect of 1 and 2 mg/mL OLE on Let-7d expression was negligible (<2-fold) in our previous study, 10 the present study revealed that T98G cells exposed to 277.5 and 555 µM OL had 125.94- and 263.97-fold increased Let-7d expression, respectively. Moreover, although the combination of 1 mg/mL OLE and 325 µM TMZ caused a 9.78-fold increase in Let-7d expression, 10 the present study showed that the combination of 277.5 or 555 µM OL and 325 µM TMZ caused 318.09- and 615.75-fold higher Let-7d expression, respectively, compared with TMZ alone. These findings suggest that the main miRNA target of OL is Let-7d.
To the best of our knowledge, the present study is the first to compare the molecular effect of OLE and its main component OL on GBM cells. Our results demonstrated that OL alone has antitumour effects on GBM cells mainly related to the regulation of Let-7d expression, whereas OLE also affected miR-181b and miR-137 expression. These results indicate that other minor substances within OLE also play important anticancer roles. Further investigation clarifying specific molecular effects of other substances within OLE on GBM tumours and comparative effects on healthy glial cells are required in future and will contribute to the production of more effective OLE-based anticancer drugs at the preclinical level.
Footnotes
Author Contributions
GT and SAA contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Uludag University Research Fund, Project No. OUAP(T)-2015/3. Preparation for publication of this article is partly supported by Turkish Neurosurgical Society.
